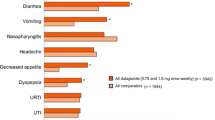
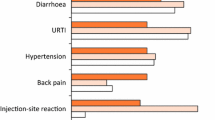
Abstract
Subcutaneous liraglutide (Victoza®), a glucagon-like peptide 1 receptor agonist, is approved for the treatment of adult patients with type 2 diabetes mellitus. Once-daily liraglutide, as monotherapy or add-on therapy to other antidiabetic agents (including basal insulin), was an effective and generally well tolerated treatment in adult patients with type 2 diabetes in several well-designed phase III trials and in the real world clinical practice setting. In addition to improving glycaemic control, liraglutide had beneficial effects on bodyweight, systolic blood pressure and surrogate measures of β-cell function in clinical trials, with these benefits maintained during long-term treatment (≤2 years). Liraglutide has a convenient once-daily administration regimen, a low potential for drug–drug interactions and low propensity to cause hypoglycaemia. Thus, liraglutide continues to be a useful option for the management of type 2 diabetes. This article reviews the therapeutic use of liraglutide in adult patients with type 2 diabetes and summarizes its pharmacological properties.

Similar content being viewed by others
References
American Diabetes Association. Standards of medical care in diabetes: 2014. Diabetes Care. 2014;37(Suppl 1):S14–80.
Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycemia in type 2 diabetes: a patient-centered approach. Position statement of the American Diabetes Association (ADA) and European Association for the Study of Diabetes (EASD). Diabetes Care. 2012;35(6):1364–79.
Garber AJ, Abrahamson MJ, Barzilay JI, et al. AACE comprehensive diabetes management algorithm. Endocr Pract. 2013;19(2):327–36.
American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2012;35(Suppl 1):S64–71.
International Diabetes Federation. Diabetes: facts and figures. 2013. http://www.idf.org/worlddiabetesday/toolkit/gp/facts-figures. Accessed 22 Aug 2014.
International Diabetes Federation. Morbidity and mortality. 2009. http://www.idf.org/diabetesatlas/diabetes-mortality. Accessed 22 Aug 2014.
International Diabetes Federation. Global diabetes plan 2011–2021. 2011. http://www.idf.org/sites/default/files/Global_Diabetes_Plan_Final.pdf. Accessed 30 Aug 2014.
Barnett AH. The role of GLP-1 mimetics and basal insulin analogues in type 2 diabetes mellitus: guidance from studies of liraglutide. Diabetes Obes Metab. 2012;14(4):304–14.
Nauck M. Incretin-based therapies for type 2 diabetes mellitus: properties, functions, and clinical implications. Am J Med. 2011;124(Suppl):S3–18.
Perry CM. Liraglutide: a review of its use in the management of type 2 diabetes mellitus. Drugs. 2011;71(17):2347–73.
Croom KF, McCormack PL. Liraglutide: a review of its use in type 2 diabetes mellitus. Drugs. 2009;69(14):1985–2004.
Novo Nordisk A/S. Victoza liraglutide (rDNA origin) injection: US prescribing information. 2013. http://www.novo-pi.com/victoza.pdf. Accessed 20 Aug 2014.
Novo Nordisk A/S. Victoza: EU summary of product characteristics. 2014. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/001026/WC500050017.pdf. Accessed 20 Aug 2014.
Flint A, Kapitza C, Zdravkovic M. The once-daily human GLP-1 analogue liraglutide impacts appetite and energy intake in patients with type 2 diabetes after short-term treatment. Diabetes Obes Metab. 2013;15(10):958–62.
Horowitz M, Flint A, Jones KL, et al. Effect of the once-daily human GLP-1 analogue liraglutide on appetite, energy intake, energy expenditure and gastric emptying in type 2 diabetes. Diabetes Res Clin Pract. 2012;97(2):258–66.
Hermansen K, Baekdal TA, During M, et al. Liraglutide suppresses postprandial triglyceride and apolipoprotein B48 elevations after a fat-rich meal in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled, cross-over trial. Diabetes Obes Metab. 2013;15(11):1040–8.
Buse JB, Garber A, Rosenstock J, et al. Liraglutide treatment is associated with a low frequency and magnitude of antibody formation with no apparent impact on glycemic response or increased frequency of adverse events: results from the Liraglutide Effect and Action in Diabetes (LEAD) trials. J Clin Endocrinol Metab. 2011;96(6):1695–702.
Chatterjee DJ, Khutooryansky N, Zdravkovic M, et al. Absence of QTc prolongation in a thorough QT study with subcutaneous liraglutide, a once-daily GLP-1 analog for treatment of type 2 diabetes. J Clin Pharmacol. 2009;49(11):1353–62.
Klein DJ, Battelino T, Chatterjee DJ, et al. Liraglutide’s safety, tolerability, pharmacokinetics, and pharmacodynamics in pediatric type 2 diabetes: a randomized, double-blind, placebo-controlled trial. Diabetes Technol Ther. 2014;16(10):1–9.
Garber A, Henry R, Ratner R, et al. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): a randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet. 2009;373(9662):473–81.
Nauck M, Frid A, Hermansen K, et al. Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin, in type 2 diabetes: the LEAD (Liraglutide Effect and Action in Diabetes)-2 study. Diabetes Care. 2009;32(1):84–90.
Marre M, Shaw J, Brandle M, et al. Liraglutide, a once-daily human GLP-1 analogue, added to a sulphonylurea over 26 weeks produces greater improvements in glycaemic and weight control compared with adding rosiglitazone or placebo in subjects with type 2 diabetes (LEAD-1 SU). Diabet Med. 2009;26(3):268–78.
Zinman B, Gerich J, Buse JB, et al. Efficacy and safety of the human glucagon-like peptide-1 analog liraglutide in combination with metformin and thiazolidinedione in patients with type 2 diabetes (LEAD-4 Met + TZD). Diabetes Care. 2009;32(7):1224–30.
Pratley RE, Nauck M, Bailey T, et al. Liraglutide versus sitagliptin for patients with type 2 diabetes who did not have adequate glycaemic control with metformin: a 26-week, randomised, parallel-group, open-label trial. Lancet. 2010;375(9724):1447–56.
Yang W, Chen L, Ji Q, et al. Liraglutide provides similar glycaemic control as glimepiride (both in combination with metformin) and reduces body weight and systolic blood pressure in Asian population with type 2 diabetes from China, South Korea and India: a 16-week, randomized, double-blind, active control trial. Diabetes Obes Metab. 2011;13(1):81–8.
Kaku K, Rasmussen MF, Clauson P, et al. Improved glycaemic control with minimal hypoglycaemia and no weight change with once-daily human glucagon-like peptide-1 analogue liraglutide as add-on to sulphonylurea in Japanese patients with type 2 diabetes. Diabetes Obes Metab. 2010;12(4):341–7.
Seino S, Rasmussen MF, Nishida T, et al. Efficacy and safety of once-daily human GLP-1 analogue, liraglutide, vs glibenclamide monotherapy in Japanese patients with type 2 diabetes. Curr Med Res Opin. 2010;26(5):1013–22.
Buse JB, Rosenstock J, Sesti G, et al. Liraglutide once a day versus exenatide twice a day for type 2 diabetes: a 26-week randomised, parallel-group, multinational, open-label trial (LEAD-6). Lancet. 2009;374(9683):39–47.
Mathieu C, Rodbard HW, Cariou B, et al. A comparison of adding liraglutide versus a single daily dose of insulin aspart to insulin degludec in subjects with type 2 diabetes (BEGIN: VICTOZA ADD-ON). Diabetes Obes Metab. 2014;16(7):636–44.
DeVries JH, Bain SC, Rodbard HW, et al. Sequential intensification of metformin treatment in type 2 diabetes with liraglutide followed by randomized addition of basal insulin prompted by A1C targets. Diabetes Care. 2012;35:1446–54.
Russell-Jones D, Vaag A, Schmitz O, et al. Liraglutide vs insulin glargine and placebo in combination with metformin and sulfonylurea therapy in type 2 diabetes mellitus (LEAD-5 met + SU): a randomised controlled trial. Diabetologia. 2009;52(10):2046–55.
Aroda VR, Bailey T, Cariou B, et al. The effect of insulin degludec in combination with liraglutide and metformin in patients with type 2 diabetes requiring treatment intensification [abstract no. 145]. Diabetologia. 2014;57(Suppl 1):S68.
Lahtela J, Ahmann A, Rodbard H, et al. Efficacy and safety of liraglutide vs. placebo when added to basal insulin analgoues in subjects with type 2 diabetes [abstract no. 37]. Diabetologia. 2014;57(Suppl 1):S21–2.
Umpierrez G, Atkin S, Bain S, et al. Efficacy and safety of liraglutide versus placebo in subjects with type 2 diabetes and moderate renal impairment (LIRA-RENAL): a randomised trial [abstract no. 182]. Diabetologia. 2014;57(Suppl 1):S84.
Garber A, Henry RR, Ratner R, et al. Liraglutide, a once-daily human glucagon-like peptide 1 analogue, provides sustained improvements in glycaemic control and weight for 2 years as monotherapy compared with glimepiride in patients with type 2 diabetes. Diabetes Obes Metab. 2011;13(4):348–56.
Kaku K, Rasmussen MF, Nishida T, et al. Fifty-two-week, randomized, multicenter trial to compare the safety and efficacy of the novel glucagon-like peptide-1 analog liraglutide vs glibenclamide in patients with type 2 diabetes. J Diabetes Invest. 2011;2(6):441–7.
Nauck M, Frid A, Hermansen K, et al. Long-term efficacy and safety comparison of liraglutide, glimepiride and placebo, all in combination with metformin in type 2 diabetes: 2-year results from the LEAD-2 study. Diabetes Obes Metab. 2013;15(3):204–12.
Pratley R, Nauck M, Bailey T, et al. One year of liraglutide treatment offers sustained and more effective glycaemic control and weight reduction compared with sitagliptin, both in combination with metformin, in patients with type 2 diabetes: a randomised, parallel-group, open-label trial. Int J Clin Pract. 2011;65(4):397–407.
Ross SA, Ballantine J. Early use of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) in type 2 diabetes. Curr Med Res Opin. 2013;29(12):1617–26.
Seino S, Rasmussen MF, Nishida T, et al. Glucagon-like peptide-1 analog liraglutide in combination with sulfonylurea safely improves blood glucose measures vs sulfonylurea monotherapy in Japanese patients with type 2 diabetes: results of a 52-week, randomized controlled trial. J Diabetes Invest. 2011;2(4):280–6.
Buse JB, Sesti G, Schmidt WE, et al. Switching to once-daily liraglutide from twice-daily exenatide further improves glycemic control in patients with type 2 diabetes using oral agents. Diabetes Care. 2010;33(6):1300–3.
Rosenstock J, Rodbard HW, Bain SC, et al. One-year sustained glycemic control and weight reduction in type 2 diabetes after addition of liraglutide to metformin followed by insulin detemir according to HbA1c target. J Diabetes Complicat. 2013;27(5):492–500.
Nyeland ME, Ploug UJ, Skovgaard R, et al. Comparative effectiveness of liraglutide versus sitagliptin in type 2 diabetes in the United Kingdom: a retrospective study in primary care [abstract no. PDB3]. Value Health. 2013;16(7):A431.
DiGenio A, Ling Z, Davis KL, et al. Real-world characteristics of patients with type 2 diabetes initiating insulin glargine plus exenatide or insulin glargine plus liraglutide [abstract no. 797]. Diabetologia. 2012;55(Suppl 1):S328.
Gupta PS, Thong KY, Armstrong M, et al. Does liraglutide therapy affect the metabolic response in patients with elevated alanine aminotransferase and type 2 diabetes mellitus? The ABCD nationwide liraglutide audit [abstract no. 1037-P]. Diabetes. 2013;62(Suppl):A267.
Heymann A, Maor Y, Goldstein I, et al. Efficacy of liraglutide in a real-life cohort. Diabetes Ther. 2014;5(1):193–206.
Lee WC, Dekoven M, Bouchard J, et al. Improved real-world glycaemic outcomes with liraglutide versus other incretin-based therapies in type 2 diabetes. Diabetes Obes Metab. 2014;16(9):819–26.
Saunders W, Hiep Nguyen H, Kalsekar I. Real-world comparative effectiveness of exenatide once-weekly and liraglutide in patients with type 2 diabetes mellitus [abstract no. 828]. Diabetologia. 2014;57(Suppl 1):S335–6.
Wangnoo SK, Mithal A, Kumar S, et al. Safety and efficacy of liraglutide in type 2 diabetes: post marketing surveillance data from India [abstract no. 732]. Diabetologia. 2012;55(Suppl 1):S299.
Levin PA, Buysman E, Thayer S, et al. The INITIATOR study: real-world treatment patterns and outcomes in patients with type 2 diabetes initiating insulin glargine or liraglutide [abstract no. 111]. Diabetologia. 2014;57(Suppl 1):S53–4.
Madani S, Charpentier G, Martinez L, et al. Effectiveness of switching from a DPP-4 inhibitor to the human GLP-1 analogue liraglutide in patients with type 2 diabetes: data from the EVIDENCE study [abstract no. 832]. Diabetologia. 2014;57(Suppl 1):S337.
Gourdy P, Penfornis A, Charpentier G, et al. EVIDENCE 1 [abstract no. 840]. Diabetologia. 2014;57(Suppl 1):S341.
Zhang Y, Hu G, Yuan Z, et al. Glycosylated hemoglobin in relationship to cardiovascular outcomes and death in patients with type 2 diabetes: a systematic review and meta-analysis. PLoS One. 2012;7(8):e42551.
Stolar M. Glycemic control and complications in type 2 diabetes mellitus. Am J Med. 2010;123:S3–11.
Stolar M. Addressing cardiovascular risk in patients with type 2 diabetes: focus on primary care. Am J Med Sci. 2011;341(2):132–40.
European Medicines Agency. Summary of opinion (initial authorization): Xultophy (insulin degludec/liraglutide). 2014. http://ec.europa.eu/health/documents/community-register/html/h947.htm#EndOfPage. Accessed 1 Oct 2014.
Marso SP, Poulter NR, Nissen SE, et al. Liraglutide effect and action in diabetes: evaluation of cardiovascular outcome and results (LEADER) trial design and methods [abstract no. 74]. Diabetologia. 2013;56(Suppl 1):S37.
Giorda CB, Sacerdote C, Nada E, et al. Incretin-based therapies and acute pancreatitis risk: a systematic review and meta-analysis of observational studies. Endocrine. 2014. doi:10.1007/s12020-014-0386-8.
Butler AE, Campbell-Thompson M, Gurlo T, et al. Marked expansion of exocrine and endocrine pancreas with incretin therapy in humans with increased exocrine pancreas dysplasia and the potential for glucagon-producing neuroendocrine tumors. Diabetes. 2013;62(7):2595–604.
Yang L, He Z, Tang X, et al. Type 2 diabetes mellitus and the risk of acute pancreatitis: a meta-analysis. Eur J Gastroenterol Hepatol. 2013;25(2):225–31.
Singh S, Chang H-Y, Richards TM, et al. Glucagonlike peptide 1-based therapies and risk of hospitalization for acute pancreatitis in type 2 diabetes mellitus: a population-based matched case-control study. JAMA Intern Med. 2013;173(7):534–9.
Nauck M. A critical analysis of the clinical use of incretin-based therapies: the benefits by far outweigh the potential risks. Diabetes Care. 2013;36(7):2126–32.
Nauck M, Friedrich N. Do GLP-1-based therapies increase cancer risk? Diabetes Care. 2013;36(Suppl 2):S245–S52.
Brodovicz KG, Kou TD, Alexander CM, et al. Impact of diabetes duration and chronic pancreatitis on the association between type 2 diabetes and pancreatic cancer risk. Diabetes Obes Metab. 2012;14(12):1123–8.
Egan AG, Blind E, Dunder K, et al. Pancreatic safety of incretin-based drugs: FDA and EMA assessment. N Engl J Med. 2014;370(9):794–7.
Zueger PM, Schultz NM, Lee TA. Cost effectiveness of liraglutide in type II diabetes: a systematic review. Pharmacoeconomics. 2014. doi:10.1007/s40273-014-0192-4.
Disclosure
The preparation of this review was not supported by any external funding. During the peer review process, the manufacturer of the agent under review was offered an opportunity to comment on this article. Changes resulting from comments received were made by the authors on the basis of scientific and editorial merit. Lesley J. Scott is a salaried employee of Adis/Springer.
Author information
Authors and Affiliations
Corresponding author
Additional information
The manuscript was reviewed by: S.C. Bain, Swansea University, Institute of Life Science, Swansea, UK; D.S.H. Bell, Southside Endocrinology, University of Alabama, Birmingham, AL, USA; J.G. Eriksson, University of Helsinki, Department of General Practice and Primary Health Care, Finland and Helsinki University Central Hospital, Unit of General Practice, Helsinki, Finland; B.B. Yeap, University of Western Australia, School of Medicine and Pharmacology, Fremantle and Fiona Stanley Hospitals, Perth, WA, Australia.
Rights and permissions
About this article
Cite this article
Scott, L.J. Liraglutide: A Review of Its Use in Adult Patients with Type 2 Diabetes Mellitus. Drugs 74, 2161–2174 (2014). https://doi.org/10.1007/s40265-014-0321-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-014-0321-6