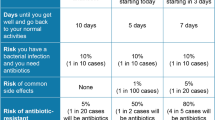
Abstract
Objectives
Identify Australian public preferences for antibiotic treatments in the context of antibiotic stewardship.
Methods
A discrete choice experiment (DCE) was conducted in Australia to investigate the importance of seven attributes associated with antibiotic treatments and related stewardship practices: contribution to antimicrobial resistance (AMR), treatment duration, side effects, days needed to recover, days before taking antibiotics, treatment failure and out-of-pocket costs. The DCE data were analysed using conditional logit, mixed logit and latent class conditional logit models. The relative importance of each attribute was calculated.
Results
A total of 1882 respondents completed the survey; the main study sample consist of 1658 respondents (mean age 48 years) who passed quality checks. All seven attributes significantly influenced respondents’ preferences for antibiotic treatments. Based on the designed attribute levels in the DCE, on average, out-of-pocket costs (32.8%) and contribution to antibiotic resistance (30.3%) were the most important attributes, followed by side effects (12.9%). Days before starting medication was least important (3.9%). Three latent classes were identified. Class 1 (including respondents who were more likely to be older and more health literate; 24.5%) gave contribution to antibiotic resistance greater importance in treatment preferences. Class 2 (including respondents more likely to report poorer health; 25.2%) gave out-of-pocket costs greater importance. The remaining (50.4%), who were generally healthier, perceived side effects as the most important attribute.
Conclusions
Despite concerted public awareness raising campaigns, our results suggest that several factors may influence the preferences of Australians when considering antibiotic use. However, for those more likely to be aware of the need to preserve antibiotics, out-of-pocket costs and limiting the contribution to antibiotic resistance are the dominant influence. Delays in starting treatment were not important for any latent class, suggesting public tolerance for this measure. These results could help inform strategies to promote prudent antibiotic stewardship.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
A large survey (n = 1658) with diverse and broadly representative sample of Australian public exploring the factors that influence preferences for antibiotic treatments. |
Three population subgroups identified, with out-of-pocket costs and contribution to antibiotic resistance the dominant factors for first two. |
Group three had far greater preference heterogeneity, within which contribution to AMR is not a key concern, such that stressing individual responsibility may have limited impacts on personal decision making. |
There is a need for more nuanced approach and targeted interventions to enhance public awareness of AMR. |
1 Introduction
Antimicrobial resistance (AMR) is a pressing global public health concern with serious health and financial consequences [1, 2]. Current estimates indicate that about 10 million people die each year from sepsis and other consequences of bacterial infections, which equates to more than 20% of deaths globally [3, 4]. AMR will significantly increase the burden of this mortality and morbidity in coming decades unless effective action is taken [5, 6]. A major driver of the development of AMR is indiscriminate or inappropriate use of antibiotics. Avoiding antibiotic misuse requires decisions to be made about how best to use these therapeutic resources [7]. The rates of AMR in Australia are low compared with other countries but are steadily rising because of high rates of community antibiotic use [1, 8]. In Australia, antibiotics must be prescribed by a medical practitioner (most frequently in a community setting by a general practitioner or GP). The cost of antibiotics is publicly subsidized on the Pharmaceutical Benefits Scheme with a means tested contribution from the patient.
Campaigns deploying social marketing techniques targeting the Australian public began in the early 2000s to raise community awareness about AMR and to encourage people not to seek antibiotics when experiencing cold or flu symptoms [9]. Given the sustained high rates of antibiotic use in the Australian community, the impact of past public awareness raising efforts has been modest [10, 11]. Recent analyses indicate that antibiotics continue to be overprescribed in Australia compared with guideline recommendations. For example, the 2021 AURA [12] report highlights that Therapeutic Guidelines: Antibiotic recommends acute bronchitis not be treated with antibiotics, yet 81% of adult patients received a prescription for this condition. Patient expectations and other social factors can influence prescribing behaviour, such that communities can have cultures of high or low antibiotic use [13, 14]. Further exploration of factors which are most important in influencing patient decision making and the preferences of the public regarding antibiotic treatments can inform strategies to promote prudent antibiotic stewardship.
A discrete choice experiment (DCE) is a quantitative methodology which has been widely utilised to study what factors influence people’s decisions [15]. The technique is an attribute-based measure of benefit, based on the assumption that services (such as antibiotic treatments) can be described by a number of key attributes (characteristics), and that an individual’s valuation of the services depends upon the levels of these attributes [16]. Participants are given hypothetical scenarios comprising different levels of attributes and asked to choose between two or more alternatives. Individuals’ stated preferences reflect their perceived benefit, where the benefit is defined in terms of the economics concept of ‘'utility’ or value [17].
This study will advance knowledge in the field by assessing and estimating the preferences of the Australian public for antibiotic treatment characteristics, costs and risks entailed by measures to support antibiotic stewardship. It has three unique contributions to the literature: (i) we include and explore more individual characteristics not considered in previous studies to potentially explain the heterogeneity of stated preferences; (ii) we use comparable attributes and levels in the choice experiments to a DCE conducted in Sweden [18], permitting comparisons between countries with low and high community antibiotic consumption [19]; and (iii) we use a sample size larger than previous studies [18, 20].
2 Methods
An online survey was developed and consisted of two key components—the choice experiments and respondents’ socio-demographic characteristics, including their experiences and attitudes towards using antibiotics. The survey preamble informed respondents that the aim of the study was to understand what is most important to the public when it comes to antibiotic use in healthcare, and to develop a better appreciation of which antibiotic stewardship measures to control antibiotic use reflect what most people will accept as fair and good for public health. To introduce the choice tasks, they were then told the survey had ten questions (including a warm-up task) comprising two imaginary yet possible versions of how antibiotics could be used in Australia. For each question, they should indicate which scenario presented in the choice task they preferred. Ethical approval to conduct this study was obtained from the University of Wollongong Human Research Ethics Committee [2021/312].
2.1 DCE Design
This DCE included seven attributes developed in adherence with methodological standards [21]. Attributes were developed based on published estimates of AMR-related burdens in Australia [22, 23], a review of DCE studies on preferences for antibiotic prescribing and treatments with Australian and Danish GPs [24, 25], as well as DCE conducted on this topic with members of the public in other jurisdictions [18, 20]. In addition, we consulted with representatives from different implicated stakeholder groups in Australia. The attributes and corresponding levels were finalised drawing on input from these groups and the extensive expertise of the research team which includes infectious disease physicians, pharmacists, health service researchers and health social scientists. The final attribute measures included (1) contribution to AMR (DrugResist), (2) treatment duration (Duration), (3) side effects (SideEff), (4) days needed to recover (Recover), (5) days before taking antibiotics (Medication), (6) treatment failure (Failure) and (7) out-of-pocket cost (Cost) (see Table 1).
We conducted an initial face-to-face piloting of the survey with nine lay people to ensure adequate comprehension and interpretation of the wording of questions, and to allow the interviewer to refine effective means of communicating the choice tasks. The wording of some attributes was subsequently revised before the soft and subsequently full launch of the survey. A D-efficient design was used to generate a total of 72 choice sets for presentation using the Ngene version 1.1.1 DCE design software package (www.choice-metrics.com). These choice sets were divided into eight versions so that each respondent completed nine choice sets to reduce cognitive burden. One dominant choice task (i.e. one alternative that is clearly better or no worse than the other alternative in all attributes) was further included to help select the respondents who make a rational choice to be included in the main analyses.
2.2 Data Analysis
The data from the DCE were analysed within the framework of random utility theory, which assumes that respondents choose the alternative that maximizes their utility [26]. The utility function could be written as U = V + ε, where V is a vector of observed components (i.e. the attributes and levels presented to respondents) and ε is an unobservable error term. Assuming an additive function form of V, it can be further written as:
The probability (P) of respondent I choose an alternative j from k candidate alternatives could be specified as, \({P}_{ij}=\mathrm{exp}\left({V}_{ij}\right)/\sum_{k}\mathrm{exp}({V}_{ij})\). In the above equation, all attributes are treated as categorical variables and effect coding was used. The omitted levels of all attributes are highlighted in Table 1 and are included in regression results. Three statistical methods were considered, including a conditional logit (CL) model that assuming a homogeneous preference among respondents, as well as methods that allow for potential preference heterogeneity, including a mixed-logit (MIXL) model and a latent-class conditional logit (LCL) model [27,28,29]. For the MIXL model, a normal distribution was specified for all effect coded variables. The Online Supplementary Material includes additional results, in which the cost attribute was specified as a continuous variable to calculate the willingness to pay (WTP). The choice of the optimal method for analysing DCE data, as well as the choice of the optimal number of latent classes for the LCL, were decided by the Bayesian information criterion (BIC). After identifying the optimal number of latent classes, we further explored the latent class memberships by including respondents’ characteristics (sex, household income levels, risk attitudes towards health, whether in the metro area and whether the respondent was born in Australia) as well as experience and attitudes about antibiotics. The final model specification only contains the statistically significant membership predictors. When membership predictors are directly included in the LCL model, the share of each latent class could slightly change as compared with the case when only attributes are included.
We presented the relative importance of each attribute, which was calculated as the percentage of the utility range of each attribute (i.e. the estimated coefficient of the most preferred attribute level minus the estimated coefficient of the least preferred attribute level) to the summary of the utility range of all seven attributes. It should be noted that the calculation and interpretation of the relative importance of each attribute is specific to the levels included in this particular DCE. All statistical analyses were performed in Stata version 16.1 (StataCorp LP, College Station, Texas, USA).
2.3 Respondents’ Recruitment
A sample of the Australian general public, aged 18 years and older, were recruited by Dynata (www.dynata.com), an online panel provider in Australia. All respondents provided informed consent before accessing the survey and they could withdraw from the study at any time before survey completion. A target sample size of 1000 adults in Australia was used. We requested the online panel company to apply demographic (age and gender) quotas to ensure that a broader representation of the views of the Australian adult population could be achieved. The study was conducted from 5 to 23 November 2021. Owing to an unexpected technical issue, the recruitment continued after the initial targeting sample size was achieved. A total of 1882 people completed the online survey (Fig. 1).
For the main analyses, we excluded respondents who either (1) completed the whole survey in less than 2.5 minutes (medium duration was 8.5 minutes for all respondents) or who completed the DCE tasks extremely fast, in less than 0.8 minutes (around 2% of all respondents) (N = 43), or (2) failed the dominant DCE task (N = 191), leaving a final study sample size of 1658 (88% of the original sample). The final study sample was overrepresented by females and older adults. Consequently, we further created a post-stratification weight [30] to align the final study sample with June 2021 population statistics of the Australian population according to age group and gender distributions. For more details on the creation of the post-stratification weight, see Online Supplementary Table 1.
3 Results
Respondents of the study sample had a mean age (range) of 48 (18–69) years and 63% were females. Around 77% of respondents were born in Australia, 14% spoke a language other than English at home and 37% had completed a Bachelor's degree or above education. Applying the post-stratification weight adjusted the age and gender distributions as expected. More details on respondents’ characteristics are presented in Table 2.
Supplementary Table 2 presents further details on respondents’ characteristics, self-reported health, experience and attitudes towards antibiotics usage. In brief, one-quarter of respondents self-assessed their health to be fair or poor, 38% of respondents had not been prescribed any antibiotic in the last 2 years, 6.5% definitely wanted an antibiotic and 33% definitely had not wanted antibiotics for a cold or flu, and 69% had heard of the need to reduce the use of unnecessary antibiotics in healthcare.
Table 3 reports the DCE results based on CL and MIXL models that include the post-stratification weight. Sensitivity analyses results without a post-stratification weight and include all respondents or a subsample of main study sample of those who spent at least 2 min on DCE tasks are presented in Supplementary Tables 3A, 3B and 4, with broadly similar findings. All seven attributes statistically and significantly influence respondents’ preferences. The statistically significant standard deviations in MIXL estimates also indicate the existence of preference heterogeneity among respondents. Regarding the attribute importance, we could see very similar results between CL and MIXL estimates. Based on the MIXL estimates (Fig. 2), we could see that out-of-pocket costs (32.8%) and contribution to antibiotic resistance (30.3%) were the two most important attributes, followed by side effects (12.9%), days needed to recover (8.7%), treatment failure (6.3%) and treatment duration (5.1%). Days before starting medication was the least important attribute (3.9%).
For the latent class conditional logit (LCL) model, we initially explored two-class, three-class and four-class scenarios and the three-class LCL model had the lowest BIC and is the preferred model. The three-class LCL model has also been found to have better performance than MIXL and CL (see the BIC values reported in Table 3 and Supplementary Table 5).
The optimal three-class LCL estimates are reported in Supplementary Table 5, whilst Table 4 further presents the LCL estimates including a set of latent class membership predictors. Overall, the results are robust, indicating that the share of each class only changed marginally (i.e. 51%–25%–24% without predictors versus 50%–25%–25% including predictors). The relative importance of the seven attributes in Fig. 2 reveals the differences among the three latent classes. Respondents in class 1 pay special attention to the contribution of antibiotic resistance (70%), respondents in class 2 focused particularly on out-of-pocket costs (61%), whilst respondents in class 3 demonstrated a more balanced preference for all seven attributes as compared with the previous two classes. This pattern is also highlighted in the more detailed coefficients reported in Table 4, suggesting that different levels of treatment duration attribute were statistically insignificant for class 1, treatment failure attribute was insignificant for class 2 and that all seven attributes were statistically significant in class 3.
We explored what types of respondents were more likely to be in the first two classes, as compared with class 3 which accounts for half of the respondents. Except for the predictors in Table 4 (which are statistically significant at a 5% significance level), we originally also considered a series of other socio-demographic characteristics, such as sex, household income levels, risk attitudes towards health, location (i.e. whether in the metro area) and whether the respondent was born in Australia. However, they were excluded from the final model because none of them were statistically significant (all non-reported results are available from the corresponding author).
Generally speaking, we could see respondents in class 1, who paid more attention to the contribution of antibiotic resistance, were more likely to be older adults, highly educated, had no antibiotic prescriptions in the last 2 years and had heard of the need to reduce unnecessary antibiotic use in healthcare. Out-of-pocket cost was most significant to the preferences of class 2. Furthermore, as compared with class 3, respondents from the first two classes were more likely to have fair or poor health, and were less likely to hold the attitude that they ‘definitely want to receive antibiotics for a cold or flu’.
4 Discussion
We investigated the preferences of members of the Australian public for different antibiotic treatment characteristics and treatment outcomes. All seven attributes had influence on participant’s choices—some have larger influence on respondents’ preference than others. We found that members of the public had strong preferences for keeping the out-of-pocket costs of antibiotics low, while also preferring antibiotic treatments that do not contribute to the further development of antibiotic resistance. Notably, unfavourable treatment outcomes such as side effects and treatment failure were not strongly disvalued by participants until the upper extremes of attribute levels (Table 2). This suggests that for most participants the current low levels of antibiotic resistance—recently estimated by Wozniak and colleagues [22] to cause approximately 600 deaths in per year Australia—and the associated impacts on treatment outcomes are not important enough to warrant changing their preferences for antibiotics, but larger negative impacts may change public preferences significantly.
Respondent preferences exhibited marked preference heterogeneity, such that three population subgroups were identified. Class 1 could be characterised as ‘health literate older people’ who were most concerned about antibiotic resistance and less likely to have had a course of antibiotics in the last few years. The higher level of education and decreased likelihood of recent antibiotic use that characterises this group is notable given that results from systematic reviews and cross-sectional surveys consistently indicate that the capacity to engage with the biological aspects of AMR is strongly associated with educational attainment [31,32,33]. Out-of-pocket cost was most significant to the preferences of class 2. Notably, in the current study, sensitivity to the influence of higher out-of-pocket costs was tied to lower levels of self-reported health, rather than lower social and economic status which has been reported elsewhere [34, 35]. These two classes clearly differ with respect to what drives their behaviours related to antibiotic use. Class 3 were generally much healthier than the other 2 classes and less likely to endorse antibiotic-preserving behaviours. Our working assumptions is that class 3 have not really thought about AMR much before taking part in survey and therefore were much more likely to be concerned about and weigh up all the attributes in making their choices.
Geographic location and rurality were not significant to latent class composition in the current study. People living in rural and remote areas of Australia often have higher levels of disease, poorer health outcomes and less access to primary and tertiary healthcare than those in metropolitan areas [36]. Yet the Australian Commission on Safety and Quality in Health Care (ACSQHC) atlas of healthcare variation shows a strong gradient in prescribing, where urban populations receive more antibiotics per capita than rural and remote populations [37]. The most recent ACSQHC report (2022) on antimicrobial use in Australia shows that areas with the lowest antibiotic dispensing rates are often near to, or contiguous with, areas with the highest dispensing rate, indicating that local physician preference could be a major influence on antibiotic use [12]. That the gradient in antibiotic prescribing between Australian urban and rural locations is not a function of consumer or patient preferences is consistent with the results of the current study.
Of direct relevance to our results, a small scale DCE and interview study conducted with Australian GPs in 2017 showed that these healthcare providers believe that patient expectation is the dominant modifiable factor influencing antibiotic prescribing [24]. However, as Table 4 shows, contribution to antibiotic resistance was not significant for about half the participants in our study (class 3), whereas the other half (classes 1 and 2) were less likely to expect antibiotics for a cold or flu. Because contribution to AMR is not a key concern for much of our sample of the Australian public, stressing individual responsibility for antibiotic resistance in clinical and societal communication may have limited impacts on personal decision making. Previous research indicates that health communications about AMR can spread inequitably through a population, such that even as awareness is increased, there is limited effect on attitudes and behaviours [38]. Our results suggest that AMR-related health messaging and social marketing has not been internalised by this cohort, which may suggest a need for a more nuanced approach targeting the concerns of different groups. Beyond this speculation, it is difficult to explain the general lack of agreement with antibiotic preserving behaviours as being significant to class 3 in the class probability model. Further research into this variable would benefit greater understanding and better targeted interventions to address these outcomes.
The sensitivity of class 2 to price signals raises a different set of concerns. Using increases in out-of-pocket costs to discourage antibiotic use can create significant equity issues and may promote undertreatment. But messaging that emphasises the cost to individuals of treatment failure (and the costs of second-line antibiotics) might discourage unnecessary use among this group. Against this background it is notable that relative to the other attributes measured in the DCE, delays in starting treatment and treatment duration were not as important to treatment choice for any of the latent classes or in general. Even though the evidence for the effectiveness of delayed prescribing is weak at best (and arguably this step shifts responsibility for antibiotic stewardship from GPs to patients) [39, 40], our result suggests a level of public tolerance for these measures to lower antibiotic resistance. This is broadly consistent with a recent DCE on antibiotic use for respiratory symptoms conducted in the UK, which found that certain groups appear to be amenable to delayed prescription, indicating a possibility for greater targeted use of this strategy [41].
Finally, participant preferences in our Australian study are very similar to those found using a DCE methodology with members of the public in Sweden (n = 378) where contribution to AMR followed by out-of-pocket costs were the most important attributes shaping decision making [18]. Similar patterns are also seen in the Swedish study with three latent classes organised around contribution to AMR, out-of-pocket costs, and side-effect rates. But the socio-demographic characteristics of the latent classes in Sweden were slightly different where, for example, contribution to AMR was most important to a group of participants more likely to be of younger ages compared with the class of older health literate participants who gave this attribute greatest significance in Australia. The third latent class found in Sweden was older and more health literate and gave greatest importance to side effects; whereas in Australia this class was characterised by greater preference heterogeneity, expressed as a more equally weighted set of concerns. Variations in latent class characteristics across national settings are to be expected and may reflect key differences in the response of health authorities in Australia and Sweden to the risks posed by antibiotic resistance [19, 42]. In Sweden, the focus has been on establishing and sustaining prescriber compliance with AMS principles. Public education has been given a much lower priority, such that communications have emphasised the negative effects of antibiotics for the individuals and the broader costs to the health system of the unnecessary use [43]. The relatively lower rates of community prescribing in Sweden compared with Australia suggest that practice-level interventions may also be required to activate prescribers and the communities they serve to make substantive reductions in inappropriate antibiotic use [11].
This study has several strengths and limitations. Public preferences were elicited using a robust quantitative method informed by expert conceptual review, stakeholder consultation and qualitative work prior to the DCE, and the DCE survey was piloted. DCE uses hypothetical real-life scenarios so that respondents can report their preference trade-offs between antibiotic treatment attributes, enabling identification of the attributes that most influence antibiotic stewardship acceptability, as well as the expected level of heterogeneity of preferences for different interventions. The group sampled was large, diverse and broadly representative of the age and geographic characteristics of the Australian population but skewed towards female gender and higher education levels. Advanced analytic modelling methods were employed to assess factors driving the heterogeneity of preferences identified by the survey.
Although several attributes could potentially be coded as continuous variables and estimate the marginal rates of substitution, the latent class analyses results indicate that specifying attributes such as treatment duration, side effects, treatment failure or cost would require imposing very strong assumptions owing to the identified non-linear effects among attribute levels. When a cost attribute is included in the DCE, researchers also commonly report the WTP estimates. Albeit the limitation we just mentioned (i.e. the need to impose a strong linear assumption), we have reported the WTP results in Supplementary Table 6 for interested readers. Readers could further use the reported coefficients of each latent class to predict and compare the probability of different type of respondents’ preferences for managing antimicrobial resistance in Australia using the equation in the Methods section. A further limitation of the study is the survey design such that the relative position of each attribute in the choice task, the number of levels, and length of definitions for attribute and levels may have had some influence of participant choices. Finally, the definition of the attribute ‘days needed to recover’ did not specify whether this was with or without antibiotics. Even though the implication is that antibiotics would be used, participants may have interpreted this attribute differently. Although the DCE method is robust, its results indicate estimations of reported preferences at a point in time. Preferences may change, including in response to an infectious disease event or a rapid escalation in AMR. A further limitation of the current study is that internet access was required for participation.
5 Conclusions
Australia has high rates of antibiotic consumption in the community compared with other comparable settings [11, 12]. Despite concerted public awareness raising campaigns, our results suggest that several factors may influence the preferences of Australians when considering antibiotic use. However, for those more likely to be aware of the need to preserve antibiotics, out-of-pocket costs and limiting the contribution to antibiotic resistance are the dominant influence. Comparisons between policy settings, the rates of antibiotic consumption and DCE results in Sweden suggest that practice-level interventions may also be required in Australia to activate prescribers and patients to make reductions in inappropriate antibiotic use.
References
Murray CJL, Ikuta KS, Sharara F, Swetschinski L, Robles Aguilar G, Gray A, et al. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. The Lancet. 2022 2022/01/19/.
O’Neill J. Tackling drug-resistant infections globally final report and recommendations. London: Wellcome Trust & HM Government; 2016.
Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. The Lancet. 2020;395(10219):200–11.
Ikuta KS, Swetschinski LR, Robles Aguilar G, Sharara F, Mestrovic T, Gray AP, et al. Global mortality associated with 33 bacterial pathogens in 2019: a systematic analysis for the Global Burden of Disease Study 2019. The Lancet. 2022;400(10369):2221–48.
Laxminarayan R, Duse A, Wattal C, Zaidi AK, Wertheim HF, Sumpradit N, et al. Antibiotic resistance—the need for global solutions. Lancet Infect Dis. 2013;13(12):1057–98.
Chokshi A, Sifri Z, Cennimo D, Horng H. Global contributors to antibiotic resistance. J Glob Infect Dis. 2019;11(1):36.
Littmann J, Viens AM, Silva DS. The Super-Wicked Problem of Antimicrobial Resistance. In: Jamrozik E, Selgelid M, editors. Ethics and drug resistance: collective responsibility for global public health. Cham: Springer International Publishing; 2020. p. 421–43.
Australian Institute of Health and Welfare. OECD Health Care Quality and Outcomes Indicators, Australia 2021,. 2022. https://www.aihw.gov.au/reports/international-comparisons/oecd-health-care-quality-and-outcomes-indicators-2021/contents/prescribing-in-primary-care
Wutzke SE, Artist MA, Kehoe LA, Fletcher M, Mackson JM, Weekes LM. Evaluation of a national programme to reduce inappropriate use of antibiotics for upper respiratory tract infections: effects on consumer awareness, beliefs, attitudes and behaviour in Australia. Health Promot Int. 2007;22(1):53–64.
Weekes L, Mackson J, Artist M, Wutzke S. An ongoing national programme to reduce antibiotic prescription and use. Microbiol Aust. 2007;28(4):201–4.
Hawkins O, Scott AM, Montgomery A, Nicholas B, Mullan J, van Oijen A, et al. Comparing public attitudes, knowledge, beliefs and behaviours towards antibiotics and antimicrobial resistance in Australia, United Kingdom, and Sweden (2010–2021): a systematic review, meta-analysis, and comparative policy analysis. PLoS One. 2022;17(1): e0261917.
ACSQHC. AURA. Fourth Australian report on antimicrobial use and resistance in human health. Sydney: Australian Commission on Safety and Quality in Health Care; 2021. p. 2021.
Broom A, Kenny K, Prainsack B, Broom J. Antimicrobial resistance as a problem of values? Views from three continents. Crit Public Health. 2020;31(4):451–63.
Hulscher ME, Grol RP, Van Der Meer JW. Antibiotic prescribing in hospitals: a social and behavioural scientific approach. Lancet Infect Dis. 2010;10(3):167–75.
Lancsar E, Louviere J. Conducting discrete choice experiments to inform healthcare decision making. Pharmacoeconomics. 2008;26(8):661–77 (2008/08/01).
de Bekker-Grob EW, Ryan M, Gerard K. Discrete choice experiments in health economics: a review of the literature. Health Econ. 2012;21(2):145–72.
Ryan M, Gerard K, Amaya-Amaya M. Discrete choice experiments in a nutshell. Using discrete choice experiments to value health and health care. Springer; 2008. p. 13–46.
Ancillotti M, Eriksson S, Andersson DI, Godskesen T, Nihlén Fahlquist J, Veldwijk J. Preferences regarding antibiotic treatment and the role of antibiotic resistance: a discrete choice experiment. Int J Antimicrob Agents. 2020;56(6): 106198 (2020/12/01/).
Andersson K, van Driel M, Hedin K, Hollingworth S, Merlo G. Antibiotic use in Australian and Swedish primary care: a cross-country comparison. Scand J Primary Health Care. 2022;40(1):95–103 (2022/01/02).
Dorgali M, Longo A, Vass C, Shields G, Harrison R, Boeri M. Exploring Antimicrobial Resistance(AMR) from a societal perspective: preferences and welfare impacts in the United Kingdom. Value Health. 2020;23(S2):S619.
Bridges JF, Hauber AB, Marshall D, Lloyd A, Prosser LA, Regier DA, et al. Conjoint analysis applications in health—a checklist: a report of the ISPOR Good Research Practices for Conjoint Analysis Task Force. Value Health. 2011;14(4):403–13.
Wozniak TM, Dyda A, Merlo G, Hall L. Disease burden, associated mortality and economic impact of antimicrobial resistant infections in Australia. Lancet Region Health Western Pacific. 2022;27: 100521.
OECD. Stemming the superbug tide: just a few dollars more. Paris: OECD Publishing; 2018.
Lum EPM, Page K, Whitty JA, Doust J, Graves N. Antibiotic prescribing in primary healthcare: dominant factors and trade-offs in decision-making. Infect Dis Health. 2018;23(2):74–86 (2018/06/01/).
Sydenham RV, Jarbøl DE, Hansen MP, Justesen US, Watson V, Pedersen LB. Prescribing antibiotics: factors driving decision-making in general practice. A discrete choice experiment. Soc Sci Med. 2022;305: 115033 (2022/07/01/).
McFadden D. Conditional logit analysis of qualitative choice behavior. In: Zarembka P, editor. Frontiers of econometrics. New York: Academic Press; 1973. p. 105–42.
McFadden D, Train K. Mixed MNL models for discrete response. J Appl Economet. 2000;15(5):447–70.
Hole AR. Fitting mixed logit models by using maximum simulated likelihood. Stand Genomic Sci. 2007;7(3):388–401.
Yoo HI. lclogit2: an enhanced command to fit latent class conditional logit models. Stand Genomic Sci. 2020;20(2):405–25.
Royal K. Survey research methods: a guide for creating post-stratification weights to correct for sample bias. Educ Health Profess. 2019;2(1):48–50 (2019 January 1).
McCullough A, Parekh S, Rathbone J, Del Mar C, Hoffmann T. A systematic review of the public’s knowledge and beliefs about antibiotic resistance. J Antimicrob Chemother. 2016;71(1):27–33.
Gualano MR, Gili R, Scaioli G, Bert F, Siliquini R. General population’s knowledge and attitudes about antibiotics: a systematic review and meta-analysis. Pharmacoepidemiol Drug Saf. 2015;24(1):2–10.
Anderson A. Online health information and public knowledge, attitudes, and behaviours regarding antibiotics in the UK: multiple regression analysis of Wellcome Monitor and Eurobarometer Data. PLoS One. 2018;13(10): e0204878.
Zanichelli V, Tebano G, Gyssens IC, Vlahović-Palčevski V, Monnier AA, Stanic Benic M, et al. Patient-related determinants of antibiotic use: a systematic review. Clin Microbiol Infect. 2019;25(1):48–53 (2019/01/01/).
Masiero G, Filippini M, Ferech M, Goossens H. Socioeconomic determinants of outpatient antibiotic use in Europe. Int J Public Health. 2010;55(5):469–78 (2010/10/01).
Australian Institute of Health and Welfare. Rural and Remote Health, Australia 2021. AIHW: Australian Government; 2022.
First Australian Atlas of Healthcare Variation. https://www.safetyandquality.gov.au/our-work/healthcare-variation/atlas-2015: Australian Commission on Safety and Quality in Health Care; 2015.
Haenssgen MJ, Xayavong T, Charoenboon N, Warapikuptanun P, Khine ZY. The Consequences of AMR education and awareness raising: outputs, outcomes, and behavioural impacts of an antibiotic-related educational activity in Lao PDR. Antibiotics. 2018;7(4):95.
Lum EPM, Page K, Nissen L, Doust J, Graves N. Australian consumer perspectives, attitudes and behaviours on antibiotic use and antibiotic resistance: a qualitative study with implications for public health policy and practice. BMC Public Health. 2017;17:799.
Sargent L, McCullough A, Del Mar C, Lowe J. Is Australia ready to implement delayed prescribing in primary care?: a review of the evidence. Aust Fam Physician. 2016;45(9):688–90.
Morrell L, Buchanan J, Roope LSJ, Pouwels KB, Butler CC, Hayhoe B, et al. Public preferences for delayed or immediate antibiotic prescriptions in UK primary care: a choice experiment. PLoS Med. 2021;18(8): e1003737.
Gunnarsson R, Ebell MH, Wächtler H, Manchal N, Reid L, Malmberg S, et al. Association between guidelines and medical practitioners’ perception of best management for patients attending with an apparently uncomplicated acute sore throat: a cross-sectional survey in five countries. BMJ Open. 2020;10(9): e037884.
Mölstad S, Löfmark S, Carlin K, Erntell M, Aspevall O, Blad L, et al. Lessons learnt during 20 years of the Swedish strategic programme against antibiotic resistance. Bull World Health Organ. 2017;95(11):764–73.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This study was funded by a University of Wollongong Global Challenges Grant. The funder had no role in the design, conduct or analyses of the study nor the decision to publish.
Conflicts of Interest
The authors have no relevant financial or non-financial interest to declare.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Gang Chen and Chris Degeling. The first draft of the manuscript was written by Chris Degeling and Gang Chen and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Data Availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request and subjected to the approval from the ethics review committee.
Ethics Approval
Ethical approval to conduct this study was obtained from the University of Wollongong Human Research Ethics Committee [2021/312].
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent to Publish
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Degeling, C., Yarwood, T., Nettel-Aguirre, A. et al. Exploring the Preferences of the Australian Public for Antibiotic Treatments: A Discrete Choice Experiment. Patient 16, 555–567 (2023). https://doi.org/10.1007/s40271-023-00640-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40271-023-00640-z