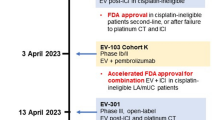
Abstract
Cadonilimab (开坦尼®), a PD-1/CTLA-4 bi-specific antibody, is being developed by Akeso, Inc. for the treatment of a range of solid tumours, including cervical cancer, lung cancer, gastric/gastroesophageal junction cancer, oesophageal squamous cell cancer, liver cancer and nasopharyngeal cancer. Cadonilimab was approved in China in June 2022 for use in patients with relapsed or metastatic cervical cancer (r/mCC) who have progressed on or after platinum-based chemotherapy. This article summarizes the milestones in the development of cadonilimab leading to this first approval for the treatment of patients with r/mCC.
Similar content being viewed by others
References
Mai HQ, Lin SJ, Chen DP, et al. A phase II study of AK104, a bispecific antibody targeting PD-1 and CTLA-4, in patients with metastatic nasopharyngeal carcinoma (NPC) who had progressed after two or more lines of chemotherapy [abstract no. 436 plus poster]. J Immunother Cancer. 2021;9(Suppl. 2):A466.
Monk BJ, Enomoto T, Kast WM, et al. Integration of immunotherapy into treatment of cervical cancer: Recent data and ongoing trials. Cancer Treat Rev. 2022;106: 102385.
Rotte A. Combination of CTLA-4 and PD-1 blockers for treatment of cancer. J Exp Clin Cancer Res. 2019. https://doi.org/10.1186/s13046-019-1259-z.
Wojtukiewicz MZ, Rek MM, Karpowicz K, et al. Inhibitors of immune checkpoints–PD-1, PD-L1, CTLA-4–new opportunities for cancer patients and a new challenge for internists and general practitioners. Cancer Metastasis Rev. 2021;40(3):949–82.
Akeso Inc. Cadonilimab: Chinese prescribing information. Zhongshan: Akeso Inc; 2022.
Wu XH, Ji JF, Lou HM, et al. Efficacy and safety of cadonilimab, an anti-PD-1/CTLA4 bi-specific antibody, in previously treated recurrent or metastatic (R/M) cervical cancer: A multicenter, open-label, single-arm, phase II trial [abstract]. Society of Gynecologic Oncology (SGO) Meeting. 2022.
Akeso Inc. Akeso’s cadonilimab (PD-1/CTLA-4), the first dual immune checkpoint inhibitor to treat cancer, approved for marketing in China [media release]. 29 Jun 2022. https://www.akesobio.com/.
National Medical Products Administration. National Medical Products Administration approval notice: cadolinimab [media release]. 29 Jun 2022. https://www.nmpa.gov.cn/yaowen/ypjgyw/20220629135936153.html.
Akeso Inc. Akeso announces clinical trial collaboration with Chipscreen Biosciences to evaluate cadonilimab in combination with chiauranib for extensive-stage small-cell lung cancer [media release]. 1 Mar 2022. http://www.akesobio.com.
Akeso Inc. Akeso, Inc. works with Pfizer to conduct a clinical research of cadonilimab (bi-specific antibody) with combination of axitinib for the first-line treatment of advanced/metastatic renal clear cell carcinoma [media release]. 27 Aug 2021. https://www.akesobio.com/.
Akeso Inc. Akesobio presents phase 1 clinical data from first-in-class bispecific drug candidate AK104 (PD1/CTLA4) as oral presentation, at 2019 SITC annual meeting [media release]. 11 Nov 2019. http://www.akesobio.com.
Huang ZL, Pang XH, Zhong TT, et al. Cadonilimab, an anti-PD1/CTLA4 bi-specific antibody with Fc effector null backbone [abstract no. 289]. J Immunother Cancer. 2021;9(Suppl. 2):A313–4.
Markman B, Tran B, Gan H, et al. A Phase 1 study of AK104, a tetrameric bispecific antibody that targets PD-1 and CTLA-4 in patients with advanced solid tumors [abstract no. O30 plus presentation]. J Immunother Cancer. 2019;7(Suppl. 1):30.
Akeso Inc. Akeso, Inc. 2021 annual results presentation, March 2022. 2022. https://www.akesobio.com/media/1643/%E6%8C%82%E7%BD%91%E7%89%88-akeso-fy21-results331_ve-1.pdf. Accessed 20 Jul 2022.
Wang J, Lou H, Cai H-B, et al. A study of AK104 (an anti-PD1 and anti-CTLA4 bispecific antibody) combined with standard therapy for the first-line treatment of persistent, recurrent, or metastatic cervical cancer (R/M CC) [abstract no. 106 plus presentation]. J Clin Oncol. 2022;40(16 Suppl.):106.
Wu L, Chen B, Yao W, et al. A phase Ib/II trial of AK104 (PD-1/CTLA-4 bispecific antibody) in combination with anlotinib in advanced NSCLC [abstract no. 1300P]. Ann Oncol. 2021;32(Suppl. 5):S1006.
Millward M, Frentzas S, Gan HK, et al. Safety and antitumor activity of AK104, a bispecific antibody targeting PD-1 and CTLA-4, in patients with mesothelioma which is relapsed or refractory to standard therapies [abstract no. 1021O plus presentation]. Ann Oncol. 2020;31(Suppl. 4):S705–6.
Ji J, Shen L, Gao X, et al. A phase Ib/II, multicenter, open-label study of AK104, a PD-1/CTLA-4 bispecific antibody, combined with chemotherapy (chemo) as first-line therapy for advanced gastric (G) or gastroesophageal junction (GEJ) cancer [abstract no. 308]. J Clin Oncol. 2022;40(4 Suppl.):308.
Bai L, Sun M, Xu A, et al. Phase 2 study of AK104 (PD-1/CTLA-4 bispecific antibody) plus lenvatinib as first-line treatment of unresectable hepatocellular carcinoma [abstract no. 4101]. J Clin Oncol. 2021;39(15 Suppl.):4101.
Akeso Inc. Akeso's cadonilimab (PD-1/CTLA-4 bi-specific antibody) combined with ivonescimab (PD-1/VEGF bi-specific antibody) combined with or without chemotherapy obtained approval to initiate a phase Ib/II clinical trial for the treatment of advanced non-small cell lung cancer [media release]. 6 Jan 2022. http://www.akesobio.com.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Authorship and Conflict of interest
During the peer review process the manufacturer of the agent under review was offered an opportunity to comment on the article. Changes resulting from any comments received were made by the authors on the basis of scientific completeness and accuracy. Susan J. Keam is a contracted employee of Adis International Ltd/Springer Nature, and declares no relevant conflicts of interest. All authors contributed to the review and are responsible for the article content.
Ethics approval, Consent to participate, Consent to publish, Availability of data and material, Code availability
Not applicable.
Additional information
This profile has been extracted and modified from the AdisInsight database. AdisInsight tracks drug development worldwide through the entire development process, from discovery, through pre-clinical and clinical studies to market launch and beyond.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Keam, S.J. Cadonilimab: First Approval. Drugs 82, 1333–1339 (2022). https://doi.org/10.1007/s40265-022-01761-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-022-01761-9