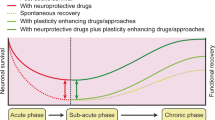
Abstract
Stroke is a leading cause of disability among older adults and more than half of stroke survivors have some residual neurological impairment. Traditionally, managing the aftermath of stroke has been by the implementation of several physical and language therapy modalities. The limitations of these rehabilitation efforts have sparked an interest in finding other ways to enhance neurological recovery. Some of these novel approaches have included pharmacological interventions, cell-derived treatments, and cortical magnetic stimulation. Mounting evidence over the last 2 decades suggests that pharmacological manipulations may have the potential to modulate practice-dependent neuroplasticity and potentially improve neurological recovery after stroke. Multiple pharmacological agents with different mechanisms of action have been evaluated, showing conflicting results. Some studies suggest some promise, yet the quality of the available studies is suboptimal overall, with most of the studies being underpowered. So far, the most promising agents include the antidepressants for motor recovery and acetylcholinesterase inhibitors and memantine for aphasia. However, large, well-designed clinical trials are needed to address the shortcomings of the available data and before any pharmacological agent can be recommended for routine use as part of the standard algorithm of stroke management.
Similar content being viewed by others
References
Truelsen T, Begg S, Mathers C. The global burden of cerebrovascular disease. 2000. WHO 21/06/2006. http://www.who.int/healthinfo/statistics/bod_cerebrovasculardiseasestroke.pdf. Accessed 23 Mar 2015.
Appelros P, Nydevik I, Viitanen M. Poor outcome after first-ever stroke. Predictors for death, dependency, and recurrent stroke within the first year. Stroke. 2003;34:122–6.
Schwamm LH, Ali SF, Reeves MJ, Smith EE, Saver JL, Messe S, et al. Temporal trends in patients characteristics and treatment with intravenous thrombolysis among acute ischemic stroke patients at Get with the Guidelines—Stroke Hospitals. Circ Cardiovasc Qual Outcomes. 2013;6:543–9.
Jauch EC, Saver JL, Adams HP Jr, Bruno A, Connors JJ, Demaerschalk BM, American Heart Association Stroke Council, Council on Cardiovascular Nursing, Council on Peripheral Vascular Disease, Council on Clinical Cardiology, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44:870–947.
Sutherland BA, Minnerup J, Balami JS, Arba F, Buchan AM, Kleinschnitz C. Neuroprotection for ischaemic stroke: translation from the bench to the bedside. Int J Stroke. 2012;7:407–18.
Langhorne P, Bernhardt J, Kwakkel G. Stroke rehabilitation. Lancet. 2011;377:1693–702.
Langhorne P, Coupar F, Pollock A. Motor recovery after stroke: a systematic review. Lancet Neurol. 2009;8:741–54.
Takeuchi N, Izumi SI. Combinations of stroke neurorehabilitation to facilitate motor recovery: perspectives on Hebbian plasticity and homeostatic metaplasticity. Front Hum Neurosci. 2015;9:349.
Hermann DM, Chopp M. Promoting brain remodeling and plasticity for stroke recovery: therapeutic promise and potential pitfalls of clinical translation. Lancet Neurol. 2012;11:369–80.
Cramer SC, Riley JD. Neuroplasticity and brain repair after stroke. Curr Opin Neurol. 2008;21:76–82.
Chollet F, Cramer SC, Stinear C, et al. Pharmacological therapies in post stroke recovery: recommendations for future clinical trials. J Neurol. 2014;261:1461–8.
Rösser N, Flöel A. Pharmacological enhancement of motor recovery in subacute and chronic stroke. Neurorehabilitation. 2008;23:95–103.
Ward NS, Cohen LG. Mechanisms underlying recovery of motor function after stroke. Arch Neurol. 2004;61:1844–8.
Celnik PA, Cohen LG. Modulation of motor function and cortical plasticity in health and disease. Restor Neurol Neurosc. 2004;22:261–8.
Hübener M, Bonhoeffer T. Neuronal plasticity: beyond the critical period. Cell. 2014;159:727–37.
Floel A, Cohen LG. Recovery of function in humans: cortical stimulation and pharmacological treatments after stroke. Neurobiol Dis. 2010;37:243–51.
Berends HI, Nijlant JMM, Movig KLL, Van Putten MJAM, Jannink MJA, Ijzerman MJ. The clinical use of drugs influencing neurotransmitters in the brain to promote motor recovery after stroke; a Cochrane systematic review. Eur J Phys Rehabil Med. 2009;45:621–30.
Rösser N, Heuschmann P, Wersching H, Breitenstein C, Knetch S, Flöel A. Levodopa improves procedural motor learning in chronic stroke patients. Arch Phys Med Rehabil. 2008;89:1633–41.
Acler M, Manganotti P. Role, indications and controversies of levodopa administration in chronic stroke patients. Eur J Phys Rehabil Med. 2013;49:243–9.
Scheidtmann K, Fries W, Muller F, Koenig E. Effects of levodopa in combination with physiotherapy on functional motor recovery after stroke: a prospective, randomized, double-blind study. Lancet. 2001;358:787–90.
Lokk J, Salman Roghani R, Delbari A. Effects of methylphenidate and/or levodopa coupled with physiotherapy on functional and motor recovery after stroke—a randomized, double-blind, placebo-controlled trial. Acta Neurol Scand. 2011;123:266–73.
Scheidtmann K. Advances in adjuvant pharmacotherapy for motor rehabilitation: effects of levodopa. Restor Neurol Neurosci. 2004;22:393–8.
Bhakta B, Hartley S, Holloway I, Couzens JA, Ford GA, Meads D, et al. The DARS (Dopamine Augmented Rehabilitation in Stroke) trial: protocol for a randomized controlled trial of Co-careldopa treatment in addition to routine NHS occupational and Physical therapy after stroke. Trials. 2014;15:316.
Berthier ML, Pulvermüller F, Davila G, Garcia Casares N, Gutierrez A. Drug therapy of post-stroke aphasia: a review of current evidence. Neuropsychol Rev. 2011;21:302–17.
Boissezon X, Peran P, Boysson C, Démonet JF. Pharmacotherapy of aphasia: myth or reality? Brain Lang. 2007;102:114–25.
Seniów J, Litwin M, Litwin T, Leśniak M, Czlonkowska A. New approach to the rehabilitation of post-stroke focal cognitive syndrome: effect of levodopa combined with speech and language therapy on functional recovery from aphasia. J Neurol Sci. 2009;28:214–8.
Breitenstein C, Korsukewitz C, Baumgärtner A, Flöel A, Zwitserlood P, Dobel C, et al. l-Dopa does not add to the success of high-intensity language training in aphasia. Restor Neurol Neurosci. 2015;33:115–20.
Martinsson L, Eksborg S. Drugs for stroke recovery: the example of amphetamines. Drugs Aging. 2004;21:67–79.
Walker-Batson D. Amphetamine and post-stroke rehabilitation: indications and controversies. Eur J Phys Rehabil Med. 2013;49:251–60.
Ortega FJ, Jolkkonen J. Restorative therapies to enhance sensorimotor recovery following cerebral ischemia. Acta Neurobiol Exp. 2013;73:66–78.
Gladstone DJ, Danells CJ, Armesto A, McIlroy WE, Staines WR, Graham SJ, Subacute Therapy with Amphetamine and Rehabilitation for Stroke Study Investigators, et al. Physiotherapy coupled with dextroamphetamine for rehabilitation after hemiparetic stroke: a randomized, double-blind, placebo-controlled trial. Stroke. 2006;37:179–85.
Schuster C, Maunz G, Lutz K, Kischka U, Sturzenegger R, Ettlin T. Dexamphetamine improves upper extremity outcome after stroke: a pilot randomized controlled trial. Neurorehabil Neural Repair. 2011;25:749–55.
Martinsson L, Hårdemark HG, Eksborg S. Amphetamines for improving recovery after stroke. Cochrane Database Syst Rev. 2007;1:CD002090. doi:10.1002/14651858.CD002090.pub2.
Walker-Batson D, Curtis S, Natarajan R, Ford J, Dronkers N, Salmeron E, et al. A double-blind, placebo-controlled study of the use of amphetamine in the treatment of aphasia. Stroke. 2001;32:2093–8.
Paolucci S. Role, indications, and controversies of antidepressant therapy in chronic stroke patients. Eur J Phys Rehabil Med. 2013;49:233–41.
Chollet F, Tardy J, Albucher JF, Thalamas C, Berard E, Lamy C, et al. Fluoxetine for motor recovery after acute ischaemic stroke (FLAME): a randomized placebo-controlled trial. Lancet Neurol. 2011;10:123–30.
Mead GE, Hsieh CF, Lee R, Kutlubaev MA, Claxton A, Hankey GJ, et al. Selective serotonin reuptake inhibitors (SSRIs) for stroke recovery. Cochrane Database Syst Rev. 2012;11:CD009286. doi:10.1002/14651858.CD009286.pub2.
Paolucci S, Bureca I, Multari M, Nocentini U, Matano A. An open-label pilot study of the use of rivastigmine to promote functional recovery in patients with unilateral spatial neglect due to first ischemic stroke. Func Neurol. 2010;25:195–200.
Birks J, McGuinness B, Craig D. Rivastigmine for vascular cognitive impairment. Cochrane Database Syst Rev. 2013;5:CD004744. doi:10.1002/14651858.CD004744.pub3.
Dichgans M, Markus HS, Salloway S, Verkkoniemi A, Moline M, Wang Q, et al. Donepezil inpatients with subcortical vascular cognitive impairment: a randomized double-blind trial in CADASIL. Lancet Neurol. 2008;7(4):310–8.
Nadeau SE, Behrman AL, Davis SE, Reid K, Wu SS, Stidham BS, et al. Donepezil as an adjuvant to constraint-induced therapy for upper-limb dysfunction after stroke: an exploratory randomized clinical trial. J Rehabil Res Dev. 2004;41:525–34.
Barrett KM, Brott TG, Brown RD Jr, Carter RE, Geske JR, Graff-Radford NR, Mayo Acute Stroke Trial for Enhancing Recovery (MASTER) Study Group, et al. Enhancing recovery after acute ischemic stroke with donepezil as an adjuvant therapy to standard medical care: results of a phase IIA clinical trial. J Stroke Cerebrovasc Dis. 2011;20:177–82.
Trotman M, Vermehren P, Gibson CL, Fern R. The dichotomy of memantine treatment for ischemic stroke: dose-dependent protective and detrimental effects. J Cereb Blood Flow Metab. 2015;35:230–9.
López-Valdés H, Clarkson AN, Ao Y, Charles AC, Carmichael ST, Sofroniew MV, et al. Memantine enhances recovery from stroke. Stroke. 2014;45:2093–100.
Larpthaveesarp A, Ferriero DM, Gonzalez FF. Growth factors for the treatment of ischemic brain injury (growth factor treatment). Brain Sci. 2015;5(2):165–77. doi:10.3390/brainsci5020165.
Bath PM, Springg N, England T. Colony stimulating factors (including erythropoietin, granulocyte colony stimulating factor and analogues) for stroke. Cochrane Database Syst Rev. 2013;6:CD005207. doi:10.1002/14651858.CD005207.pub4.
Ringelstein EB, Thijs V, Norrving B, Chamorro A, Aichner F, Grond M, et al. AXIS 2 Investigators. Granulocyte colony-stimulating factor in patient with acute stroke: results of the AX200 for ischemic stroke trial. Stroke. 2013;44:2681–7.
Lagarto Parra A, Garcia Rodriguez JC. Nasal Neuro EPO could be a reliable choice for neuroprotective stroke treatment. Cent Nerv Syst Agents Med Chem. 2012;12:60–8.
Engelter ST. Safety in pharmacological enhancement of stroke rehabilitation. Eur J Phys Rehabil Med. 2013;49:261–7.
Kessler J, Thiel A, Karbe H, Heiss WD. Piracetam improves activated blood flow and facilitates rehabilitation of poststroke aphasic patients. Stroke. 2000;31:2112–6.
Greener J, Enderby P, Whurr R. Pharmacological treatment for aphasia following stroke. Cochrane Database Syst Rev. 2001;4:CD000424.
Grieb P. Neuroprotective properties of citicoline: facts, doubts and unresolved issues. CNS Drugs. 2014;28:185–93.
Alvarez-Sabin J, Roman GC. Citicoline in vascular cognitive impairment and vascular dementia after stroke. Stroke. 2011;56(suppl 1):S40–3.
Davalos A, Alvarez-Sabın J, Castillo J, et al. Citicoline in the treatment of acute ischaemic stroke: an international, randomised, multicentre, placebo-controlled study (ICTUS trial). Lancet. 2012;380:349–57.
Fioravanti M, Yanagi M. Cytidinediphosphocholine (CDP-choline) for cognitive and behavioural disturbances associated with chronic cerebral disorders in the elderly. Cochrane Database Syst Rev. 2005;18:CD000269.
Guekht A, Skoog I, Korczyn AD, Zakharov V, Eeg M, Vigonius U. A randomised, double-blind, placebo-controlled trial of actovegin in patients with post-stroke cognitive impairment: ARTEMIDA study design. Dement Geriatr Cogn Disord Extra. 2013;3:459–67.
Skoog I, Korczyn AD, Guekht A. Neuroprotection in vascular dementia: a future path. J Neurol Sci. 2012;322:232–6.
Bennett A, Majersik J. Memantine for enhanced stroke recovery [ClinicalTrials.gov identifier NCT02144584]. US National Institutes of Health, ClinicalTrials.gov. https://clinicaltrials.gov. Accessed 31 Aug 2015.
Takeda. Efficacy and safety of actovegin in post-stroke cognitive impairment (PSCI) (ARTEMIDA) [ClinicalTrials.gov identifier NCT01582854]. US National Institutes of Health, ClinicalTrials.gov. https://clinicaltrials.gov. Accessed 31 Aug 2015.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for the publication of this article.
Conflict of interest
Dr. Beristain and Dr. Golombievski have no conflicts of interest to report.
Rights and permissions
About this article
Cite this article
Beristain, X., Golombievski, E. Pharmacotherapy to Enhance Cognitive and Motor Recovery Following Stroke. Drugs Aging 32, 765–772 (2015). https://doi.org/10.1007/s40266-015-0299-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-015-0299-0