Abstract
Background
Prescription sleep medications are often utilized to manage sleep problems among older adults even though these drugs are associated with multiple risks.
Objective
The aim was to determine the prevalence and characteristics of new-onset compared to chronic sleep medication users and to examine factors associated with the conversion from new to chronic use. A secondary objective was to investigate the impact of sleep medications on health outcomes of injurious falls and patterns of healthcare utilization and expenditures.
Methods
A 25% random sample of adults ≥ 65 years with 3-year continuous AARP® Medicare Supplement medical and AARP® MedicareRx drug plan enrollment was utilized to identify new-onset and chronic sleep medication users. Prescription sleep medication drugs were defined using National Drug Codes (NDCs); falls or hip fractures were identified from diagnosis codes. New users had no sleep medication use in 2014, but initiated medication use in 2015; chronic users had at least one sleep medication prescription in 2014 and in 2015; both groups had follow-up through 2016. Characteristics associated with new users, new users who converted to chronic use, and chronic users were determined using multivariate logistic regression. Prevalence of falls, healthcare utilization and expenditures were regression adjusted.
Results
Among eligible insureds, 3 and 9% were identified as new-onset and chronic sleep medication users, respectively. New-onset sleep medication prescriptions were often associated with an inpatient hospitalization. The strongest characteristics associated with new users, those who converted to chronic use, and chronic users were sleep disorders, depression and opioid use. About 50% of new users had > 30 days’ supply; 25% converted to chronic use with ≥ 90 days’ supply. The prevalence of falls for new-onset users increased by 70% compared to a 22% increase among chronic users.
Conclusion
New-onset and chronic sleep medication users were characterized by sleep disorders, depression and pain. Addressing the underlying problems associated with sleep problems among older adults may decrease the need for sleep medications and thus reduce the risk of sleep medication-related adverse events.
Similar content being viewed by others
This study was designed to identify new-onset and chronic sleep medication users, establish characteristics of users and explore evidence of risks associated with the utilization of prescription sleep medications among older adults. |
New-onset and chronic sleep medication users were identified from pharmaceutical records. Users were characterized by sleep disorders, depression and pain and were at increased risk for falls, emergency room visits and inpatient admissions. |
This study highlights the importance of assessing underlying conditions associated with sleep problems among older adults. Addressing these issues may lead to a decrease in use of prescription sleep medications and thus reduce the risk of sleep medication-related adverse events. |
1 Introduction
Sleep problems are common among older adults, with about half reporting less than optimal sleep, including difficulty falling asleep, frequent awakenings, early waking or waking not rested [1,2,3,4,5,6,7,8,9]. The consequences of poor sleep include difficulty sustaining attention, slowed response times, difficulty with memory and decreased physical performance [10]. Poor sleep among older adults is also associated with poor mental and physical health outcomes [2, 3, 11]. Longer-term, poor sleep is associated with an increased risk of disabilities, including activities of daily living (ADLs) and mobility limitations [3, 8].
Prescription sleep medications are often utilized as a first-line treatment option to manage the sleep problems of older adults even though these drugs are associated with multiple risks and are generally discouraged [11,12,13,14,15,16,17]. Consistently, about 10–14% of older adults use prescription sleep medications [1, 3, 11, 18, 19]. Safety concerns have been raised as some sleep medications are considered inappropriate for use among older adults, either by drug category or dosages that exceed levels recommended for them [14,15,16,17]. Risk factors associated with sleep medication use among older adults include increased prevalence of falls and cognitive problems (e.g., memory loss, disorientation, drowsiness) [6, 9, 10, 13, 14, 16, 20,21,22]. Characteristics associated with sleep medication use include being older, female, and white, having a higher income, but also being in poorer health and suffering from more depression, disabilities and sleep problems [4, 11, 12, 18, 20, 23].
Poor sleep is often associated with common chronic medical conditions, including cardiovascular diseases, hypertension, respiratory problems [e.g., chronic obstructive pulmonary disease (COPD), asthma], diabetes, musculoskeletal diseases, psychiatric disorders (e.g., depression, anxiety), chronic pain and disability [3, 7, 8, 10, 24,25,26]. In addition, as the number of chronic conditions increases, the likelihood of sleep difficulties also increases [10]. Of these conditions, depression and comorbid chronic pain are perhaps most consistently associated with poor sleep [3, 7, 8, 10, 24,25,26,27,28,29,30,31]. Studies focused on associations between depression, pain and sleep problems would recommend that depression and pain management programs incorporate sleep management strategies [23, 28, 29].
Poor sleep quality exacerbated by side effects associated with sleep medications, including drowsiness, fatigue, impaired cognitive and motor function as well as postural sway, is associated with an increased risk of falls [6, 9, 20]. While non-benzodiazepine hypnotics were introduced as incurring lower levels of falls risk, subsequent research has not substantiated those claims [20, 21]. The risk for falls and/or fractures associated with non-benzodiazepine hypnotics was demonstrated to be as high or higher than the more common benzodiazepines they were designed to replace [20, 21]. Risk of falls is most notable with drug initiation and tends to dissipate over time for chronic users [4, 16, 21].
Sleep problems exacerbate common chronic conditions such as cardiovascular disease and respiratory problems and contribute to increased healthcare cost profiles [25]. Poor sleep may be a marker for poor health and is associated with more chronic conditions, polypharmacy, increased hospitalizations and emergency room use [2, 26]. Furthermore, the common practice of in-hospital use of sleep medications may be a risk factor for post-hospitalization medication use [32].
Only one research study to date was found focusing on predictors of new-onset sleep medication use, and none were found that included information on conversion rates from new to chronic use among older adults with Medicare Supplement plans (i.e., Medigap) [33]. In the US, government-funded Medicare covers adults aged 65 years and older as well as those under 65 and disabled. Medicare fee-for-service plans (about 70% of all Medicare plans) pay about 80% of medical expenditures for these individuals, but offer no prescription drug benefits. Those enrolled in these Medicare plans are personally responsible for obtaining additional insurance plans to cover the remaining 20% of medical expenses (i.e., Medicare Supplement or Medigap plans) and prescription drug coverage (Medicare Part D plans). While most (about 90%) of those with original fee-for-service Medicare coverage have some type of supplemental insurance coverage, about 28% (currently about 10.2 million adults) have purchased Medigap coverage [33]. Of these coverage plans, AARP® Medicare Supplement plans constitute about 40% of the US market share. Since this population may differ from general older adult and/or specifically overall Medicare populations, it was of interest to determine the prevalence of and characteristics associated with new-onset and chronic sleep medication use among these older adults. With current recommendations from the Centers for Disease Control and Prevention (CDC) that older adults only use prescription sleep medications for short time periods (less than 14 days) and not in combination with opioid pain medications, those older adults who continue sleep medication use and certainly those using sleep medication in combination with pain medications could be considered at-risk and targeted for intervention programs [34]. This study adds to the research literature on prescription sleep medication use among older adults in utilizing administrative drug claims to detail identification of new-onset and chronic sleep medication users and establish relevant characteristics of new-onset and chronic users as well as new users who converted to chronic use.
Thus, our primary objective was to estimate the prevalence and characteristics of new-onset and chronic prescription sleep medication users, along with an examination of factors associated with the conversion from new to chronic sleep medication use. The secondary objective was to investigate the impact of new-onset and chronic sleep medication use on health outcomes of injurious falls and patterns of healthcare utilization and expenditures. This research was covered under the New England IRB No. 120160532.
2 Methods
2.1 Study Sample
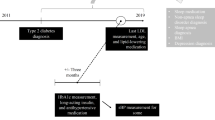
In 2015, approximately 4 million Medicare insureds were covered by an AARP® Medicare Supplement plan insured by UnitedHealthcare Insurance Company. These plans are offered in all 50 states, Washington DC, and various US territories. A 25% random sample of 2015 AARP® Medicare Supplement insureds with AARP® MedicareRx plans (insured through UnitedHealthcare; about 55% of insureds) who were at least 65 years of age with 3-year continuous enrollment, 2014–2016, was utilized to define new-onset and chronic sleep medication users along with a control group of non-users randomly selected in a 1:5 ratio of users to non-users.
2.2 Sleep Medication Use and Diagnosed Sleep Disorders
New-onset and chronic sleep medication users were defined from National Drug Codes (NDCs) for insomnia recommended by the CDC [24]. Sleep medications included benzodiazepines (estazolam, flurazepam, quazepam, temazepam, and triazolam) and non-benzodiazepine hypnotics (chloral hydrate, doxepin, doxylamine, eszopiclone, ramelteon, secobarbital, suvorexant, trazodone, zaleplon, and zolpidem). New-onset users had no sleep medication use in 2014, but initiated medication use at any time in 2015; chronic users had at least one sleep medication prescription in 2014, with continued use of at least one prescription medication in 2015. Both groups had follow-up through 2016. Since this study design was intended to promote an intervention, exact timing of sleep medication index dates relative to health outcomes was not included in these analyses. Identified sleep disorders in 2015 were defined from International Classification of Diseases, Ninth Revision (ICD-9) or ICD-10 diagnoses codes for sleep-related problems recommended by the CDC [24].
2.3 Covariates
Covariates were included to characterize individuals with new-onset and chronic sleep medication use and to adjust for other risk factors. These covariates included measures of demographics, socioeconomic factors, health status, and other characteristics taken from health plan eligibility and administrative medical claims.
Demographic questions included age and gender. Age groups were defined as 64–69, 70–79 and ≥ 80 years. Geographic regions (Northeast, South, Midwest, and West); low, medium, and high minority areas; and low, medium, and high income levels were geocoded from zip codes. AARP® Medicare Supplement plan types were grouped by cost-sharing levels, including high-level coverage plans with minimal copayments or deductibles, less comprehensive medium-level coverage, and all other plans. A measure of health services access was calculated as acute hospital beds per 100,000 capita. Opioid use defined from NDCs was utilized as a surrogate measure for pain.
Inappropriate sleep medications for older adults were identified from Beers Criteria [35]. The Beers Criteria for Potentially Inappropriate Medication Use in Older Adults are guidelines for healthcare professionals to help improve the safety of prescribing medications for older adults by reducing the problems of polypharmacy, drug interactions, and adverse drug reactions.
2.4 Prevalence of Common Chronic Conditions
Six chronic conditions identified using Evidence-Based Medicine (Symmetry EBM Connect®, Version 8.3) software (heart problems, COPD, depression, diabetes, osteoporosis and rheumatoid arthritis) were included in these analyses. This software program was developed to calculate utilization of care patterns from healthcare claims and medications from pharmaceutical claims using a defined set of rules for evidence-based care associated with various chronic conditions.
Dementia was defined from the Charlson Comorbidity Index (CCI) diagnoses codes. The CCI is a measure of the risk of 1-year all-cause mortality attributable to selected comorbidities that has also been shown to be highly predictive of morbidity and healthcare expenditures [36]. While the CCI identifies other chronic conditions similar to those listed above, only dementia from the CCI was included in these analyses.
2.5 Health Outcomes: Injurious Falls/Hip Fractures and Healthcare Utilization and Expenditures
One side effect of the use of sleep medications is an increased risk of falls. Thus, injurious falls requiring medical services or hip fractures, as a combined measure, were defined from suggested Healthcare Effectiveness Data and Information Set (HEDIS) diagnoses codes [37]. Falls or hip fractures were documented from these selected diagnoses codes at any time during each calendar year, 2014, 2015 and 2016.
A second health outcome associated with sleep medication initiation and use is a defined medical event evident in healthcare utilization and subsequent expenditures. Healthcare utilization was defined from administrative medical claims as an inpatient admission or emergency room visit at any time during each calendar year, 2014, 2015 and 2016. Healthcare expenditures were defined as annual paid claims aggregated from Medicare, Medicare Supplement, and patient out-of-pocket paid amounts. Prescription drug expenditures included Medicare Part D plan paid and patient drug copayments.
2.6 Propensity Matching
Propensity score matching was used to minimize casemix differences between sleep medication users and non-users for those analyses focused on health outcomes: falls/hip fractures; healthcare utilization and expenditures. In this set of analyses, a logistic model was used to estimate the likelihood of using a sleep medication. The variables used in the model were those listed in Table 1. The propensity score for each sample individual was defined as each individual’s predicted probability of being a new-onset or chronic sleep medication user. This probability was then used to match respective users to similar non-users. Propensity score matching is a convenient and acceptable way to remove casemix differences when evaluating user and non-user subgroups [38, 39].
2.7 Statistical Models
Characteristics associated with those with new-onset and chronic sleep medication users compared to non-users were determined using logistic regression models. Covariates included those variables listed in Table 1.
Injurious falls/hip fractures, healthcare utilization (inpatient admissions and emergency room visits) and healthcare expenditures (medical and prescription drugs) were determined for each calendar year and regression adjusted for demographic, socioeconomic, and health status variables listed in Table 1. Variables with high correlations (e.g., > 0.5) were dropped from regression models. All analyses were completed using SAS Enterprise Guide Version 6.1 (SAS Institute Inc., Cary, NC, USA).
3 Results
Overall, among the 25% random study sample (N = 446,192), about 12% (N = 51,669) were identified as sleep medication users. Of these, 3% (N = 12,888) were new-onset users and 9% (N = 38,781) were chronic users. Randomly selected non-users constituted the control group (N = 266,828). After study inclusion criteria were applied (i.e., ≥ 65 years; 3-year continuous enrollment), the final study populations included 10,017 new-onset users, 32,986 chronic users and 220,399 non-user controls. New and chronic sleep medication users were mostly female (64%; 67%, respectively), were 70–79 years of age (48%; 50%, respectively), had higher incomes (53%; 54%, respectively) and lived in the South (42%; 43%, respectively). Overall, the most commonly used sleep medications were zolpidem (52%), trazodone (36%), and temazepam (15%). Sleep disorders were evident in 37 and 31% of the new-onset and chronic users, respectively. Over 50% of new and chronic users also were using opioids for pain management. Inappropriate prescription sleep medications were used by 48 and 61% of new and chronic users, respectively (Table 1).
3.1 Characteristics Associated with New-Onset and Chronic Sleep Medication Users
Characteristics associated with new-onset sleep medication users included sleep disorders, opioid use, being younger, being female, higher income, and having chronic conditions of dementia, depression, COPD and heart problems. Chronic users had sleep disorders, had opioid use, were younger, white and female, had higher incomes, were in higher coverage medical plans and had chronic conditions of depression, COPD, heart problems and rheumatoid arthritis (Table 2).
3.2 Health Outcomes: Injurious Falls and Healthcare Utilization and Expenditure Patterns
Injurious falls for new-onset sleep medication users increased from 6% in the year prior to initiation (2014) to 9% in the year of sleep medication initiation (2015) compared to an increase of 5–6% for chronic medication users (Table 3). Likewise, inpatient admissions increased from 17 to 30% for new users compared to a 17–18% increase for chronic users. Consequently, healthcare expenditures for new-onset users increased by US$5689 in 2015 compared to 2014 before returning to near 2014 levels in 2016. Chronic users’ annual healthcare expenditure pattern was relatively stable, decreasing somewhat over the time period. Matched control healthcare expenditures were stable, reflecting minimal changes over the 3-year study period.
3.3 Factors Associated with Conversion of New-Onset to Longer-Term Sleep Medication Use
About 50% of new-onset sleep medication users discontinued use of sleep medication within the recommended 30 days or less; however, the remaining 50% continued use of the medications: 14% for 31–60 days, 11% for 61–90 days and 25% for more than 90 days. Sleep disorders and dementia were consistently associated with all longer-term use of sleep medication. For those using for more than 90 days, depression and opioid use (i.e., pain) were also evident (Table 4).
4 Discussion
In this population of AARP® Medicare Supplement insureds, 3 and 9% were identified as new-onset and chronic sleep medication users in 2015, respectively. As in a previous report, the most commonly used sleep medications were zolpidem and trazodone [18]. About 25% of new users converted to chronic use (> 90 days supply) of these medications. New-onset sleep medication prescriptions were frequently associated with inpatient admissions or emergency room visits, indicating some type of medical event or negative change in health status [11]. Opioid use, although higher than previously reported (50 vs 25% opioid use), was also evident [18]. Thus, sleep medication initiation may be a marker for a seminal medical event for these patients [11].
Characteristics associated with new-onset sleep medication users included being female, being younger (65–69 years), having a higher income, using opioids for pain and having sleep disorders, depression, dementia, COPD and heart problems. Chronic users had a similar profile, but did not include dementia as a condition associated with longer-term use. These results are generally consistent with previous studies, with the exception of age [4, 11, 20, 23]. In our study population, the younger age group of 65–69 years rather than older age groups was more likely to use sleep medications. While most new users did not convert to chronic use, the conversion to chronic use was more strongly associated with sleep disorders, depression, dementia and pain than with specific demographic or socioeconomic characteristics. To date, we have found no other studies that considered conversion rates or characteristics that might be influential in determining chronic use of these medications.
Despite the CDC recommendation [35] that sleep medications and opioids not be used concurrently among older adults, over 50% of new-onset and chronic sleep medication users were also prescribed opioids for pain management. Besides sleep disorders, pain and depression were the strongest predictors of sleep medication use, indicating noteworthy underlying conditions that may contribute to poor sleep [4]. Chronic pain has been estimated to impact as many as 76% of older adults, with almost half suffering from poor sleep associated with back, joint, chest or gastrointestinal pain conditions [5]. Furthermore, as the number of pain conditions increases, so also does the likelihood of poor sleep [5]. The prevalence of diagnosed depression among sleep medication users in this study was almost three times higher than non-users (12–13 vs 5%). Similar to previous results, depression is strongly associated with poor sleep quality and sleep problems [9, 30, 31].
Consumption of inappropriate medications continues to be problematic among older adults. Overall, the prevalence of Beers Criteria medications is estimated among older adults at about 30% [17]. In this study, inappropriate sleep medications were prescribed among 48 and 61% of new and chronic users, respectively. Routine use in hospitals and dosages recommended based on younger adults may contribute to the problem [16, 32].
The use of sleep medication for dementia and/or Alzheimer’s disease is even more controversial than general use of sleep medications among older adults [40]. Sleep disturbances are common among these patients, but use of sleep medications and appropriate dosages are difficult to manage and thus are discouraged [40]. In these results, while dementia was associated with new-onset sleep medication use, the condition was not significantly associated with chronic use. This may imply that dementia caregivers dealing with disruptive night-time behaviors, including wandering and increased night-time wakefulness, attempted short-term use of sleep medications, but then opted to discontinue use.
The prevalence of falls for new-onset sleep medication users increased by 70% from 2014 to 2015 with the initiation of the sleep medication compared to a 22% increase for chronic users or a 28–31% increase for matched control subgroups. These results are in general agreement with other studies that show about 50–100% increases in falls rates associated with initiating selected benzodiazepines or non-benzodiazepine hypnotic drugs [6, 12, 13, 20, 21]. Falls rates associated with sleep medication may be even higher if poor sleep quality can also be documented [6, 9, 22]. Thus, it is not always clear whether the medications per se or the poor sleep exacerbated by the medications is the cause of the falls.
Poor sleep, sleep disorders and sleep duration (i.e., short sleep 1–5 h or long sleep ≥ 10 h) have been associated with increased hospitalizations and emergency room use [2, 11, 25, 26]. In our results, new-onset sleep medication use was associated with significantly increased inpatient admissions, emergency room use and increased healthcare expenditures. About 30% of new-onset users experienced a hospital event; a 75% increase from the year prior to initiation [11]. These data would be in agreement with the suggestion that in-hospital use of sleep medication may be an independent risk factor for subsequent sleep medication use [32]. In addition, the use of opioids also increased with the initiation of sleep medications, implying both sleep and pain management as a result of the medical event (data not shown).
While prescription sleep medications are often used as a first-line treatment for sleep problems, non-pharmacological options are preferred by up to 80% of older adults [11, 19]. These options primarily include cognitive behavioral therapy for insomnia (CBT-I) and general sleep hygiene awareness [10, 13, 14, 23, 24, 29, 41]. To minimize the utilization of sleep medications, clinicians may need to re-evaluate the effectiveness of sleep medications relative to other non-pharmacological strategies [26]. Integration of sleep management into depression and/or pain management programs may be beneficial [29, 41]. De-prescribing programs focused primarily on benzodiazepines used for insomnia have demonstrated some success [42, 43]. These programs suggest that sleep medications may be one class of drugs that older adults are willing to forego given other options that address their sleep problems [4, 42, 43].
This study has some limitations. The study population of AARP® Medicare Supplement insureds may not generalize to all older adults or other Medicare or Medicare Supplement beneficiaries. Prescription sleep medications were defined from NDCs, but we had no measure of over-the-counter non-prescription sleep medication utilization. In addition, we had no measure of sleep quality or information on the reasons for use of prescription sleep medications, which may have enhanced the study results. Strengths of the study include a large study population that identified new-onset and chronic sleep medication users, potentially serving sleep management intervention strategies.
5 Conclusion
Overall, in this population of Medicare Supplement insureds, 3 and 9% were identified as new-onset and chronic sleep medication users, respectively. New-onset sleep medication use was associated with increased falls and often with a medical event. The strongest characteristics associated with new-onset and chronic sleep medication use were sleep disorders, depression, and pain. The findings of our study highlight the importance of assessing underlying disorders in older patients with sleep problems. This may lead to a decrease in prescription sleep medications and, therefore, a reduction in the risk of sleep medication-related adverse events.
References
National Sleep Foundation. 2015 Sleep in America® Poll: Sleep and Pain. https://sleepfoundation.org/sleep-polls-data/sleep-in-america-poll/2015-sleep-and-pain. Accessed 2 Sept 2017.
Kaufmann CN, Canham SL, Mojtabai R, et al. Insomnia and health services utilization in middle-aged and older adults: results from the Health and Retirement Study. J Gerontol A Biol Sci Med Sci. 2013;68:1512–7.
Zimmerman ME, Bigal ME, Katz MJ, Derby CA, Lipton RB. Are sleep onset/maintenance difficulties associated with medical or psychiatric comorbidities in nondemented community-dwelling older adults? J Clin Sleep Med. 2013;9:363–9.
Bourgeois J, Elseviers MM, Van Bortel L, Petrovic M, Vander Stichele RH. Sleep quality of benzodiazepine users in nursing homes: a comparative study with nonusers. Sleep Med. 2013;14:614–21.
Blay SL, Andreoli SB, Gastal FL. Chronic painful physical conditions, disturbed sleep and psychiatric morbidity: results from an elderly survey. Ann Clin Psychiatry. 2007;19:169–74.
Min Y, Nadpara PA, Slattum PW. The association between sleep problems, sleep medication use, and falls in community-dwelling older adults: results from the Health and Retirement Study 2010. J Aging Res. 2016;2016:3685789.
Wu CY, Su TP, Fang CL, Yeh Chang M. Sleep quality among community-dwelling elderly people and its demographic, mental, and physical correlates. J Chin Med Assoc. 2012;75:75–80.
Park M, Buchman AS, Lim AS, Leurgans SE, Bennett DA. Sleep complaints and incident disability in a community-based cohort study of older persons. Am J Geriatr Psychiatry. 2014;22:718–26.
St George RJ, Delbaere K, Williams P, Lord SR. Sleep quality and falls in older people living in self- and assisted-care villages. Gerontology. 2009;55:162–8.
Ancoli-Israel S. Sleep and aging: prevalence of disturbed sleep and treatment considerations in older adults. J Clin Psychiatry. 2005;66(Suppl 9):24–30.
Leggett A, Pepin R, Sonnega A, Assari S. Predictors of new onset sleep medication and treatment utilization among older adults in the United States. J Gerontol A Biol Sci Med Sci. 2016;71:954–60.
Stowell KR, Chang CC, Bilt J, Stoehr GP, Ganguli M. Sustained benzodiazepine use in a community sample of older adults. J Am Geriatr Soc. 2008;56:2285–91.
Glass J, Lanctôt KL, Herrmann N, Sproule BA, Busto UE. Sedative hypnotics in older people with insomnia: meta-analysis of risks and benefits. BMJ. 2005;331(7526):1169 (Epub 2005 Nov 11).
Schroeck JL, Ford J, Conway EL, et al. Review of safety and efficacy of sleep medicines in older adults. Clin Ther. 2016;38:2340–72.
Donoghue J, Lader M. Usage of benzodiazepines: a review. Int J Psychiatry Clin Pract. 2010;14:78–87.
Blachman NL, Leipzig RM, Mazumdar M, Poeran J. High-risk medications in hospitalized elderly adults: are we making it easy to do the wrong thing? J Am Geriatr Soc. 2017;65:603–7.
Davidoff AJ, Miller GE, Sarpong EM, Yang E, Brandt N, Fick DM. Prevalence of potentially inappropriate medication use in older adults using the 2012 Beers criteria. J Am Geriatr Soc. 2015;63:486–500.
Bertisch SM, Herzig SJ, Winkelman JW, Buettner C. National use of prescription medications for insomnia: NHANES 1999–2010. Sleep. 2014;37:343–9.
Venn S, Arber S. Understanding older peoples’ decisions about the use of sleeping medication: issues of control and autonomy. Sociol Health Illn. 2012;34:1215–29.
Diem SJ, Ewing SK, Stone KL, et al. Use of non-benzodiazepine sedative hypnotics and risk of falls in older men. J Gerontol Geriatr Res. 2014;3:158.
Finkle WD, Der JS, Greenland S, et al. Risk of fractures requiring hospitalization after an initial prescription for zolpidem, alprazolam, lorazepam, or diazepam in older adults. J Am Geriatr Soc. 2011;59:1883–90.
Min Y, Kirkwood CK, Mays DP, Slattum PW. The effect of sleep medication use and poor sleep quality on risk of falls in community-dwelling older adults in the US: a prospective cohort study. Drugs Aging. 2016;33:151–8.
Cho HJ, Lavretsky H, Olmstead R, Levin MJ, Oxman MN, Irwin MR. Sleep disturbance and depression recurrence in community-dwelling older adults: a prospective study. Am J Psychiatry. 2008;165:1543–50.
Ford ES, Wheaton AG, Cunningham TJ, Giles WH, Chapman DP, Croft JB. Trends in outpatient visits for insomnia, sleep apnea, and prescriptions for sleep medications among US adults: findings from the National Ambulatory Medical Care survey 1999–2010. Sleep. 2014;37:1283–93.
Tarasiuk A, Greenberg-Dotan S, Simon-Tuval T, Oksenberg A, Reuveni H. The effect of obstructive sleep apnea on morbidity and health care utilization of middle-aged and older adults. J Am Geriatr Soc. 2008;56:247–54.
Choi NG, DiNitto DM, Marti CN, Choi BY. Too little sleep and too much sleep among older adults: associations with self-reported sleep medication use, sleep quality and healthcare utilization. Geriatr Gerontol Int. 2017;17:545–53.
Potvin O, Lorrain D, Belleville G, Grenier S, Préville M. Subjective sleep characteristics associated with anxiety and depression in older adults: a population-based study. Int J Geriatr Psychiatry. 2014;29:1262–70.
Smith MT, Quartana PJ, Okonkwo RM, Nasir A. Mechanisms by which sleep disturbance contributes to osteoarthritis pain: a conceptual model. Curr Pain Headache Rep. 2009;13:447–54.
McCurry SM, Shortreed SM, Von Korff M, et al. Who benefits from CBT for insomnia in primary care? Important patient selection and trial design lessons from longitudinal results of the Lifestyles trial. Sleep. 2014;37:299–308.
Bourgeois J, Elseviers MM, Van Bortel L, Petrovic M, Vander Stichele RH. One-year evolution of sleep quality in older users of benzodiazepines: a longitudinal cohort study in Belgian nursing home residents. Drugs Aging. 2014;31:677–82.
Naughton F, Ashworth P, Skevington SM. Does sleep quality predict pain-related disability in chronic pain patients? The mediating roles of depression and pain severity. Pain. 2007;127:243–52.
Zisberg A, Shadmi E, Sinoff G, Gur-Yaish N, Srulovici E, Shochat T. Hospitalization as a turning point for sleep medication use in older adults: prospective cohort study. Drugs Aging. 2012;29:565–76.
AHIP Center for Policy and Research. Trends in Medigap enrollment and coverage options, 2014. https://www.ahip.org/wp-content/uploads/2016/04/MedigapEnrollmentReport_Linked.pdf. Accessed 1 May 2017.
Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain—United States, 2016. MMWR Recomm Rep. 2016;65:1–49.
American Geriatrics Society 2012 Beers Criteria Update Expert Panel. American Geriatrics Society updated Beers Criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2012;2012(60):616–31.
Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83.
National Committee for Quality Assurance. HEDIS® measures included in the 2016 Quality Rating System (QRS). http://www.ncqa.org/hedis-quality-measurement/hedis-measures/hedis-2016. Accessed 13 Apr 2017
Fairies DEL, Haro JM, Obenchain RL, Leon AC. Analysis of observational healthcare data using SAS. Cary: SAS Institute, Inc.; 2010.
Seeger JD, Williams PL, Walker AM. An application of propensity score matching using claims data. Pharmacoepidemiol Drug Saf. 2005;14:465–76.
Vitiello MV, Borson S. Sleep disturbances in patients with Alzheimer’s disease: epidemiology, pathophysiology and treatment. CNS Drugs. 2001;15:777–96.
Salwen JK, Smith MT, Finan PH. Mid-treatment sleep duration predicts clinically significant knee osteoarthritis pain reduction at 6 months: effects from a behavioral sleep medicine clinical trial. Sleep. 2017;40:1–8.
Reeve E, Ong M, Wu A, Jansen J, Petrovic M, Gnjidic D. A systematic review of interventions to deprescribe benzodiazepines and other hypnotics among older people. Eur J Clin Pharmacol. 2017;73:927–35.
Tannenbaum C, Martin P, Tamblyn R, Benedetti A, Ahmed S. Reduction of inappropriate benzodiazepine prescriptions among older adults through direct patient education: the EMPOWER cluster randomized trial. JAMA Intern Med. 2014;174:890–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was funded by the Medicare Supplement Insurance Program. The research did not receive any specific grant or grant number from funding agencies.
Conflicts of interest
Shirley Musich, Shaohung Wang, Lynn Saphire and Luke Slindee are employed by UnitedHealth Group and hold stock in UnitedHealth Group. Ellen Wicker is employed by AARP Services, Inc. However, their compensation was not dependent upon the results obtained in this research, and the investigators retained full independence in the conduct of this research.
Ethical approval
This research is covered under New England IRB No. 120160532.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Musich, S., Wang, S.S., Slindee, L.B. et al. Characteristics of New-Onset and Chronic Sleep Medication Users Among Older Adults: A Retrospective Study of a US Medigap Plan Population using Propensity Score Matching. Drugs Aging 35, 467–476 (2018). https://doi.org/10.1007/s40266-018-0543-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-018-0543-5