Abstract
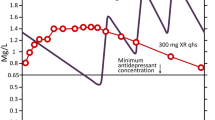
Once-daily prolonged-release trazodone [Triticum® OD (Europe), Oleptro™ (USA)] offers an option for the treatment of depression in adult patients. It is effective in reducing the symptoms of depression and improving the quality of sleep, and is generally well tolerated. The prolonged release of trazodone over 24 h provided by the novel once-daily trazodone formulation may reduce the risk of adverse effects (particularly daytime sedation) associated with the early high-peak trazodone plasma concentrations obtained with immediate-release formulations of the drug, and may also enhance adherence to antidepressant treatment. The prolonged-release tablets can broken in half, providing flexibility in dosing.

Similar content being viewed by others
References
Fagiolini A, Comandini A, Dell’Osso MC, et al. Rediscovering trazodone for the treatment of major depressive disorder. CNS Drugs. 2012;26(12):1033–49.
Haria M, Fitton A, McTavish D. Trazodone: a review of its use pharmacology, therapeutic use in depression and therapeutic potential in other disorders. Drugs Aging. 1994;4(4):331–55.
Triticum OD 150 and 300 mg prolonged-release tablets: summary of product characteristics. Cruz Quebrada-Dafundo: Angelini Farmacêutica, Lda; 2014.
Oleptro™ (trazodone hydrocloride tablet, extended release): US prescribing information. Gaithersburg (MD): Angelini Pharma Inc.; 2014.
Papakostas GI, Fava M. A meta-analysis of clinical trials comparing the serotonin (5HT)-2 receptor antagonists trazodone and nefazodone with selective serotonin reuptake inhibitors for the treatment of major depressive disorder. Eur Psychiatry. 2007;22(7):444–7.
Gartlehner G, Hansen RA, Morgan LC, et al. Comparative benefits and harms of second-generation antidepressants for treating major depressive disorder: an updated meta-analysis. Ann Intern Med. 2011;155(11):772–85.
Beasley CM Jr, Dornseif BE, Pultz JA, et al. Fluoxetine versus trazodone: efficacy and activating-sedating effects. J Clin Psychiatry. 1991;52(7):294–9.
Cunningham LA, Borison RL, Carman JS, et al. A comparison of venlafaxine, trazodone, and placebo in major depression. J Clin Psychopharmacol. 1994;14(2):99–106.
Kasper S, Olivieri L, Di Loreto G, et al. A comparative, randomised, double-blind study of trazodone prolonged-release and paroxetine in the treatment of patients with major depressive disorder. Curr Med Res Opin. 2005;21(8):1139–46.
Moon CA, Laws D, Stott PC, et al. Efficacy and tolerability of controlled-release trazodone in depression: a large multicentre study in general practice. Curr Med Res Opin. 1990;12(3):160–8.
Munizza C, Olivieri L, Di Loreto G, et al. A comparative, randomized, double-blind study of trazodone prolonged-release and sertraline in the treatment of major depressive disorder. Curr Med Res Opin. 2006;22(9):1703–13.
Weisler RH, Johnston JA, Lineberry CG, et al. Comparison of bupropion and trazodone for the treatment of major depression. J Clin Psychopharmacol. 1994;14(3):170–9.
Blacker R, Shanks NJ, Chapman N, et al. The drug treatment of depression in general practice: a comparison of nocte administration of trazodone with mianserin, dothiepin and amitriptyline. Psychopharmacology. 1988;95(Suppl):S18–24.
Feighner JP, Boyer WF. Overview of USA controlled trials of trazodone in clinical depression. Psychopharmacology. 1988;95(Suppl):S50–3.
Owens MJ, Morgan NW, Plott SJ, et al. Neurotransmittter receptor and transporter binding profile of antidepressants and their metabolites. J Pharmacol Exp Ther. 1997;283(3):1305–22.
Sheehan DV, Croft HA, Gossen ER, et al. Extended-release trazodone in major depressive disorder: a randomized, double-blind, placebo-controlled study. Psychiatry (Edgmont). 2009;6(5):20–33.
Sheehan DV, Rozova A, Gossen ER, et al. The efficacy and tolerability of once-daily controlled-release trazodone for depressed mood, anxiety, insomnia, and suicidality in major depressive disorder. Psychopharmacol Bull. 2009;42(4):5–22.
Clayton AH, Favit A. Concerns in depression treatment: sexual dysfunction and weight gain. Prim Psychiatry. 2007;14:66–75.
Ashton AK, Jamerson BD, Weinstein W, et al. Antidepressant-related adverse effects impacting treatment compliance: results of a patient survey. Curr Ther Res Clin Exp. 2005;66(2):96–106.
Acknowledgments
The manuscript was reviewed by: M. Hahn, Vitos Rheingau, Eltville, Germany; S. Kasper, Department of General Psychiatry and Psychotherapy, Medical University of Vienna, Vienna, Austria; S. Saluja, Saran Ashram Hospital, Dayalbagh, Agra, India; R. B. Shah, Department of Pharmacology, GMERS Medical College and Hospital, Gandhinagar, Gujarat, India.
Disclosure
This article was written by a salaried employee of Adis/Springer and was not supported by any external funding. During the peer review process, the manufacturer of the agent under review was offered an opportunity to comment on the article. Changes resulting from comments received were made by the author on the basis of scientific merit.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lyseng-Williamson, K.A. Trazodone prolonged release: a guide to its use in depression. Drugs Ther Perspect 31, 137–142 (2015). https://doi.org/10.1007/s40267-015-0207-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40267-015-0207-z