Abstract
Background and Objectives
As regression approaches have been used more recently to model the effectiveness and health-related quality of life (HRQOL) of novel migraine treatments, an example is provided for fremanezumab. The objective is to estimate the distribution of mean monthly migraine days (MMD) as a continuous variable and corresponding migraine-specific utility values as a function of the MMD, to inform health states in a cost-effectiveness model (CEM).
Methods
Three longitudinal regression models (zero-adjusted gamma [ZAGA], zero-inflated beta-binomial [ZIBB], and zero-inflated negative binomial [ZINBI]) were fitted to Japanese-Korean clinical trial data of episodic (EM) and chronic migraine (CM) patients treated with fremanezumab or placebo, to estimate MMD over a period of 12 months. The EQ-5D-5L and the migraine-specific quality-of-life (MSQ), mapped to the EQ-5D-3L, questionnaires were used to measure HRQOL. Migraine-specific utility values were estimated as a function of MMD using a linear mixed effects model.
Results
The ZIBB models fitted the data best in estimating the distribution of mean MMD over time. MSQ-derived values were more sensitive than the EQ-5D-5L values for the effect of the number of MMD on HRQOL, with higher values for less MMD and more time on treatment.
Conclusions
Using longitudinal regression models to estimate MMD distributions and linking utility values as a function is an appropriate method to inform CEMs and capture inter-patient heterogeneity. The observed distribution shifts demonstrated fremanezumab’s effect at reducing MMD for both EM and CM patients, while treatment effect on HRQOL was captured by MMD and time on treatment.
Plain Language Summary
The current study provides an example of an approach that can be used to estimate the number of migraine days per month and the quality of life of migraine patients. The outcomes of this approach can give an impression of how well a patient reacts to a new migraine treatment called fremanezumab. In this study, different mathematical equations were used to measure the migraine days per month and quality of life over a period of 1 year. The data came from Japanese-Korean patients that participated in clinical trials. The patients reported the number of days that they had migraine and their quality of life was measured with two validated questionnaires. With the gathered data, the quality of life was calculated for the number of migraine days that a patient could have per month. Patients who had the fewest migraine days and were treated the longest with the new treatment reported the best quality of life. The investigated approach is an appropriate method to measure the impact of fremanezumab on the number of monthly migraine days and a patient’s quality of life. The measurements of this approach can be linked to other parameters in an economic model to estimate the costs required to reach a certain level of treatment effect with this new migraine treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Longitudinal regression models can be used to estimate the distribution of the mean monthly migraine days (MMD) to inform cost-effectiveness analysis, by selecting the best-fitting model based on the Akaike information criteria and visual inspections. |
The distribution shifts compared with baseline between fremanezumab and placebo demonstrated fremanezumab’s effect at reducing MMD for both episodic and chronic migraine patients. |
Patients with fewer MMD and more time on treatment show a higher health-related quality of life (HRQOL); The migraine-specific quality-of-life questionnaire is a suitable tool to capture the HRQOL change related to MMD reduction compared with the EQ-5D-5L. |
1 Introduction
Newly emerging drugs are becoming available for the treatment of migraine but it can be challenging to capture their effectiveness and impact on health-related quality of life (HRQOL) due to the widely varying number of monthly migraine days (MMD) of patients [1]. Longitudinal regression approaches have recently been used in migraine to populate decision analytic models. These approaches are beneficial over conventional methods as they are capable of modeling MMD as a continuous outcome and thus capturing the effectiveness of migraine drugs on a patient level [1,2,3].
Migraine is a neurological condition, characterized by a throbbing headache, that can be classified as either episodic migraine (EM) (< 15 monthly headache days [MHD] including ≥ 4 MMD) and chronic migraine (CM) (≥ 15 MHD including ≥ 8 MMD over the preceding 3 months) [4, 5]. Despite the high prevalence worldwide and the burden on patients’ daily life, migraine remains underrecognized and undertreated and there is a significant medical need for treatments designed specifically to prevent migraine. Fremanezumab is an investigational calcitonin gene-related peptide monoclonal antibody that has been demonstrated to be effective in preventing EM and CM [6,7,8]. As such, fremanezumab has been approved in many countries, such as the United States, major countries in Europe, and Japan, for the treatment of migraine [9,10,11]. However, a decision analytic model to evaluate the effectiveness and cost effectiveness of fremanezumab in EM and CM patients is needed. This cost-effectiveness model (CEM) needs to capture the effect of fremanezumab on disease severity as observed in clinical trials. Conventional methods that have been used for modeling the effectiveness of migraine drugs in a CEM consist of decision tree and Markov model-based approaches that use health states with predefined ranges of MMD or headache day frequencies [12,13,14]. A downside of these approaches is that they compartmentalize patients that seem to have a similar response to the treatment or predefined categories of MMD, which can lead to a loss of information and introduce bias when the relation between model parameters and the number of MMD is non-linear in a heterogeneous population such as migraine patients [1, 2, 15,16,17].
A suggested approach to estimate clinical efficacy of fremanezumab in migraine on a patient level is estimating MMD distributions using parametric modeling techniques [3, 15, 18]. As such, it is possible to effectively model all 29 potential health states each cycle (assuming 28 days per month, including 0 MMD) and avoid grouping of patients by number of MMD. The main advantage of using parametric models is that health state membership can be estimated with only a few parameters to drive the statistical distribution. In contrast, conventional approaches would need numerous transition probabilities, which can be highly demanding on the data, and migraine models with many health states can quickly become unwieldy. Additionally, using a parametric model described by a mean and standard deviation makes extrapolating more straightforward and easier to interpret than doing so with numerous transition probabilities. Although extrapolating data always involves assumptions, it is more transparent when it concerns a ‘mean value over time’ rather than ‘many transition probabilities over time’. Another advantage of modeling MMD as a continuous outcome is that it allows for other model parameters, such as utility values, to be linked to the number of MMD on an individual level [15]. In this way, marginal differences (i.e., the difference between a patient with 25 MMD compared with 26 MMD) can be identified, and therefore the model is more sensitive to QALY changes and can potentially capture smaller QALY gains than if the patients were grouped together and assumed to have the same utility. This has a benefit on its own as a non-linear association was shown between decreasing utility values and increasing number of MMD with a potential ceiling effect [2]. However, linking utility values to individual MMD frequencies requires migraine-specific HRQOL that is sensitive enough to the MMD distributions.
Therefore, the objective of the current study is to estimate the MMD distribution as a continuous variable and corresponding migraine-specific utility values as a function of the MMD to inform health states in a CEM based on Japanese-Korean clinical trial data.
2 Methods
2.1 Overview
The current study uses patient-level clinical trial data of Japanese-Korean EM and CM patients who were treated with fremanezumab or placebo [19, 20]. Longitudinal regression models were used to estimate MMD over a period of 12 months, and the EQ-5D-5L and the migraine-specific quality-of-life (MSQ) questionnaires were used to measure HRQOL. The MSQ was mapped to the EQ-5D-3L and the HRQOL of both questionnaires were correlated to the MMD and compared with each other. Subsequently, migraine-specific utility values were estimated as a function of MMD using a linear mixed effects model. Analyses were performed in R-statistical software.
2.2 Trial Data
The trial data that was used for estimating MMD and HRQOL consisted of three sets of Japanese-Korean patient-level data. The 406-102-00001 and 406-102-00002 trials included data of CM and EM patients, respectively, gathered at different time points: baseline (i.e., 1 month prior to trial initiation), month 1, month 2, and month 3 [19, 20]. Long-term data was gathered in the 406-102-00003 trial at month 6 and month 12 [21].
The CM and EM clinical trials included 571 and 357 patients, respectively, aged between 18 and 70 years that were treated with a monthly (675 mg at baseline and 225 mg at week 4 and week 8) or quarterly dose (675 mg at baseline and placebo at week 4 and week 8) of fremanezumab or with placebo over a period of 3 months [19, 20]. The long-term trial concerned the same population but only included 34 CM and 16 EM patients who were treated with a monthly or quarterly dose of fremanezumab [21]. More details on the clinical trials are listed in Table 1 in the electronic supplementary material (ESM).
For the current study, all available patient and treatment characteristic variables were extracted from the clinical trials: age, gender, country, treatment arm, prior migraine medication use, number of migraine days for each time point, and HRQOL data measured with the EQ-5D-5L and the MSQ questionnaires over time. The MMD data from the clinical trials were normalized to 28 days of data, assuming 28 days per month. Both the EQ-5D-5L and MSQ data were collected at baseline, month 3, month 6, and month 12, while the MSQ was also collected at month 1 and month 2. More details on the baseline characteristics of the study populations of the clinical trials are reported elsewhere [19, 20].
2.3 Quality-of-Life Instruments
The EQ-5D-5L is a generic HRQOL preference-based instrument developed for clinical and economic evaluations [22], and the MSQ is a disease-specific HRQOL instrument [23]. The EQ-5D-5L measures five health dimensions: mobility, self-care, usual activity, pain/discomfort, and anxiety/depression. Each dimension contains five levels of responses to reflect the degree of problems patients have experienced: no problems (level 1), slight problems (level 2), moderate problems (level 3), severe problems (level 4), and extreme problems (level 5). The utility values were generated from the EQ-5D-5L dimension scores in the patient-level data [24].
The MSQ is designed to measure how migraines affect and/or limit daily functioning across three domains: role function–restrictive (RR) (7 items assessing how migraines limit one’s daily social and work-related activities), role function–preventive (RP) (4 items assessing how migraines prevent these activities), and emotional function (EF) (3 items assessing the emotions associated with migraines). Participants responded to items using a 6-point scale: ‘none of the time’, ‘a little bit of the time’, ‘some of the time’, ‘a good bit of the time’, ‘most of the time’, and ‘all of the time’, which are assigned scores of 1–6, respectively. Raw dimension scores are computed as a sum of item responses and rescaled from a 0–100 scale such that higher scores indicate better HRQOL [23].
2.4 Analyses
2.4.1 Mapping MSQ to EQ-5D-3L
The MSQ does not generate utility values itself. Therefore, mapping was performed using a regression model to convert MSQ responses to EQ-5D-3L utility values [25]. This published mapping algorithm was validated for this purpose and has been used before in the migraine setting [2, 25]. The EQ-5D-3L is a version of the EQ-5D with dimensions that only consist of three levels: no problems (level 1), some or moderate problems (level 2) and severe or extreme problems (level 3). Whilst mapping-generated utilities would not be the preferred approach by health technology agencies, mapping is an acceptable second-best method when no other data are available or EQ-5D is not appropriate to measure the condition, but should be performed only after sufficient assessment of the statistical validity [26]. More details on the mapping function can be found in the Appendix in the ESM.
2.4.2 Descriptive Analysis
The following MMD and EQ-5D utility value (both raw and MSQ-derived) characteristics were explored separately for the EM and CM populations: number of observations, mean, standard deviation (SD), median, interquartile range (IQR), minimum and maximum. These characteristics were estimated separately by treatment arm (placebo, fremanezumab monthly, and fremanezumab quarterly) and by time point. Histograms were generated to explore the distribution of EQ-5D scores, both raw and MSQ-derived. The correlation between MMD and raw EQ-5D-5L was graphically compared with the correlation between MMD and MSQ-derived EQ-5D-3L values. The HRQOL values that were considered to be sufficiently sensitive to MMD (i.e., showed most variation in outcomes across the number of MMD) were used to build the linear mixed effect models.
2.4.3 Modeling Monthly Migraine Days
Parametric distributions were used to estimate MMD over 12 months separately for EM and CM patients, using the data from every time point for all arms of the three trials. Three different statistical distributions were fitted for all patients to describe the MMD: the zero-inflated beta-binomial (ZIBB), the zero-inflated negative binomial (ZINBI), and the zero-adjusted gamma (ZAGA) distribution. The choice of distributions was guided by previous work that used similar approaches to model MMD [11, 27].
Zero-inflated distributions allow for the distributions to have additional weight on the zero value. Preliminary work showed that without this inflation, zero migraine days would be severely underrepresented in the modeling. These distributions have been shown to provide reasonable approximations for the observed distributions of migraine-day count data over other clinical trials, with a negative binomial distribution implemented in a recent migraine prophylaxis CEM [3, 18]. More details on the parametric distributions are described in the Appendix in the ESM.
The models (i.e., parametric distributions) were fitted to the data using generalized additive models for location scale and shape (GAMLSS), using the R package “gamlss” [28]. GAMLSS allows for separate models for distribution parameters that describe the respective location, shape, and scale (e.g., \(\mu , \sigma\) and \(\nu\)). Due to non-convergence issues with the random-effects models, fixed-effects models were used.
The following covariates were considered in the models: treatment group, coded as dummy variables for each treatment arm (e.g., placebo, monthly injection, quarterly injection); baseline MMD, as a centered variable (placebo data after baseline was not included for baseline but was used as a proxy for standard of care); scheduled visits as dummy variables (for modeling the MMD over time); the interaction of treatment group with scheduled visit (e.g., month 1, month 2, etc.) coded as dummy variables; additional baseline stratification factors of interest (age, gender, prior migraine medication use); and country (to allow for Japan-specific analyses). Forward and backward selections were used to determine which coefficients were considered as candidates for the final model. The summation of the coefficient estimates transformed with the link functions in Table 3 in the ESM calculated the parameters for each distribution that can be used to inform the distribution of MMD.
2.4.4 Modeling Utilities
To generate utility values that correspond to the MMD health states, utilities needed to be modeled as a linear function of MMD (and other covariates) due to data constraints. Therefore, the data from both the EM and CM patients, all time points, and all trial arms were stacked together to increase the power of the analysis. Since the data was collected repeatedly over time for the same patient, observations were correlated between time points, resulting in non-independence of the data. Therefore, it was necessary to account for the repeated nature of the data using linear mixed effects. These models factor in the correlation between repeated measurements and provide the option to include fixed- and random-effect terms [29].
The following covariates were considered in the models: baseline MMD; MMD value at the time of the HRQOL questionnaire; treatment arm—to allow a separate set of utilities specific to fremanezumab; scheduled visits as dummy variables; any additional baseline stratification factors of interest (age, gender, prior migraine medication use); and country. Like the MMD models, forward and backward selections were used to determine which coefficients were considered as candidates for the final models. An example specification is presented in the Appendix in the ESM.
2.4.5 Model Selection
For the MMD, model selection was primarily determined by the generalized Akaike information criterion (AIC) [30]. The AIC is based on the likelihood function with a penalty term for the number of parameters in the model. The model with the lowest AIC was preferred. Furthermore, each fitted model was compared with the underlying observed data on visual fit.
For the utilities, the optimal model was defined as the model which best reflected reality and generated plausible results (based on external data from the literature [2]). This meant that the optimal model was selected based on the level of significance, the magnitude of each estimated coefficient, and the model’s AIC statistic.
3 Results
3.1 Descriptive Results
In trials 406-102-00001 and 406-102-00003, there were 2431 MMD measurements taken from 600 patients with CM. In trials 406-102-00002 and 406-102-00003, there were 1495 MMD measurements taken from 370 patients with EM. In trial 406-102-00003, there were only 15 observations after 6 and 12 months. The distributions of all observed MMD are displayed in Figs. 1 and 2. The statistical breakdown of these assessments are reported elsewhere [19, 20].
In all trials combined, there were 1944 complete EQ-5D-5L measurements from 970 patients that were included in the utility analyses. The distribution of all EQ-5D-5L scores is displayed in Fig. 1 in the ESM. There was little variance in the reported EQ-5D-5L scores, as most scores were either ~ 0.9 or ~ 1.0. The mean value was 0.905 (SD 0.106) with a minimum–maximum range of 0.144–1.000, and a median of 0.898 (IQR 0.0860–1.000). A baseline score was available for all patients. For month 3, there were 883 observations. As only trial 406-102-00003 included measurements after 6 months and 12 months, the number of observations for these time points were 46 and 45, respectively. There was no correlation between treatment and EQ-5D-5L score observed at any of the time points.
There were 3743 MSQ questionnaires completed by 970 patients. In total 970, 910, 889, and 883 observations could be included in the analyses for baseline, month 1, month 2, and month 3, respectively. The distribution of MSQ-derived EQ-5D-3L values are displayed in Fig. 2 in the ESM. As a result of the mapping algorithm, the mean value was 0.668 (SD 0.144) with a minimum–maximum range of − 0.022 to 0.856, and a median of 0.698 (IQR 0.506–0.778). Utility values in the treated patients were higher than in the placebo group. More details on the descriptive statistics of the utility analysis can be found in Tables 4 and 5 in the ESM.
3.2 Modeling Monthly Migraine Days
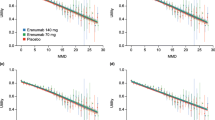
Due to small numbers, month 6 and month 12 measurements were excluded from the MMD analyses. The covariates for scheduled visits (baseline, month 2, month 3), treatment (quarterly injection, monthly injection), and interactions between treatment group and scheduled visit were required for use in an accompanying economic model. The AIC, the covariates included and excluded in the final model fits, and their level of significance are listed in Table 1. The location parameter ‘Mu’ indicated the mean MMD, with positive values resulting in more MMD and negative values in fewer MMD. Therefore, the treatment effect was reflected in the Mu coefficient values, especially when they were significant (Table 1). Temporal trends were only observed in time-related parameters (i.e., baseline, month 2, month 3).
Visual comparison of the models and the observed data for the CM and EM patients are displayed in Figs. 1 and 2, respectively. It is shown that over time, the distributions shift towards lower values of MMD compared with the baseline for EM and CM patients treated with fremanezumab, while this shift was less visible in the placebo arms.
Based on the AIC and visual comparisons, the ZIBB distribution fitted the data best, followed by ZINBI and ZAGA. The covariate values of the parametric distributions for EM and CM patients are listed in Tables 6 to 11 in the ESM.
3.3 Modeling Health State Utilities
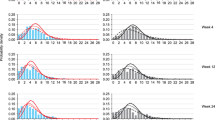
The variance in MSQ-derived EQ-5D-3L scores (Fig. 2 in the ESM) is much larger than in the raw EQ-5D-5L data (Fig. 1 in the ESM), suggesting the former might be more sensitive to the effect of MMD on HRQOL. This was further explored with correlations between patients’ HRQOL measured with the EQ-5D-5L and the MSQ-derived EQ-5D-3L with the number of MMD (Fig. 3).
The mean HRQOL as measured by the EQ-5D-5L ranged from 0.963 for 0 MMD to 0.765 for 28 MMD, and by the MSQ-derived EQ-5D-3L from 0.826 for 0 MMD and 0.512 for 28 MMD (Fig. 3). Therefore, the range was larger for MSQ-derived EQ-5D-3L values than for raw EQ-5D-5L values. Furthermore, the decline in EQ-5D scores observed with MMD 0–14 was steeper with the MSQ-derived utility values (0.826–0.611) than with the raw EQ-5D-5L scores (0.963–0.892).
Based on the lowest AIC, the MSQ-derived utility model, with the covariates baseline MMD, MMD (at the time of the HRQOL questionnaire), scheduled visits, and country, fitted the data best and its results are presented in Table 12 in the ESM. The covariates age, gender, and prior migraine medication were not included in the model as they did not improve the model’s performance. Furthermore, treatment arm was also not included in the model, which suggests that the inclusion of MMD captures the effect of treatment on HRQOL. The coefficient for MMD is − 0.009, suggesting that for every day increase in MMD, the HRQOL of patients decreases. All coefficients for visits are positive (between 0.047 and 0.070), indicating that the utility values at baseline were lower than during the trial. Additionally, the higher the baseline MMD, the lower the HRQOL, even when accounting for the current MMD. Lastly, Korean patients had, on average, a slightly lower HRQOL compared with Japanese patients (− 0.029).
Figure 4 presents the estimated health-state-specific utility values by scheduled visit when the baseline MMD value was equal to the average baseline value of all patients in all three trials (13.05) and the target population was Japanese. Results of the EQ-5D-5L utility model are listed in Table 13 in the ESM.
Estimated MSQ-derived utility values for each health state, used for the base-case analysis. A pattern can be seen as the tiles in the heatmap have the brightest green color (i.e., highest utilities) in the top right where patients have the least MMD, and more time has passed that they were on treatment. The tiles in the heatmap have the darkest red color (i.e., lowest utilities) in the bottom left where patients have the most MMD and the least time has passed that they were on treatment. There is also a clear difference in color (i.e., increase of utility values) between baseline and month 1, where patients were on treatment. An interruption of this pattern can be seen between month 3 and month 6, as most data was only available up until month 3. MMD monthly migraine days, MSQ migraine-specific quality-of-life questionnaire
4 Discussion
The objective was to estimate the MMD distribution as a continuous variable and corresponding migraine-specific utility values as a function of the MMD to inform health states in a CEM of fremanezumab based on Japanese-Korean clinical trial data. The results indicated that the ZIBB models fitted the data best. Covariates included in the final model, selected based on significance, were treatment, baseline MMD, scheduled visit, age, gender, previous medication use, and country. Due to the small response range in raw scores, we found that the EQ-5D-5L was unable to detect an impact of treatment on HRQOL with enough sensitivity in a heterogenous population such as migraine patients. Therefore, the MSQ was considered more appropriate to measure HRQOL in the current patient population. MSQ-derived EQ-5D-3L values were more sensitive for the effect of the number of MMD on HRQOL. The covariates that were selected for model inclusion were MMD, baseline MMD, scheduled visit, and country. Including treatment as a covariate did not improve model performance, indicating that the effectiveness of fremanezumab on HRQOL could be fully captured by including MMD as a continuous variable in the linear mixed effects model. We found that higher utility values were reached if patients had fewer MMD and more time had passed that they were on treatment.
The longitudinal regression models were capable of estimating MMD distributions over time that fitted the observed data well. The distribution shifts compared with the baseline between fremanezumab and placebo showed that fremanezumab had an effect on reducing MMD for both EM and CM patients, reconfirming what was already found in earlier publications [19, 20]. Modeling MMD as a continuous variable enabled us to estimate the marginal differences between patients more accurately, as this approach is more sensitive compared with conventional methods, whilst requiring less data to power it. A previous study also concluded that longitudinal regression models were a valid approach to estimate MMD distributions in migraine patients over time and highlighted the importance of model selection in relation to the data [1]. The study also showed that this approach allows for linking other outcomes such as HRQOL to be quantified directly by estimating it as a function of MMD [1]. This was done in the current research and it was found that the number of MMD and time spent on treatment had the most influence on the utility values. This is in line with the findings of another study that also modeled utility values as a function of MMD [2]. Additionally, this study also found mean utility values for patients with 0–28 MMD at baseline that were comparable to our results [2]. Our findings could indicate that the treatment provided to the patients might have a positive effect on HRQOL. However, this result should be interpreted with caution, as utility values over time are often better than the baseline, as patients who are not doing well on treatment are likely to not report their HRQOL due to the disease. We did not account for this informative censoring.
A limitation of applying longitudinal regression models to estimate MMD was that it rests on the assumption that the data follows a statistical distribution, which we cannot conclusively prove to be true and could lead to introducing bias [1]. Therefore, we tested the model fit to the observed data by looking at the AIC and visual inspection. Another limitation was that we did not externally validate our modeled results with clinical experts. However, despite the importance of validating data, it is less important in the current research as we did not use the models to interpolate or extrapolate the outcomes of interest. To make optimal use of modeling MMD as a continuous variable to inform health states in decision analytic models, it would also require patient-level data of other parameters (e.g., HRQOL, resource use, productivity losses) that could be linked to the MMD. Especially, given that these parameters are likely to have a non-linear relationship with MMD and the often limited availability of patient-level data, the alternative of using aggregated data would likely introduce bias to the model [1, 2, 17]. Another limitation in relation to the likely non-linear relationship between HRQOL and MMD was that we were limited to a linear mixed effects model to estimate utility values due to data constraints [2]. Therefore, the estimated health utilities could be slightly under- or overestimated and should be interpreted with caution. Finally, the UK tariffs used in the mapping algorithm from Gillard et al. were also considered as a limitation, as we used it to map MSQ derived values into EQ-5D-3L utilities in a Japanese-Korean population [25].
A strength of the investigated parametric approach to estimate MMD was that it is more sensitive than conventional methods, while requiring relatively little data. This would also make it easy to model the effectiveness of other migraine drugs in a CEM as well, by using published endpoints such as mean change in MMD (although with the assumption that the compared study populations are similar) [1]. Another strength of the current research is that both efficacy data of fremanezumab and HRQOL measured with a general and disease-specific questionnaire were available for the same patient population. This enabled us to compare the accuracy of capturing the HRQOL in relation to the estimated MMD with the well-established EQ-5D-5L with a mapped disease-specific questionnaire. The investigated approach proved that it was capable of modeling MMD-specific health states and link HRQOL which enabled us to capture the heterogeneity of patients with different amounts of MMD.
An implication of the current study is that using longitudinal regression models can be applied in many disease areas that have continuous data in relation to treatment efficacy and health states available that are sensitive to inter-patient heterogeneity. The investigated approach can capture this heterogeneity and reduces the chance of introducing bias. Similar regression approaches could be used to estimate disease progression in HIV/AIDS where health states are based on cell counts [31] or asthma where the number of health states are determined by intermediate results that have effect on each other [32, 33]. Our results also showed that using a disease-specific HRQOL questionnaire can sometimes be more sensitive in capturing the health utilities compared with the generally accepted EQ-5D-5L, especially in diseases with a paroxysmal nature such as migraine, where the period of awareness of symptoms can be short. This leads to higher utility values when using the EQ-5D, compared with questionnaires with a longer recall period like the MSQ [34]. This is likely not specific to our data as it was confirmed by two other studies in EM and CM patients that were conducted in the UK [35, 36].
Further research should explore whether other model parameters that can be linked to MMD can be modeled as a function of MMD in a linear and or non-linear way. Furthermore, more research should be focused on whether disease-specific HRQOL questionnaires are more sensitive in capturing health utilities compared with the EQ-5D-5L, especially in heterogeneous diseases.
5 Conclusions
Using longitudinal regression models to estimate MMD distributions and linking utility values as a function is an appropriate method to inform CEMs and capture inter-patient heterogeneity. The distribution shifts demonstrated fremanezumab’s effect at reducing MMD for both EM and CM patients. Utility values were higher as patients had fewer MMD and more time on treatment, indicating fremanezumab’s treatment effect on HRQOL.
References
Di Tanna GL, et al. Migraine day frequency in migraine prevention: longitudinal modelling approaches. BMC Med Res Methodol. 2019;19:20. https://doi.org/10.1186/s12874-019-0664-5.
Di Tanna GL, et al. Longitudinal assessment of utilities in patients with migraine: an analysis of erenumab randomized controlled trials. Health Qual Life Outcomes. 2019;17:171. https://doi.org/10.1186/s12955-019-1242-6.
Lipton RB, Brennan A, Palmer S, Jansen JP, Hatswell AJ, Porter JK, Di Tanna GL, Villa G, Shah N, Sapra S. Novel biologics versus conventional preventive therapies in migraine: a framework for economic evaluation. Value Health. 2017;20:A732. https://doi.org/10.1016/j.jval.2017.08.1997.
Katsarava Z, Buse DC, Manack AN, Lipton RB. Defining the differences between episodic migraine and chronic migraine. Curr Pain Headache Rep. 2012;16:86–92. https://doi.org/10.1007/s11916-011-0233-z.
Buse DC, et al. Chronic migraine prevalence, disability, and sociodemographic factors: results from the American Migraine Prevalence and Prevention Study. Headache. 2012;52:1456–70. https://doi.org/10.1111/j.1526-4610.2012.02223.x.
Dodick DW, et al. Effect of fremanezumab compared with placebo for prevention of episodic migraine: a randomized clinical trial. JAMA. 2018;319:1999–2008. https://doi.org/10.1001/jama.2018.4853.
Silberstein SD, et al. Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med. 2017;377:2113–22. https://doi.org/10.1056/NEJMoa1709038.
Ferrari MD, et al. Fremanezumab versus placebo for migraine prevention in patients with documented failure to up to four migraine preventive medication classes (FOCUS): a randomised, double-blind, placebo-controlled, phase 3b trial. Lancet. 2019;394:1030–40. https://doi.org/10.1016/S0140-6736(19)31946-4.
Lee MJ, Al-Karagholi MA, Reuter U. New migraine prophylactic drugs: current evidence and practical suggestions for non-responders to prior therapy. Cephalalgia. 2023;43:3331024221146315. https://doi.org/10.1177/03331024221146315.
Messina R, et al. Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: a systematic review and network meta-analysis. Cephalalgia. 2023;43:3331024231152169. https://doi.org/10.1177/03331024231152169.
National institute for Health and Care Excellence. Fremanezumab for preventing migraine. (2020).
Batty AJ, et al. The cost-effectiveness of onabotulinumtoxinA for the prophylaxis of headache in adults with chronic migraine in the UK. J Med Econ. 2013;16:877–87. https://doi.org/10.3111/13696998.2013.802694.
Brown JS, et al. Cost-effectiveness of topiramate in migraine prevention: results from a pharmacoeconomic model of topiramate treatment. Headache. 2005;45:1012–22. https://doi.org/10.1111/j.1526-4610.2005.05182.x.
Yu J, Smith KJ, Brixner DI. Cost effectiveness of pharmacotherapy for the prevention of migraine: a Markov model application. CNS Drugs. 2010;24:695–712. https://doi.org/10.2165/11531180-000000000-00000.
Lipton RB, et al. Estimating the clinical effectiveness and value-based price range of erenumab for the prevention of migraine in patients with prior treatment failures: a US societal perspective. J Med Econ. 2018;21:666–75. https://doi.org/10.1080/13696998.2018.1457533.
Royston P, Altman DG, Sauerbrei W. Dichotomizing continuous predictors in multiple regression: a bad idea. Stat Med. 2006;25:127–41. https://doi.org/10.1002/sim.2331.
Porter JK, Di Tanna GL, Lipton RB, Sapra S, Villa G. Costs of acute headache medication use and productivity losses among patients with migraine: insights from three randomized controlled trials. Pharmacoecon Open. 2019;3:411–7. https://doi.org/10.1007/s41669-018-0105-0.
Porter J. Parametric modelling of migraine day frequency in migraine prevention: a case study of erenumab clinical trial data. Value Health. 2017;20:A733.
Sakai F, et al. Efficacy and safety of fremanezumab for chronic migraine prevention: multicenter, randomized, double-blind, placebo-controlled, parallel-group trial in Japanese and Korean patients. Headache. 2021;61:1092–101. https://doi.org/10.1111/head.14169.
Sakai F, et al. Efficacy and safety of fremanezumab for episodic migraine prevention: multicenter, randomized, double-blind, placebo-controlled, parallel-group trial in Japanese and Korean patients. Headache. 2021;61:1102–11. https://doi.org/10.1111/head.14178.
Sakai F, et al. Long-term safety and tolerability of fremanezumab for migraine preventive treatment in Japanese outpatients: a multicenter, randomized, open-label study. Drug Saf. 2021;44:1355–64. https://doi.org/10.1007/s40264-021-01119-2.
EuroQolResearchFoundation. EQ-5D-5L User Guide. Version 3.0. September. (2019).
Rendas-Baum R, Bloudek LM, Maglinte GA, Varon SF. The psychometric properties of the Migraine-Specific Quality of Life Questionnaire version 2.1 (MSQ) in chronic migraine patients. Qual Life Res. 2013;22:1123–33. https://doi.org/10.1007/s11136-012-0230-7.
Shiroiwa T, et al. Comparison of value set based on DCE and/or TTO data: scoring for EQ-5D-5L health states in Japan. Value Health. 2016;19:648–54. https://doi.org/10.1016/j.jval.2016.03.1834.
Gillard PJ, Devine B, Varon SF, Liu L, Sullivan SD. Mapping from disease-specific measures to health-state utility values in individuals with migraine. Value Health. 2012;15:485–94. https://doi.org/10.1016/j.jval.2011.12.007.
Kamae I, Thwaites R, Hamada A, Fernandez JL. Health technology assessment in Japan: a work in progress. J Med Econ. 2020;23:317–22. https://doi.org/10.1080/13696998.2020.1716775.
The National Insitute for Health and Care Excellence. Erenumab for preventing migraine (TA682). https://www.nice.org.uk/guidance/ta682 (2021).
Rigby RA, Stasinopoulos DM. Generalized additive models for location, scale and shape. Appl Stat. 2005;54:507–54.
Harrison XA, et al. A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ. 2018;6: e4794. https://doi.org/10.7717/peerj.4794.
Akaike H. In: Parzen E, Tanabe K, Kitagawa G, editors. Selected Papers of hirotugu akaike. New York: Springer; 1998. p. 199–213.
Grover G, Gadpayle AK, Swain PK. A multistate markov model based on CD4 cell count for HIV/AIDS patients on antiretroviral therapy (ART). Int J Stat Med Res. 2013;2:144–51.
Exuzides A, et al. Statistical modeling of disease progression for chronic obstructive pulmonary disease using data from the ECLIPSE study. Med Decis Making. 2017;37:453–68. https://doi.org/10.1177/0272989x15610781.
Hoogendoorn M, Corro Ramos I, Baldwin M, Gonzalez-Rojas Guix N, Rutten-van Mölken M. Broadening the perspective of cost-effectiveness modeling in chronic obstructive pulmonary disease: a new patient-level simulation model suitable to evaluate stratified medicine. Value Health. 2019;22:313–21. https://doi.org/10.1016/j.jval.2018.10.008.
Powell LC, et al. Health state utility mapping of rimegepant for the preventive treatment of migraine: double-blind treatment phase and open label extension (BHV3000-305). Adv Ther. 2022. https://doi.org/10.1007/s12325-022-02369-x.
Tepper SJ, et al. Effects of galcanezumab on health-related quality of life and disability in patients with previous failure of 2–4 migraine preventive medication categories: results from a phase IIIb randomized, placebo-controlled, multicenter clinical trial (CONQUER). Clin Drug Investig. 2022;42:263–75. https://doi.org/10.1007/s40261-021-01115-5.
Khan K, et al. Mapping between headache specific and generic preference-based health-related quality of life measures. BMC Med Res Methodol. 2022;22:277. https://doi.org/10.1186/s12874-022-01762-y.
Acknowledgements
The authors would like to thank Dr Claire Simons from OPEN Health for providing technical advice on the analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by Otsuka Pharmaceuticals Co., Ltd. The funding source had a supervising role in conducting the study and in preparing the manuscript. The open access fee was paid by Otsuka Pharmaceuticals Co., Ltd.
Conflict of interest
Martijn Simons, Josef Paris, Elisabeth Peterse and Craig Bennison are employees of OPEN Health and report consulting fees from Otsuka during the conduct of the study.
Availability of data and materials
The datasets generated during and/or analyzed during the current study are not publicly available due to confidentiality.
Ethics approval
This work reports on results of a study that was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Data analyses were performed in R statistical software.
Authors’ contributions
Conceptualization and design: XW, KY, YK, JP, EP, MS, CB. Data acquisition: XW, YK. Methodology: XW, KY, YK, JP, EP, CB. Formal analysis and interpretation of the data: XW, KY, YK, JP, EP, MS, CB. Validation: XW, KY, YK, JP, EP, MS, CB. Writing original draft: EP, MS, CB. Writing review and editing: XW, KY, YK, MS, CB. Critical revision of the article for important intellectual content: XW, KY, YK, JP, EP, MS, CB.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Wang, X., Yamato, K., Kojima, Y. et al. Modeling Monthly Migraine-Day Distributions Using Longitudinal Regression Models and Linking Quality of Life to Inform Cost-Effectiveness Analysis: A Case Study of Fremanezumab in Japanese-Korean Migraine Patients. PharmacoEconomics 41, 1263–1274 (2023). https://doi.org/10.1007/s40273-023-01288-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-023-01288-1