Abstract
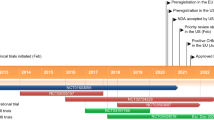
Subcutaneous vosoritide (Voxzogo®), a modified recombinant human C-type natriuretic peptide (CNP) analogue, is the first precision therapy approved for the treatment of achondroplasia in the EU, USA and multiple other countries. It is indicated for patients aged ≥ 2 years with open epiphyses in the EU and those aged ≥ 5 years with open epiphyses in the USA. In a phase 3 trial (study 111-301), vosoritide 15 μg/kg/day significantly improved annualized growth velocity and height Z-score compared with placebo, without worsening upper-to-lower body segment ratio in children with achondroplasia. The growth-promoting effects of vosoritide persisted with continued treatment for up to 60 months. Vosoritide is generally well tolerated, with most adverse events being mild in severity. Longer-term safety data revealed no new safety signals.
Plain Language Summary
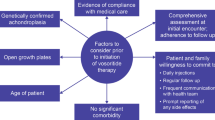
Achondroplasia is the most common form of skeletal dysplasia and is characterized by short stature with disproportionate bone growth. The condition is caused by a gain-of-function mutation in the gene for fibroblast growth factor receptor 3 (FGFR3), which is a negative regulator of bone growth. With increased understanding of the pathogenesis of achondroplasia, vosoritide (Voxzogo®), a modified recombinant human C-type natriuretic peptide (CNP) analogue, has been approved as the first precision therapy for the treatment of achondroplasia in patients aged ≥ 2 years with open epiphyses in the EU and those aged ≥ 5 years with open epiphyses in the USA. In children with achondroplasia, daily subcutaneous administration of vosoritide was associated with growth-promoting effects. Vosoritide is generally well tolerated, with most adverse events being mild in severity. Thus, vosoritide is a valuable, effective and generally well-tolerated treatment option for children with achondroplasia.

Similar content being viewed by others
References
Wrobel W, Pach E, Ben-Skowronek I. Advantages and disadvantages of different treatment methods in achondroplasia: a review. Int J Mol Sci. 2021;22(11):5573.
Semler O, Rehberg M, Mehdiani N, et al. Current and emerging therapeutic options for the management of rare skeletal diseases. Paediatr Drugs. 2019;21(2):95–106.
Constantinides C, Landis SH, Jarrett J, et al. Quality of life, physical functioning, and psychosocial function among patients with achondroplasia: a targeted literature review. Disabil Rehabil. 2021. https://doi.org/10.1080/09638288.2021.1963853.
Legeai-Mallet L, Savarirayan R. Novel therapeutic approaches for the treatment of achondroplasia. Bone. 2020;141: 115579.
Shediac R, Moshkovich O, Gerould H, et al. Experiences of children and adolescents living with achondroplasia and their caregivers. Mol Genet Genomic Med. 2022;10(4): e1891.
Clark PA, Ramireddy K, Koneru M, et al. Achondroplasia: to treat or not to treat. Internet J Intern Med. 2021;14(1).
Hogler W, Ward LM. New developments in the management of achondroplasia. Wien Med Wochenschr. 2020;170(5–6):104–11.
Kitoh H, Matsushita M, Mishima K, et al. Disease-specific complications and multidisciplinary interventions in achondroplasia. J Bone Miner Metab. 2022;40(2):189–95.
Cormier-Daire V, AlSayed M, Ben-Omran T, et al. The first European consensus on principles of management for achondroplasia. Orphanet J Rare Dis. 2021;16(333):1–7.
Savarirayan R, Ireland P, Irving M, et al. International consensus statement on the diagnosis, multidisciplinary management and lifelong care of individuals with achondroplasia. Nat Rev Endocrinol. 2022;18(3):173–89.
Fafilek B, Bosakova M, Krejci P. Expanding horizons of achondroplasia treatment: current options and future developments. Osteoarthr Cartil. 2021;30(4):535–44.
BioMarin Pharmaceutical. Voxzogo (vosoritide) for injection, for subcutaneous use: US prescribing information. 2021. https://www.accessdata.fda.gov/. Accessed 20 July 2022.
BioMarin Pharmaceutical. BioMarin announces the Ministry of Health, Labor and Welfare (MHLW) in Japan granted approval for Voxzogo® (vosoritide) for injection for the treatment of children with achondroplasia, whose growth plates are not closed [media release]. 21 June 2022. https://investors.biomarin.com/.
BioMarin Pharmaceutical. Australian product information—voxzogo (vosoritide). 2022. https://www.tga.gov.au/. Accessed 22 July 2022.
BioMarin Pharmaceutical. Voxzogo: EU summary of product characteristics. 2021. https://www.ema.europa.eu/. Accessed 20 July 2022.
Wendt DJ, Dvorak-Ewell M, Bullens S, et al. Neutral endopeptidase-resistant C-type natriuretic peptide variant represents a new therapeutic approach for treatment of fibroblast growth factor receptor 3-related dwarfism. J Pharmacol Exp Ther. 2015;353(1):132–49.
Lorget F, Kaci N, Peng J, et al. Evaluation of the therapeutic potential of a CNP analog in a Fgfr3 mouse model recapitulating achondroplasia. Am J Hum Genet. 2012;91(6):1108–14.
Mackenzie D, Bell S, Heglar B, et al. In vitro signaling characteristics and dosing evaluation of vosoritide, a CNP-variant for the treatment of achondroplasia [abstract no. MON-944]. J Bone Miner Res. 2019;34(Suppl 1):374.
Chan ML, Qi Y, Larimore K, et al. Pharmacokinetics and exposure-response of vosoritide in children with achondroplasia. Clin Pharmacokinet. 2021;61(2):263–80.
Savarirayan R, Irving M, Bacino CA, et al. C-type natriuretic peptide analogue therapy in children with achondroplasia. N Engl J Med. 2019;381(1):25–35.
Hoover-Fong J, Irving M, Bacino C, et al. Vosoritide for children with achondroplasia: a 60-month update from an ongoing phase 2 clinical trial [abstract no. eP151]. Mol Genet Metab. 2021;132(Suppl 1):S101.
Polgreen LE, Irving M, Hoover-Fong J, et al. Vosoritide for children with achondroplasia: a 60-month update from an ongoing phase 2 clinical trial. Horm Res Paediatr. 2021;94(Suppl 2):134.
Savarirayan R, Tofts L, Irving M, et al. Once-daily, subcutaneous vosoritide therapy in children with achondroplasia: a randomised, double-blind, phase 3, placebo-controlled, multicentre trial. Lancet. 2020;396(10252):684–92.
Savarirayan R, Tofts L, Irving M, et al. Safe and persistent growth-promoting effects of vosoritide in children with achondroplasia: 2-year results from an open-label, phase 3 extension study. Genet Med. 2021;23(12):2443–7.
Irving M, Hoover-Fong J, Bacino C, et al. Vosoritide for children with achondroplasia: growth velocity and pubertal milestones [abstract no. OPO21]. Mol Genet Metab. 2022;24(3 Suppl):S352–3.
Savarirayan R, Wilcox W, Harmatz P, et al. A randomized controlled trial of vosoritide in infants and toddlers with achondroplasia [abstract]. In: ENDO, 2022.
Savarirayan R, Baratela W, Butt T, et al. Literature review and expert opinion on the impact of achondroplasia on medical complications and health-related quality of life and expectations for long-term impact of vosoritide: a modified Delphi study. Orphanet J Rare Dis. 2022;17(1):224.
Acknowledgements
The manuscript was reviewed by: H. Kitoh, Department of Orthopaedic Surgery, Aichi Children's Health and Medical Center, Obu, Japan; K. Mohnike, Otto-von-Guericke-Universität, Magdeburg, Germany. During the peer review process, BioMarin Pharmaceuticals Inc., the marketing authorization holder of vosoritide, was also offered an opportunity to provide a scientific accuracy review of their data. Changes resulting from comments received were made on the basis of scientific and editorial merit.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Authorship and conflict of interest
Young-A Heo, a salaried employee of Adis International Ltd/Springer Nature and an editor of Drugs & Therapy Perspectives, was not involved in any publishing decisions for the manuscript and declares no relevant conflicts of interest. All authors contributed to the review and are responsible for the article content.
Ethics approval, Consent to participate, Consent for publication, Availability of data and material, Code availability
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Heo, YA. Vosoritide in achondroplasia: a profile of its use. Drugs Ther Perspect 38, 417–422 (2022). https://doi.org/10.1007/s40267-022-00942-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40267-022-00942-6