Abstract
Objectives
Longitudinal prescription patterns of antiepileptic drugs (AEDs) have not been described to date in Korea. Here we aimed to describe AED prescribing trends over a 12-year period and assess age differences in AED prescribing patterns in a pediatric epilepsy population.
Methods
We retrieved and analyzed all AED prescribing and dispensing data in 2001–2012 in patients aged 0–18 years with an established diagnosis of epilepsy at the largest tertiary children’s hospital in Korea. AEDs included for analysis were classified as older (i.e., carbamazepine, ethosuximide, phenobarbital, phenytoin, and valproic acid) and newer (i.e., gabapentin, lamotrigine, levetiracetam, oxcarbazepine, pregabalin, topiramate, vigabatrin, zonisamide, lacosamide, and rufinamide) on the basis of market availability before versus after 1991.
Results
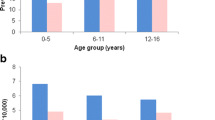
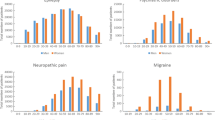
A total of 5593 patients with epilepsy were prescribed an AED during the 12-year period. The proportion of newer AED prescriptions was 52.6 % in 2001 and continuously increased to 74.3 % in 2012. Oxcarbazepine was most widely used, followed by valproic acid. While carbamazepine and vigabatrin use progressively decreased over the 12-year period, those of lamotrigine and topiramate rapidly increased. Age differences in prescribing patterns were observed. Polytherapy was observed in 49.7 % of the total population, while 83.9 % of new users were prescribed monotherapy.
Conclusion
This study provided updated information on AED prescription trends for childhood epilepsy. We found a progressive increase in the use of newer AEDs. However, valproic acid, the only prevalent older AED, continued to be widely prescribed. A high rate of polytherapy among the prescriptions overall raises some safety concerns.



Similar content being viewed by others
References
Camfield PR, Camfield CS. What happens to children with epilepsy when they become adults? Some facts and opinions. Pediatr Neurol. 2014;51:17–23.
Kotsopoulos IA, van Merode T, Kessels FG, de Krom MC, Knottnerus JA. Systematic review and meta-analysis of incidence studies of epilepsy and unprovoked seizures. Epilepsia. 2002;43:1402–9.
Savica R, Beghi E, Mazzaglia G, Innocenti F, Brignoli O, Cricelli C, Caputi AP, Musolino R, Spina E, Trifiro G. Prescribing patterns of antiepileptic drugs in Italy: a nationwide population-based study in the years 2000–2005. Eur J Neurol. 2007;14:1317–21.
van de Vrie-Hoekstra NW, de Vries TW, van den Berg PB, Brouwer OF, de Jong-van den Berg LT. Antiepileptic drug utilization in children from 1997–2005–a study from the Netherlands. Eur J Clin Pharmacol. 2008;64:1013–20.
Alacqua M, Trifiro G, Spina E, Moretti S, Tari DU, Bramanti P, Caputi AP, Arcoraci V. Newer and older antiepileptic drug use in Southern Italy: a population-based study during the years 2003–2005. Epilepsy Res. 2009;85:107–13.
Bielen I, Sruk A, Planjar-Prvan M, Cvitanovic-Sojat L, Kosicek M, Bergman-Markovic B, Baraba R, Butkovic-Soldo S. Age-related pattern of the antiepileptic drug utilization in active epilepsy: a population-based survey. Coll Antropol. 2009;33:659–63.
Hsia Y, Neubert A, Sturkenboom MC, Murray ML, Verhamme KM, Sen F, Giaquinto C, Ceci A, Wong IC. Comparison of antiepileptic drug prescribing in children in three European countries. Epilepsia. 2010;51:789–96.
Malerba A, Ciampa C, De Fazio S, Fattore C, Frassine B, La Neve A, Pellacani S, Specchio LM, Tiberti A, Tinuper P, Perucca E. Patterns of prescription of antiepileptic drugs in patients with refractory epilepsy at tertiary referral centres in Italy. Epilepsy Res. 2010;91:273–82.
Landmark CJ, Fossmark H, Larsson PG, Rytter E, Johannessen SI. Prescription patterns of antiepileptic drugs in patients with epilepsy in a nation-wide population. Epilepsy Res. 2011;95:51–9.
Hamer HM, Dodel R, Strzelczyk A, Balzer-Geldsetzer M, Reese JP, Schoffski O, Graf W, Schwab S, Knake S, Oertel WH, Rosenow F, Kostev K. Prevalence, utilization, and costs of antiepileptic drugs for epilepsy in Germany–a nationwide population-based study in children and adults. J Neurol. 2012;259:2376–84.
Nicholas JM, Ridsdale L, Richardson MP, Ashworth M, Gulliford MC. Trends in antiepileptic drug utilisation in UK primary care 1993–2008: cohort study using the General Practice Research Database. Seizure. 2012;21:466–70.
Strzelczyk A, Haag A, Reese JP, Nickolay T, Oertel WH, Dodel R, Knake S, Rosenow F, Hamer HM. Trends in resource utilization and prescription of anticonvulsants for patients with active epilepsy in Germany. Epilepsy Behav. 2013;27:433–8.
Italiano D, Capuano A, Alibrandi A, Ferrara R, Cannata A, Trifirò G, Sultana J, Ferrajolo C, Tari M, Tari DU, Perrotta M, Pagliaro C, Rafaniello C, Spina E, Arcoraci V. Indications of newer and older anti-epileptic drug use: findings from a southern Italian general practice setting from 2005–2011. Br J Clin Pharmacol. 2015;79:1010–9.
Bourgeois FT, Olson KL, Poduri A, Mandl KD. Comparison of drug utilization patterns in observational data: antiepileptic drugs in pediatric patients. Pediatr Drugs. 2015;13:401–410. doi:10.1007/s40272-015-0139-z.
Kwong KL, Tsui KW, Wu SP, Yung A, Yau E, Eva F, Ma CK, Cherk S, Liu KT, Cheng WW, Yau MM. Utilization of antiepileptic drugs in Hong Kong children. Pediatr Neurol. 2012;46:281–6.
Tan WW, Kong ST, Chan DW, Ho PC. A retrospective study on the usage of antiepileptic drugs in Asian children from 2000 to 2009 in the largest pediatric hospital in Singapore. Pharmacoepidemiol Drug Saf. 2012;21:1074–80.
Mattsson P, Tomson T, Edebol Eeg-Olofsson K, Brannstrom L, Ringback Weitoft G. Association between sociodemographic status and antiepileptic drug prescriptions in children with epilepsy. Epilepsia. 2012;53:2149–55.
Oteri A, Trifiro G, Gagliostro MS, Tari DU, Moretti S, Bramanti P, Spina E, Caputi AP, Arcoraci V. Prescribing pattern of anti-epileptic drugs in an Italian setting of elderly outpatients: a population-based study during 2004–07. Br J Clin Pharmacol. 2010;70:514–22.
Chin RF, Ajetunmobi O, Weir CJ, Wood R. Prescription of emergency antiepileptic medication after a first childhood seizure: analysis of routine administrative data. Epileptic Disord. 2015;17:172–6.
Lee SY, Jung KY, Lee IK, Yi SD, Cho YW, Kim DW, Hwang SS, Kim S, Korean Epilepsy S. Prevalence of treated epilepsy in Korea based on national health insurance data. J Korean Med Sci. 2012;27:285–90.
Lyons B, Abdelmagid E, Banerjee S. Use of newer antiepileptic drugs in children: how NICE are we? Prog Neurol Psychiatry. 2010;14:6–10.
Williams K, Thomson D, Seto I, Contopoulos-Ioannidis DG, Ioannidis JP, Curtis S, Constantin E, Batmanabane G, Hartling L, Klassen T, Sta RCHG. Standard 6: age groups for pediatric trials. Pediatrics. 2012;129(Suppl 3):S153–60.
Bhatt KM, Malhotra SD, Patel KP, Patel VJ. Drug utilization in pediatric neurology outpatient department: a prospective study at a tertiary care teaching hospital. J Basic Clin Pharm. 2014;5:68–73.
Schmidt D, Sachdeo R. Oxcarbazepine for treatment of partial epilepsy: a review and recommendations for clinical use. Epilepsy Behav. 2000;1:396–405.
Glauser T, Ben-Menachem E, Bourgeois B, Cnaan A, Guerreiro C, Kalviainen R, Mattson R, French JA, Perucca E, Tomson T. Updated ILAE evidence review of antiepileptic drug efficacy and effectiveness as initial monotherapy for epileptic seizures and syndromes. Epilepsia. 2013;54:551–63.
Yang MS, Kang MG, Jung JW, Song WJ, Kang HR, Cho SH, Min KU. Clinical features and prognostic factors in severe cutaneous drug reactions. Int Arch Allergy Immunol. 2013;162:346–54.
Chen SY, Wu N, Boulanger L, Sacco P. Antiepileptic drug treatment patterns and economic burden of commercially-insured patients with refractory epilepsy with partial onset seizures in the United States. J Med Econ. 2013;16:240–8.
Rosenow F, Schade-Brittinger C, Burchardi N, Bauer S, Klein KM, Weber Y, Lerche H, Evers S, Kovac S, Hallmeyer-Elgner S, Winkler G, Springub J, Niedhammer M, Roth E, Eisensehr I, Berrouschot J, Arnold S, Schröder M, Beige A, Oertel WH, Strzelczyk A, Haag A, Reif PS, Hamer HM. The LaLiMo Trial: lamotrigine compared with levetiracetam in the initial 26 weeks of monotherapy for focal and generalised epilepsy–an open-label, prospective, randomised controlled multicenter study. J Neurol Neurosurg Psychiatry. 2012;83:1093–8.
Ackers R, Murray ML, Besag FM, Wong IC. Prioritizing children’s medicines for research: a pharmaco-epidemiological study of antiepileptic drugs. Br J Clin Pharmacol. 2007;63:689–97.
Freitas-Lima P, Baldoni Ade O, Alexandre V, Pereira LR, Sakamoto AC. Drug utilization profile in adult patients with refractory epilepsy at a tertiary referral center. Arq Neuropsiquiatr. 2013;71:856–61.
Kim JM, Kwon S, Seo HE, Choe BH, Cho MH, Park SP. Long-term effectiveness and tolerability of topiramate in children with epilepsy under the age of 2 years: 4-year follow-up. J Korean Med Sci. 2009;24:1078–82.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No significant financial support was received for this study.
Conflict of interest
Y.S. Cho, Y.-M. Ah, A.H. Jung, K.J. Kim, and J.-Y. Lee declare that they have no conflict of interest.
Additional information
Y. S. Cho and Y.-M. Ah contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cho, Y.S., Ah, YM., Jung, A.H. et al. Trends in Antiepileptic Drug Prescriptions for Childhood Epilepsy at a Tertiary Children’s Hospital in Korea, 2001–2012. Pediatr Drugs 17, 487–496 (2015). https://doi.org/10.1007/s40272-015-0147-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40272-015-0147-z