Abstract
Monoclonal antibodies (mAbs) are a recent addition to multiple myeloma (MM) therapies and a number of mAbs directed at myeloma cell surface molecules are in development. Daratumumab is a CD38 mAb that has demonstrated substantial activity and good tolerability in four phase I, phase I/II and phase II studies as monotherapy, as well as in combination with current standard treatments in MM. The positive results obtained in the relapsed/refractory setting in patients with advanced-stage disease and in a small number of patients with newly diagnosed disease provide the rationale for the investigation of the agent in a number of ongoing phase III trials. mAbs are generally better tolerated than conventional chemotherapy; however, their use requires other special considerations. Such factors include those common to all mAbs, namely infusion-related reactions, but also factors that are observed with mAbs used in myeloma, such as interference with response assessment, or factors that are related to CD38 mAbs such as daratumumab, for instance blood typing interference. Our review provides an overview of the results from the daratumumab clinical trials conducted to date, as well as practical management considerations for the use of daratumumab based on our experience with the agent.
Similar content being viewed by others
Monoclonal antibodies (mAbs) present a new development in the treatment of multiple myeloma. |
Daratumumab is a fully human mAb directed at CD38 that has shown significant clinical activity and good tolerability as monotherapy and in combination with current standard therapies in patients with relapsed/refractory multiple myeloma, which was approved by the US FDA in November 2015. |
mAb therapy, in myeloma as in other diseases, is associated with specific management considerations. Practical recommendations regarding these, as well as the administration and scheduling of daratumumab, are provided in this review. |
Extensive clinical development of daratumumab is currently ongoing, which will help define the place for the agent in the treatment of multiple myeloma. |
1 Introduction
Immunotherapy, aimed at engaging the immune system in the fight against malignant cells, is an attractive concept in cancer treatment. A number of different strategies can be distinguished, such as mAbs, chimeric antigen receptor (CAR) T cells, checkpoint inhibition and vaccination. mAbs are a widely used treatment modality in hematology, with rituximab being the most prominent example. The agent was approved for the treatment of lymphoma in 1997 by the US FDA and in 1998 by the European Medicines Agency (EMA) [1], and is now considered a standard therapy in lymphoma that has had a major impact on survival [2]. A large body of experience has been accumulated regarding the appropriate management of patients receiving rituximab.
In multiple myeloma (MM), the introduction of proteasome inhibitors (PIs) (e.g. bortezomib and carfilzomib) and immunomodulatory drugs (IMiDs; e.g. lenalidomide and pomalidomide) heralded a major change in the management of the disease. These agents form the backbone of current treatment strategies and have led to significant improvements in patient survival [3]. However, for patients who are refractory to these agents, the prognosis remains poor, with a median overall survival (OS) of only 9 months [3, 4]. Novel strategies are therefore needed and immunotherapy, particularly the mAbs, presents an exciting new approach. mAbs combine a high specificity for antigens on the surface of the neoplastic cell or in the microenvironment and the capability to engage immune cells.
In MM, the search for mAbs to target the tumor cell has been elusive until the recent past; however, a large number of mAbs are currently under investigation. Among them, elotuzumab, a humanized immunoglobulin (Ig) G1 kappa mAb that targets the signaling lymphocytic activation molecule-F7 (SLAMF7, also known as CS1), has advanced furthest in clinical testing. Recently, CD38 has been identified as an attractive target for mAb therapy in myeloma, and three CD38 mAbs are currently being investigated in clinical trials—the chimeric IgG1 kappa mAb isatuximab (SAR650984) and two human mAbs, daratumumab (IgG1 kappa) and MOR202 (IgG1 lambda). Daratumumab is the CD38 mAb for which the clinical development has progressed furthest and is the focus of our review. It has shown highly promising results in preclinical and clinical studies. It was granted breakthrough status by the FDA in 2013 [5] and was approved by the FDA in November 2015 for the treatment of patients who have received three or more prior lines of therapy, including a PI and an immunomodulatory agent, or who are double refractory to a PI and an immunomodulatory agent [6]. Daratumumab is currently undergoing regulatory review by the EMA [7].
Our aim was to provide a summary of the daratumumab clinical trials conducted in multiple myeloma to date on the one hand, and to outline management considerations on the other hand, with the addition of practical recommendations based on our experience with the agent.
2 Target and Mode of Action of Daratumumab
CD38 is a transmembrane glycoprotein that combines adhesion, receptor and enzymatic functions [8–12]. As an ectoenzyme, it exerts its catalytic function on the external surface of the cell membrane. It catalyzes the conversion of nicotinamide adenine dinucleotide (NAD) + to adenosine diphosphate ribose (ADPR) and nicotinic acid adenine dinucleotide phosphate (NAADP), and is thus involved in the mobilization of calcium, a key factor in signaling pathways of cell growth, survival and differentiation [8, 10]. CD38 is normally expressed by precursor and activated B and T cells, as well as myeloid cells, natural killer (NK) cells, erythrocytes, platelets and plasma cells [10]. In leukocytes, it functions as a plasma membrane signaling receptor and acts as a co-receptor on B cells, where it modulates B cell receptor signals [13]. It is also involved in the chemotaxis of neutrophils and monocytes [14, 15], and mediates the production of cytokines by effector cells, the proliferation of T lymphocytes, and the protection of mature B lymphocytes and dendritic cells from apoptosis [16]. CD38 is not expressed by pluripotent hematopoietic precursor cells, which may be important for bone marrow recovery, in particular, when CD38 mAbs are used in combination with myelotoxic drugs [17, 18].
While the expression on normal lymphoid and myeloid cells is relatively low, CD38 is highly and uniformly expressed on myeloma cells [10, 11, 19, 20]. Through the production of adenosine, CD38 is thought to play an important role in the survival of the myeloma cell in the bone marrow environment [12, 21]. The high expression on myeloma cells combined with important enzymatic and signaling functions make CD38 an attractive target for immunotherapy [10, 21].
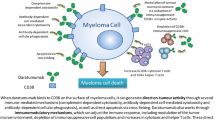
Following binding to CD38, daratumumab is understood to exert its cytotoxic effects through a number of mechanisms, including complement-dependent cytotoxicity (CDC), antibody-dependent cell-mediated cytotoxicity (ADCC), and antibody-dependent cell-mediated phagocytosis (ADCP), as well as the direct induction of apoptosis upon secondary crosslinking [22–24]. In vitro, daratumumab was found to be the most active CDC-activating mAb of the CD38 mAbs currently undergoing investigation [25]. In addition, the modulation of cellular enzymatic activities associated with calcium mobilization and signaling is thought to contribute to the cytotoxic effect [26]. Moreover, an immunomodulatory effect of daratumumab was recently described following an analysis of patient samples from two clinical trials in which patients with advanced-stage disease received daratumumab [27]. Following daratumumab treatment, increased adaptive immune responses were observed, evidenced by T-cell increases alongside increases in CD8+:CD4+ ratios, antiviral responses, anti-allotypic responses and T-cell clonality. Interestingly, subpopulations of regulatory T cells, B cells and monocytes were identified that expressed high levels of CD38 and were found to be highly immunosuppressive and sensitive to treatment with daratumumab. These results suggest that daratumumab may have an important immunomodulatory role by eliminating cells that suppress the immune system in myeloma and potentially releasing an antimyeloma response, as reflected by the emergence of spikes of clonal T cells and improved anti-allotypic responses.
3 Antimyeloma Activity of Daratumumab
3.1 Preclinical Activity
Daratumumab demonstrated potent in vitro activity against myeloma cells isolated from patients and myeloma cell lines [22, 23]. Of note, in preclinical studies, synergy in inducing ADCC between lenalidomide or bortezomib and daratumumab was shown [28, 29]. In addition, the benefit of combining daratumumab and lenalidomide could also be shown in lenalidomide/bortezomib-resistant multiple myeloma cell lines and even in primary multiple myeloma cells from bone marrow mononuclear cells derived from lenalidomide- and/or bortezomib-refractory patients [30].
3.2 Clinical Activity: Daratumumab Monotherapy
To date, the results of four clinical studies with daratumumab have been published or presented at congresses (Table 1). In the first-in-human study of daratumumab (GEN501), designed as a phase I/II trial, patients with relapsed/refractory MM who had received two or more prior lines of therapy and who were ineligible for autologous stem cell transplantation were treated with daratumumab single agent administered in a dose-escalation scheme [31]. In the first part of the study, involving 32 patients, the dose of daratumumab, administered intravenously once weekly, was increased gradually, with three patients treated at each dose level from 0.005 to 24 mg/kg without observation of a dose-limiting toxicity. Among 12 patients who received daratumumab 4–24 mg/kg, a partial response (PR) was seen in four patients and a minimal response (MR) was seen in three patients. Overall, daratumumab showed a favorable tolerability profile. Serious adverse events (AEs) occurred in 37 % of patients and included pyrexia, infections (9 % each), bronchospasm (6 %), anemia, thrombocytopenia, atrial fibrillation, abdominal pain, and hepatobiliary disorders (3 % each). Infusion-related reactions (IRRs) were observed in 63 % of patients, of which 6 % were grade 3 or 4 (bronchospasm and hypersensitivity, 3 % each).
In part 2 of the study, two dosing cohorts were selected (8 mg/kg and 16 mg/kg) and 30 and 42 patients, who had received a median of four prior therapies, were treated, respectively [31]. The overall response rate (ORR) was 36 % in the 16 mg/kg cohort [two patients complete response (CR), two patients very good PR (VGPR), and 11 patients PR], and 10 % in the 8 mg/kg group (three patients PR). The median time to response was 0.9 months in the 16 mg/kg group. The median duration of response was longer in the 16 mg/kg group (not reached for the 16 mg/kg group and 6.9 months for the 8 mg/kg group). The median progression-free survival (PFS) was 5.6 months in the 16 mg/kg group and 2.4 months in the 8 mg/kg group, while the 1-year OS was 77 % in both groups. Most AEs were grade 1 or 2, with the most common, defined as occurring in at least 25 % of patients, being fatigue, allergic rhinitis, and pyrexia. Overall, grade 3 or 4 AEs were seen in 26 % of patients in the 16 mg/kg group and 53 % of patients in the 8 mg/kg group. Grade 3 or 4 events that occurred in two or more patients were pneumonia (five patients), thrombocytopenia (four patients), and neutropenia, leukopenia, anemia, and hyperglycemia (two patients each). IRRs were seen in 71 % of patients and were grade 1 and 2, except for one patient with a grade 3 reaction. The majority of IRRs occurred during the first infusion. Of note, there were no discontinuations due to IRRs. In this trial, different infusion rates in both the 8 and 16 mg/kg groups were investigated and a higher infusion rate was found to be associated with a higher incidence of IRRs, pointing to the role of the infusion rate in the management of IRRs. This trial was the first to demonstrate the substantial activity and good tolerability of daratumumab monotherapy in patients with advanced disease and limited further treatment options, and paved the way for additional trials investigating this drug.
Lonial et al. recently published the results of a second daratumumab monotherapy study involving a larger number of patients (n = 106) [MMY2002, SIRIUS trial] [32]. Patients had substantially advanced disease, were heavily pretreated, and had highly refractory disease. The median time since the initial diagnosis of myeloma was 4.8 years and the patients had received a median of five prior lines of therapy. Eighty percent had undergone autologous stem cell transplantation previously and 95 % had disease refractory to a PI and an IMiD. In addition, 63 % of patients were refractory to pomalidomide, 48 % were refractory to carfilzomib, 66 % were refractory to three of four therapies (bortezomib, lenalidomide, carfilzomib, pomalidomide) and 31 % were refractory to all four agents.
Initially, a small number of patients were randomized 1:1 to receive daratumumab either 8 mg/kg every 4 weeks (18 patients) or 16 mg/kg every week for 8 weeks, then every 2 weeks for 16 weeks, then every 4 weeks thereafter (16 patients). Following a response evaluation, the 16 mg/kg dose was established as the recommended dose for further study and an additional 90 patients were enrolled at this level. Considering all patients dosed at 16 mg/kg (n = 106), the ORR was 29.2 % (3 stringent CRs [sCRs], 10 VGPRs, 18 PRs), with a median time to response of 1 month and a median duration of response of 7.4 months. Of note, responses were seen across the different subgroups investigated, e.g. age ≥75 years, more than three prior lines of therapy, and refractory to PIs and IMiDs. With a median follow-up of 9.3 months, the median PFS was 3.7 months and the 1-year survival rate was 64.8 %. At a subsequent cutoff, the median OS was 17.5 months. Serious treatment-emergent AEs were noted in 30 % of patients and grade 3/4 AEs were noted in 23 % of patients. There were no discontinuations due to AEs. The most frequent AEs of any grade were fatigue (40 %) and anemia (33 %). IRRs, which occurred in 42 % of patients, did so predominantly during the first infusion, were usually grade 1 or 2 (5 % grade 3, no grade 4), and were manageable. Only 6 % of patients had an infusion-related reaction beyond the first infusion. The most common IRRs included nasal congestion (12 %), throat irritation (7 %), cough, dyspnea, chills and vomiting (6 % each). Of note, no patients discontinued treatment due to IRRs. The investigators concluded that the impressive efficacy results combined with the good tolerability make daratumumab a new standard of care in this setting.
A combined efficacy analysis of the two monotherapy studies (GEN501, SIRIUS) was conducted, which included only patients who had received daratumumab 16 mg/kg, which is the recommended dose [33]. Patients (n = 148) had received a median of five prior lines of therapy, 86 % were refractory to both a PI and an IMiD and, notably, 39 and 55 % were refractory to carfilzomib or pomalidomide, respectively. In this combined analysis, the ORR was 31 %, with a VGPR rate of 13 % or better. Of note, two patients had CR and three patients had an sCR. The median duration of response was 7.6 months and, at a median follow-up of 14.8 months, 50 % of responders were progression-free at 12 months. The median PFS was not estimable for responders (≥PR), but was 3.2 months for those achieving MR or stable disease (SD) and 0.9 months for those with progressive disease (PD). For the overall group, the median OS was 19.9 months, with a 1-year OS rate of 69 %. For responders (≥PR), the median OS was not estimable, but was 17.5 months for those achieving MR or SD and 3.7 months for those with PD. The AE profile was consistent with those of the individual studies. The most frequent grade 3 or higher AEs were anemia (18 %), thrombocytopenia (14 %), and neutropenia (10 %). Forty-eight percent of patients had IRRs; 46 % occurred during the first infusion, 4 % during the second infusion, and 3 % during subsequent infusions. These results further illustrate the remarkable single-agent activity of daratumumab in patients with very advanced-stage disease who are refractory to current treatment options, including PIs and IMiDs, and who typically only survive for 8–9 months at this stage of the disease [4, 34].
3.3 Clinical Activity: Daratumumab in Combination with Current Standard Regimens
Daratumumab has also been investigated in combination with current standard regimens. In a phase I/II study involving 45 patients with relapsed and refractory MM following a median of two prior therapies, daratumumab was combined with lenalidomide and dexamethasone [35, 36]. Daratumumab was dose-escalated from 2 to 16 mg/kg in part 1 of the study (n = 13), and then administered at 16 mg/kg in the expansion cohort (n = 32). Lenalidomide was administered at 25 mg on days 1–21, and dexamethasone was administered at 40 mg weekly. With a mean duration of follow-up of 12.9 months for part 1, the overall best response was 100 % (31 % CR, 46 % VGPR, 23 % PR) [35]. In the most recent update of part 2 of the study, data for 32 patients who had received a median of two prior lines were presented with a median follow-up of 15.6 months. In this group, the ORR was 81 %, including 19 % PR, 28 % VGPR, 9 % CR, and 25 % sCR (63 % VGPR or better). The clinical benefit rate (≥MR) was 88 %, and 91 % of patients were progression-free at 12 months. Responses were found to deepen over time, with a time to first response of 1 month and a time to best response of 5.1 months. The 18-month PFS and OS rates were 72 and 90 %, respectively. The most frequent treatment-emergent AEs were neutropenia (84 %), cough (50 %), diarrhea (44 %) and muscle spasms (44 %), and the most frequent grade 3 or higher AEs were neutropenia (78 %), thrombocytopenia (13 %) and anemia (13 %). Sixteen patients had serious AEs, eight of which were due to infection. IRRs were noted in 18 patients [56 %; cough (25 %), allergic rhinitis, nausea, vomiting (9 % each), dyspnea, nasal congestion (6 % each)]; these were grade 2 or lower in the majority of patients. In all patients who experienced an IRR, this occurred during the first infusion, with three patients having IRRs in the second or subsequent infusions. In two patients, grade 3 IRRs were noted (laryngeal edema and hypertension), but no grade 4 IRRs were reported [36].
An accelerated infusion program, which was investigated in the trial, was found to be tolerable, but was associated with a higher incidence of grade 1/2 AEs and will require further investigation [35]. Based on the positive results of this phase I/II trial, the combination of daratumumab, lenalidomide and dexamethasone is being investigated in a randomized phase III trial in the relapsed/refractory setting (POLLUX; http://www.clinicaltrials.gov) (Table 2). Furthermore, a phase III trial has also been initiated in the front-line setting to assess the combination, prospectively, in newly diagnosed patients who are not eligible for transplantation (MAIA) (http://www.clinicaltrials.gov).
To date, one further daratumumab combination study has been presented. In this phase Ib trial, Mateos et al. treated 25 patients with newly diagnosed MM with daratumumab plus bortezomib and dexamethasone (VD), bortezomib, melphalan and prednisone (VMP) or bortezomib, thalidomide and dexamethasone (VTD), while 24 patients with relapsed/refractory MM were treated with the combination of pomalidomide–dexamethasone (Pom–Dex) plus daratumumab [37]. Among the patients with relapsed/refractory MM, the median number of prior therapies was four, all patients had received prior treatment with PIs, IMiDs and steroids, and 92 % were refractory to their last line of treatment. The ORR in 35 evaluable patients was 100 % in the newly diagnosed group and 54.5 % in those with relapsed or relapsed and refractory disease. The combinations were well tolerated and the addition of daratumumab did not result in significant additional toxicity other than IRRs, which occurred in 24 patients (49 %). The IRRs were generally grade 1 or 2 (three grade 3 and no grade 4) and the majority occurred on the first day of the first cycle. Across all patients, grade 3 AEs occurred in 22 (45 %) patients, with the most frequent being neutropenia (25 %), thrombocytopenia (10 %), anemia (8 %), and pneumonia (6 %). Furthermore, daratumumab was not found to have a negative impact on stem cell mobilization [38].
Recently updated results of the Pom–Dex plus daratumumab arm including 98 patients with a median of four prior therapies, 67 % of whom were refractory to both a PI and an IMiD, confirmed the previous results [39]. The ORR was 71 %, including 5 % sCR, 4 % CR, 33 % VGPR, and 28 % PR. The ORR in patients with double-refractory disease was 67 %. At a median follow-up time of 4.2 months, 47 of 53 responders (89 %) had not progressed, and the 6-month PFS rate was 66 %. The most frequent grade 3 or higher AEs were neutropenia (n = 60), anemia (n = 25), leukopenia (n = 20), and thrombocytopenia (n = 15). Serious AEs occurred in 42 % of patients. Overall, the rate of grade 3 or higher AEs was similar to that observed with Pom–Dex alone, and no new safety signals were observed with daratumumab + Pom–Dex. IRRs occurred in 52 patients and were predominately grade 2 or lower. Six patients had grade 3 IRRs and two patients discontinued because of an IRR. The IRRs occurred in 53, 1, and 0 % of patients during the first, second, and subsequent infusions, respectively.
Taken together, daratumumab presents an exciting new addition to the existing treatment options for myeloma. It has demonstrated impressive activity in patients with advanced disease and limited remaining options. In addition, the tolerability of the agent is remarkable, making it a suitable option across the patient spectrum. A number of phase III trials in the relapsed/refractory setting, as well as the upfront setting, are ongoing, the results of which are eagerly awaited (Table 2).
4 Optimal Dose and Schedule
4.1 Optimal Daratumumab Dose and Schedule
Based on the available clinical evidence, the optimal dose of daratumumab single agent is 16 mg/kg as an intravenous infusion administered weekly during the first 8 weeks, every 2 weeks for the following 16 weeks and monthly thereafter. This was the optimal dose defined in the GEN501 and SIRIUS trials [31, 32]. The optimal duration of treatment is as yet unknown and, in the currently ongoing trials, daratumumab is being administered until disease progression or unacceptable toxicity, but, to date, no sign of long-term toxicity has been observed with daratumumab.
4.2 Daratumumab Infusion
The infusion solution is prepared as a 1000 mL (first dose only) or 500 mL dilution of daratumumab in sterile, pyrogen free 0.9 % NaCl on the day of the planned infusion [31, 40]. Daratumumab is administered as an intravenous infusion through a well-functioning intravenous catheter using an infusion set with a flow regulator to control the infusion rate. The drug must be filtered using an inline filter (0.2 μM) during the infusion.
4.3 Daratumumab Administration and Infusion Rate
Daratumumab is administered as an intravenous infusion, with each patient’s dose calculated based on the patient’s weight rounded to the nearest kilogram. The first infusion of daratumumab (dilution volume 1000 ml) should be administered at an initial rate of 50 ml/h in the first hour, with subsequent increases at the rate of 50 ml/h every hour up to a maximum infusion rate of 200 ml/h possible (Table 3) [31]. The same should be undertaken for the second infusion (dilution volume 500 ml). For subsequent infusions (dilution volume 500 ml), the initial rate for the first hour can be 100 ml/h. Increases in the infusion rate of 50 ml/h every hour up to a maximum infusion rate of 200 ml/h are possible. Importantly, increases in infusion rates should only be carried out if the prior infusion was well tolerated. Of note, any increase in infusion rate can lead to the occurrence of IRRs. Therefore, careful monitoring of the patient is needed to detect IRRs as early as possible. If IRRS are observed, the infusion should be paused promptly, symptoms should be treated accordingly, and the infusion can be resumed at a lower rate when the symptoms have abated.
4.3.1 Practical Recommendations Based on our Experience
If no infusion reactions occur during the first two infusions, the infusion time can be reduced to 3.5 h, but not less, by the third infusion.
5 Managing Daratumumab Therapy in Myeloma
There are special management considerations concerning the use of therapeutic mAbs in hematology in general and MM in particular (Table 4), which we outline in the following sections.
5.1 Blood Typing in Patients Receiving CD38 mAb Therapy
A frequent problem in laboratory medicine is the interference of endogenous and exogenous substances with assays for clinical analytes. Major endogenous compounds that can interfere with laboratory results are hemoglobin, bilirubin, lipids, and paraproteins [41]. For example, paraproteins can result in precipitation or chemical or immunological interference from specific or non-specific binding of a particular analyte [42]. The main exogenous sources of interference are therapies administered to the patient [41]. For many drugs, interference with laboratory tests are unknown and are discovered by chance when unexpected laboratory results are found that do not conform with the patient’s condition [43]. The increasing use of diagnostic and therapeutic mAbs has highlighted the possible interference of these agents with routine laboratory tests. In a review on the interference of mAbs with laboratory diagnostic procedures, Ostrov and Amsterdam explain that interference, apart from being categorized into endogenous or exogenous, can be due to direct or indirect mechanisms [44]. While direct interference occurs when the mAb itself appears as the analyte in the assay, indirect interference develops when antibodies interact with reagents or with the analyte target, thus altering test performance. They describe that, particularly in immunoassays, which rely on the precise binding of mAb assay reagents with other complex biologic antigen/analyte reagents, exogenous indirect interference may occur. For example, in transplant patients, concerns have been raised that mAbs may interfere with human leukocyte antigen (HLA) typing [44].
When daratumumab was investigated in the phase I and II studies described above, an interference with routine laboratory tests used in blood transfusion medicine was found. In patients who were receiving daratumumab, the indirect antiglobulin test (IAT; Coombs test), which is used for the detection of irregular blood group antibodies, was found to be false positive [43, 45]. This was found to be not daratumumab-specific but rather a class effect of CD38 mAbs [43]. Importantly, daratumumab does not interfere with ABO/RhD typing [45]. Daratumumab, bound to red blood cells (RBCs), only masks the detection of antibodies to minor antigens in the patient’s serum.
The agglutination that is caused by CD38 mAbs in vitro and which leads to the false-positive results in the IAT can be explained by the expression of CD38 on erythrocytes, which has previously been described [8, 11, 46, 47]. Indeed, in vitro, the binding of daratumumab to RBCs could be shown [45] and results remain positive for up to 5 months [43]. In vivo, the administration of daratumumab was shown to be associated with a clinically not significant decrease in hemoglobin of approximately 1.6 g/dL and a compensatory increase in reticulocytes; however, daratumumab did not lead to anemia [43]. The small decrease in hemoglobin is not considered to be due to complement-mediated lysis, but rather a result of Fc-receptor-mediated clearance of daratumumab-loaded RBCs in the spleen. Only a small number of patients required transfusions, which were due to the underlying disease and not a result of the binding of daratumumab to CD38 on erythrocytes. In these patients, no major transfusion-related events were observed [43, 45]. In addition, an analysis of the frequency of RBC transfusions and any transfusion-related AEs in the SIRIUS trial, in which 124 patients with very advanced disease were treated with daratumumab monotherapy, showed that there were no transfusion-related reactions in the 47 patients who required transfusions [48].
Importantly, strategies to overcome the interference of CD38 mAbs with IATs have been developed, including the denaturation of cell surface CD38 by the reducing agent dithiothreitol (DTT) and the addition of an excess of soluble CD38 or neutralizing anti-idiotype antibodies. Using these methods, irregular antibody screening and identification could be restored [43, 45].
The interference of CD38 mAbs presents an in vitro clinical laboratory challenge. In order to conduct reliable blood compatibility testing, immunohematology laboratories/transfusion medicine departments will need to be informed when a patient is receiving CD38 mAbs so that strategies can be chosen. Furthermore, it has been suggested that patients undergoing CD38 mAb therapy carry a blood transfusion card to indicate the specific therapy they are receiving [43]. One option to avoid delays in providing compatible RBC transfusions is to obtain an RBC phenotype prior to initiating daratumumab therapy, and providing phenotypically matched blood thereafter [48].
5.1.1 Practical Recommendations Based on our Experience
Daratumumab does not interfere with ABO/RhD typing; however, immunohematology laboratories/transfusion medicine departments need to be informed if patients are receiving CD38 mAbs so that the reliable strategies outlined above to overcome the interference of CD38 mAbs with IATs can be implemented. We support the suggestion by Oostendorp et al. of a patient wallet card [43]. Such a card should specify the blood profile (ABO, Rh, and IAT) determined before the first infusion of daratumumab, along with information on the IAT interference for healthcare providers/transfusion medicine departments. Patients should carry this card throughout the treatment period and for at least 6 months after the treatment ends.
The possible methods for immunohematology laboratories/transfusion medicine departments to provide safe RBCs for transfusion to patients receiving daratumumab include (1) providing ABO/RhD compatible, phenotypically or genotypically matched units, and (2) providing ABO/RhD compatible, K-negative units after ruling out or identifying allo-antibodies using DTT-treated reagent RBCs.
Uncrossmatched, ABO/RhD compatible RBC units should be administered if a transfusion is needed as an emergency, as per local transfusion medicine department practice.
5.2 Assessment of Response for Patients Receiving Daratumumab
According to the International Myeloma Working Group (IMWG) criteria, the absence of M protein on serum protein electrophoresis (SPE) and serum immunofixation electrophoresis (IFE) are requirements for a CR.
An interference with SPE has been described for several therapeutic agents, for example antibiotics and radio-opaque agents [49]. In addition, therapeutic mAbs, including chimeric human-mouse immunoglobulins (rituximab, siltuximab, infliximab, cetuximab), humanized mAbs (trastuzumab, bevacizumab, adalimumab), as well as human mAbs (ofatumumab) have been shown to lead to false-positive results on SPE [50, 51]. mAbs, as immunoglobulins, can appear on SPE and IFE, thereby preventing a distinction between therapeutic antibody and a patient’s clonal immunoglobulin, and making the validation of a CR unfeasible. It has been described that therapeutic mAbs become undetectable in patients approximately 3 months after the cessation of therapy [50].
Approximately 50 % of patients with MM produce an IgG kappa M protein, and daratumumab, an IgG1 kappa mAb, has been detected on SPE and IFE assays [52]. The concentration of daratumumab used clinically is equivalent to approximately 1 g/L and thus, for an M spike of less than 2 g/L, a distinction between daratumumab and remaining M protein is not possible, obscuring the assessment of CR and, in some patients, depending on the level of the M protein at the start of therapy, VGPRs. Similarly, for elotuzumab, which is also an IgG1 mAb, an interference of the antibody with the assessment of CR and sCR has been suggested [53]. The same is expected for the other CD38 antibodies, SAR650984 (isatuximab) and MOR202.
Therefore, a specific assay is needed to discriminate between mAb and M protein. Such an assay has been developed and validated for daratumumab [54]. It involves the addition of an anti-idiotype mAb to patient samples, which binds daratumumab. As a result, the migration of daratumumab on IFE is shifted, enabling the confirmation of CR. Further work is needed to implement the assay in routine clinical practice.
5.2.1 Practical Recommendations Based on our Experience
Consider that persisting M component may be due to the therapeutic mAb and presents a false-positive result that does not indicate recurrence or resistance to treatment. In some patients, it may be possible to identify two M spikes at different locations by carefully examining the SPE and IFE, which help to monitor the disease response, and a patient can qualify for CR if it is evident that the M spike present in the SPE or immunofixation results from the mAb interference [55]. If the patient is receiving daratumumab therapy, a specific assay has been devised to distinguish M protein and therapeutic antibody.
5.3 Infusion-Related Reactions of Monoclonal Antibody Therapy
Virtually all systemic chemotherapeutic agents can be associated with IRRs, and symptoms can vary in severity from mild to life-threatening. Patients may experience chills, fever, nausea, asthenia, headache, skin rash, or pruritus. In addition, bronchospasm, hypotension, urticaria and angioedema may also occur. Of the cytotoxic agents used in cancer therapy, taxanes and platinum agents are among those most frequently associated with infusion reactions [56, 57]. Of the agents used in myeloma, hypersensitivity reactions have been described for melphalan, pegylated liposomal doxorubicin, and carfilzomib [58–61].
The IRRs seen with mAbs are generally mild and only a small fraction of patients develop severe reactions [60, 62]. The highest risk of a reaction is during the first or second exposure to the mAb, with the risk declining with subsequent infusions [60, 62, 63]. With rituximab, approximately 77 % of patients experience IRRs during the first infusion, however only 10 % of these are severe (grade 3/4) [60]. The following infusion reactions have been described with rituximab: urticaria, hypotension, angioedema, hypoxia, pulmonary infiltrates, acute respiratory distress syndrome, myocardial infarction, ventricular fibrillation, or cardiogenic shock [57]. The majority of rituximab IRRs occur during the first infusion and decline substantially during subsequent infusions (30 % with the fourth infusion; 14 % with the eighth infusion) [60]. In clinical practice, the application of premedication, which typically consists of corticosteroids and antihistamines, is routine to reduce the risk of IRRs occurring. Close monitoring of patients is required and in the case of IRRs, an interruption of the infusion and symptom management is essential. Upon resolution of the symptoms, a restart of the infusion at 50 % of the infusion rate can be considered [57, 60].
With daratumumab, IRRs were seen in 42–71 % of patients, and occurred mostly during the first infusion [31, 32, 64]. IRRs were grade 1/2 in almost all cases, and discontinuations due to IRRs were rare. With daratumumab monotherapy, no discontinuations and no grade 4 IRRs have been seen [31, 32]. Daratumumab IRRs are characterized by nasal congestion, throat irritation, cough, dyspnoea, chills, and vomiting [31, 32]. The occurrence of IRRs can be influenced by the infusion rate, as suggested by an analysis of different infusion regimens [31]. In order to prevent the occurrence of IRRs with daratumumab, the following premedication regimen is recommended approximately 1 h prior to every daratumumab infusion: intravenous corticosteroid (methylprednisolone 100 mg or an equivalent long-acting corticosteroid for the first two infusions, and 60 mg thereafter [in the absence of IRRs in the first two infusions]), oral antipyretics (paracetamol 650–1000 mg) plus an oral or intravenous antihistamine (diphenhydramide 25–50 mg or equivalent). In our experience, the leukotriene receptor antagonist montelukast (10 mg) can be beneficial as an optional premedication. The doses of steroids used as premedication are substantially lower than the therapeutic doses typically used, such that the antimyeloma activity observed in the daratumumab single-agent studies can be attributed to daratumumab alone [64].
If IRRs occur despite the implementation of such a regimen, it is crucial to stop the daratumumab infusion immediately, even if only mild symptoms are detectable. The infusion should be halted until symptoms have resolved and the appropriate symptom management should be instigated. The administration of an antihistamine can be considered, which may shorten the time to the restart of the infusion and may also result in faster resolution of the symptoms. The restart of daratumumab infusions at a lower infusion rate following the resolution of symptoms is feasible [64]. Table 5 outlines recommendations for the management of IRRs [64].
The application of oral corticosteroids on the first and second day after all infusions (methylprednisolone, 20 mg; or equivalent in accordance with local standards) is useful to prevent the occurrence of delayed IRRs.
Special care should be taken in patients with a history of obstructive pulmonary disease for whom medications, including short- and long-acting bronchodilators and inhaled corticosteroids, can be considered. In addition, these at-risk patients may be hospitalized for monitoring for up to 2 nights after an infusion. Physicians may prescribe bronchodilators, antihistamines, and corticosteroids that are deemed necessary to provide adequate supportive care in case a bronchospasm occurs after patients are released from the hospital. The postmedication may be discontinued after the first four infusions if the patient has not experienced any major IRRs.
5.3.1 Practical Recommendations Based on our Experience
A premedication regimen administered approximately 1 h prior to every daratumumab infusion should be used to reduce the risk of IRRs occurring. The premedication should consist of an intravenous corticosteroid (methylprednisolone 100 mg or an equivalent long-acting corticosteroid), an oral antipyretic (paracetamol 650–1000 mg) plus an oral or intravenous antihistamine (diphenhydramide 25–50 mg or equivalent). For the daratumumab infusion, the use of an infusion set with a flow regulator is recommended to control the infusion rate. As IRRs mainly occur during the first infusion, it is important to be alert in order to detect early mild symptoms that mostly affect the upper respiratory tract. Prompt action is essential in case of IRRs, even if symptoms are only mild; the daratumumab infusion should be stopped immediately. The use of an antihistamine may shorten the period of treatment interruption and may lead to a faster resolution of symptoms. The infusion can be restarted at a lower rate once symptoms have resolved (Table 5). Dose modification of daratumumab is not recommended, but dose delay is the primary method for the management of side effects.
Patients with known chronic obstructive pulmonary disease (COPD) with a forced expiratory volume in 1 s (FEV1) <50 % of predicted normal, or with moderate or severe persistent asthma within the past 2 years, should not receive daratumumab. In fact, FEV1 testing should be performed for patients in whom COPD is suspected, and they must be excluded if FEV1 is <50 % of the predicted value.
6 Other Practical Recommendations
6.1 Herpes Zoster Prophylaxis
Patients with multiple myeloma have a significantly increased risk of developing viral infections, including herpes zoster (varicella zoster virus reactivation) compared with healthy controls [65]. For patients receiving daratumumab, it is recommended that herpes zoster prophylaxis be initiated within 1 week of starting daratumumab, and to continue for 3 months following treatment [40].
6.2 Fever
Following the administration of daratumumab, patients are not expected to develop fever; however, they should be advised to contact their treating physician should fever occur.
6.3 Liver Function
No dosage adjustments of daratumumab are necessary for patients with mild hepatic impairment based on a population pharmacokinetic analysis [40]. No data were available for moderate or severe hepatic impairment.
6.4 Patients with Lung Disease
In the clinical trials conducted to date, patients with lung disease were excluded because of the expression of CD38 on endothelial cells; however, it can be speculated that with careful monitoring it may be possible to treat these patients with daratumumab. Further data are needed to provide firm recommendations.
6.5 Daratumumab in Special Populations
Daratumumab single agent seems to be effective in the following special patient populations, although the low number of patients precludes strong recommendations.
6.5.1 Renal Failure
Daratumumab is not metabolized by the kidney, therefore renal failure is not a contraindication for treatment with this agent. Twelve patients with moderate renal failure (creatinine clearance ≥30 and <60 ml/min) were included in the GEN501 trial, 33 % of whom responded to treatment with daratumumab single agent [31]. In the SIRIUS trial, 42 patients had a creatinine clearance of 30–60 ml/min, and the ORR in these patients was 26.2 % [32]. The number of patients with severe renal impairment was too low in the two studies to make any statement.
6.5.2 Advanced Age
In the GEN501 study, daratumumab was administered to 16 patients aged 65–74 years, 56 % of whom responded [31], while none of the four patients who were 75 years or older responded. In the SIRIUS trial, 36 patients were aged between 65 and <75 years, while 12 patients were 75 years or older, respectively. The ORR in these subgroups of patients was 25 and 33.3 %, respectively, indicating that the efficacy of daratumumab is comparable in young or elderly MM patients [32].
6.5.3 Extramedullary Disease
Fourteen patients included in the SIRIUS trial had extramedullary disease, 21.4 % of whom achieved at least PR, indicating that daratumumab single agent might be effective in patients with disease involvement outside the bone marrow [32].
6.6 Retreatment with Daratumumab
Early results indicate that retreatment with daratumumab after previous treatment with this agent may be feasible. Alici et al. reported the results for two patients with triple-refractory disease (IMiDs, PIs, cytostatic drugs) who achieved a response to initial daratumumab therapy but relapsed after treatment [66]. They rechallenged with daratumumab and observed a PR in both patients. Although these results will need to be confirmed in larger patient groups, they suggest that retreatment with daratumumab is feasible and effective.
7 Future Directions
Daratumumab is currently being investigated in a number of trials (Table 2) [67]. In the relapsed/refractory setting, two large, international, phase III trials investigating daratumumab in combination with current standard treatments have been initiated. In the CASTOR trial, the combination of daratumumab, bortezomib, and dexamethasone is being compared with bortezomib/dexamethasone alone. Approximately 480 participants will be randomly assigned to receive either daratumumab plus bortezomib/dexamethasone or bortezomib/dexamethasone alone. The second large, international trial in the relapsed/refractory setting is the POLLUX trial, in which daratumumab is being added to lenalidomide/dexamethasone and compared with lenalidomie/dexamethasone alone. It is planned to enrol approximately 570 patients. In both trials, the primary outcome measure is PFS.
Large, international, phase III trials are also ongoing in the frontline setting; two trials have been initiated in the non-transplant setting. In the ALCYONE trial, in which it is planned to enrol approximately 700 patients, daratumumab is being added to VMP and compared with VMP alone. In both arms, nine cycles of therapy are initially administered. In the daratumumab arm, this is followed by continuous daratumumab until PD, unacceptable toxicity, or study end. In the MAIA study, daratumumab is combined with lenalidomide/dexamethasone and compared with lenalidomide/dexamethasone alone. In both arms, treatment will be administered until PD or unacceptable toxicity, and approximately 730 patients will be enrolled. PFS is the primary outcome measure in both trials.
The Intergroupe Francophone du Myelome (IFM) and the Dutch/Belgium Haemato-Oncology Foundation for Adults in the Netherlands (HOVON) are cooperating in a trial to investigate daratumumab in the transplant setting (CASSIOPEIA). Patients will be randomized to receive either VTD or VTD + daratumumab as induction and consolidation. Following a second randomization step, patients will either receive daratumumab maintenance therapy or will only be observed. Over 1000 patients will be included in the trial and the primary outcome measures are sCR after consolidation therapy and PFS after maintenance therapy.
There are also ongoing trials to investigate daratumumab in smoldering myeloma and other hematological malignancies, as well as a subcutaneous formulation of the agent.
This expansive clinical development illustrates that daratumumab is expected to have a significant impact on the treatment of multiple myeloma across the different treatment lines and patient spectra, and the trials outlined above will help to define the optimal place for daratumumab in the treatment of myeloma.
8 Conclusions
CD38 mAbs represent an exciting recent addition to the therapeutic armamentarium in multiple myeloma. It is hoped that their incorporation into current treatment strategies will enable a more effective and targeted approach with improved outcomes. Results from daratumumab clinical trials conducted to date have been overwhelmingly positive, demonstrating the substantial efficacy and good tolerability of the agent. There are a number of particular considerations regarding the administration and management of daratumumab and the CD38 mAbs in general in the treatment of myeloma. We have outlined some of these in the present article with the aim of providing our recommendations and learnings from the clinical trials for their management so that the incorporation of these agents into routine practice is as straightforward as possible to enable our patients to benefit from this highly effective new strategy.
References
Grillo-López AJ, White CA, Dallaire BK, et al. Rituximab: the first monoclonal antibody approved for the treatment of lymphoma. Curr Pharm Biotechnol. 2000;1(1):1–9.
Sehn LH, Donaldson J, Chhanabhai M, Fitzgerald C, Gill K, Klasa R, et al. Introduction of combined CHOP plus rituximab therapy dramatically improved outcome of diffuse large B-cell lymphoma in British Columbia. J Clin Oncol. 2005;23(22):5027–33.
Majithia N, Rajkumar SV, Lacy MQ, Buadi FK, Dispenzieri A, Gertz MA, et al. Outcomes of primary refractory multiple myeloma and the impact of novel therapies. Am J Hematol. 2015;90(11):981–5.
Kumar SK, Lee JH, Lahuerta JJ, et al. Risk of progression and survival in multiple myeloma relapsing after therapy with IMiDs and bortezomib: a multicenter International Myeloma Working Group study. Leukemia. 2012;26:149–57.
Laubach JP, Tai YT, Richardson PG, Anderson KC. Daratumumab granted breakthrough drug status. Expert Opin Investig Drugs. 2014;23(4):445–52.
US FDA. FDA approves Darzalex for patients with previously treated multiple myeloma. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm472875.htm. Accessed 25 Mar 2016.
The Myeloma Beacon™. Janssen submits marketing authorisation application for daratumumab for European patients with heavily pre-treated multiple myeloma. http://www.myelomabeacon.com/pr/2015/09/09/daratumumab-european-marketing-authorization-application/. Accessed 25 Mar 2016.
Mehta K, Shahid U, Malavasi F. HumanCD38, a cell-surface protein with multiple functions. FASEB J. 1996;10(12):1408–17.
Funaro A, Malavasi F. Human CD38, a surface receptor, an enzyme, an adhesion molecule and not a simple marker. J Biol Regul Homeost Agents. 1999;13(1):54–61.
Malavasi F, Deaglio S, Funaro A, Ferrero E, Horenstein AL, Ortolan E, et al. Evolution and function of the ADP ribosyl cyclase/CD38 gene family in physiology and pathology. Physiol Rev. 2008;88(3):841–86.
Deaglio S, Mehta K, Malavasi F. Human CD38: a (r)evolutionary story of enzymes and receptors. Leuk Res. 2001;25(1):1–12.
Quarona V, Ferri V, Chillemi A, Bolzoni M, Mancini C, Zaccarello G, et al. Unraveling the contribution of ectoenzymes to myeloma life and survival in the bone marrow niche. Ann N Y Acad Sci. 2015;1335:10–22.
Deaglio S, Vaisitti T, Aydin S, Ferrero E, Malavasi F. In-tandem insight from basic science combined with clinical research: CD38 as both marker and key component of the pathogenetic network underlying chronic lymphocytic leukemia. Blood. 2006;108(4):1135–44.
Partida-Sánchez S, Cockayne DA, Monard S, Jacobson EL, Oppenheimer N, Garvy B, et al. Cyclic ADP-ribose production by CD38 regulates intracellular calcium release, extracellular calcium influx and chemotaxis in neutrophils and is required for bacterial clearance in vivo. Nat Med. 2001;7(11):1209–16.
Partida-Sánchez S, Randall TD, Lund FE. Innate immunity is regulated by CD38, an ecto-enzyme with ADP-ribosyl cyclaseactivity. Microbes Infect. 2003;5(1):49–58.
Fedele G, Di Girolamo M, Recine U, Palazzo R, Urbani F, Horenstein AL, et al. CD38 ligation in peripheral blood mononuclear cells of myeloma patients induces release of protumorigenic IL-6 and impaired secretion of IFNγ cytokines and proliferation. Mediat Inflamm. 2013;2013:564687.
Terstappen LW, Huang S, Safford M, Lansdorp PM, Loken MR. Sequential generations of hematopoietic colonies derived from single nonlineage-committed CD34 + CD38-progenitor cells. Blood. 1991;77(6):1218–27.
Theilgaard-Mönch K, Raaschou-Jensen K, Schjødt K, Heilmann C, Vindeløv L, Jacobsen N, et al. Pluripotent and myeloid-committed CD34+ subsets in hematopoietic stem cell allografts. Bone Marrow Transpl. 2003;32(12):1125–33.
Lin P, Owens R, Tricot G, Wilson CS. Flow cytometric immunophenotypic analysis of 306 cases of multiple myeloma. Am J Clin Pathol. 2004;121(4):482–8.
Santonocito AM, Consoli U, Bagnato S, Milone G, Palumbo GA, Di Raimondo F, et al. Flow cytometric detection of aneuploidCD38(++) plasmacells and CD19(+) B-lymphocytes in bone marrow, peripheral blood and PBSC harvest in multiple myeloma patients. Leuk Res. 2004;28(5):469–77.
Chillemi A, Zaccarello G, Quarona V, Ferracin M, Ghimenti C, Massaia M, et al. Anti-CD38 antibody therapy: windows of opportunity yielded by the functional characteristics of the target molecule. Mol Med. 2013;19:99–108.
de Weers M, Tai YT, van der Veer MS, Bakker JM, Vink T, Jacobs DC, et al. Daratumumab, a novel therapeutic human CD38 monoclonal antibody, induces killing of multiple myeloma and other hematological tumors. J Immunol. 2011;186(3):1840–8.
Overdijk MB, Verploegen S, Bögels M, van Egmond M, Lammerts van Bueren JJ, Mutis T, et al. Antibody-mediated phagocytosis contributes to the anti-tumor activity of the therapeutic antibody daratumumab in lymphoma and multiplemyeloma. MAbs. 2015;7(2):311–21.
Jansen JH, Boross P, Overdijk MB, van Bueren JJ, Parren PW, Leusen JH. Daratumumab, a human CD38 antibody induces apoptosis of myeloma tumor cells via Fc receptor-mediated crosslinking [abstract no. 2974]. American Society of Hematology Proceedings 2012. Blood. 2012;120(21):2974.
Lammerts van Bueren J, Jakobs D, Kaldenhoven N, Roza M, Hiddingh S, Meesters J, et al. Direct in vitro comparison of daratumumab with surrogate analogs of CD38 antibodies MOR03087, SAR650984 and Ab79 [abstract no. 3474]. Blood. 2014;124(21):3474.
Phipps C, Chen Y, Gopalakrishnan S, Tan D. Daratumumab and its potential in the treatment of multiple myeloma: overview of the preclinical and clinical development. Ther Adv Hematol. 2015;6(3):120–7.
Krejcik J, Casneuf T, Nijhof I, Verbist B, Bald J, Plesner T, et al. Immunomodulatory effects and adaptive immune response to daratumumab in multiple myeloma [abstract no. 3037]. In: American society of hematology 57th annual meeting and exposition, 5–8 Dec 2015, Orlando (FL).
van der Veer MS, de Weers M, van Kessel B, Bakker JM, Wittebol S, Parren PW, et al. Towards effective immunotherapy of myeloma: enhanced elimination of myeloma cells by combination of lenalidomide with the human CD38 monoclonal antibody daratumumab. Haematologica. 2011;96(2):284–90.
van der Veer MS, de Weers M, van Kessel B, Bakker JM, Wittebol S, Parren PW, et al. The therapeutic human CD38 antibody daratumumab improves the anti-myeloma effect of newly emerging multi-drug therapies. Blood Cancer J. 2011;1(10):e41.
Nijhof IS, Groen RW, Noort WA, van Kessel B, de Jong-Korlaar RA, Bakker JM, et al. Preclinical evidence for the therapeutic potential of CD38-targeted immuno-chemotherapy in multiple myeloma patients refractory to lenalidomide and bortezomib. Clin Cancer Res. 2015;21(12):2802–10.
Lokhorst HM, Plesner T, Laubach JP, Nahi H, Gimsing P, Hansson M, et al. Targeting CD38 with daratumumab monotherapy in multiple myeloma. N Engl J Med. 2015;373(13):1207–19.
Lonial S, Weiss BM, Usmani SZ, Singhal S, Chari A, Bahlis NJ, et al. Daratumumab monotherapy in patients with treatment-refractory multiple myeloma (SIRIUS): an open-label, randomised, phase 2 trial. Lancet. Epub. 2016. doi:10.1016/S0140-6736(15)01120-4.
Usmani S, Weiss B, Bahlis NJ, Belch A, Lonial S, Lokhorst H, et al. Clinical efficacy of daratumumab monotherapy in patients with heavily pretreated relapsed or refractory multiple myeloma [abstract no. 29]. In: American Society of Hematology 57th Annual Meeting and Exposition, 5–8 Dec 2015, Orlando (FL).
Usmani S, Ahmadi T, Ng Y, Lam A, Potluri R, Mehra M. Analyses of real world data on overall survival in multiple myeloma patients with at least 3 prior lines of therapy including a PI and an IMiD, or double refractory to a PI and an IMiD [abstract no. 4498]. In: American Society of Hematology 57th Annual Meeting and Exposition, 5–8 Dec 2015, Orlando (FL).
Plesner T, Arkenau HT, Lokhorst HM, Gimsing P, Krejcik J, Lemech C, et al. Safety and efficacy of daratumumab with lenalidomide and dexamethasone in relapsed or relapsed, refractory multiple myeloma [abstract no. 84]. Blood. 2014;124(21):84.
Plesner T, Arkenau HT, Gimsing P, Krejcik J, Lemech C, Minnema MC, et al. Daratumumab in combination with lenalidomide and dexamethasone in patients with relapsed or relapsed and refractory multiple myeloma: updated results of a phase 1/2 study (GEN503) [abstract no. 507]. Blood. 2015;126(23):507.
Mateos MV, Moreau P, Comenzo R, Bladé J, Benboubker L, de la Rubia J, et al. An open-label, multicenter, phase 1b study of daratumumab in combination with pomalidomide-dexamethasone and with backbone regimens in patients with multiple myeloma [abstract no. P275]. In: European Hematology Association 20th Congress, 11–14 Jun 2015, Vienna.
Moreau P, Mateos MV, Bladé J, et al. An open-label, multicenter, phase 1b study of daratumumab in combination with backbone regimens in patients with multiple myeloma [abstract no. 176]. In: American Society of Hematology 56th Annual Meeting and Exposition, 6–9 Dec 2014, San Francisco (CA).
Chari A, Lonial S, Suvannasankha A, Fay JW, Arnulf B, Ifthikharuddin JJ, et al. Open-label, multicenter, phase 1b study of daratumumab in combination with pomalidomide and dexamethasone in patients with at least 2 lines of prior therapy and relapsed or relapsed and refractory multiple myeloma [abstract no. 508]. In: American Society of Hematology 57th Annual Meeting and Exposition, 5–8 Dec 2015, Orlando (FL).
Darzalex™ (daratumumab) injection for intravenous infusion 100 mg/5 mL, 400 mg/20 mL. https://www.darzalex.com. Accessed 25 Mar 2016.
Kroll MH, Elin RJ. Interference with clinical laboratory analyses. Clin Chem. 1994;40(11 Pt 1):1996–2005.
King RI, Florkowski CM. How paraproteins can affect laboratory assays: spurious results and biological effects. Pathology. 2010;42(5):397–401.
Oostendorp M, Lammerts van Bueren JJ, Doshi P, Khan I, Ahmadi T, Parren PW, et al. When blood transfusion medicine becomes complicated due to interference by monoclonal antibody therapy. Transfusion. 2015;55(6 Pt 2):1555–62.
Ostrov BE, Amsterdam D. The interference of monoclonal antibodies with laboratory diagnosis: clinical and diagnostic implications. Immunol Invest. 2013;42(8):673–90.
Chapuy CI, Nicholson RT, Aguad MD, Chapuy B, Laubach JP, Richardson PG, et al. Resolving the daratumumab interference with blood compatibility testing. Transfusion. 2015;55(6 Pt 2):1545–54.
Zocchi E, Franco L, Guida L, Benatti U, Bargellesi A, Malavasi F, et al. A single protein immunologically identified as CD38 displays NAD1 glycohydrolase, ADP-ribosylcyclase and cyclic ADP-ribosehydrolase activities at the outer surface of human erythrocytes. Biochem Biophys Res Commun. 1993;196:1459–65.
Albeniz I, Demir O, Türker-Sener L, Yalçintepe L, Nurten R, Bermek E. Erythrocyte CD38 as a prognostic marker in cancer. Hematology. 2007;12(5):409–14.
Chari A, Satta T, Tayal A, Jagannath S, Cho HJ, Parekh S, et al. Outcomes and management of red blood cell transfusions in multiple myeloma patients treated with daratumumab [abstract no. 3571]. In: American Society of Hematology 57th Annual Meeting and Exposition, 5–8 Dec 2015, Orlando (FL).
Bossuyt X. Interferences in clinical capillary zone electrophoresis of serum proteins. Electrophoresis. 2004;25:1485–7.
McCudden CR, Voorhees PM, Hainsworth SA, Whinna HC, Chapman JF, Hammett-Stabler CA, et al. Interference of monoclonal antibody therapies with serum protein electrophoresis tests. Clin Chem. 2010;56(12):1897–9.
Genzen JR, Kawaguchi KR, Furman RR. Detection of a monoclonal antibody therapy (ofatumumab) by serum protein and immunofixation electrophoresis. Br J Haematol. 2011;155(1):123–5.
Axel AE. McCudden CR, Xie H, Hall BM, Sasser AK. Development of clinical assay to mitigate daratumumab, an IgG1κ monoclonal antibody, interference with serum immunofixation (IFE) and clinical assessment of M-protein response in multiple myeloma [abstract no. 2563]. In: Proceedings of the American Association for Cancer Research (AACR) Annual Meeting 2014, 5–9 April 2014, San Diego (CA).
Lonial S, Dimopoulos M, Palumbo A, White D, Grosicki S, Spicka I, et al. ELOQUENT-2 Investigators. Elotuzumab therapy for relapsed or refractory multiple myeloma. N Engl J Med. 2015;373(7):621–31.
McCudden C, Axel A, Slaets D, Frans S, Bald J, Schecter JM, et al. Assessing clinical response in multiple myeloma (MM) patients treated with monoclonal antibodies (mAbs): validation of a daratumumab IFE reflex assay (DIRA) to distinguish malignant M-protein from therapeutic antibody [abstract no. 8590]. J Clin Oncol. 2015;33:8590.
Durie BGM, San Miguel JF, Blade J, Rajkumar SV, on behalf of the International Myeloma Working Group. Clarification of the definition of complete response in multiple myeloma. Leukemia. 2015;29(12):2416–7.
Zanotti KM, Markman M. Prevention and management of antineoplastic-induced hypersensitivity reactions. Drug Saf. 2001;24:767–79.
Lenz HJ. Management and preparedness for infusion and hypersensitivity reactions. Oncologist. 2007;12(5):601–9.
Cornwell GG 3rd, Pajak TF, McIntyre OR. Hypersensitivity reactions to ivmelphalan during treatment of multiple myeloma: cancer and Leukemia Group B experience. Cancer Treat Rep. 1979;63(3):399–403.
Chanan-Khan A, Szebeni J, Savay S, Liebes L, Rafique NM, Alving CR, et al. Complement activation following first exposure to pegylated liposomal doxorubicin (Doxil): possible role in hypersensitivity reactions. Ann Oncol. 2003;14(9):1430–7.
Chung CH. Managing premedications and the risk for reactions to infusional monoclonal antibody therapy. Oncologist. 2008;13(6):725–32.
Siegel DS. From clinical trials to clinical practice: single-agent carfilzomib adverse events and their management in patients with relapsed and/or refractory multiple myeloma. Ther Adv Hematol. 2013;4(6):354–65.
Guan M, Zhou YP, Sun JL, Chen SC. Adverse events of monoclonal antibodies used for cancer therapy. Biomed Res Int. 2015;2015:428169.
Meisel K, Rizvi S. Complications of monoclonal antibody therapy. Med Health R I. 2011;94(11):317–9.
Voorhees P, Weiss B, Usmani S, Huaibao F, Uhlar CM, Khan I, et al. Management of infusion-related reactions following daratumumab monotherapy in patients with ≥3 lines of prior therapy or double refractory multiple myeloma (MM): 54767414MMY2002 (SIRIUS) [abstract no. 1829]. Blood. 2015;126(23):1829
Blimark C, Holmberg E, Mellqvist UH, Landgren O, Björkholm M, Hultcrantz M, et al. Multiple myeloma and infections: a population-based study on 9253 multiple myeloma patients. Haematologica. 2015;100(1):107–13.
Alici E, Chrobok M, Lund J, Ahmadi T, Khan I, Duru AD, et al. Re-challenging with anti-CD38 monotherapy in triple-refractory multiple myeloma patients is a feasible and safe approach. Br J Haematol. Epub. 2015. doi:10.1111/bjh.13776.
ClinicalTrials.gov, a service of the US National Institutes of Health. http://www.clinicaltrials.gov. Accessed 25 Mar 2016.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Medical writing and editorial support was provided by Pia Sondergeld (University of Giessen, Germany) and was funded by Janssen.
Conflicts of interest
Philippe Moreau received fees for advisory board participation from Janssen, Celgene, Bristol–Myers Squibb, Novartis, Takeda, and Amgen. Niels W. C. J. van de Donk received a research Grant from Celgene, Janssen Pharmaceuticals, and Amgen. Jesus San Miguel received fees for advisory board participation from Millennium, Celgene, Novartis, Onyx, Janssen, Bristol–Myers Squibb, and Merck Sharp and Dohme. Henk Lokhorst received fees for attending advisory boards on daratumumab (Janssen). Michele Cavo received honoraria from Janssen, Cilag, Takeda, and Bristol–Myers Squibb, and payment for lectures from Janssen, Cilag, and Takeda. Gordon Cook received Grants from Janssen, Celgene, Takeda, and Chugai-Pharmaceutical Co.; consulting fees or honorarium from Janssen, Celgene, Takeda, Sanofi, Bristol–Myers Squibb, Glycomimetics, and Amgen; support for travel from Janssen, Celgene, Takeda, Sanofi; fees for participation in review activities from Celgene, and Glycomimetics; and payment for lectures from Janssen, Celgene, Takeda, Sanofi, Bristol–Myers Squibb, and Amgen. Michel Delforge received a Grant from Celgene (phase IV clinical trial), and honoraria from Amgen, Celgene, Janssen, Novartis, and Takeda. Hermann Einsele received consulting fees or honorarium, payment for lectures, and support for travel from Janssen, Celgene, Amgen, and Novartis. Sonja Zweegman received honoraria for advisory board participation from Celgene, Takeda, Janssen, and Novartis (honorarium for departmental use, not personal use), and lectures for Celgene and Janssen (payment for departmental use only). Heinz Ludwig received fees for participation in data monitoring committees of early daratumumab studies. Christoph Driessen received consulting honorarium related to daratumumab from Janssen–Cilag (not related to the current manuscript). Antonio Palumbo received consulting fees and honoraria from Janssen–Cilag (not related to the current manuscript). Thierry Facon received fees for advisory board participation, as well as support for travel and payment for lectures from Janssen. He undertakes advisory board activities with Janssen and is on the speaker’s bureau for Janssen. Torben Plesner received a research Grant from Janssen, honoraria for advisory board participation from Genmab and Janssen, and payment for lectures from Janssen. Meletios Dimopoulos has received consulting fees or honoraria from Janssen, Celgene, Novartis, and Amgen. Pia Sondergeld received payment from Janssen for the writing of this manuscript. Pieter Sonneveld received Grants from Amgen, Celgene, Janssen, and Karyopharm; consulting fees or honoraria from Amgen, Celgene, Janssen, and Karyopharm; fees for participation in review activities from Celgene and Janssen; provision of writing assistance from Janssen; and payment for lectures from Amgen, Celgene, and Janssen. He has been involved in drug research, i.e. clinical trials, both investigator-initiated and company-initiated, and has served on advisory boards and given lectures in the context of new drug development for multiple myeloma. Maria-Victoria Mateos has received honoraria for lectures and participation in advisory boards from Janssen, Celgene, Takeda, Amgen, and Bristol–Myers Squibb. Hareth Nahi and Dina Ben-Yehuda have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Moreau, P., van de Donk, N.W.C.J., Miguel, J.S. et al. Practical Considerations for the Use of Daratumumab, a Novel CD38 Monoclonal Antibody, in Myeloma. Drugs 76, 853–867 (2016). https://doi.org/10.1007/s40265-016-0573-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-016-0573-4