Abstract
Cefepime/enmetazobactam (EXBLIFEP®), an intravenous (IV) antibacterial fixed-dose combination of a 4th generation cephalosporin and an extended-spectrum β-lactamase (ESBL) inhibitor, is being developed by Allecra Therapeutics and ADVANZ PHARMA for the treatment of infections caused by multi-drug-resistant (MDR) Gram-negative bacteria. In February 2024, cefepime/enmetazobactam was approved in the USA for use in adults with complicated urinary tract infections (cUTI) including pyelonephritis, caused by susceptible strains of Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, Proteus mirabilis, and Enterobacter cloacae complex. In March 2024, cefepime/enmetazobactam was approved in the EU for use in adults for the treatment of cUTI, including pyelonephritis, and hospital-acquired pneumonia, including ventilator associated pneumonia, and the treatment of patients with bacteraemia occurring in association with or suspected to be associated with any of these infections. This article summarizes the milestones in the development of cefepime/enmetazobactam leading to this first approval for the treatment of adults with infections caused by MDR Gram-negative bacteria.
Similar content being viewed by others
References
Papp-Wallace KM, Bethel CR, Caillon J, et al. Beyond piperacillin-tazobactam: cefepime and AAI101 as a potent β-lactam-β-lactamase inhibitor combination. Antimicrob Agents Chemother. 2019;63(5):e00105-e119.
Belley A, Morrissey I, Hawser S, et al. Third-generation cephalosporin resistance in clinical isolates of Enterobacterales collected between 2016–2018 from USA and Europe: genotypic analysis of β-lactamases and comparative in vitro activity of cefepime/enmetazobactam. J Glob Antimicrob Resist. 2021;25:93–101.
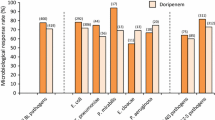
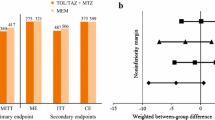
Kaye KS, Belley A, Barth P, et al. Effect of cefepime/enmetazobactam vs piperacillin/tazobactam on clinical cure and microbiological eradication in patients with complicated urinary tract infection or acute pyelonephritis: a randomized clinical trial. JAMA. 2022;328(13):1304–14.
Isler B, Harris P, Stewart AG, Paterson DL. An update on cefepime and its future role in combination with novel β-lactamase inhibitors for MDR Enterobacterales and Pseudomonas aeruginosa. J Antimicrob Chemother. 2021;76(3):550–60.
Magill SS, O’Leary E, Ray SM, et al. Antimicrobial use in US hospitals: comparison of results from emerging infections program prevalence surveys, 2015 and 2011. Clin Infect Dis. 2021;72(10):1784–92.
Advani SD, Claeys K. Cefepime/enmetazobactam for complicated urinary tract infections. JAMA. 2022;328(13):1299–301.
Vázquez-Ucha JC, Lasarte-Monterrubio C, Guijarro-Sánchez P, et al. Assessment of activity and resistance mechanisms to cefepime in combination with the novel β-lactamase inhibitors zidebactam, taniborbactam, and enmetazobactam against a multicenter collection of carbapenemase-producing Enterobacterales. Antimicrob Agents Chemother. 2022;66(2): e0167621.
European Medicines Agency. Cefepime/enmetazobactam: EU prescribing information. 2024. https://www.ema.europa.eu/. Accessed 11 Apr 2024.
Allecra Therapeutics. Cefepime/enmetazobactam: US prescribing information. 2024. https://www.fda.gov/. Accessed 26 Feb 2024.
Allecra Therapeutics. Allecra Therapeutics announces US FDA approval for EXBLIFEP® for the treatment of complicated urinary tract infections [media release]. 27 Feb 2024. https://www.allecra.com. Accessed 27 Feb 2024.
US Food & Drug Administration. Exblifep (cefepime and enmetazobactam): approval letter. 2024. https://www.fda.gov/. Accessed 20 Mar 2024.
ADVANZ PHARMA Limited, Allecra Therapeutics. ADVANZ PHARMA and Allecra Therapeutics announce approval in the European Union for the new antibiotic combination EXBLIFEP® (cefepime/enmetazobactam) for the treatment of severe infections [media release]. 27 Mar 2024. https://www.advanzpharma.com/. Accessed 27 Mar 2024.
European Medicines Agency. Cefepime/enmetazobactam: EU assessment report. 2024. https://www.ema.europa.eu/. Accessed 11 Apr 2024.
Allecra Therapeutics. Allecra Therapeutics and ADVANZ PHARMA sign exclusive license and supply agreement for Allecra’s antibiotic cefepime/enmetazobactam in Europe [media release]. 13 Jan 2022. https://www.allecra.com. Accessed 13 Jan 2022.
Allecra Therapeutics GmbH, Shanghai Haini Pharmaceutical Co Ltd. Allecra Therapeutics and Shanghai Haini Pharmaceutical announce exclusive licensing agreement for cefepime/enmetazobactam for Greater China [media release]. 21 Dec 2020. https://www.allecra.com. Accessed 21 Dec 2020.
Orchid Chemicals & Pharmaceuticals. Orchid enters into strategic partnership with Allecra Therapeutics for development of novel antibiotics [media release]. 18 Apr 2013. https://www.orchidpharma.com. Accessed 18 Apr 2013.
Principe L, Lupia T, Andriani L, et al. Microbiological, clinical, and PK/PD features of the new anti-Gram-negative antibiotics: β-lactam/β-lactamase inhibitors in combination and cefiderocol—an all-inclusive guide for clinicians. Pharmaceuticals (Basel). 2022;15:463.
Morrissey I, Magnet S, Hawser S, et al. In vitro activity of cefepime-enmetazobactam against Gram-negative isolates collected from US and European hospitals during 2014–2015. Antimicrob Agents Chemother. 2019;63(7):e00514-e519.
Tselepis L, Langley GW, Aboklaish AF, et al. In vitro efficacy of imipenem-relebactam and cefepime-AAI101 against a global collection of ESBL-positive and carbapenemase-producing Enterobacteriaceae. Int J Antimicrob Agents. 2020;56(1): 105925.
Hawser S, Morrissey I, Kothari N, et al. Influence of genotype on the activity of cefepime/enmetazobactam against beta-lactam resistant Enterobacterales collected from Europe in 2019-2021 [abstract and poster no. P0097]. In: 33rd European Congress of Clinical Microbiology & Infectious Diseases (ECCMID). 2023.
Knechtle P, Shapiro S, Morrissey I, et al. Sigmoid Emax modeling to define the fixed concentration of enmetazobactam for MIC testing in combination with cefepime. Antimicrob Agents Chemother. 2021;65(8): e0092621.
Belley A, Kothari N, Monti F, et al. Antibacterial activity of cefepime/enmetazobactam and comparator agents against US enterobacterales clinical isolates collected from 2019–2021. Open Forum Infect Dis. 2023;10(Suppl 2):S60–70.
Hawser S, Morrissey I, Kothari N, et al. Surveillance of cefepime/enmetazobactam against European isolates of Enterobacterales collected between 2019 and 2021, including cephalosporin-resistant phenotypes [Abstract and poster no. P0289]. In: 33rd European Congress of Clinical Microbiology & Infectious Diseases (ECCMID). 2023.
Crandon JL, Nicolau DP. In vivo activities of simulated human doses of cefepime and cefepime-AAI101 against multidrug-resistant Gram-negative Enterobacteriaceae. Antimicrob Agents Chemother. 2015;59(5):2688–94.
Bernhard F, Odedra R, Sordello S, et al. Pharmacokinetics-pharmacodynamics of enmetazobactam combined with cefepime in a neutropenic murine thigh infection model. Antimicrob Agents Chemother. 2020;64(6):e00078-e120.
Johnson A, McEntee L, Farrington N, et al. Pharmacodynamics of cefepime combined with the novel extended-spectrum-β-lactamase (ESBL) inhibitor enmetazobactam for murine pneumonia caused by ESBL-producing Klebsiella pneumoniae. Antimicrob Agents Chemother. 2020;64(6):e00180-e220.
Das S, Fitzgerald R, Ullah A, et al. Intrapulmonary pharmacokinetics of cefepime and enmetazobactam in healthy volunteers: towards new treatments for nosocomial pneumonia. Antimicrob Agents Chemother. 2020;65(1):e01468-e1520.
Carmeli Y, Knechtle P, Hardenberg J, et al. A randomized phase 2 study of cefepime combined with the novel extended spectrum β-lactamase inhibitor enmetazobactam in hospitalized adults with complicated urinary tract infections (cUTI) including acute pyelonephritis (AP) [abstract no 1479]. OFID. 2019;6(Suppl 2):S539.
Funding
The preparation of this review was not supported by any external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Authorship and Conflict of Interest
During the peer review process the manufacturer of the agent under review was offered an opportunity to comment on the article. Changes resulting from any comments received were made by the authors on the basis of scientific completeness and accuracy. Susan J. Keam is a contracted employee of Adis International Ltd/Springer Nature, and declares no relevant conflicts of interest. All authors contributed to this article and are responsible for its content.
Ethical approval, Consent to Participate, Consent to Publish, Availability of Data and Material, Code Availability
Not applicable.
Additional information
This profile has been extracted and modified from the AdisInsight database. AdisInsight tracks drug development worldwide through the entire development process, from discovery, through pre-clinical and clinical studies to market launch and beyond.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Keam, S.J. Cefepime/Enmetazobactam: First Approval. Drugs (2024). https://doi.org/10.1007/s40265-024-02035-2
Accepted:
Published:
DOI: https://doi.org/10.1007/s40265-024-02035-2