Abstract
Background
Ensuring safe and effective analgesic use in residential aged care services is important because older adults are susceptible to analgesic-related adverse drug events (ADEs).
Objective
The aim of this study was to identify the proportion and characteristics of residents of aged care services who may benefit from analgesic review based on indicators in the 2021 Society for Post-Acute and Long-Term Care Medicine (AMDA) Pain Management Guideline.
Methods
Cross-sectional analyses of baseline data from the Frailty in Residential Sector over Time (FIRST) study (N = 550 residents) across 12 South Australian residential aged care services in 2019 were conducted. Indicators included the proportion of residents who received > 3000 mg/day of acetaminophen (paracetamol), regular opioids without a documented clinical rationale, opioid doses > 60 mg morphine equivalents (MME)/day, more than one long-acting opioid concurrently, and a pro re nata (PRN) opioid on more than two occasions in the previous 7 days. Logistic regression was performed to investigate factors associated with residents who may benefit from analgesic review.
Results
Of 381 (69.3%) residents charted regular acetaminophen, 176 (46.2%) were charted > 3000 mg/day. Of 165 (30%) residents charted regular opioids, only 2 (1.2%) had no prespecified potentially painful conditions in their medical record and 31 (18.8%) received > 60 MME/day. Of 153 (27.8%) residents charted long-acting opioids, 8 (5.2%) received more than one long-acting opioid concurrently. Of 212 (38.5%) residents charted PRN opioids, 10 (4.7%) received more than two administrations in the previous 7 days. Overall, 196 (35.6%) of 550 residents were identified as potentially benefiting from analgesic review. Females (odds ratio [OR] 1.87, 95% confidence interval [CI] 1.20–2.91) and residents with prior fracture (OR 1.62, 95% CI 1.12–2.33) were more likely to be identified. Observed pain (OR 0.50, 95% CI 0.29–0.88) was associated with a lower likelihood of being identified compared with residents with no observed pain. Overall, 43 (7.8%) residents were identified based on opioid-related indicators.
Conclusions
Up to one in three residents may benefit from a review of their analgesic regimen, including 1 in 13 who may benefit from a specific review of their opioid regimen. Analgesic indicators represent a new approach to target analgesic stewardship interventions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Analgesic indicators represent a new approach to improving the safe and effective use of analgesic medications in residential aged care services. |
Up to one in three residents may benefit from a review of their analgesic medications, predominantly due to acetaminophen dosing. In total, 1 in 13 residents may benefit from a specific review of their opioid regimen. |
1 Introduction
Optimizing analgesic use in residential aged care services is complex and involves assessing each resident’s treatment benefits and risks. Unmanaged or undermanaged pain is associated with reduced quality of life, while overuse of analgesics poses a risk to resident wellbeing [1, 2]. Complex multimorbidity, polypharmacy and renal impairment correspond to increased susceptibility to analgesic-related adverse drug events (ADEs) [3, 4].
Evidence-based guidelines recommend acetaminophen (paracetamol) as the first-line analgesic for mild-to-moderate pain in older adults [5, 6]. Despite a favourable safety profile, earlier pharmacokinetic studies suggest volume of distribution and clearance are reduced by 20% and 38% in frail older adults, potentially increasing the risk of hepatotoxicity at standard therapeutic doses (4000 mg/day) [7]. Recent guidelines recommend a maximum dose of 3000 mg/day for frail older adults or those with low body weight (< 50 kg) [8,9,10]. Opioids are highly prevalent, with five Australian studies reporting that 28–34% of residents are prescribed regular opioids over periods from 1 week to 1 month [11]. A meta-analysis of previous research reported an association between opioids and falls (odds ratio [OR] 1.6, 95% confidence interval [CI] 1.4–1.9) [12], with opioids being one of three medication classes implicated in 60% of ADE-related emergency department presentations among older adults in the United States (US) [13]. An observational study reported older adults receiving ≥ 60 mg morphine equivalents (MME)/day were at increased odds of ADEs compared with those receiving < 60MME/day (OR 4.2, 95% CI 1.9–9.0) [14]. A US cohort study found the risk of opioid-related ADEs is heightened when receiving multiple opioids and other central nervous system (CNS) medications [15]. Opioids are now more prevalent than nonsteroidal anti-inflammatory drugs (NSAIDs) in most countries due to the risk of ADEs associated with NSAIDs in older adults [16].
Initiatives to ensure analgesic appropriateness include frequent pain assessment, medication review, and consideration of resident cognitive impairment, frailty, therapeutic goals and preferences [17,18,19,20]. The 2021 Society for Post-Acute and Long-Term Care Medicine (AMDA) ‘Pain Management in the Post-Acute and Long-Term Care Setting’ guideline provides facility-wide guidance on how to monitor, review and improve pain management practices [8]. This includes possible indicators for identifying residents who may benefit from an analgesic review. The objective of this study was to identify the proportion and characteristics of residents of aged care services who may benefit from an analgesic review based on indicators in the 2021 AMDA Pain Management Guideline.
2 Methods
2.1 Study Design and Setting
A secondary cross-sectional analysis of baseline data from the Frailty in Residential Sector over Time (FIRST) study, a prospective 3-year cohort study across 12 residential aged care services in South Australia, was conducted [21]. In Australia, residential aged care services provide supported accommodation for older adults with long-term needs and are synonymous with ‘nursing homes’ and ‘long-term care facilities’ internationally [22]. Analgesics are predominantly charted by general medical practitioners (GPs), dispensed by off-site community pharmacies and administered by nurses or care workers [23].
2.2 Study Sample
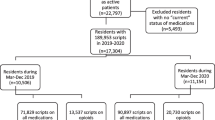
Permanent residents living in the residential aged care services for at least 8 weeks were eligible to participate, regardless of their pain levels. Residents who were deemed by clinical staff to be medically unstable (e.g., experiencing delirium) or at the end of life (< 3 months to live) were excluded, as were residents who had difficulty understanding English. Across 1243 residents living in the 12 services (representing 6.8% of total residential aged care residents [n = 18,375] in South Australia), 1060 (85.3%) residents were eligible for the FIRST study. A total of 472 (44.5%) of those eligible either declined (n = 367, 34.6%) or their substitute decision maker was not available (n = 105, 9.9%), resulting in 588 residents (55.5%) being included in the FIRST study and 550 (51.9%) completing baseline assessments.
2.3 Data Collection
Data were collected between March and October 2019 using a combination of resident medical records and observations, physical assessments and questionnaires administered by trained study nurses. The study nurse interviewed site registered nurses (RNs) on the day of data collection, or within 5 days of the interview date, to complete dementia and frailty assessments. Site RNs were required to have known the respective resident for at least 2 weeks before conducting assessments. Trained study nurses scanned each resident’s medication chart and researchers with pharmacy training extracted medication data. A full audit of each medication entry was completed for all residents. Data on pro re nata (PRN) administrations in the preceding 7 days were extracted.
2.3.1 Resident Characteristics
The Pain Assessment in Advanced Dementia (PAINAD) scale was used to assess possible pain in residents with and without cognitive impairment [24]. The trained study nurse observed each resident for 5 min before applying the scale, as recommended in the PAINAD guideline [24]. Possible scores range from 0 to 10 and were classified as no (0), mild (1–3), moderate (4–6) and severe (7–10) pain. These cut-off points were consistent with published literature and have been demonstrated to have good concurrent validity (Kendall’s τ = 0.73, p < 0.0001) and inter-rater agreement (ĸ = 0.74, p < 0.0001) compared with the numerical rating scale [25]. In our study, residents were dichotomized into those with no (0) versus mild/moderate (1–6) pain. This was because no residents were observed to be in severe (7–10) pain.
Data on a series of prespecified potentially painful conditions were extracted from resident medical records and classified as musculoskeletal (arthritis or osteoporosis or gout), cancer (any cancer or any tumour), prior fracture (any fracture) or vascular (peripheral artery disease or leg ulcers or ulcer disease) pain [26, 27]. The 12-item Dementia Severity Rating Scale (DSRS) was used to assess dementia severity [28]. Possible scores ranged from 0 to 54 and were classified as no (0–11), mild (12–18), moderate (19–36) and severe (37–54) dementia. The 7-item FRAIL-NH screening tool was used to assess frailty [29]. FRAIL-NH scores ranged from 0 to 14 and were classified as non-frail (0–2), frail (3–6) or most frail (7–14) [21].
2.3.2 Medications
Analgesics were categorized using the Anatomical Therapeutic Chemical (ATC) Classification System recommended by the World Health Organization [30]. This included acetaminophen (ATC code: N02BE01), NSAIDs (M01A), gabapentinoids (pregabalin [N03AX16] and gabapentin [N03AX12]) and opioids (buprenorphine [N02AE01], codeine [R05DA04], codeine and acetaminophen [N02AJ06], fentanyl [N02AB03], hydromorphone [N02AA03], morphine [N02AA01], oxycodone [N02AA05], oxycodone and naloxone [N02AA55], tapentadol [N02AX06] and tramadol [N02AX02]). MME/day was calculated by multiplying the prescribed daily dose of opioids by the conversion factor [31].
2.4 Indicators
We applied all five analgesic-related indicators included in the AMDA Pain Management Guideline indicators for identifying residents who may benefit from an in-depth review of their pain management (Table 34, p. 73) [8]. The analgesic indicators were applied to identify residents who may benefit from an analgesic review. These included the proportion of residents receiving:
-
1.
> 3000 mg/day of acetaminophen.
-
2.
A regular opioid without a documented clinical rationale. As clinical indications for opioid use were unavailable, medical records were examined for potentially painful conditions.
-
3.
Opioid doses of > 60 MME/day.
-
4.
More than one long-acting opioid concurrently.
-
5.
A PRN opioid on more than two occasions in the previous 7 days.
2.5 Statistical Analysis
Descriptive statistics were used to report the characteristics of residents who comprised the denominator for each indicator. Normally distributed variables were presented as mean values and standard deviations (SD), non-normally distributed variables were presented as median values and interquartile ranges (IQRs), and categorical variables were presented as frequencies and percentages. The proportion of residents who met the threshold for each indicator was calculated. For indicators with > 30 residents meeting the indicator, the characteristics of residents who met and did not meet the indicator were compared using independent t tests for normally distributed continuous variables, Mann–Whitney U tests for non-normally distributed continuous variables, and Pearson’s Chi-square tests for categorical variables. Residents who met one or more indicator (i.e., identified as potentially benefiting from an analgesic review) were also compared with the 550 participating residents using the same method. Logistic regression analyses were performed to compute unadjusted and adjusted ORs and 95% CIs for resident and clinical factors associated with potentially benefiting from review. Variables with p values < 0.25 in univariate analyses were included in the final regression model, along with variables that have been significantly associated with analgesic use in residential aged care services in the literature (age, sex, DSRS and FRAIL-NH) [32,33,34]. For the regression model, DSRS was classified into a binary variable (i.e., no dementia [0–11] or dementia [12–54]). Statistical significance was set at p < 0.05. Analyses were performed using SAS version 9.4 (SAS Institute, Inc., Cary, NC, USA).
2.6 Ethical Considerations
The study was approved by the University of Adelaide Human Research Ethics Committee (HREC-2018-247), South Australian Department for Health and Wellbeing Human Research Ethics Committee (HREC/20/SAH/15) and Department of Human Services External Request Evaluation Committee (EREC/RMS0432), and was registered with the Monash University Human Research Ethics Committee (23620). All residents or their substitute decision maker provided written informed consent to participate.
3 Results
3.1 Characteristics
In total, 550 residents completed baseline assessments. The median age was 89 years (IQR 84–92) and 403 (73.3%) residents were female (Table 1). The majority (n = 470, 85.5%) of residents were not observed to be in pain using the PAINAD scale, 417 (75.8%) had mild, moderate, or severe dementia severity using the DSRS, and 482 (87.6%) had frailty using FRAIL-NH. Of 550 residents, 381 (69.3%) were charted regular acetaminophen, 299 (54.4%) were charted PRN acetaminophen, 165 (30%) were charted a regular opioid and 212 (38.5%) were charted a PRN opioid. In total, 57 (10.4%) residents were charted regular pregabalin and 4 (0.7%) were charted regular gabapentin. The prevalence of regular NSAIDs was low (n = 8, 1.5%).
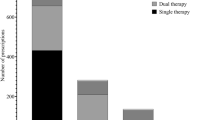
3.2 Indicator 1: The Proportion of Residents Charted > 3000 mg/day of Acetaminophen
Of 381 (69.3%) residents charted regular acetaminophen by their prescriber, 205 (53.8%) were charted ≤ 3000 mg/day and 176 (46.2%) were charted > 3000 mg/day (Fig. 1). Of residents charted > 3000mg/day, 101 (57.4%) were charted 3990 mg/day and 75 (42.6%) were charted 4000 mg/day. Of residents charted > 3000 mg/day, 155 (88.1%) were classified as either frail or most frail using FRAIL-NH (electronic supplementary material [ESM] Table S1). Residents charted > 3000 mg/day had lower median DSRS scores compared with those charted ≤ 3000 mg/day (18 [IQR 11–31] vs. 25 [IQR 14–39]; p = 0.002) [ESM Table S1). Of 299 (54.4%) residents charted PRN acetaminophen, 4 (1.3%) were administered PRN acetaminophen in the preceding 24 h, but none received > 3000 mg/day.
3.3 Indicator 2: The Proportion of Residents Receiving a Regular Opioid without a Documented Clinical Rationale (i.e., Potentially Painful Condition)
Of 165 (30%) residents charted a regular opioid, 2 (1.2%) had none of the potentially painful conditions listed in their medical records (Fig. 1). The most common potentially painful condition was arthritis (n = 152, 93.3%); however, when excluding arthritis, 22 (13.3%) residents received a regular opioid with none of the prespecified potentially painful conditions.
3.4 Indicator 3: The Proportion of Residents Receiving Opioid Doses of > 60 MME/Day
Of 165 (30%) residents charted a regular opioid, 31 (18.8%) received > 60 MME/day (Fig. 1, ESM Table S1). Of 212 (38.5%) residents charted a PRN opioid, 6 (2.8%) were administered a PRN opioid in the preceding 24 h but none received > 60 MME/day.
3.5 Indicator 4: The Proportion of Residents Receiving More Than One Long-Acting Opioid Concurrently
Of 153 (27.8%) residents charted any long-acting opioid, 8 (5.2%) received more than one long-acting opioid concurrently (Fig. 1). For six of the eight residents, this involved using both a long-acting patch and long-acting oral formulation.
3.6 Indicator 5: The Proportion of Residents Receiving a PRN Opioid on More Than Two Occasions in the Previous 7 Days
Of 212 (38.5%) residents charted a PRN opioid, 10 (4.7%) were administered a PRN opioid on more than two occasions in the previous 7 days (Fig. 1).
3.7 Residents Identified for an Analgesic Review
Overall, 196 (35.6%) of 550 residents were identified as potentially benefiting from an analgesic review. Identified residents had lower median DSRS scores compared with those who were not identified (19 [IQR 12–31] vs. 24 [IQR 12–37]; p = 0.02) [Table 2]. Female residents (OR 1.87, 95% CI 1.20–2.91) and residents with prior fracture (OR 1.62, 95% CI 1.12–2.33) were more likely to be identified for review (Table 3). Observed pain (OR 0.50, 95% CI 0.29–0.88) meant residents were less likely to be identified for review, compared with residents with no observed pain. When applying the four opioid-related indicators, 43 (7.8%) of 550 residents were identified as potentially benefiting from an analgesic review.
4 Discussion
This was the first study to apply analgesic indicators derived from the new AMDA Pain Management Guideline in residential aged care services. In total, 35.6% of all residents were identified as potentially benefiting from a review of their analgesic regimen. While the majority of residents were identified based on the prescribed dose of acetaminophen, 7.8% were identified based on four opioid-related indicators. Female residents and those with a prior fracture were more likely to be identified as potentially benefiting from analgesic review. Residents observed to be in any mild/moderate pain were less likely to be identified compared with residents with no observed pain.
Almost half of residents charted regular acetaminophen were prescribed > 3000 mg/day, with 88.1% of these residents classified as frail or most frail. The AMDA and other pain management guidelines recommend acetaminophen dose for frail older adults should not exceed 3000 mg/day (2000 mg/day for individuals with impaired liver function) [8,9,10]. Our research suggests dose adjustment of acetaminophen was uncommon when our data were collected in 2019. Our findings are comparable with another Australian study in 2019 that reported 150 (62.5%) of 240 older hospital inpatients were prescribed acetaminophen at higher than recommended doses (> 3000 mg/day or ≥ 60 mg/kg/day for patients with a body weight < 50 kg) [35]. Overall, 18.8% of residents received opioid doses > 60 MME/day. Despite increasing long-term dose requirements due to opioid tolerance, there is little evidence to support improved clinical outcomes with higher doses and more evidence regarding harms, including falls and fractures [5, 14].
Of residents charted a PRN opioid, 4.7% received a PRN opioid on two or more occasions in the previous 7 days. In contrast, previous Australian studies have reported that opioids were among the most administered PRN medications [36, 37]. Picton et al. reported that of 118 residents prescribed a PRN opioid at the index date, 76.0% were administered an opioid on one or more occasions over 12 months [36]. This discrepancy is likely due to the short lookback period of 7 days in our study. In Australia, GPs visit residential aged care services periodically and PRN medications ensure staff have timely access to manage resident symptoms, such as breakthrough pain. Sharma et al. reported opioid administrations were more common among residents in non-metropolitan areas, where GP presence is often limited [37]. In our study, 3 of 12 services were located in regional areas. The exclusion of individuals with < 3 months to live in our study is an important consideration, given that PRN administrations are more frequent at the end-of-life [38].
Females were more likely to be identified for analgesic review compared with males. Females report pain more frequently, receive more analgesics and are more susceptible to opioid-related ADEs [26]. Residents with prior fracture in their medical records were more likely to be identified, which highlights the importance of medication review post-hospitalization and potential deprescribing of analgesics post-resolution of pain [39]. Interestingly, observed pain meant residents were less likely to be identified compared with residents with no observed pain. Prevalence of mild/moderate observed pain was low (14.5%) in our study compared with previous research. A systematic review of five studies reported observed pain ranged from 19.1 to 43.2% in residents with dementia, and 27.1–67.0% in residents without dementia [40]. This may have been because pain management strategies were effective in reducing pain for residents included in our study. The aged care provider organization had a structured approach to identifying and managing pain for all residents when our study was conducted.
4.1 Future Directions
Analgesic indicators can be used to identify residents who may benefit from an individualized assessment of their analgesic regimen. Components of analgesic reviews and stewardship interventions include multidisciplinary medication reviews, systematic and thorough assessment of pain, and/or introduction of non-pharmacological approaches to pain management [19, 41]. Given these interventions can be resource and time intensive, indicators implemented at the system-level could be used to target analgesic stewardship interventions at the resident level.
Among other benefits, advances in electronic medication management systems may represent unique opportunities to efficiently implement analgesic indicators and reform analgesic optimization in residential aged care services. Given the complexity of pain, the AMDA emphasizes the importance of adopting an interprofessional approach to pain management [8]. Interventions involving enhanced roles for nurses and pharmacists within the interprofessional team can improve analgesic use and appropriateness in residential aged care services [41]. Following recommendations from the Royal Commission into Aged Care Quality and Safety, the Australian Government has committed AUS$345.7 million (US$239.5 million) over 4 years for aged care provider organizations to employ embedded on-site pharmacists starting in 2023 [42, 43]. Analgesic reviews could be a key role undertaken by these pharmacists in partnership with the interprofessional team. Analgesic reviews may assist in reducing polypharmacy, because an increase in the use of analgesic medications has been identified as one of the top five factors contributing to the increasing prevalence of polypharmacy in Australian residential aged care services [44]. Embedded on-site pharmacists may complement or replace the pre-existing model for residential medication management reviews (RMMRs).
In total, only 1 in 13 residents were identified for review based on opioid-related indicators. Application of opioid-related indicators provides a possible mechanism for aged care provider organizations to monitor safe and effective opioid use. Australian aged care provider organizations operate medication advisory committees (MACs) that oversee and monitor high-risk medication use [45]. It is possible that further residents may have been identified for review if other analgesic indicators were applied. Other potential indicators include the proportion of opioid users administered laxatives, the concurrent use of opioids with other CNS depressants, and the proportion of opioid users experiencing pain [46]. Although not a focus of this study, frequent pain assessment helps to address potential underprescribing of opioids associated with resident and prescriber reluctance, hence improving uncontrolled pain [47]. Chronic opioid use, defined as continuous opioid use for at least 90 days or for 120 non-consecutive days, has been identified as a potential future quality indicator for addition to Australia’s National Aged Care Mandatory Quality Indicator Programme [48, 49]. Further research is needed to explore the predictive validity of analgesic indicators at identifying residents most susceptible to analgesic-related ADEs.
4.2 Strengths and Limitations
We analysed data for a well-characterized sample of residents. The AMDA Pain Management Guideline recommends an analgesic review for residents receiving opioids without a clearly documented clinical rationale (i.e., indicator 2) [8]. However, not all potentially painful conditions should be managed with opioids. For example, we included arthritis as a potentially painful condition, yet the Royal Australian College of General Practitioners (RACGP) do not recommend opioids for managing osteoarthritis [50]. Additionally, some conditions may cause pain and were not considered as one of the prespecified potentially painful conditions (e.g., people living with diabetes). Nursing progress notes were not accessed for this study but may have contained information related to each resident’s current pain status and pain management strategy. Although comprehensive pain assessments were undertaken by trained study nurses using a scale for which inter-rater reliability has been demonstrated, we did not compute inter-rater reliability of pain assessments undertaken by the nurses in our study. The prevalence of pain and pain intensity can vary according to the pain assessment method used [40]. Future studies may analyse both self-reported and observed pain scores [6]. It was not possible to determine whether pain management practices have changed following publication of the guideline or with increased awareness regarding acetaminophen dose, as the data were collected 2 years prior to publication of the AMDA Pain Management Guideline. The study was conducted in partnership with a single aged care provider organization in South Australia, meaning the generalizability to the wider aged care population is unknown.
5 Conclusions
Up to one in three residents may benefit from a review of their analgesic regimen, including 1 in 13 who may benefit from a specific review of their opioid regimen. Analgesic indicators represent a new approach to target analgesic stewardship interventions in residential aged care services. Analgesic indicators may also assist aged care provider organizations to monitor safe and effective opioid use.
References
Helvik A-S, Bergh S, Šaltytė Benth J, et al. Pain in nursing home residents with dementia and its association to quality of life. Aging Ment Health. 2022;26(9):1787–97. https://doi.org/10.1080/13607863.2021.1947968.
Brandauer A, Berger S, Freywald N, et al. Quality of life in nursing home residents with pain: pain interference, depression and multiple pain-related diseases as important determinants. Qual Life Res. 2020;29(1):91–7. https://doi.org/10.1007/s11136-019-02290-x.
Yoshikawa A, Ramirez G, Smith ML, et al. Opioid use and the risk of falls, fall injuries and fractures among older adults: a systematic review and meta-analysis. J Gerontol A. 2020;75(10):1989–95. https://doi.org/10.1093/gerona/glaa038.
Swart LM, van der Zanden V, Spies PE, et al. The comparative risk of delirium with different opioids: a systematic review. Drugs Aging. 2017;34(6):437–43. https://doi.org/10.1007/s40266-017-0455-9.
American Geriatrics Society Panel. Pharmacological management of persistent pain in older persons. J Am Geriatr Soc. 2009;57:1331–46. https://doi.org/10.1111/j.1532-5415.2009.02376.x.
Schofield P, Dunham M, Martin D, et al. Evidence-based clinical practice guidelines on the management of pain in older people—a summary report. Br J Pain. 2022;16(1):6–13. https://doi.org/10.1177/2049463720976155.
Mian P, Allegaert K, Spriet I, et al. Paracetamol in older people: towards evidence-based dosing? Drugs Aging. 2018;35(7):603–24. https://doi.org/10.1007/s40266-018-0559-x.
Levenson S, Resnick B, Cryst S, et al. Pain management in the post-acute and long-term care setting: a clinical practice guideline (CPG) from the Society for Post-Acute and Long-Term Care Medicine (AMDA). J Am Med Dir Assoc. 2021;22(12):2407. https://doi.org/10.1016/j.jamda.2021.10.008.
Freo U, Ruocco C, Valerio A, et al. Paracetamol: a review of guideline recommendations. J Clin Med. 2021;10(15):3420. https://doi.org/10.3390/jcm10153420.
Kolasinski SL, Neogi T, Hochberg MC, et al. 2019 American College of Rheumatology/Arthritis foundation guideline for the management of osteoarthritis of the hand, hip, and knee. Arthritis Rheumatol. 2020;72(2):220–33. https://doi.org/10.1002/art.41142.
Dowd LA, Reynolds L, Cross AJ, et al. A systematic review of opioid prevalence in Australian residential aged care facilities. Australas J Ageing. 2022;41(4):501–12. https://doi.org/10.1111/ajag.13071.
Seppala LJ, van de Glind EMM, Daams JG, et al. Fall-risk-increasing drugs: a systematic review and meta-analysis: III. Others. J Am Med Dir Assoc. 2018;19(4):372.e1-372.e8. https://doi.org/10.1016/j.jamda.2017.12.099.
Shehab N, Lovegrove MC, Geller AI, et al. US emergency department visits for outpatient adverse drug events, 2013–2014. JAMA. 2016;316(20):2115–25. https://doi.org/10.1001/jama.2016.16201.
Kim JY, Kim JH, Yee J, et al. Risk factors of opioid-induced adverse reactions in elderly male outpatients of Korea Veterans Hospital. BMC Geriatr. 2018;18(1):293. https://doi.org/10.1186/s12877-018-0990-1.
Herzig SJ, Anderson TS, Jung Y, et al. Risk factors for opioid-related adverse drug events among older adults after hospital discharge. J Am Geriatr Soc. 2022;70(1):228–34. https://doi.org/10.1111/jgs.17453.
La Frenais FL, Bedder R, Vickerstaff V, et al. Temporal trends in analgesic use in long-term care facilities: a systematic review of international prescribing. J Am Geriatr Soc. 2018;66(2):376–82. https://doi.org/10.1111/jgs.15238.
Schofield P, Abdulla A. Pain assessment in the older population: what the literature says. Age Ageing. 2018;47(3):324–7. https://doi.org/10.1093/ageing/afy018.
Tsai YIP, Browne G, Inder KJ. The effectiveness of interventions to improve pain assessment and management in people living with dementia: a systematic review and meta-analyses. J Adv Nurs. 2021;77(3):1127–40. https://doi.org/10.1111/jan.14660.
Sheikh F, Brandt N, Vinh D, Elon RD. Management of chronic pain in nursing homes: navigating challenges to improve person-centered care. J Am Med Dir Assoc. 2021;22(6):1199–205. https://doi.org/10.1016/j.jamda.2020.12.029.
Husebø BS, Ballard C, Aarsland D, et al. The effect of a multicomponent intervention on quality of life in residents of nursing homes: a randomized controlled trial (COSMOS). J Am Med Dir Assoc. 2019;20(3):330–9. https://doi.org/10.1016/j.jamda.2018.11.006.
Jadczak AD, Robson L, Cooper T, et al. The frailty in residential sector over time (FIRST) study: methods and baseline cohort description. BMC Geriatr. 2021;21(1):1–12. https://doi.org/10.1186/s12877-020-01974-1.
Sanford AM, Orrell M, Tolson D, et al. An international definition for “nursing home.” J Am Med Dir Assoc. 2015;16(3):181–4. https://doi.org/10.1016/j.jamda.2014.12.013.
Sluggett JK, Ilomäki J, Seaman KL, et al. Medication management policy, practice and research in Australian residential aged care: current and future directions. Pharmacol Res. 2017;116:20–8. https://doi.org/10.1016/j.phrs.2016.12.011.
Warden V, Hurley AC, Volicer L. Development and psychometric evaluation of the Pain Assessment in Advanced Dementia (PAINAD) scale. J Am Med Dir Assoc. 2003;4(1):9–15. https://doi.org/10.1097/01.JAM.0000043422.31640.F7.
Mosele M, Inelmen EM, Toffanello ED, et al. Psychometric properties of the pain assessment in advanced dementia scale compared to self assessment of pain in elderly patients. Dement Geriatr Cogn Disord. 2012;34(1):38–43. https://doi.org/10.1159/000341582.
Tan EC, Visvanathan R, Hilmer SN, et al. Analgesic use and pain in residents with and without dementia in aged care facilities: a cross-sectional study. Australas J Ageing. 2016;35(3):180–7. https://doi.org/10.1111/ajag.12295.
Veal F, Williams M, Bereznicki L, et al. A retrospective review of pain management in Tasmanian residential aged care facilities. Br J Gen Pract. 2019. https://doi.org/10.3399/bjgpopen18X101629.
Clark CM, Ewbank DC. Performance of the dementia severity rating scale: a caregiver questionnaire for rating severity in Alzheimer disease. Alzheimer Dis Assoc Disord. 1996;10(1):31–9. https://doi.org/10.1097/00002093-199601010-00006.
Liau SJ, Lalic S, Visvanathan R, et al. The FRAIL-NH scale: systematic review of the use, validity and adaptations for frailty screening in nursing homes. J Nutr Health Aging. 2021;25(10):1205–16. https://doi.org/10.1007/s12603-021-1694-3.
World Health Organization (WHO). Anatomical Therapeutic Chemical (ATC) Classification. WHO; 1982. https://www.who.int/tools/atc-ddd-toolkit/atc-classification. Accessed 22 July 2022.
The Centers for Medicare and Medicaid Services. Opioid oral morphine milligram equivalent (MME) conversion factors. 2020. https://www.cms.gov/Medicare/Prescription-Drug-Coverage/PrescriptionDrugCovContra/Downloads/Oral-MME-CFs-vFeb-2018.pdf. Accessed 1 Jan 2022.
Veal FC, Bereznicki LR, Thompson AJ, Peterson GM. Pharmacological management of pain in Australian aged care facilities. Age Ageing. 2014;43(6):851–6. https://doi.org/10.1093/ageing/afu072.
Fain KM, Alexander GC, Dore DD, et al. Frequency and predictors of analgesic prescribing in U.S. Nursing home residents with persistent pain. J Am Geriatr Soc. 2017;65(2):286–93. https://doi.org/10.1111/jgs.14512.
La Frenais F, Vickerstaff V, Cooper C, et al. Factors influencing prescription and administration of analgesic medication: a longitudinal study of people with dementia living in care homes. Int J Geriatr Psychiatry. 2021;36(9):1354–61. https://doi.org/10.1002/gps.5526.
Reid O, Ngo J, Lalic S, et al. Paracetamol dosing in hospital and on discharge for older people who are frail or have low body weight. Br J Clin Pharmacol. 2022;88(10):4565–72. https://doi.org/10.1111/bcp.15394.
Picton L, Ilomäki J, Keen CS, et al. Rates of PRN medication administration in Australian residential aged care. J Am Med Dir Assoc. 2021;22(1):117-123.e1. https://doi.org/10.1016/j.jamda.2020.04.033.
Sharma M, Wong XY, Bell JS, et al. Trajectories of pro re nata (PRN) medication prescribing and administration in long-term care facilities. Res Social Adm Pharm. 2021;17(8):1463–8. https://doi.org/10.1016/j.sapharm.2020.11.003.
Dörks M, Allers K, Hoffmann F. Pro re nata drug use in nursing home residents: a systematic review. J Am Med Assoc. 2019;20(3):287-293.e287. https://doi.org/10.1016/j.jamda.2018.10.024.
Parekh N, Ali K, Stevenson JM, et al. Incidence and cost of medication harm in older adults following hospital discharge: a multicentre prospective study in the UK. Br J Clin Pharmacol. 2018;84(8):1789–97. https://doi.org/10.1111/bcp.13613.
Tan ECK, Jokanovic N, Koponen M, et al. Prevalence of analgesic use and pain in people with and without dementia or cognitive impairment in aged care facilities: a systematic review and meta-analysis. Curr Clin Pharmacol. 2015;10(3):194–203. https://doi.org/10.2174/157488471003150820144958.
Dowd LA, Cross AJ, Veal F, et al. A systematic review of interventions to improve analgesic use and appropriateness in long-term care facilities. J Am Med Dir Assoc. 2022;23(1):33-43.e33. https://doi.org/10.1016/j.jamda.2021.09.024.
Australian Government Aged Care Quality and Safety Commission. Royal Commission into Aged Care Quality and Safety. Final report—list of recommendations; 2021. https://agedcare.royalcommission.gov.au/sites/default/files/2021-03/final-report-recommendations.pdf. Accessed 7 Oct 2022.
Cross AJ, La Caze A, Steeper M, Bell JS. Embedding pharmacists in residential aged care: why it’s important to integrate resident- and system-level services. J Pharm Pract Res. 2022;52(4):263–5. https://doi.org/10.1002/jppr.1821.
Jokanovic N, Tan ECK, Dooley MJ, et al. Why is polypharmacy increasing in aged care facilities? The views of Australian health care professionals. J Eval Clin Pract. 2016;22(5):677–82. https://doi.org/10.1111/jep.12514.
Picton L, Lalic S, Ryan-Atwood TE, et al. The role of medication advisory committees in residential aged care services. Res Soc Adm Pharm. 2020;16(10):1401–8. https://doi.org/10.1016/j.sapharm.2020.01.008.
Wei Y-JJ, Schmidt S, Chen C, et al. Quality of opioid prescribing in older adults with or without Alzheimer disease and related dementia. Alzheimers Res Ther. 2021;13(1):78. https://doi.org/10.1016/j.trci.2018.10.012.
Pringle J, Mellado ASAV, Haraldsdottir E, et al. Pain assessment and management in care homes: understanding the context through a scoping review. BMC Geriatr. 2021;21(1):431. https://doi.org/10.1186/s12877-021-02333-4.
Inacio MC, Lang C, Caughey GE, et al. The Registry of Senior Australians outcome monitoring system: quality and safety indicators for residential aged care. Int J Qual Health Care. 2020;32(8):502–10. https://doi.org/10.1093/intqhc/mzaa078.
Australian Government Aged Care Quality and Safety Commission. National aged care mandatory quality indicator program; 2019. https://www.agedcarequality.gov.au/consumers/national-aged-care-mandatory-quality-indicator-program. Accessed 28 June 2022.
Royal Australian College of General Practitioners. Guideline for the management of knee and hip osteoarthritis. 2nd ed; 2018. https://www.racgp.org.au/download/Documents/Guidelines/Musculoskeletal/guideline-for-the-management-of-knee-and-hip-oa-2nd-edition.pdf. Accessed 20 June 2022.
Acknowledgements
The authors would like to acknowledge Terry Short for supporting the study coordination and contributing to the study design, as well as Hilary Ashby who supported the study coordination. They would also like to thank their study nurses Jean Zhang, Victoria Foy and Kim Testi who conducted the data collection and collected informed consent. Finally, the authors would like to acknowledge Tina Cooper and Leonie Robson for contributing to the study design, and thank Tina Cooper and Merridy Baylis for reviewing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The FIRST study and ADJ were funded through project funding to RV from the Healthy Aging Research Consortium funded by the South Australian Department for Innovation and Skills, Hospital Research Foundation and Resthaven Inc. LAD and SJL were supported by postgraduate research scholarships funded by Monash University. AJC was supported by a National Health and Medical Research Council (NHMRC) Emerging Leadership Fellowship. SJL, RV, and JSB were supported by the NHMRC Centre of Research Excellence in Frailty and Healthy Ageing. SJL was supported by the Australian Government Research Training Program Scholarship. JSB was supported by the NHMRC Boosting Dementia Research Leadership Fellowship. Open Access funding enabled and organized by CAUL and its Member Institutions.
Conflict of interest
Renuka Visvanathan was previously a board member and member of the clinical governance committee of Resthaven Inc. She recently received honorarium for participation in workgroups from HealthConsult Pty Ltd in relation to a Commonwealth Funded project, and in the past has received honoraria and speakers and educational grants in various combinations from Nutricia, Abbott and Nestlé. J. Simon Bell has received grant or consulting funds from the NHMRC, Medical Research Future Fund, Victorian Government Department of Health and Human Services, Dementia Australia Research Foundation, Yulgilbar Foundation, Aged Care Quality and Safety Commission, Australian Commission on Safety and Quality in Health Care, Dementia Centre for Research Collaboration, Pharmaceutical Society of Australia, Society of Hospital Pharmacists of Australia, GlaxoSmithKline Supported Studies Programme, Amgen, and several aged care provider organizations, all paid to Monash University. Laura A. Dowd, Amanda J. Cross, Shing J. Liau, Agathe D. Jadczak and Felicity C. Veal declare no conflicts of interest.
Ethics approval
This study was approved by the University of Adelaide Human Research Ethics Committee (HREC-2018-247), South Australian Department for Health and Wellbeing Human Research Ethics Committee (HREC/20/SAH/15) and Department of Human Services External Request Evaluation Committee (EREC/RMS0432), and was registered with the Monash University Human Research Ethics Committee (23620).
Consent to participate
Informed consent was obtained from all residents or their substitute decision maker.
Consent for publication
All authors approved the current manuscript for publication. All participants and or their family provided consent for publication of de-identified data.
Availability of data and material
The authors confirm that all relevant data are included in the article and/or its supplementary information files.
Code availability
Not applicable.
Author contributions
Conceptualization: LAD, AJC, ADJ, RV, JSB. Acquisition of data: SJL, ADJ, JSB, RV. Analysis and interpretation of data: LAD, AJC, SJL, JSB. Drafting of the manuscript: LAD, AJC, SJL, FCV, JSB. Critical revision of the manuscript for important intellectual content: All authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Dowd, L.A., Cross, A.J., Liau, S.J. et al. Identifying Residents Who May Benefit from an Analgesic Review: Applying Analgesic Indicators in Residential Aged Care Services. Drugs Aging 40, 449–459 (2023). https://doi.org/10.1007/s40266-023-01025-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-023-01025-5