Abstract
Background
Psychotropic medications are commonly prescribed to people with dementia (PwD) for non-cognitive symptoms of dementia (NCSD), but have significant risks. A national audit was performed in acute hospitals in the Republic of Ireland (ROI) to establish baseline practice prior to the launch and implementation of a National Clinical Guideline on the appropriate prescribing of psychotropic medications for NCSD. The objective of this study was to analyse psychotropic prescribing patterns and compare these with international data and with existing (limited) data from a previous audit round.
Methods
The pooled anonymous dataset from the second round of the Irish National Audit of Dementia Care (INAD-2) was analysed. The audit had collected retrospective data from 30 random healthcare records from each of 30 acute hospitals in 2019. Inclusion criteria were a clinical diagnosis of dementia of any type, hospital stay of 72 hours or more, and discharge or death within the audit period. Most hospitals (87%) self-audited their healthcare records, but a random sample of six healthcare records (20%) from each hospital were re-audited by a highly trained healthcare auditor. The audit tool was based on a tool used in the England and Wales National Audit of Dementia audit rounds (Royal College of Psychiatrists), adapted to the Irish healthcare setting and national priorities.
Results
In total, 893 cases were included, as one hospital could not retrieve 30 cases even within a more prolonged audit period. The sample comprised 55% females and 45% males; the median age was 84 years (interquartile range 79–88 years) and the majority (89.6%) were >75 years of age. Only 52% of healthcare records specified the type of dementia; within these, the most common diagnosis was Alzheimer’s disease (45%). Most PwD (83%) were receiving psychotropic medication on admission; 40% were prescribed new or increased psychotropic medication during admission, mainly for medical indications, including end-of-life care and delirium. Anticonvulsants or cognitive enhancers were rarely prescribed for NCSD in hospital. However, new/increased antipsychotic medication was prescribed for NCSD in 11.8–17.6% of the total cohort, while 4.5–7.7% were prescribed a benzodiazepine for anxiety or NCSD. Overall, there was poor documentation of risk/benefit, or of discussion with the patient/family, and apparently inadequate review for efficacy and tolerability. Concurrently, acetylcholinesterase inhibitors appeared to be underused for cognitive impairment in the community.
Conclusion
This audit provides baseline data on psychotropic medication prescription for NCSD in Irish hospitals prior to a specific Irish guideline on this topic. Reflecting this, most PwD were receiving psychotropic medications on admission, and many were prescribed new/increased psychotropic medication in hospital, often without evidence of appropriate decision making and prescribing processes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Psychotropic prescribing is very common in people with dementia admitted to acute hospitals and is more common in people admitted from residential care than from home or other care settings. |
The majority of new psychotropics prescribed are antipsychotics, and to a much lesser extent benzodiazepines, while antidepressants, anticonvulsants, and cognitive enhancers are rarely commenced for non-cognitive symptoms of dementia in acute hospitals. |
Antipsychotic prescribing in acute hospitals in Ireland for non-cognitive symptoms of dementia appears to occur without sufficient comprehensive assessment, trials of non-pharmacological alternatives, and with insufficient review for effectiveness and side effects. |
1 Background
Non-cognitive symptoms of dementia (NCSD), or neuropsychiatric symptoms, are common in people with dementia (PwD). These symptoms include anxiety, depression, apathy, hallucination, etc. Many people with dementia will experience NCSD [1], reported to be 90% by Cerejeira et al. [2], and reported to be more common as dementia progresses [3].
Psychotropic medication is any drug that affects behaviour and mood [4]. This includes a variety of drugs such as antidepressants, antipsychotics, benzodiazepines, and hypnotics. The use of psychotropic medications in PwD can result in severe adverse effects including but not limited to cerebrovascular events and death [5,6,7,8]. Other side effects include confusion, drowsiness, extrapyramidal symptoms, increased risk of falls, lethargy, peripheral oedema, arrhythmias, and respiratory depression [9, 10]. In addition to their significant risk profile, many psychotropic medications are prescribed ‘off label’ as they are not licensed for use for NCSD (e.g., quetiapine, olanzapine, lorazepam) [9].
Within psychotropic medications overall, the risks of antipsychotic medications specifically have been well highlighted [11, 12]. Antipsychotics have demonstrated modest efficacy in treating certain NCSD symptoms (namely psychosis, aggression and agitation) but they have a significant adverse effect profile [13]. Despite this, the use of antipsychotics for treating NCSD has grown over the past two decades [13]. In April 2005, the United States (US) Food and Drug Administration (FDA) [14] published an independent, pooled analysis of 17 randomised controlled trials, which reported a 1.7-fold increased risk of all-cause mortality associated with atypical antipsychotic use compared with placebo, leading to a ‘black box warning’. More recent studies indicate an increased risk of mortality by up to 3.5-fold [5], with increased risk of sudden cardiac death and all‐cause mortality rates [15]. There is also a 3-fold increased risk of stroke compared with placebo [16]. The use of antipsychotic medication in people with dementia was reported to cause approximately an additional 1600 strokes per year and 1800 deaths per year in the United Kingdom (UK) [11]. Following this pivotal report, the National Health Service (NHS) endeavoured to reduce atypical antipsychotic use in PwD by two-thirds.
Similar concerns about the risk–benefit profile of benzodiazepines exist, with a reported 40% increase in hip fracture among people with Alzheimer's disease (AD) [17] and increased mortality for benzodiazepines [18], and for anticonvulsants and antidepressants [5]. There are also concerns that psychotropic medications are associated with lower quality of life [19].
A National Clinical Guideline (NCG) titled “Appropriate prescribing of psychotropic medication for non-cognitive symptoms in people with dementia” was launched by the Department of Health in the Republic of Ireland (ROI) in December 2019 [20]. This NCG fulfilled a priority action of the Irish National Dementia Strategy Implementation Plan that the Health Service Executive (HSE) would develop guidance material on the appropriate management of medication for PwD, in particular, the management of psychotropic medication. The NCG applies to a person with dementia in any setting of care. This NCG was closely based on three existing guidelines: the 2018 (UK) National Institute for Health and Care Excellence (NICE) guideline “Dementia: assessment, management and support for people living with dementia and their carers” [21]; the 2016 (Australian) Cognitive Decline Partnership Centre’s “Clinical practice guidelines and principles of care for people with dementia” [22] approved by the National Health and Medical Research Council (NHMRC); and “The American Psychiatric Association practice guideline on the use of antipsychotics to treat agitation or psychosis in patients with dementia” published in 2016 [23]. The Irish NCG remains, to the best of the authors’ knowledge, the most recent guideline on this topic.
Prior to the launch and national implementation of this NCG, it was necessary to establish baseline practice with regards to prescribing psychotropic medications for NCSD. This would inform the implementation of the guideline, identifying areas of suboptimal practice that could benefit from evidence-based guidelines and quality improvement, and also allow an accurate evaluation of the change in practice after the NCG was implemented. This current paper describes the results of this baseline snapshot of the usage of psychotropic medications for PwD in acute hospitals in Ireland in 2019. Although this baseline data cannot be used as evidence of the degree of adherence to the NCG, as the audit preceded the NCG, the other international guidelines were already available to guide practice at the time.
2 Methods
Healthcare record audit data was collected from 30 (out of 34) acute, publicly funded hospitals admitting PwD in the Republic of Ireland (ROI) in 2019 (thus excluding elective, private, and exclusively paediatric/maternity hospitals). One hospital group, comprising four acute hospitals, had declined to participate in the audit; thus 88% of all eligible hospitals were included. With permission, the audit tool was adapted and updated from the tool used in the first round of the UK National Audit of Dementia (Royal College of Psychiatrists) [24], through consultation with the Second Irish National Audit of Dementia care in acute hospitals (INAD-2) Steering Committee to consider changes made to this tool in subsequent rounds of the UK audit and also the context of the Irish healthcare system. Questions deemed to be irrelevant were removed and additional relevant questions for the Irish context were added.
The final audit tool is available at: https://www.ucc.ie/en/media/research/irishnationalauditofdementia/INAD-2CaseNoteAuditfinalwithpsychotropicmedicationsection.pdf, and as Online Resource 1 (see electronic supplementary material [ESM]).
The audit tool was used to scrutinise 30 healthcare records from each hospital. Data collection was carried out between June 2019 and November 2019. Most hospitals (87%) self-audited their healthcare records. All staff involved in the audit data collection completed audit training with the Audit Coordinator prior to commencing the audit and a detailed guidance document was prepared for their use. A quality assurance process was executed in all hospitals whereby a random sample of six healthcare records (20%) were re-audited by a HSE Healthcare auditor (or by a second HSE Healthcare auditor where the hospital was entirely audited by HSE Healthcare auditors).
Eligible healthcare records were identified through the Hospital Inpatient Enquiry (HIPE) system using the following inclusion criteria:
-
a diagnosis of dementia of any type;
-
minimum length of stay (LOS) of 72 hours;
-
dates of discharge (or death) between 1 January and 30 April 2019.
The diagnosis of dementia was based on there being a documented clinical diagnosis of dementia anywhere in the healthcare record, as coded by any of an inclusive list of relevant International Classification of Disease, 10th revision (ICD-10) codes for dementia. Of note, a prescription of dementia medications was not used as a surrogate for a diagnosis of dementia, partly because this is not accurate, but mainly because the practice around prescription of cognitive enhancers was part of the audit. The minimum LOS of 72 hours was chosen by the national audit team to allow time for comprehensive assessment and collateral gathering, after any required emergency treatment or medical stabilisation was achieved.
All audit data was collected between June and November 2019, based on eligible healthcare records with dates of discharge (or death) between 1 January and 30 April 2019. Where more than 30 eligible healthcare records were identified, a random sample was generated by the hospital’s coding department (using a randomisation function of the HIPE coding software system). Where needed, the eligible discharge date was extended retrogradely (up to one year) until the required number of cases was reached (e.g., from September 1, 2018 to April 30, 2019). Of note, all wards in the hospital were eligible for inclusion if a person with dementia was admitted there within the 30 selected healthcare records.
The audit collected information on demographics, assessments carried out during admission, referral to specialist services, and discharge planning. It examined the use of psychotropic medications overall, both existing prescriptions at the time of admission, and in-hospital prescriptions. Prescribing a medication for regular and/or for pro re nata ‘prn’ (as needed) prescribing was considered, without differentiation between these. All medications prescribed and administered to a hospital patient in Ireland are recorded in a standardised ‘drug cardex’—a standalone medication prescription and administration record. There are only minor differences in the structure of these between hospitals, and the documentation within these is tightly audited, so that the medication record can be taken to be fully accurate and complete. The decision making around commencing or altering a medication by a doctor, or to administer a prescribed ‘prn’ medication by a nurse, should be documented in the healthcare record, in the narrative clinical notes, and nursing notes, respectively. This documentation is not standardised.
The following medications were included in the audit (the Anatomical Therapeutic Chemical [ATC] classification code is shown in brackets):
-
Antipsychotic medications (N05A, excluding N05AN01 Lithium)
-
Benzodiazepines (N05BA)
-
Hypnotics and sedatives including Z-drugs (N05CF), benzodiazepine-derivatives (N05CD) and melatonin (N05CH01)
-
Acetylcholinesterase inhibitors (N06DA) and memantine (N06DX)
-
Antidepressant medications (N06A)
-
Anticonvulsant medications (N03A)
A psychotropic medication was judged to be prescribed for a ‘medical indication’ when there was documentation of it being prescribed for seizures, delirium, respiratory distress, hiccoughs, pre-operatively, for end-of-life care, etc. Decision making around such prescribing was not analysed. Further details on the audit methodology are available within the audit report [25].
Where relevant, data are compared with that of the first round of the Irish National Audit of Dementia Care (INAD), performed in 2013 [26], noting that only the prescribing of antipsychotic medication was included in that audit.
2.1 Data Analysis
Most data were categorical in nature, and valid percentages are reported (with the numerator/denominator value also reported whenever this is < 90% of the total data set). The actual denominator values for instances where there was < 10% of the data missing can be found in Supplementary Table 1. To compare categorical data between different groups, chi-square tests were performed (at a statistical threshold of p < 0.05). Median and interquartile ranges were used to describe the distribution of continuous variables, including age and LOS, as the data did not have a normal distribution. Inter-rater reliability was assessed using Cohen’s kappa coefficient in conjunction with percentage agreement for each item on the audit tool (based on the six healthcare records per site with dual auditing by a clinician–auditor or auditor–auditor pair). Any items with poor inter-rater reliability are indicated in the text. Of note, it was not possible to calculate inter-rater reliability for some of the questions on individual medications in the psychotropic audit due to the small number of participants receiving the medication. Data were analysed using SPSS version 24.
2.2 Ethics
Ethical approval was given by the Clinical Research Ethics Committee (CREC) of the Cork Teaching Hospitals, University College Cork (UCC).
3 Results
3.1 Demographics
A total of 893 healthcare records were audited from 30 hospitals. The sample comprised 55% females and 45% males; the median age was 84 years (interquartile range [IQR] 79–88 years) and the majority (89.6%) of patients were > 75 years of age. Only 52% of healthcare records specified the type of dementia; within these, the most common diagnosis was AD (45%). Where recorded, most patients were of Irish ethnicity (98% of 427 cases) and English was the most commonly documented first language (98.8%; 646/654). Most (93%) of the cases audited were unplanned admissions. Respiratory infection was the most common primary cause of admission (21%), while 8% of cases had dementia documented as the primary reason for admission.
Most of the patients (64%) were living at home (i.e., their own home, a carer’s home, or admitted from respite care), while 30% were admitted from residential care and approximately 6% from another service, such as a rehabilitation hospital, psychiatric unit, or another general hospital.
Patients spent the longest period during their admission on a general medical ward (69.8%), geriatric medicine ward (13.6%), surgical ward (7.8%), or an orthopedic ward (2.8%), with very few patients on other types of wards. Similarly, 64.1% of patients spent the longest period of their admission under the care of a general medicine physician, while 26.2% spent the longest period under a geriatrician and 7.9% under the care of a surgeon. The median duration of stay was 10 days (IQR 6–20 days). Only 49.5% admitted from home were discharged directly home, and 24.7% were newly-admitted directly to residential care (the remainder went to rehabilitation, another hospital, convalescence etc.).
3.2 Prescription of Psychotropic Medication
Overall, 83% of patients were receiving at least one of the following psychotropic medications on hospital admission: antipsychotic medications, benzodiazepines, hypnotics or sedatives including Z-drugs, benzodiazepine-derivatives, melatonin, acetylcholinesterase inhibitors, memantine, antidepressant medications, or anticonvulsant medication. Excluding acetylcholinesterase inhibitors and memantine, 53.5% were receiving at least one psychotropic medication on admission.
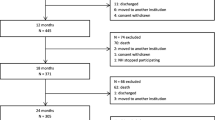
Almost 40% of the 893 cases were prescribed a new psychotropic medication or an increased dose of existing psychotropic medication (shortened to ‘new or extra’ medication) during the admission. Figure 1 provides detailed additional information in this regard.
The NCG recommends that before prescribing any psychotropic medication to a person with dementia, a trained healthcare professional must perform a comprehensive assessment (for an example, please see http://dementiapathways.ie/permacache/fdd/cf3/7a6/4c1450ec131d1454675ada815fe8d8e5.pdf).
At this baseline timepoint, 57% (174/304) had a comprehensive assessment documented for all new or extra prescriptions. This was more commonly performed in people admitted from home (66%) than in those admitted from residential care (51%) (χ2 = 4.09, p = 0.03; n = 285). A comprehensive assessment was more often performed in people on geriatric medicine wards (74%) than in those on other wards (56%) (χ2 = 4.64, p = 0.03; n = 289). The location of the hospital did not affect this practice, with 55% of patients in city-based hospitals (i.e., located in settlements with populations >50,000, according to the Irish Census 2016 [www.cso.ie]) and 59% of patients in other hospitals having a comprehensive assessment documented prior to psychotropic prescribing (χ2 = 1.23, p = 0.27; n = 295).
The NCG also recommends that non-pharmacological interventions should be used primarily to treat a person’s NCSD unless there is risk of harm to the person, or to others, or the person is in severe distress. Only 15.5% of cases prescribed psychotropics for NCSD (11/71 in bigger dataset) had documentation that non-pharmacological interventions were initiated before medication changes, the most common being the use of ‘enhanced care’ (i.e., one-to-one supervision). Where non-pharmacological interventions were not tried first (n = 60), only 14 healthcare records had a reason documented for this approach, and only 10 of these were a valid indication (i.e., risk of harm or severe distress).
3.3 Antipsychotic Medication
In total, 39.3% of patients were receiving antipsychotic medication on admission, which was higher in people admitted from residential care (55%) than in those admitted from home (30.4%) (χ2 = 43.93, p < 0.001; n = 834). Of note, these proportions were increased since the first audit round in 2013 (n = 660 healthcare records; 46% in the cohort from residential care; 19% from home) [26].
A new or increased-dose antipsychotic was prescribed to 25% of patients during their admission. Almost half (48%) of these were already receiving antipsychotic medication on admission, while 52% received a new prescription. In total, 52.2% of PwD were prescribed antipsychotic medication at some point during the admission.
The NCG recommends that the antipsychotic medications must be used with caution and only where there is risk of harm to the person themselves or others, and there is agitation, psychosis or aggression involved. Amongst the 223 patients who had been prescribed a new or increased dose of antipsychotic medication during their admission, there was an explicit indication documented in 79.5% of cases (Table 1).
If we assume that where the indication for the new or increased-dose antipsychotic medication was not documented, this was not for NCSD, then 105/893 (11.8%) of the sample received new or extra antipsychotic medication for NCSD. If we assume that where the indication was not documented, this was for NCSD, then 157/893 (17.6%) of the sample received new or extra antipsychotic medication for NCSD. Thus, between 11.8 and 17.6% of the total cohort received a new or increased-dose antipsychotic medication for NCSD. Of note, there were similar rates of antipsychotic prescribing for NCSD between patients cared for within geriatric medicine wards and patients on other wards (χ2 = 1.07, p = 0.30; n = 812).
The cases prescribed an antipsychotic for end-of-life care (n = 35) or a ‘stat’ dose for a medical procedure (n = 4) are not included in any further analysis below, as the risk, benefit, and context for these scenarios is very different to prescribing for NCSD. Within the 105 cases where the antipsychotic was prescribed for NCSD (i.e., also excluding patients with delirium), there was documented severe distress, or an identifiable risk of harm to the PwD and/or others in almost 71% of cases. Amongst this cohort, the documented indications included severe distress in 71%, risk of harm to the person (47%), and risk of harm to others in almost 32% of cases.
The NCG also recommends that a complete discussion must take place with the relevant person or their ‘decision supporter’ about the risks and benefits of using antipsychotic medication, including a higher risk of death, transient ischemic attack, and stroke. At this baseline period, the risks and benefits were documented in the healthcare records in < 10% of cases where new or extra medication was prescribed for NCSD and there was documentation of discussion with the person and/or their family or relevant decision maker in only 4% of cases.
Where an antipsychotic medication is required, it should be used at the minimum possible dose and increased slowly to the minimum effective dose. The initial antipsychotic dose was at, or close to, the lowest available dose in 91% of cases. The audit also attempted to determine the appropriateness of any subsequent increases in dosage, but this audit item had a Kappa value of 0.08 (p = 0.5), with only 42% agreement across raters, so these results are not presented.
The NCG stated that “Atypical (second generation) antipsychotic medications are associated with fewer extrapyramidal effects and risks than typical (first generation) antipsychotics, and therefore second generation medication should be used if antipsychotic therapy is necessary for the management of non-cognitive symptoms”. Where documented by the auditor, a second-generation antipsychotic was chosen in 71.6% of cases (131/183). Where a first-generation drug was prescribed (n = 52), there was no documented reason for choosing first generation.
Where new or extra antipsychotic medication was prescribed, there was a documented review for both effectiveness and side effects during the admission in just 2.3% of cases; there was a documented review for effectiveness only in 33.5% and a review for side effects only in 1.7% of cases. Of note, these findings should be interpreted with some caution as this audit item had only moderate agreement between auditors (Kappa = 0.3, p = 0.02; 68% agreement).
According to the NCG, if the person responds positively to antipsychotic medication, they should be reviewed for a possible reduction in dosage within 3 months, with a discussion with the person and/or their decision supporter. There was documentation that the antipsychotic was effective in almost one-third of cases. However, there was evidence of a planned review date within 3 months of the first prescription in only approximately 10% of cases. In addition, documentation suggested that the antipsychotic was ineffective in a small proportion of cases (7.5%). It was tapered down in two of these cases and stopped in two others (i.e., eight cases appeared to have continued with the medication despite lack of effect). However, this audit item had only moderate inter-rater agreement, so results should be interpreted with caution (Kappa = 0.4, p = 0.39; 66.7% agreement).
3.4 Benzodiazepine Medication
According to the NCG, benzodiazepines should be generally avoided for the treatment of NCSD and their use limited strictly to the short-term management of severe episodes of anxiety, because of the high risk of dependency.
Approximately 12.1% of the sample were receiving benzodiazepine medication on admission (noting this figure excludes nocturnal benzodiazepines, which are discussed with hypnotics in the next section). This figure was almost 22% for those admitted from residential care, compared with 7.7% of those admitted from home (χ2 = 31.47, p < 0.001; n = 799). In total, 11.2% of patients receiving benzodiazepine medication on admission were receiving more than one benzodiazepine.
Approximately one sixth of patients were prescribed new or extra benzodiazepines during the admission, with the majority of these, 81.7%, being a new prescription. Eight patients were prescribed more than one new and/or increased-dose benzodiazepine during the admission (seven were prescribed two; one was prescribed three). There was a documented indication for the prescription in 81.6% of cases, as per Table 2. Assuming that all other prescriptions were not for NCSD or anxiety, 40/893 PwD (4.5%) would have been prescribed a benzodiazepine for NCSD or anxiety. Conversely, if all other prescriptions were for NCSD or anxiety, then 69/893 (7.7%) would have been prescribed a benzodiazepine for NCSD or anxiety. Thus, 4.5–7.7% received a prescription in hospital for a benzodiazepine for anxiety or other NCSD.
Where the benzodiazepine was prescribed for severe anxiety (n = 19), excluding single doses, there was a maximum duration of treatment documented in 67% of cases. Where the benzodiazepine was prescribed for NCSD (n = 21), there was a justification of why it was chosen over other medication (given that the NCG says they “should be generally avoided for the treatment of NCSD”) in a quarter of cases. There was documentation that the risks and benefits were discussed with the person and/or their family or decision supporter in only one case.
3.5 Hypnotics, Z-Type Medication and Melatonin
The NCG recommends that Z-type medications should only be prescribed for a few days, and a maximum of 2 weeks, for insomnia. It also suggests that melatonin should not be prescribed to PwD and sleep disorders; instead, a personalised sleep regimen should be initiated.
In total, approximately 13% of the PwD were receiving a Z-type medication or a night-time benzodiazepine on admission. This rose to almost 19% in those admitted from residential care compared with 10.4% admitted from home (χ2 = 9.79, p = 0.002; n = 819).
Approximately 1 in 20 patients (5.3%) were prescribed a new or increased dose z-type medication or a night-time benzodiazepine during the admission. Excluding cases where the auditor felt this was not applicable (n = 3; prescribed for end-of-life care), a sleep regimen/care plan had been documented as trialled prior to trial of the medication in only 5% of cases. Only three patients were newly prescribed melatonin during their admission, while one was prescribed an increased dose.
3.6 Acetylcholinesterase (AChE) Inhibitors and Memantine
Just over one quarter of patients (27.5%) were receiving an acetylcholinesterase (AChE) inhibitor on admission, even though they are indicated for cognitive impairment in AD and in Lewy body dementias (LBD) of any severity. According to the National Institute for Health and Care Excellence (NICE) guideline (2018) [21], this should not be stopped unless life expectancy is limited. However, only 33% of the patients with AD or a LBD were receiving this medication on admission. In addition, 23 patients had an AChE inhibitor newly prescribed during their admission. Where this occurred, it was primarily prescribed for cognitive dysfunction (96%) rather than for NCSD.
As per the NICE guideline (2018) [21], memantine monotherapy is an option in moderate AD where AChE inhibitors are not well tolerated, or in severe AD. For people with AD who are already taking an AChE inhibitor, it is recommended to consider adding memantine in moderate disease and to offer memantine in severe disease. Currently, memantine has a limited prescribing licence in Ireland (for moderate or severe AD only) and hence the type and severity of dementia must be known in order to make a prescribing decision. In the audit, 35% of patients were receiving memantine on admission. Additionally, 16 patients were newly prescribed this during admission. In these 16 patients, the type of dementia was recorded in only seven records and the severity of dementia was recorded in 7/11 records (missing this data in five cases). The indication was documented in 11 cases, and here it was more commonly used for cognitive symptoms (n = 7) than for NCSD (n = 4). In the four cases where the memantine was newly prescribed for NCSD, there was no documentation that the risks and benefits were discussed with the PwD and/or decision supporter in any case.
3.7 Antidepressant Medication
The NCG suggests that antidepressants may be prescribed to treat PwD who have moderate to severe depressive episodes or people with moderate depression who have not responded to psychological treatment.
In total, 38% of patients were receiving antidepressant medication on admission, and 17% of these were receiving more than one antidepressant. Overall, 43% of those admitted from residential care were receiving antidepressant medication, versus 34% of those admitted from home (χ2 = 5.45, p = 0.02; n = 819). A new or increased dose of antidepressant medication was prescribed to only 4% of the sample. The indication was specified in almost 72% of cases, as listed in Table 3.
There was documentation that the risks and benefits had been discussed with the person living with dementia and/or their decision supporter in only 14% of cases. This question had poor inter-rater reliability due to small numbers, but good percentage agreement between auditors (Kappa = 0.36, p = 0.28; 78% agreement).
3.8 Anticonvulsant Medication
Within the NCG, anticonvulsant medication is not recommended as a treatment for NCSD, but only for the treatment of seizures, pain, or bipolar disorder.
In total, 9% of patients were receiving anticonvulsant medication on admission. This was most common in people admitted from residential care (14.3%), while 6.6% of those admitted from home were receiving anticonvulsant medication on admission (χ2 = 11.79, p < 0.001; n = 819). Within the cohort receiving anticonvulsant medication on admission, 12.5% were receiving more than one anticonvulsant.
In total, only 3% of patients were prescribed a new or increased-dose anticonvulsant during the admission, with 21/27 of these being a new prescription in a patient not previously prescribed an anticonvulsant. The majority of prescriptions for the new/increased doses of anticonvulsants during the admission were for seizures (76%) or pain (8%), and further analysis was not conducted.
4 Discussion
This study provides valuable insights into the extensive use of psychotropic medications among PwD at the time of admission to acute hospitals in Ireland. It also provides baseline data on prescribing processes in hospitals before the launch and implementation of a key national clinical guideline (NCG 21) [27]. The study authors [28] had previously reported from a prospective cohort study that psychotropic medication use and polypharmacy was highly prevalent within individuals with dementia, aged 70 years and older, at the time of admission to one of six hospitals in Cork County (Ireland) in 2012/2013. Overall, 56% of the 150 PwD in that study were receiving psychotropic medication on admission, compared with 83% in our audit sample; while the rates of antipsychotic prescribing were 13.6% [27] versus 39% in our sample. This apparent large increase in antipsychotic and psychotropic prescribing rates likely relates in a large part to the case finding methods used, where cases in the audit had well-documented dementia, whereas the prospective study cases were often newly diagnosed as part of the study, and so had a lower likelihood of needing psychotropic medications.
Within our sample, 30% of cases were admitted from residential care, and this population had particularly high rates of prescription of psychotropic medications. This may simply reflect a higher prevalence of NCSD in PwD that require residential care. Equally, the rates of prescribing of antipsychotic medication (55%), benzodiazepines (22%), and antidepressants (43%) are significantly higher than those reported in residential care from other countries. For example, a systematic review of 37 studies on antipsychotic drug use and 27 studies on antidepressant drug use, with a participant mean number of 3109 (SD 13,466), across 12 different European countries, reported that antipsychotic use in nursing homes varied from 12 to 59% and the use of antidepressants from 19 to 68% [28]. In England, antipsychotic use was estimated to be 19% in 31,619 residents across 616 residential care units in a 4-year observational study [30]. In Australia, 41% of 12,157 residents within 150 residential aged care facilities were prescribed antidepressants, 22% antipsychotics, and 22% of residents were taking benzodiazepines at baseline in an interventional study [31]. A point prevalence study in the Netherlands showed that prevalence of antipsychotic prescribing in 1090 PwD (overall 31%) was higher in large urban facilities, with below-average staffing levels and poorer resident rating of personal care and recreational activities [32].
The apparent higher rate of prescription in Ireland may reflect differences in the populations of PwD within residential care between countries, or the population admitted to hospital may be atypical of the overall population of PwD in residential care in Ireland. However, it does appear that prescribing rates may be particularly high in Ireland. It is known that there are many drivers of psychotropic prescribing in residential care where overall staff training, numbers, and culture are important, and many staff (nurses requesting medications for a resident and the general practitioner (GP)/geriatrician/psychiatrist prescribing it) are simply not aware of the risks and lack of benefit of the medications in question [32, 34]. The prescribing of psychotropic medication should be explored further within a specific audit of residential care, where the prescribing rates can be confirmed in the overall population (not just those hospitalised) and where the documented indications and prescribing processes can be explored.
There was an 11% higher rate of prescription of antipsychotic medication (52% vs 41%) during an admission to hospital as compared with 2013 but this was fully explained by an increased prevalence of prescribing in the community, that is, on admission (39% vs 29%), and by increased prescribing for end-of-life care and delirium within the hospitals. The rate of prescription of new/extra antipsychotic medication for NCSD in hospital (estimated range 11.8–17.6%) was lower than in 2013 (estimated range 18.3–20.4%) [35].
It is known that it is possible to reduce the prescribing of antipsychotic medications through national initiatives. In the United States of America (USA), there was a 33% reduction in antipsychotic prescribing in residential care from 2012 to 2017, from 24 to 16% [36], reported to be due to a multifaceted strategy of stakeholder engagement and education, public reporting of prescribing metrics, and enhanced reinforcement of regulations [36]. In the UK, a considerable reduction in overall prescription of antipsychotic drugs in dementia from 22.1% in 2005 to 11.4% by 2015 was recorded [37]. Within acute hospitals in the UK, there was a 33% reduction in the use of new/extra antipsychotic medications for hospitalised PwD in England and Wales between audit rounds in 2008 to 2011 [24]. In addition to public reporting of prescribing metrics, financial penalties/rewards might encourage reduced prescribing in acute hospitals, although it is important to focus on appropriate prescribing rather than absolute prescribing rates [20]. Most research on this topic focuses on residential care settings. A recent review of reviews by our group synthesised the evidence for interventions, in residential care settings, to improve the appropriateness of psychotropic prescribing [38]. Education-only interventions and clinical protocols alone have uncertain evidence, while multicomponent interventions that include education, specialist outreach, audit and feedback, and psychosocial interventions for the residents have good evidence (with reported reductions in prescribing between 5 and 66% in one systematic review), as do medication review and efforts to improve the culture of the site [38]. Within acute hospitals, computerised interventions (electronic prescribing system alerts, clinical decision support, etc.) show some benefit for reduced inappropriate prescribing of medications, including psychotropics (not specifically for people with dementia), although the need for attitude and culture change to accept these recommendations is also evident in these studies [39].
Within our sample, almost 16% (144/893) were prescribed a new or increased dose of benzodiazepine during admission. Although most of these were for a medical indication, we estimated that between 4.4 and 7.7% of our sample were prescribed a benzodiazepine for NCSD. The NCG 21 was not yet published during the time period of this audit, but the Irish Health Service Executive (HSE) had published guidance the previous year on prescribing benzodiazepines and Z-drugs appropriately for the treatment of insomnia and anxiety (Medicines Management Programme, 2018 [updated 2021]) [40]. This may have promoted the relatively low rates of prescribing of benzodiazepines for NCSD. Where a benzodiazepine was prescribed for NCSD (n = 21), there was a justification of why a benzodiazepine was chosen over other medication in only 25% of cases, and there was documentation that the risks and benefits of use of a benzodiazepine were discussed with the PwD or their decision supporter in just one case. Similarly, about one in twenty patients were prescribed a new or increased dose of a z-type drug or a night-time benzodiazepine. Only 5% of these had documentation that non-pharmacological approaches had been trialled first. Thus, the practice change required here relates more to prescribing practices than rates.
Our study suggests that AChE inhibitors may be underused within Ireland for treating cognitive symptoms in PwD, despite there being a relevant NICE guideline published since 2018, and the accumulating evidence that they are associated with reduced mortality in dementia [41]. In particular, only one third of the patients with AD or a LBD were prescribed AChE inhibitors on admission, despite their indication for these dementia types in all dementia stages. However, this finding should be studied further in non-hospitalised cohorts because contra-indications, intolerance, and discontinuations due to limited life expectancy may be higher in a hospitalised cohort than the general population of PwD. In the audit cohort, there was insufficient documentation regarding the severity and type of dementia to be able to judge memantine prescription appropriateness (as this has a more limited indication). In general, there was insufficient evidence of discussion about the risk and benefit of AChE inhibitors and memantine with the person concerned and their decision supporter, when these were newly prescribed in hospital.
4.1 Strengths and Limitations
This audit included data from 30/34 acute hospitals in Ireland and included 893 healthcare records overall. The data collectors received specific training in the data collection, and there was a detailed accompanying manual, while the inclusion of a 20% dual-auditor sample allowed us to identify audit items with poorer inter-rater reliability (these are included in this paper but with an attached caution). However, the sample size for individual psychotropic medication classes was smaller, especially when prescriptions for medical indications were excluded, and where practice around prescribing in certain situations was of interest. In addition, the audit tool focussed on medication classes rather than sub-classes (e.g., tricyclic antidepressants were not differentiated from other types, or long-acting from short-acting benzodiazepines). This reflected the focus of the NCG on medication classes rather than individual medications, but future audits could include exact medication names to allow further depth of analysis.
Like all audit data, the limitations of analysing retrospectively collected data that has been entered in the healthcare record by busy clinical staff must be acknowledged, where documentation may under-represent actual practice. All included cases were in hospital for at least 72 hours, so the data does not represent practice for cases with shorter hospital stays.
Of note, the audit period pre-dated the COVID-19 pandemic, which caused further challenges in the care of PwD and in communicating care decisions with the person and their families during hospital and community outbreaks.
5 Conclusion
The audit reported in this paper was designed to provide baseline data on psychotropic medication prescription in acute hospitals in Ireland before the implementation of a national clinical guideline, in order to facilitate the implementation and the evaluation of implementation of the NCG. However, it does demonstrate that most PwD admitted to hospital are in receipt of psychotropic medications on admission, and that many are prescribed new or extra psychotropic medication in hospital, often without documentation of a risk/benefit analysis, trials of non-pharmacological interventions, discussion with the person and their family, or careful review for efficacy and side effects. This demonstrates the need for practice change via a fully implemented NCG and a quality improvement focus on this topic within acute hospitals.
References
Kwon CY, Lee B. Prevalence of behavioral and psychological symptoms of dementia in community-dwelling dementia patients: a systematic review. Front Psychiatry. 2021;12:1809.
Cerejeira J, Lagarto L, Mukaetova-Ladinska EB. Behavioral and psychological symptoms of dementia. Front Neurol. 2012;3:73.
Huang SS, Wang WF, Liao YC. Severity and prevalence of behavioral and psychological symptoms among patients of different dementia stages in Taiwan. Arch Clin Psychiatry (São Paulo). 2017;44:89–93.
Keltner NL, Folks DG. Psychotropic drugs. Maryland: Mosby; 1993.
Maust DT, Kim HM, Seyfried LS, Chiang C, Kavanagh J, Schneider LS, et al. Antipsychotics, other psychotropics, and the risk of death in patients with dementia: number needed to harm. JAMA Psychiat. 2015;72:438–45.
Briggs R, Dyer A, Nabeel S, Collins R, Doherty J, Coughlan T, et al. Dementia in the acute hospital: the prevalence and clinical outcomes of acutely unwell patients with dementia. QJM. 2017;110:33–7.
Bjerre LM, Farrell B, Hogel M, Graham L, Lemay G, McCarthy L, et al. Deprescribing antipsychotics for behavioural and psychological symptoms of dementia and insomnia: evidence-based clinical practice guideline. Can Fam Physician. 2018;64:17–27.
Pink J, O’Brien J, Robinson L, Longson D. Dementia: assessment, management and support: summary of updated NICE guidance. BMJ. 2018;361:k2438.
El-Saifi N, Moyle W, Jones C, Tuffaha H. Quetiapine safety in older adults: a systematic literature review. J Clin Pharm Ther. 2016;41:7–18.
Henry G, Williamson D, Tampi RR. Efficacy and tolerability of antidepressants in the treatment of behavioral and psychological symptoms of dementia, a literature review of evidence. Am J Alzheimers Dis Other Demen®. 2011;26:169–83.
Banerjee S. The use of antipsychotic medication for people with dementia: time for action. London: Department of Health; 2009.
Nørgaard A, Jensen-Dahm C, Wimberley T, Svendsen JH, Ishtiak-Ahmed K, Laursen TM, et al. Effect of antipsychotics on mortality risk in patients with dementia with and without comorbidities. J Am Geriatr Soc. 2022;70:1169–79.
Tampi RR, Tampi DJ, Balachandran S, Srinivasan S. Antipsychotic use in dementia: a systematic review of benefits and risks from meta-analyses. Ther Adv Chronic Dis. 2016;7:229–45.
US Food and Drug Administration. Deaths with antipsychotics in elderly patients with behavioral disturbances. Washington: Public Health Advisory. US Department of Health and Human Services; 2005.
Newcomer JW. Antipsychotic medications: metabolic and cardiovascular risk. J Clin Psychiatry. 2007;64:8–13.
Schneider LS, Dagerman KS, Insel P. Risk of death with atypical antipsychotic drug treatment for dementia: meta-analysis of randomized placebo-controlled trials. JAMA. 2005;294:1934–43.
Saarelainen L, Tolppanen AM, Koponen M, Tanskanen A, Sund R, Tiihonen J, et al. Risk of hip fracture in benzodiazepine users with and without Alzheimer disease. J Am Med Dir Assoc. 2017;18:87-e15.
Jennum P, Baandrup L, Ibsen R, Kjellberg J. Increased all-cause mortality with use of psychotropic medication in dementia patients and controls: a population-based register study. Eur Neuropsychopharmacol. 2015;25(11):1906–13.
Wetzels RB, Zuidema SU, de Jonghe JFM, Verhey FRJ, Koopmans RTCM. Determinants of quality of life in nursing home residents with dementia. Dement Geriatr Cogn Disord. 2010;29:189–97.
Department of Health. Appropriate prescribing of psychotropic medication for non-cognitive symptoms in people with dementia. Dublin: Department of Health; 2019.
National Institute for Health and Care Excellence. Dementia: assessment, management and support for people living with dementia and their carers. 20 June 2018. www.nice.org.uk/guidance/ng97. Accessed 25 Aug 2022.
National Health and Medical Research Council. Clinical practice guidelines and principles of care for people with dementia. Sydney: Guideline Adaptation Committee; 2016.
Reus VI, Fochtmann LJ, Eyler AE, Hilty DM, Horvitz-Lennon M, Jibson MD, et al. The American Psychiatric Association practice guideline on the use of antipsychotics to treat agitation or psychosis in patients with dementia. Am J Psychiatry. 2016;173(5):543–6.
Royal College of Psychiatrists. Report of the National Audit of Dementia Care in General Hospitals. Editors: Young J, Hood C, Woolley R, Gandesha A, Souza R. London: Healthcare Quality Improvement Partnership. 2011.
Scally M, Timmons S, O’Shea E, Gallagher P, Kennelly SP, Hamilton V, O’Neill D. Second Irish National Audit of Dementia Care in Acute Hospitals. Offaly: National Dementia Office; 2020.
de Siún A, O’Shea E, Timmons S, McArdle D, Gibbons P, O’Neill D, et al. Irish National Audit of Dementia Care in Acute Hospitals. Cork: National Audit of Dementia Care; 2014.
Department of Health. Appropriate prescribing of psychotropic medication for non-cognitive symptoms in people with dementia (NCEC National Clinical Guideline No. 21). 2019. https://www.gov.ie/en/collection/c9fa9a-national-clinical-guidelines/. Accessed 25 Aug 2022.
Walsh KA, O’Regan NA, Byrne S, Browne J, Meagher DJ, Timmons S. Patterns of psychotropic prescribing and polypharmacy in older hospitalized patients in Ireland: the influence of dementia on prescribing. Int Psychogeriatr. 2016;28:1807–20.
Janus SIM, van Manen JG, Ijzerman MJ, Zuidema SU. Psychotropic drug prescriptions in Western European nursing homes. Int Psychogeriatr. 2016;28:1775–90.
Szczepura A, Wild D, Khan AJ, Owen DW, Palmer T, Muhammad T, et al. Antipsychotic prescribing in care homes before and after launch of a national dementia strategy: an observational study in English institutions over a 4-year period. BMJ Open. 2016;6: e009882.
Westbury JL, Gee P, Ling T, Brown DT, Franks KH, Bindoff I, et al. RedUSe: reducing antipsychotic and benzodiazepine prescribing in residential aged care facilities. Med J Aust. 2018;208(9):398–403.
Kleijer BC, van Marum RJ, Frijters DHM, Jansen PAF, Ribbe MW, Egberts ACG, et al. Variability between nursing homes in prevalence of antipsychotic use in patients with dementia. Int Psychogeriatr. 2014;26:363–71.
Walsh KA, Dennehy R, Sinnott C, Browne J, Byrne S, McSharry J, Coughlan E, Timmons S. Influences on decision-making regarding antipsychotic prescribing in nursing home residents with dementia: a systematic review and synthesis of qualitative evidence. J Am Med Dir Assoc. 2017;18(10):897.e1-897.e12. https://doi.org/10.1016/j.jamda.2017.06.032.
Walsh KA, Sinnott C, Fleming A, Mc Sharry J, Byrne S, Browne J, Timmons S. Exploring antipsychotic prescribing behaviors for nursing home residents with dementia: a qualitative study. J Am Med Dir Assoc. 2018;19(11):948-958.e12. https://doi.org/10.1016/j.jamda.2018.07.004.
Gallagher P, Curtin D, De Siún A, O’Shea E, Kennelly S, O’Neill D, et al. Antipsychotic prescription amongst hospitalized patients with dementia. QJM. 2016;109:589–93.
Gurwitz JH, Bonner A, Berwick DM. Reducing excessive use of antipsychotic agents in nursing homes. JAMA. 2017;318(2):118–9.
Donegan K, Fox N, Black N, Livingston G, Banerjee S, Burns A. Trends in diagnosis and treatment for people with dementia in the UK from 2005 to 2015: a longitudinal retrospective cohort study. Lancet Public Health. 2017;2:e149–56.
Wiggin DA, Timmons S, Rukundo A, Walsh KA. Improving the appropriateness of psychotropic prescribing for nursing home residents with dementia: an overview of reviews. Aging Ment Health. 2022;26(6):1087–94. https://doi.org/10.1080/13607863.2021.1922601.
Dalton K, O’Brien G, O’Mahony D, Byrne S. Computerised interventions designed to reduce potentially inappropriate prescribing in hospitalised older adults: a systematic review and meta-analysis. Age Ageing. 2018;47(5):670–8. https://doi.org/10.1093/ageing/afy086.
Health Service Executive. Guidance on appropriate prescribing of benzodiazepines and z-drugs (BZRA) for the treatment of anxiety and insomnia. Dublin: Health Service Executive. 2021. https://www.hse.ie/eng/about/who/cspd/ncps/medicines-management/. Accessed 25 Aug 2022.
Mueller C, Perera G, Hayes RD, Shetty H, Stewart R. Associations of acetylcholinesterase inhibitor treatment with reduced mortality in Alzheimer’s disease: a retrospective survival analysis. Age Ageing. 2018;47:88–94.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Open Access funding provided by the IReL Consortium. This article reports on an analysis of a pooled anonymous dataset from the second round of the Irish National Audit of Dementia Care (INAD-2) audit, which was funded within the Irish National Health Service, by the Health Service Executive (HSE). There was no funding for this secondary data analysis.
Conflicts of interest/competing interests
All authors declare that they have no financial conflicts of interest. ST had been the Clinical Lead and Emer Begley the Acting General Manager for the HSE National Dementia Office at the time of the audit; Mairead Bracken-Scally was the National Audit Coordinator; Vida Hamilton was the National Clinical and Group Lead for acute hospitals in Ireland at the time of the audit.
Ethics approval
Ethical approval was granted by the Clinical Research Ethics Committee (CREC) of the Cork Teaching Hospitals, University College Cork (UCC).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Source data is not available from the authors for sharing, as the primary data was collected for quality improvement purposes by the Health Service Executive and permission for sharing does not lie with the authors of this paper. The National Dementia Office can be contacted at dementia.office@hse.ie. The audit tool detailed in this study that is presented in the manuscript as Online Resource 1 is available at: https://www.ucc.ie/en/media/research/irishnationalauditofdementia/INAD-2CaseNoteAuditfinalwithpsychotropicmedicationsection.pdf.
Code availability
Not applicable.
Author contributions
All authors—ST, MB-S, SC, PG, VH, EB, and EOS—participated in the writing and revision of this manuscript. ST, VH, EB, and PG had the idea for the article and had collected the primary dataset; ST, MB-S, SC, and EOS contributed to data analysis; MB-S wrote a preliminary partial draft and SC/ST wrote the first full draft; all authors read and contributed to draft refinements. All authors have read and approve the final submitted manuscript, and agree to be accountable for the work.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Timmons, S., Bracken-Scally, M., Chakraborty, S. et al. Psychotropic Medication Prescribing to Patients with Dementia Admitted to Acute Hospitals in Ireland. Drugs Aging 40, 461–472 (2023). https://doi.org/10.1007/s40266-023-01020-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-023-01020-w