Abstract
Background
To reduce prescribing cascades occurring in clinical practice, healthcare providers require information on the prescribing cascades they can recognize and prevent.
Objective
This systematic review aims to provide an overview of prescribing cascades, including dose-dependency information and recommendations that healthcare providers can use to prevent or reverse them.
Methods
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) was followed. Relevant literature was identified through searches in OVID MEDLINE, OVID Embase, OVID CINAHL, and Cochrane. Additionally, Web of Science and Scopus were consulted to analyze reference lists and citations. Publications in English were included if they analyzed the occurrence of prescribing cascades. Prescribing cascades were included if at least one study demonstrated a significant association and were excluded when the adverse drug reaction could not be confirmed in the Summary of Product Characteristics. Two reviewers independently extracted and grouped similar prescribing cascades. Descriptive summaries were provided regarding dose-dependency analyses and recommendations to prevent or reverse these prescribing cascades.
Results
A total of 95 publications were included, resulting in 115 prescribing cascades with confirmed adverse drug reactions for which at least one significant association was found. For 52 of these prescribing cascades, information regarding dose dependency or recommendations to prevent or reverse prescribing cascades was found. Dose dependency was analyzed and confirmed for 12 prescribing cascades. For example, antipsychotics that may cause extrapyramidal syndrome followed by anti-parkinson drugs. Recommendations focused on dosage lowering, discontinuing medication, and medication switching. Explicit recommendations regarding alternative options were given for three prescribing cascades. One example was switching to ondansetron or granisetron when extrapyramidal syndrome is experienced using metoclopramide.
Conclusions
In total, 115 prescribing cascades were identified and an overview of 52 of them was generated for which recommendations to prevent or reverse them were provided. Nonetheless, information regarding alternative options for managing prescribing cascades was scarce.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
We identified 115 distinct potential prescribing cascades for which at least one study demonstrated a significant positive association. |
For 52 prescribing cascades, recommendations for preventing and reversing prescribing cascades were provided or could be derived from a dose-dependency analysis. |
Dosage lowering was often recommended without confirmation of a dose-dependent relationship and switching or discontinuation of medication was often recommended without specifying an alternative option. |
1 Introduction
A prescribing cascade occurs when medication causes an adverse drug reaction (ADR), which is subsequently addressed by prescribing additional medications [1, 2]. This is often due to a misinterpretation of the ADR as a new medical condition or symptom [1, 2]. Prescribing cascades may lead to unnecessary medication therapies, decreased quality of life, and increased healthcare costs [3,4,5]. They can occur in any patient using one or more medications but are more likely in patients taking multiple medications [3].
Prescribing cascades can be prevented by timely recognition of the ADR or reversed by either lowering the dose, discontinuing the medication causing the ADR, or switching to (pharmacotherapeutic) alternatives [3]. To facilitate this, healthcare providers (HCPs) need to have information about prescribing cascades related to recognizable ADRs that can occur in practice. In addition, HCPs need information on how to reverse ADRs and thereby the prescribing cascade.
Many prescribing cascades have been described in the literature and several reviews have been published [3, 6,7,8]. One of the reviews was limited by including only prescribing cascades that were identified through a prescription sequence symmetry analysis [7], whereas another review identified prescribing cascades occurring with pain medication [6]. A review published in 2022 aimed to identify all published prescribing cascades in community-dwelling adults, including hypothesis-free studies employing data-mining techniques and studies with negative associations or inverse causality [8]. This review identified 78 publications, resulting in an overview of 25 commonly identified prescribing cascades. However, by including hypothesis-free studies and studies with negative associations or inverse causality, the evidence of the related ADRs can be limited. This reduces the evidence of an overview for assisting HCPs in preventing prescribing cascades. In addition, information on how to prevent or reverse specific prescribing cascades has not been summarized.
Our aim is to conduct a systematic review and provide an overview of prescribing cascades including dose-dependency information and recommendations that HCPs can use to prevent or reverse prescribing cascades. Therefore, our focus is on prescribing cascades related to confirmed ADRs (i.e., prescribing cascades showing a significant positive association in at least one study and confirmed ADRs in the Summary of Product Characteristics (SmPC) of the involved medication) occurring in clinical practice that are recognizable for HCPs.
2 Methods
In this systematic review, the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement has been followed (Table 1 of the Electronic Supplementary Material [ESM]) [9] and the protocol was published in Prospero (CRD42021253004) [10].
2.1 Search Strategy and Data Sources
Based on a previous scoping review [3], an initial literature search was developed (OA, SPV, FKC, AM,) and performed in PubMed to identify relevant publications. The initial search revealed that many (older) publications did not use the term ‘prescribing cascade’. Therefore, the indexed keywords and text terms were extracted from the publications identified in the initial search. This was done using PubReMiner [11] and OVID Embase [12] to create an extensive search strategy to identify as many publications as possible that described prescribing cascades. No restriction was made regarding the study design. This extensive search strategy included: (1) publications that mention the term ‘prescribing cascade’; (2) publications that mention terms referring to ADRs or side effects and use a sequence symmetry analysis to quantify the occurrence of a prescribing cascade; and (3) reports or case-control studies of an ADR or side effect treated with medication. This extensive search strategy was checked for the inclusion of relevant publications from the initial search, reviewed by two information specialists and adjusted accordingly. The final search strategy can be found in Table 2 of the ESM, and was performed in OVID MEDLINE, OVID Embase, OVID CINAHL, The Cochrane Database of Systematic Reviews, and the Cochrane Central Register of Controlled Trials from inception until 2 September, 2021.
We conducted backward citation [13], checking on all publications describing a prescribing cascade, including (systematic) reviews, case reports, and case series. An additional forward citation [13] analysis was performed in Web of Science (core collection) and Scopus in January 2022 for these publications. This approach was adopted to maximize the identification of prescribing cascades.
2.2 Inclusion and Exclusion of Publications
Publications in English and reporting a statistical analysis (e.g., prescription sequence symmetry analysis, regression) to identify or confirm prescribing cascades in adults (aged ≥ 18 years) were included. Case reports and case series as well as (systematic) reviews were excluded at this point. Furthermore, we excluded publications with hypothesis-free or discovery-driven analyses. For example, pharmacovigilance studies using data-mining techniques to detect unconfirmed potential ADRs.
First, titles and abstracts were reviewed for eligibility by reviewers individually, divided over five reviewers (AM, OA, MY, FKC, and JH). Second, full texts were independently screened by two reviewers (OA, AM) according to the inclusion and exclusion criteria. Any disagreements between the reviewers were resolved by consensus, if needed with the support of a senior reviewer (FKC).
2.3 Inclusion and Exclusion of Prescribing Cascades
A prescribing cascade was defined as the description of a first medication (index medication), an ADR that could be confirmed in the SmPC [14] of the index medication, and the initiation of a second medication that could ‘treat’ the ADR (marker medication), for which an association between index and marker medication was confirmed in at least one study. Prescribing cascades used as positive controls were included.
A prescribing cascade was excluded if the treatment with the marker medication was intentional. Intentional treatment was either as stated in the publication itself or derived from guidelines (e.g., prescribing a laxative to prevent ADRs when prescribing opioids) [15]. Prescribing cascades were also excluded if a medical device or a medical product (e.g., urinary incontinence products) [16] was used to treat the ADR. In addition, prescribing cascades were excluded if the index medication was described at an unspecific level. For example, the highest level of the World Health Organization Anatomical Therapeutic Chemical (ATC) classification for medication (e.g., Cardiovascular system; ATC code C) [17]. Finally, prescribing cascades were excluded if the medication could not be found in the European Medicines Agency [18] or US Food and Drug Administration [19] websites between July and November 2022, indicating the medication was no longer available.
2.4 Data Extraction
A data extraction form in MS Excel 2016 (Microsoft Corporation, Redmond, WA, USA) was used. The senior reviewers (FKC, JH, PD, PvdB, LM) checked the data extracted from 45 publications to refine the data extraction form. Extracted data included information about study characteristics, the identified prescribing cascade, the patient population, and the method of statistical analysis (Table 3 of the ESM). Results of the main statistical analysis regarding the prescribing cascade were extracted as showing: (1) a positive significant association; (2) a non-significant association; or (3) an inverse significant association. Additionally, analyses on dose dependency regarding the occurrence of prescribing cascades along with the corresponding results were extracted. Finally, information and recommendations on how to prevent or reverse the prescribing cascades as provided by the authors were extracted and classified as a dose reduction, a medication switch, or a discontinuation of the index medication (including the recommended alternatives). Data were extracted by one of three reviewers and checked by one of the other reviewers (OA, AM, or RV). Disagreements between the reviewers were resolved by consensus, if needed with the support of a senior reviewer (FKC). An extra check was performed by FKC to confirm that all recommendations were extracted.
2.5 Data Synthesis
To provide a concise overview that HCPs can use to prevent or reverse prescribing cascades, similar prescribing cascades were grouped at the medication and ADR levels. To group prescribing cascades that were described both at the chemical substance level (e.g., lisinopril) and at the related pharmacological or chemical subgroup level (e.g., angiotensin-converting enzyme inhibitor), the index and marker medication were grouped using the World Health Organization Anatomical Therapeutic Chemical classification system [17]. However, when the ADR could not be confirmed for at least half of the chemical substances within a pharmacological or chemical subgroup, the prescribing cascades were presented for the individual substances. In addition, similar ADRs (e.g., dribbling of urine and urinary incontinence) were grouped using the Medical Dictionary for Regulatory Activities classification system [20]. In this way, reports of distinct prescribing cascades were created (for further details, see Table 4 of the ESM).
3 Results
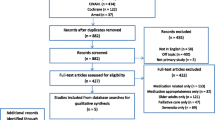
A total of 6834 publications were screened on titles and abstracts, 375 of which required full-text eligibility assessment, resulting in 95 publications that met the eligibility criteria and were included (see Fig. 1). Reasons for excluding 280 full text publications can be found in Table 5 of the ESM.
Of the included publications, 80 concerned cohort studies and 15 were case-control studies. The prescription sequence symmetry analysis was used as the main analysis in 45 publications, and most studies were conducted in the primary care setting (Table 1) [16, 21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114].
3.1 Prescribing Cascades
In the 95 publications, 571 reports of non-distinct prescribing cascades were identified that were grouped into 214 distinct prescribing cascades. Of these, 29 were excluded for various reasons, most commonly because the ADR was not found in the SmPC (n = 10) [see Table 6 of the ESM for all exclusion reasons). For the remaining 185 prescribing cascades, 70 showed no significant positive association (Table 7 of the ESM), whereas at least one significant positive association was found for 115 prescribing cascades (Tables 8 and 9 of the ESM). For 52 (45.2%) of these 115 prescribing cascades, information was available on either dose dependency of the occurrence of prescribing cascades or a recommendation to prevent or reverse the prescribing cascades (Table 2).
3.2 Dose Dependency and Recommended Actions
An analysis on the dose dependency was presented for 12 distinct prescribing cascades in 14 publications. For all prescribing cascades, the dose dependency was confirmed, although for one prescribing cascade conflicting results were found (i.e., inhaled glucocorticosteroids causing oral candidiasis treated with anti-infective agents). Other dose dependencies were found with metoclopramide/antipsychotics causing extrapyramidal syndrome, thiazides causing gout, amiodarone causing both hypo- and hyperthyroidism, calcium channel blockers causing peripheral edema, acitretin causing candidiasis, glucocorticoids causing hyperglycemia, non-steroidal anti-inflammatory drugs/diflunisal causing hypertension, and pregabalin/gabapentin causing peripheral edema (Table 2). Differences were found between studies regarding cut-offs used for defining a high dose. For example, Avorn et al. [54] used > 150 mg of chlorpromazine-equivalent per day as a high dose for antipsychotics whereas Schillevoort et al. [55] used > 375 mg of chlorpromazine-equivalent.
For 22 prescribing cascades, recommendations were made to switch the index medication but only for three it was mentioned to which medication (Table 2). Avorn et al. [21] recommended the switch to ondansetron or granisetron when extrapyramidal syndrome is experienced using metoclopramide. Vegter et al. [26, 28] recommended the switch to an angiotensin-receptor-blocker when a cough is experienced using angiotensin-converting enzyme inhibitors. Kirwan et al. [72] recommended the switch to betaxolol when an obstructive airways disorder is experienced when using antiglaucoma preparations, whereas Avorn et al. [71] recommended to switch to any other antiglaucoma preparation.
4 Discussion
This systematic review resulted in 115 distinct prescribing cascades, involving an ADR that was confirmed for the index medication in the SmPC, for which a significant positive association was found between the index and marker medication in at least one study. For 52 of these confirmed prescribing cascades, recommendations on how to prevent or reverse it were provided or could be derived from a dose-dependency analysis. Dose dependency was tested and shown for 12 prescribing cascades. Dosage lowering was often recommended without confirmation of a dose-dependent association. Switching to another medication and discontinuation of medication were often recommended without specifying an alternative.
4.1 Comparison with Previous Publications
Several reviews on prescribing cascades have been published but none provided information on how to prevent or reverse them. The most recently published systematic review (2022) of Doherty et al. aimed to identify prescribing cascades in community-dwelling adults [8]. In their review, they identified 76 publications, whereas our review included 95 publications. Our review was not limited to community-dwelling adults like Doherty et al. or to a specific medication group as done by Nunnari et al. [6], resulting in the inclusion of 33 additional publications compared to both. Of note, Doherty et al. identified nine publications and Nunnari et al. identified 11 publications that were excluded from our review, as they concerned hypothesis-free studies, case reports, or ADRs that could not be confirmed in the SmPC of the index medication. A direct comparison of identified prescribing cascades is complicated by differences in the level at which the prescribing cascades are presented in the reviews. Nonetheless, our review included three prescribing cascades not identified by Doherty et al. [8]. This concerned calcium channel blockers causing depression treated with antidepressants, glucocorticosteroids causing hyperglycemia treated with diabetes drugs, and diabetes drugs causing depression treated with antidepressants. Based on the opinion of experts, McCarthy et al. recently provided an overview of nine clinically important prescribing cascades [115]. All of these were also included in our review.
4.2 Implications for Policy, Practice, and Research
An overview of clinically relevant prescribing cascades can serve as a tool for HCPs to raise awareness on prescribing cascades [115]. The overview presented can be the starting point for specific interventions or policies on preventing and reversing prescribing cascades. The 52 confirmed prescribing cascades with information about dose dependency and recommendations can aid HCPs in recognizing and addressing potentially preventable prescribing cascades. Confirmation that the ADR can be caused by the index medication is relevant for HCPs because it is often difficult to recognize whether a new symptom might be an ADR, particularly in patients using multiple medications [5].
Additionally, knowing whether the ADR and the related prescribing cascade have been confirmed for multiple substances within a pharmacological subgroup can help HCPs determine whether switching a medication within a subgroup is a viable strategy for reversing a prescribing cascade. For 12 confirmed prescribing cascades, evidence was found that they were dose dependent. This implies that dosage lowering may reverse these prescribing cascades.
For prescribing cascades with consistent positive associations in the studies and concrete recommendations, development of electronic decision support tools could help manage these prescribing cascades (e.g., angiotensin-converting enzyme inhibitors induced cough, metoclopramide induced extrapyramidal syndrome, calcium channel blocker induced peripheral edema). For others, it may be helpful to create alerts so that the prescriber can reconsider the benefit-risk balance of the index medication. In some cases, particularly among the very old and frail patients, discontinuation may be an option.
More research is needed to assess whether using low dosages in patients at a high risk of developing an ADR, such as older people or women [75, 86, 116], can prevent prescribing cascades and is also sufficient to reverse existing ADRs. Furthermore, recommendations to switch a medication without specific advice are not helpful for HCPs. Knowledge synthesis is needed to create an overview of prescribing cascades with specific details on alternative options.
4.3 Strengths
This systematic review has several strengths. The extensive literature search strategy without limitations on medication groups, patient population, setting, study design, or method of analysis increased the chance of finding prescribing cascades relevant for HCPs. Publications on this topic do not necessarily use the term ‘prescribing cascade’ or relevant indexed terms. Therefore, forward and backwards citation searches were conducted for included studies, systematic reviews, and case reports/series, which led to the inclusion of additional publications. Recommendations for conducting systematic reviews were followed and in all steps at least two researchers were involved. Furthermore, SmPCs providing information as certified by marketing authorities were used to confirm ADRs, and existing classification systems were used to group medications and ADRs.
4.4 Limitations
Prescribing cascades were included that showed a significant association in at least one study. This indicates that the prescribing cascade occurred in clinical practice but it does not imply that an ADR actually occurred in all cases nor that the marker medication was prescribed for that reason. Some studies included small sample sizes or used suboptimal data or designs, which can result in missing some relevant prescribing cascades. To address this limitation, we included all identified prescribing cascades in the supplementary material. Finally, some prescribing cascades were assessed in many studies, whereas others were assessed in one study or may not have been studied at all. It should be noted that the occurrence of prescribing cascades can be country, culture, or care setting dependent. Treatment guidelines and available and approved medication can differ per country. Whether people visit their doctor with specific complaints may also differ depending on the culture or setting. For these reasons, not all of the confirmed prescribing cascades presented in our review may be relevant for all HCPs. Furthermore, some of the prescribing cascades for which no significant association has been shown might still be relevant in specific settings. Our overview of confirmed prescribing cascades should therefore be seen as a first step to support HCPs.
5 Conclusions
From 185 distinct prescribing cascades identified in 95 publications, 115 showed a significant positive association between the index and marker medication in at least one study. Of these, an overview of 52 prescribing cascades was created, for which information on dose dependency or recommendations on how to prevent or reverse the prescribing cascade was provided. This review illustrates that specific recommendations on how to reverse and prevent prescribing cascades is often lacking.
References
Rochon PA, Gurwitz JH. Drug therapy. Lancet. 1995;346(8966):32–6.
Farrell B, Galley E, Jeffs L, Howell P, McCarthy LM. “Kind of blurry”: deciphering clues to prevent, investigate and manage prescribing cascades. PLoS ONE. 2022;17(8): e0272418.
Brath H, Mehta N, Savage RD, Gill SS, Wu W, Bronskill SE, et al. What is known about preventing, detecting, and reversing prescribing cascades: a scoping review. J Am Geriatr Soc. 2018;66(11):2079–85.
Kalisch LM, Caughey GE, Roughead EE, Gilbert AL. The prescribing cascade. Aust Prescr. 2011;34(6):162–6.
McCarthy LM, Visentin JD, Rochon PA. Assessing the scope and appropriateness of prescribing cascades. J Am Geriatr Soc. 2019;67(5):1023–6.
Nunnari P, Ceccarelli G, Ladiana N, Notaro P. Prescribing cascades and medications most frequently involved in pain therapy: a review. Eur Rev Med Pharm Sci. 2021;25(2):1034–41.
Morris EJ, Hollmann J, Hofer AK, Bhagwandass H, Oueini R, Adkins LE, et al. Evaluating the use of prescription sequence symmetry analysis as a pharmacovigilance tool: a scoping review. Res Social Adm Pharm. 2022;18(7):3079–93.
Doherty AS, Shahid F, Moriarty F, Boland F, Clyne B, Dreischulte T, et al. Prescribing cascades in community-dwelling adults: a systematic review. Pharm Res Perspect. 2022;10(5): e01008.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372: n71.
O Adrien AM, Hugtenburg JG, McCarthy L, Priester-Vink S, Visscher R, van den Bemt PMLA, et al. Prescribing cascades: a systematic review. PROSPERO 2021 CRD42021253004. https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42021253004. Accessed 27 Sep 2023.
Koster J. PubMed PubReMiner 2023. https://hgserver2.amc.nl/cgi-bin/miner/miner2.cgi. Accessed 27 Sep 2023.
Ovid Embase. 2023. https://tools.ovid.com/embase/. Accessed 27 Sep 2023.
Nursing - Literature Reviews & Systematic Reviews. Backward & Forward Reference Searching. Florida Atlantic University; 2023 [updated 03-03-2023]. https://libguides.fau.edu/c.php?g=966176&p=7013756. Accessed 27 Sep 2023.
MEB 27: MEB commentary on the Guideline on Summary of Product Characteristics, 23 August 2017. https://www.cbg-meb.nl/documenten/beleidsdocumenten/2023/01/01/meb-27. Accessed 27 Sep 2023.
NHG. NHG richtlijnen. https://richtlijnen.nhg.org/. Accessed 27 Sep 2023.
Monane M, Bohn RL, Gurwitz JH, Glynn RJ, Choodnovskiy I, Avorn J. Topical glaucoma medications and cardiovascular risk in the elderly. Clin Pharmacol Ther. 1994;55(1):76–83.
WHO Collaborating Centre for Drug Statistics Methodology. ATC/DDD index 2023 2023 [updated 01-23-2023]. https://www.whocc.no/atc_ddd_index/. Accessed 27 Sep 2023.
Agency EM. Summary of product characteristics 2023. https://www.ema.europa.eu/en/glossary/summary-product-characteristics. Accessed 30 Apr 2023.
US Food and Drug Administration (FDA). Index drug specific information 2023 [updated 28-02-2023]. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/index-drug-specific-information. Accessed 30 Feb 2023.
Whetzel PL NN, Shah NH, Alexander PR, Nyulas C, Tudorache T, Musen MA. Medical Dictionary for Regulatory Activities terminology (MedDRA). Nucleic Acids Res. https://bioportal.bioontology.org/ontologies/MEDDRA. Accessed 27 Sep 2023.
Avorn J, Gurwitz JH, Bohn RL, Mogun H, Monane M, Walker A. Increased incidence of levodopa therapy following metoclopramide use. JAMA. 1995;274(22):1780–2.
Huh Y, Kim DH, Choi M, Park JH, Kwon DY, Jung JH, et al. Metoclopramide and levosulpiride use and subsequent levodopa prescription in the Korean elderly: the prescribing cascade. J Clin Med. 2019;8(9):1496.
Janetzki JL, Sykes MJ, Ward MB, Pratt NL. Proton pump inhibitors may contribute to progression or development of chronic obstructive pulmonary disease: a sequence symmetry analysis approach. J Clin Pharm Ther. 2021;46(6):1687–94.
Hashimoto M, Hashimoto K, Ando F, Kimura Y, Nagase K, Arai K. Prescription rate of medications potentially contributing to lower urinary tract symptoms and detection of adverse reactions by prescription sequence symmetry analysis. J Pharm Health Care Sci. 2015;1:7.
Caughey GE, Roughead EE, Pratt N, Shakib S, Vitry AI, Gilbert AL. Increased risk of hip fracture in the elderly associated with prochlorperazine: is a prescribing cascade contributing? Pharmacoepidemiol Drug Saf. 2010;19(9):977–82.
Vegter S, de Jong-van den Berg LT. Misdiagnosis and mistreatment of a common side-effect--angiotensin-converting enzyme inhibitor-induced cough. Br J Clin Pharmacol. 2010;69(2):200–3.
Chen Y, Huang ST, Hsu TC, Peng LN, Hsiao FY, Chen LK. Detecting suspected prescribing cascades by prescription sequence symmetry analysis of nationwide real-world data. J Am Med Dir Assoc. 2022;23(3):468–74.
Vegter S, de Boer P, van Dijk KW, Visser S, de Jong-van den Berg LT. The effects of antitussive treatment of ACE inhibitor-induced cough on therapy compliance: a prescription sequence symmetry analysis. Drug Saf. 2013;36(6):435–9.
Bowman L, Carlstedt BC, Miller ME, McDonald CJ. Evaluation of ACE-inhibitor (ACE-I) associated cough using modified prescription sequence analysis (PSA). Pharmacoepidemiol Drug Saf. 1995;4(1):17–22.
Gurwitz JH, Kalish SC, Bohn RL, Glynn RJ, Monane M, Mogun H, et al. Thiazide diuretics and the initiation of anti-gout therapy. J Clin Epidemiol. 1997;50(8):953–9.
Zhang H, Wu J, Zhang Z, Qian H, Wang Y, Yang M, et al. Association of atorvastatin with the risk of hepatotoxicity: a pilot prescription sequence symmetry analysis. Ther Clin Risk Manage. 2019;15:803–10.
Nishtala PS, Chyou TY. Exploring New Zealand prescription data using sequence symmetry analyses for predicting adverse drug reactions. J Clin Pharm Ther. 2017;42(2):189–94.
Bouvy ML, Heerdink ER, Hoes AW, Leufkens HG. Amiodarone-induced thyroid dysfunction associated with cumulative dose. Pharmacoepidemiol Drug Saf. 2002;11(7):601–6.
Pratt N, Chan EW, Choi NK, Kimura M, Kimura T, Kubota K, et al. Prescription sequence symmetry analysis: assessing risk, temporality, and consistency for adverse drug reactions across datasets in five countries. Pharmacoepidemiol Drug Saf. 2015;24(8):858–64.
Yokoyama S, Tanaka Y, Hosomi K, Takada M. Polypharmacy is associated with amiodarone-induced hypothyroidism. Int J Med Sci. 2021;18(15):3574–80.
Garrison SR, Dormuth CR, Morrow RL, Carney GA, Khan KM. Nocturnal leg cramps and prescription use that precedes them: a sequence symmetry analysis. Arch Intern Med. 2012;172(2):120–6.
Savage RD, Visentin JD, Bronskill SE, Wang X, Gruneir A, Giannakeas V, et al. Evaluation of a common prescribing cascade of calcium channel blockers and diuretics in older adults with hypertension. JAMA Intern Med. 2020;180(5):643–51.
Singh S, Cocoros NM, Haynes K, Nair VP, Harkins TP, Rochon PA, et al. Identifying prescribing cascades in Alzheimer’s disease and related dementias: the calcium channel blocker-diuretic prescribing cascade. Pharmacoepidemiol Drug Saf. 2021;30(8):1066–73.
Vouri SM, van Tuyl JS, Olsen MA, Xian H, Schootman M. An evaluation of a potential calcium channel blocker-lower-extremity edema-loop diuretic prescribing cascade. J Am Pharm Assoc (2003). 2018;58(5):534–9.e4.
Vouri SM, Jiang X, Manini TM, Solberg LM, Pepine C, Malone DC, et al. Magnitude of and characteristics associated with the treatment of calcium channel blocker-induced lower-extremity edema with loop diuretics. JAMA Netw Open. 2019;2(12): e1918425.
Trenaman SC, Bowles SK, Kirkland S, Andrew MK. An examination of three prescribing cascades in a cohort of older adults with dementia. BMC Geriatr. 2021;21(1):297.
Kalisch Ellett LM, Pratt NL, Barratt JD, Rowett D, Roughead EE. Risk of medication-associated initiation of oxybutynin in elderly men and women. J Am Geriatr Soc. 2014;62(4):690–5.
Thiessen BQ, Wallace SM, Blackburn JL, Wilson TW, Bergman U. Increased prescribing of antidepressants subsequent to beta-blocker therapy. Arch Intern Med. 1990;150(11):2286–90.
Avorn J, Everitt DE, Weiss S. Increased antidepressant use in patients prescribed beta-blockers. JAMA. 1986;255(3):357–60.
Hallas J. Evidence of depression provoked by cardiovascular medication: a prescription sequence symmetry analysis. Epidemiology. 1996;7(5):478–84.
Johnell K, Fastbom J. The association between use of cardiovascular drugs and antidepressants: a nationwide register-based study. Eur J Clin Pharmacol. 2008;64(11):1119–24.
van Dijk KN, de Vries CS, van den Berg PB, Dijkema AM, Brouwers JR, de Jong-van den Berg LT. Constipation as an adverse effect of drug use in nursing home patients: an overestimated risk. Br J Clin Pharmacol. 1998;46(3):255–61.
Pouwels KB, Visser ST, Bos HJ, Hak E. Angiotensin-converting enzyme inhibitor treatment and the development of urinary tract infections: a prescription sequence symmetry analysis. Drug Saf. 2013;36(11):1079–86.
Sturkenboom MC, Middelbeek A, de Jong van den Berg LT, van den Berg PB, Stricker BH, Wesseling H. Vulvo-vaginal candidiasis associated with acitretin. J Clin Epidemiol. 1995;48(8):991–7.
Monane M, Avorn J, Beers MH, Everitt DE. Anticholinergic drug use and bowel function in nursing home patients. Arch Intern Med. 1993;153(5):633–8.
Gurwitz JH, Bohn RL, Glynn RJ, Monane M, Mogun H, Avorn J. Glucocorticoids and the risk for initiation of hypoglycemic therapy. Arch Intern Med. 1994;154(1):97–101.
Gurwitz JH, Avorn J, Bohn RL, Glynn RJ, Monane M, Mogun H. Initiation of antihypertensive treatment during nonsteroidal anti-inflammatory drug therapy. JAMA. 1994;272(10):781–6.
Hirano Y. Risk of extrapyramidal syndromes associated with psychotropic polypharmacy: a study based on large-scale Japanese claims data. Ther Innov Regul Sci. 2020;54(2):259–68.
Avorn J, Bohn RL, Mogun H, Gurwitz JH, Monane M, Everitt D, et al. Neuroleptic drug exposure and treatment of parkinsonism in the elderly: a case-control study. Am J Med. 1995;99(1):48–54.
Schillevoort I, Herings RM, Hugenholtz GW, de Boer A, Nolen WA, Roos RA, et al. Antipsychotic-induced extrapyramidal syndromes in psychiatric practice: a case-control study. Pharm World Sci. 2005;27(4):285–9.
De Hert M, Wampers M, van Winkel R, Peuskens J. Anticholinergic use in hospitalised schizophrenic patients in Belgium. Psychiatry Res. 2007;152(2–3):165–72.
Schillevoort I, de Boer A, Herings RM, Roos RA, Jansen PA, Leufkens HG. Antipsychotic-induced extrapyramidal syndromes: risperidone compared with low- and high-potency conventional antipsychotic drugs. Eur J Clin Pharmacol. 2001;57(4):327–31.
Kalish SC, Bohn RL, Mogun H, Glynn RJ, Gurwitz JH, Avorn J. Antipsychotic prescribing patterns and the treatment of extrapyramidal symptoms in older people. J Am Geriatr Soc. 1995;43(9):967–73.
Touré JT, Brandt NJ, Limcangco MR, Briesacher BA. Impact of second-generation antipsychotics on the use of antiparkinson agents in nursing homes and assisted-living facilities. Am J Geriatr Pharmacother. 2006;4(1):25–35.
Lai EC, Hsieh CY, Kao Yang YH, Lin SJ. Detecting potential adverse reactions of sulpiride in schizophrenic patients by prescription sequence symmetry analysis. PLoS ONE. 2014;9(2): e89795.
Read SH, Giannakeas V, Pop P, Bronskill SE, Herrmann N, Chen S, et al. Evidence of a gabapentinoid and diuretic prescribing cascade among older adults with lower back pain. J Am Geriatr Soc. 2021;69(10):2842–50.
Marras C, Herrmann N, Fischer HD, Fung K, Gruneir A, Rochon PA, et al. Lithium use in older adults is associated with increased prescribing of Parkinson medications. Am J Geriatr Psychiatry. 2016;24(4):301–9.
Gill SS, Mamdani M, Naglie G, Streiner DL, Bronskill SE, Kopp A, et al. A prescribing cascade involving cholinesterase inhibitors and anticholinergic drugs. Arch Intern Med. 2005;165(7):808–13.
Lampela P, Taipale H, Hartikainen S. Use of cholinesterase inhibitors increases initiation of urinary anticholinergics in persons with Alzheimer’s disease. J Am Geriatr Soc. 2016;64(7):1510–2.
Venäläinen O, Bell JS, Kirkpatrick CM, Nishtala PS, Liew D, Ilomäki J. Adverse drug reactions associated with cholinesterase inhibitors: sequence symmetry analyses using prescription claims data. J Am Med Dir Assoc. 2017;18(2):186–9.
Kröger E, Van Marum R, Souverein P, Carmichael PH, Egberts T. Treatment with rivastigmine or galantamine and risk of urinary incontinence: results from a Dutch database study. Pharmacoepidemiol Drug Saf. 2015;24(3):276–85.
Henriksen DP, Davidsen JR, Christiansen A, Laursen CB, Damkier P, Hallas J. Inhaled corticosteroids and systemic or topical antifungal therapy: a symmetry analysis. Ann Am Thorac Soc. 2017;14(6):1045–7.
van Boven JF, de Jong-van den Berg LT, Vegter S. Inhaled corticosteroids and the occurrence of oral candidiasis: a prescription sequence symmetry analysis. Drug Saf. 2013;36(4):231–6.
Kennedy WA, Laurier C, Gautrin D, Ghezzo H, Paré M, Malo JL, et al. Occurrence and risk factors of oral candidiasis treated with oral antifungals in seniors using inhaled steroids. J Clin Epidemiol. 2000;53(7):696–701.
Petri H, Kessels F, Kamakura T. Markers of adverse drug reactions in medication histories. An analysis of inhaled steroid utilization. Pharm Weekbl Sci. 1991;13(2):97–106.
Avorn J, Glynn RJ, Gurwitz JH, Bohn RL, Monane M, Everitt DE, et al. Adverse pulmonary effects of topical Beta blockers used in the treatment of glaucoma. J Glaucoma. 1993;2(3):158–65.
Kirwan JF, Nightingale JA, Bunce C, Wormald R. Do selective topical beta antagonists for glaucoma have respiratory side effects? Br J Ophthalmol. 2004;88(2):196–8.
Kirwan JF, Nightingale JA, Bunce C, Wormald R. Beta blockers for glaucoma and excess risk of airways obstruction: population based cohort study. BMJ. 2002;325(7377):1396–7.
Roughead EE, Kalisch LM, Pratt NL, Killer G, Barnard A, Gilbert AL. Managing glaucoma in those with co-morbidity: not as easy as it seems. Ophthal Epidemiol. 2012;19(2):74–82.
Adimadhyam S, Schumock GT, Calip GS, Smith Marsh DE, Layden BT, Lee TA. Increased risk of mycotic infections associated with sodium-glucose co-transporter 2 inhibitors: a prescription sequence symmetry analysis. Br J Clin Pharmacol. 2019;85(1):160–8.
Brandt-Christensen M, Kvist K, Nilsson FM, Andersen PK, Kessing LV. Treatment with antidepressants and lithium is associated with increased risk of treatment with antiparkinson drugs: a pharmacoepidemiological study. J Neurol Neurosurg Psychiatry. 2006;77(6):781–3.
Chang KJ, Hong CH, Lee Y, Lee KS, Roh HW, Back JH, et al. Effect of psychotropic drugs on development of diabetes mellitus in patients with Alzheimer’s disease. Medicine. 2015;94(23): e919.
Clifford GM, Farmer RD. Drug or symptom-induced depression in men treated with alpha 1-blockers for benign prostatic hyperplasia? A nested case-control study. Pharmacoepidemiol Drug Saf. 2002;11(1):55–61.
Corrao G, Botteri E, Bagnardi V, Zambon A, Carobbio A, Falcone C, et al. Generating signals of drug-adverse effects from prescription databases and application to the risk of arrhythmia associated with antibacterials. Pharmacoepidemiol Drug Saf. 2005;14(1):31–40.
Dunvald AD, Henriksen DP, Hallas J, Christensen MMH, Lund LC. Selective serotonin reuptake inhibitors and the risk of restless legs syndrome: a symmetry analysis. Eur J Clin Pharmacol. 2020;76(5):719–22.
Dyson TE, Cantrell MA, Lund BC. Lack of Association between 5α-reductase inhibitors and depression. J Urol. 2020;204(4):793–8.
Ko HHT, Lareu RR, Dix BR, Hughes JD, Parsons RW. A sequence symmetry analysis of the interrelationships between statins, diabetes and skin infections. Br J Clin Pharmacol. 2019;85(11):2559–67.
Lai EC, Hsieh CY, Wong MB, Lin SJ, Yang YK, Kao Yang YH, et al. Comparative risk of oral ulcerations among antipsychotics users: population-based retrospective cohort study. Pharmacoepidemiol Drug Saf. 2016;25(2):123–32.
Lai EC, Yang YH, Lin SJ, Hsieh CY. Use of antiepileptic drugs and risk of hypothyroidism. Pharmacoepidemiol Drug Saf. 2013;22(10):1071–9.
Lega IC, Bronskill SE, Campitelli MA, Guan J, Stall NM, Lam K, et al. Sodium glucose cotransporter 2 inhibitors and risk of genital mycotic and urinary tract infection: a population-based study of older women and men with diabetes. Diabetes Obes Metab. 2019;21(11):2394–404.
Li XX, Cheng YC, Zhai SD, Yao P, Zhan SY, Shi LW. Risk of liver injury associated with intravenous lipid emulsions: a prescription sequence symmetry analysis. Front Pharmacol. 2021;12: 589091.
Lindberg G, Hallas J. Cholesterol-lowering drugs and antidepressants: a study of prescription symmetry. Pharmacoepidemiol Drug Saf. 1998;7(6):399–402.
MacDonald TM, Beardon PH, McGilchrist MM, Duncan ID, McKendrick AD, McDevitt DG. The risks of symptomatic vaginal candidiasis after oral antibiotic therapy. Quarterly J Med. 1993;86(7):419–24.
Man KK, Shao S-C, Chaiyakunapruk N, Dilokthornsakul P, Kubota K, Li J, et al. Metabolic events associated with the use of antipsychotics in children, adolescents and young adults: a multinational sequence symmetry study. Eur Child Adolesc Psychiatry. 2022;31(1):99–120.
Masurkar PP, Chatterjee S, Sherer JT, Aparasu RR. Antimuscarinic cascade across individual cholinesterase inhibitors in older adults with dementia. Drugs Aging. 2021;38(7):593–602.
Maura G, Billionnet C, Coste J, Weill A, Neumann A, Pariente A. Non-bleeding adverse events with the use of direct oral anticoagulants: a sequence symmetry analysis. Drug Saf. 2018;41(9):881–97.
Nakhleh A, Zloczower M, Gabay L, Shehadeh N. Effects of sodium glucose co-transporter 2 inhibitors on genital infections in female patients with type 2 diabetes mellitus: real world data analysis. J Diabetes Compl. 2020;34(7): 107587.
Park KR, Kim KB, Baek YH, Sung HG, Park JT, Lee EK, et al. Signal detection of benzodiazepine use and risk of dementia: sequence symmetry analysis using South Korean national healthcare database. Int J Clin Pharm. 2018;40(6):1568–76.
Park SK, Baek YH, Pratt N, Kalisch Ellett L, Shin JY. The uncertainty of the association between proton pump inhibitor use and the risk of dementia: prescription sequence symmetry analysis using a Korean healthcare database between 2002 and 2013. Drug Saf. 2018;41(6):615–24.
Petri H, de Vet HC, Naus J, Urquhart J. Prescription sequence analysis: a new and fast method for assessing certain adverse reactions of prescription drugs in large populations. Stat Med. 1988;7(11):1171–5.
Petri H, Leufkens H, Naus J, Silkens R, van Hessen P, Urquhart J. Rapid method for estimating the risk of acutely controversial side effects of prescription drugs. J Clin Epidemiol. 1990;43(5):433–9.
Pratt N, Andersen M, Bergman U, Choi NK, Gerhard T, Huang C, et al. Multi-country rapid adverse drug event assessment: the Asian Pharmacoepidemiology Network (AsPEN) antipsychotic and acute hyperglycaemia study. Pharmacoepidemiol Drug Saf. 2013;22(9):915–24.
Rahimtoola H, Buurma H, Tijssen CC, Leufkens HG, Egberts AC. Incidence and determinants of antidepressant drug use in migraine patients. Int Clin Psychopharmacol. 2003;18(6):331–9.
Rasmussen L, Hallas J, Madsen KG, Pottegård A. Cardiovascular drugs and erectile dysfunction: a symmetry analysis. Br J Clin Pharmacol. 2015;80(5):1219–23.
Roughead EE, Chan EW, Choi NK, Griffiths J, Jin XM, Lee J, et al. Proton pump inhibitors and risk of Clostridium difficile infection: a multi-country study using sequence symmetry analysis. Expert Opin Drug Saf. 2016;15(12):1589–95.
Roughead EE, Chan EW, Choi NK, Kimura M, Kimura T, Kubota K, et al. Variation in association between thiazolidinediones and heart failure across ethnic groups: retrospective analysis of large healthcare claims databases in six countries. Drug Saf. 2015;38(9):823–31.
Silwer L, Petzold M, Hallas J, Lundborg CS. Statins and nonsteroidal anti-inflammatory drugs-an analysis of prescription symmetry. Pharmacoepidemiol Drug Saf. 2006;15(7):510–1.
Singh S, Cocoros NM, Haynes K, Nair VP, Harkins TP, Rochon PA, et al. Antidopaminergic-antiparkinsonian medication prescribing cascade in persons with Alzheimer’s disease. J Am Geriatr Soc. 2021;69(5):1328–33.
Takada M, Fujimoto M, Hosomi K. Association between benzodiazepine use and dementia: data mining of different medical databases. Int J Med Sci. 2016;13(11):825–34.
Takada M, Fujimoto M, Yamazaki K, Takamoto M, Hosomi K. Association of statin use with sleep disturbances: data mining of a spontaneous reporting database and a prescription database. Drug Saf. 2014;37(6):421–31.
Wang Y, Bos JH, Schuiling-Veninga CC, Boezen HM, van Boven JF, Wilffert B, et al. Neuropsychiatric safety of varenicline in the general and COPD population with and without psychiatric disorders: a retrospective cohort study in a real-world setting. BMJ Open. 2021;11(5): e042417.
Wang Y, van Boven J, Bos J, Schuiling-Veninga C, Boezen H, Wilffert B, et al. Risk of neuropsychiatric adverse events associated with varenicline treatment for smoking cessation among Dutch population: a sequence symmetry analysis. Pharmacoepidemiol Drug Saf. 2022;31(2):158–66.
Winkel JS, Damkier P, Hallas J, Henriksen DP. Treatment with montelukast and antidepressive medication: a symmetry analysis. Pharmacoepidemiol Drug Saf. 2018;27(12):1409–15.
Yokoyama S, Ieda S, Nagano M, Nakagawa C, Iwase M, Hosomi K, et al. Association between oral anticoagulants and osteoporosis: real-world data mining using a multi-methodological approach. Int J Med Sci. 2020;17(4):471–9.
Yokoyama S, Wakamoto S, Tanaka Y, Nakagawa C, Hosomi K, Takada M. Association between antipsychotics and osteoporosis based on real-world data. Ann Pharmacother. 2020;54(10):988–95.
Dial S, Delaney JA, Schneider V, Suissa S. Proton pump inhibitor use and risk of community-acquired Clostridium difficile-associated disease defined by prescription for oral vancomycin therapy. Can Med Assoc J. 2006;175(7):745–8.
Erickson SC, Le L, Zakharyan A, Stockl KM, Harada AS, Borson S, et al. New-onset treatment-dependent diabetes mellitus and hyperlipidemia associated with atypical antipsychotic use in older adults without schizophrenia or bipolar disorder. J Am Geriatr Soc. 2012;60(3):474–9.
Gurwitz JH, Bohn RL, Glynn RJ, Monane M, Mogun H, Avorn J. Antihypertensive drug therapy and the initiation of treatment for diabetes mellitus. Ann Intern Med. 1993;118(4):273–8.
Hagberg KW, Divan HA, Nickel JC, Jick SS. Risk of incident antidepressant-treated depression associated with use of 5α-reductase inhibitors compared with use of α-blockers in men with benign prostatic hyperplasia: a population-based study using the Clinical Practice Research Datalink. Pharmacotherapy. 2017;37(5):517–27.
McCarthy LM, Savage R, Dalton K, Mason R, Li J, Lawson A, et al. ThinkCascades: a tool for identifying clinically important prescribing cascades affecting older people. Drugs Aging. 2022;39(10):829–40.
Rochon PA, Gurwitz JH. The prescribing cascade revisited. Lancet. 2017;389(10081):1778–80.
Acknowledgements
We thank Mustafa Yasar for his help in the preparation of this review and Hanneke Wessemius for her extra validation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The Royal Dutch Association of Pharmacists (KNMP) has supported the study with a non-conditional grant (Grant number PR20_0103). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflicts of interest/competing interests
The authors declare that none of them have received honoraria, reimbursement, or fees from any pharmaceutical companies related to this study.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
The dataset generated during and/or analyzed during the current study is available from the corresponding author on reasonable request.
Code availability
Not applicable.
Authors’ contributions
Study concept and design: FKC, PD, PvdB, JH. Study selection and data extraction: FKC, PD, AM, OA, RV, LM, SPV. Data, analysis: FKC, PD, PvdB, JH, AM, OA, RV. Interpretation of data: FKC, PD, PvdB, JH, AM, OA, RV. Preparation of manuscript: AM, OA. Critically reviewing the manuscript: all authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Adrien, O., Mohammad, A.K., Hugtenburg, J.G. et al. Prescribing Cascades with Recommendations to Prevent or Reverse Them: A Systematic Review. Drugs Aging 40, 1085–1100 (2023). https://doi.org/10.1007/s40266-023-01072-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-023-01072-y