Abstract
Introduction
Biosimilar CT-P13 was approved with limited data from clinical trials compared to the originator infliximab in biologic-naïve patients with rheumatoid arthritis. Three prospective post-marketing surveillance studies have been conducted in Japanese biologic-naïve patients and switched patients from biologics including the originator infliximab.
Objective
We performed an integrated analysis of final data from three post-marketing studies to provide long-term safety and efficacy data of CT-P13 in a real-world clinical setting.
Methods
A total of 1816 patients consisting of 987 patients with rheumatoid arthritis, 342 patients with Crohn’s disease, 322 patients with ulcerative colitis, and 165 patients with psoriasis were evaluated for safety. Efficacy was assessed in 1150 patients whose disease parameter values were serially collected.
Results
Adverse drug reactions were reported in 24.2% of all patients. The incidence of adverse drug reactions differed by the prior treatment status with biologics: 30.5% in patients naïve to biologics, 17.0% in patients switched from the originator infliximab, and 33.5% in patients switched from other biologics. Infusion reactions were the most frequent adverse drug reactions (8.2%), and its incidence was significantly higher in patients with ulcerative colitis and an allergy history in a multivariable Cox regression analysis. Infection was the second most frequent (6.1%), but tuberculosis only occurred in four patients (0.2%). The incidence of infection was low in patients with Crohn’s disease and psoriasis, and significant risk factors were an allergy history, comorbidities, and concomitant steroid use. Interstitial lung disease occurred in 16 patients (0.9%), including 11 patients with rheumatoid arthritis. With CT-P13 therapy, disease activity parameters decreased similarly in all four diseases, although long-term drug discontinuation rates because of inefficacy varied by disease. In naïve patients, the disease activity parameters decreased rapidly and the proportion of patients in remission increased. Patients switched from infliximab maintained lowered parameter levels with infliximab pretreatment. Decreases were also observed in patients switched from other biologics, but discontinuations were most often because of insufficient efficacy.
Conclusions
The integrated analysis of a large number of patients detected no new safety signals with long-term CT-P13 treatment. Efficacy in rheumatoid arthritis, psoriasis, Crohn’s disease, and ulcerative colitis cases was confirmed in biologic-naïve patients and switched patients from the originator infliximab or other biologics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The integrated analysis of 1816 patients treated with biosimilar CT-P13 showed a similar safety profile to the originator infliximab and no additional safety concerns. Risk factors for infusion reactions and infections were identified by a multivariable analysis. |
The primary efficacy of CT-P13, accessed by changes in disease activity parameters, was confirmed across four immune-mediated inflammatory diseases. Long-term efficacy, accessed by drug discontinuation because of inefficacy, differed by disease. |
In biologic-naïve patients, CT-P13 exhibited a sufficient effect. In patients who switched from infliximab, the reduced disease activity was maintained, adverse reactions were infrequent, and drug persistence was high. Even in patients who relapsed on other biologics, CT-P13 also showed good primary efficacy, although drug persistence was relatively low. |
1 Introduction
The pathogenesis of immune-mediated inflammatory diseases is characterized by dysregulation of key cytokines such as tumor necrosis factor (TNF)-α, interleukin-23, and interleukin-17. The introduction of biologics targeting these molecules, beginning with infliximab (IFX), has revolutionized the treatment of these diseases. However, the high cost of biologics has limited patient accessibility. In response, biosimilars have emerged after the expiration of patents as a healthcare economics-driven solution aimed at reducing the financial burden on patients and enabling broader access to effective treatment options. CT-P13 is the first biosimilar of IFX, a therapeutic chimeric antibody to TNF-α. Its intravenous formulation showed similar pharmacokinetic/pharmacodynamic profiles with IFX and was approved in 2013 in the European Union [1, 2]. The subsequently developed subcutaneous formulation of CT-P13 demonstrated an improved pharmacokinetic profile compared with intravenous IFX and is convenient for patients because of its suitability for self-injection obviating the need for hospital visits, which is of special importance in the COVID-19 era [3, 4]. It was first approved in the European Union in 2019, but approval requirements and approved indications differ among regulatory authorities in each country because of the differences in administration routes and pharmacokinetics. On the contrary, all indications for the originator IFX were granted for the intravenous formulation based on limited data from clinical trials conducted in a small number of patients with rheumatoid arthritis (RA) [5, 6]. Therefore, post-approval studies of intravenous CT-P13 are needed to confirm a similar safety profile to that of the originator IFX in real-world settings and therapeutic efficacy in patients with various types of disease. The post-marketing surveillance (PMS) of CT-P13 was initiated in 2014 in patients with four immune-mediated inflammatory diseases as an approval condition in Japan. One PMS was conducted in a large number of patients with RA to confirm the results of the small clinical trials in patients with RA. Other PMSs were conducted in patients with Crohn’s disease (CD), ulcerative colitis (UC), and psoriasis (PS), for which CT-P13 use was approved by extrapolation without clinical trials. Three interim reports on the PMS in patients with CD, UC, and RA, and one final report on the PMS in patients with PS had been published [7,8,9,10]. In these reports, the safety profile was shown to be similar to the safety profile of the originator IFX, and adverse drug reactions (ADRs) of special interest were confirmed to be infusion reactions (IRs), infections including tuberculosis, and interstitial lung disease (ILD). However, the number of patients in each PMS was not sufficient to provide definitive results on infrequent or long-term ADRs. Therefore, we performed an integrated analysis of the anonymized final data after completion of all PMSs, which were conducted with similar protocols (especially for safety) and by the same operating procedures. Our aim in this study was to address the following clinical issues that are important for biosimilar use but have not been thoroughly investigated in clinical trials: (1) to determine the long-term safety profile of CT-P13 and the incidence of rare ADRs such as tuberculosis and ILD, (2) to identify risk factors for frequent ADRs such as IRs and infection by multivariable analyses, (3) to confirm the efficacy of CT-P13 in immune-mediated inflammatory diseases other than RA, and (4) to investigate the safety and efficacy of the biosimilar in patients who switched from the originator IFX or other biologics.
2 Methods
2.1 Study Design
In this study, patient data collected in three independent PMSs of CT-P13 were pooled and analyzed. In the PMS of RA (Code IFX 11), patients who received the first intravenous administration of CT-P13 between January 2015 and November 2020 were monitored during the surveillance period of 1 year. Patients with CD and UC who started CT-P13 treatment between January 2015 and April 2017 were followed for 2 years in one PMS (Code IFX 21). The PMS of PS (Code IFX 22) enrolled patients from May 2015 to January 2020 and followed them for 1 year. All three PMSs were conducted by Nippon Kayaku Co., Ltd. (Tokyo, Japan) after approval of CT-P13 in Japan. The participating patients should have been unsuccessfully treated with non-biologic therapies, and CT-P13 was administered intravenously at the approved dosage per the guidelines for use of a TNF inhibitor for each disease (see Electronic Supplementary Material [ESM] for details). To minimize patient selection bias, we prospectively enrolled all consecutive patients at contracted clinical sites. Information on prior treatment and use of concomitant medications with CT-P13 was collected. The patients were classified into three groups based on biologic use prior to PMS entry: (1) patients naïve to biologics and treated with CT-P13 as the first biologic therapy; (2) patients who had been treated with the originator IFX and switched to CT-P13 for mainly non-medical reasons such as the reduction in drug cost burden and hospital policy, and (3) patients who had received other biologics and switched to CT-P13 for medical reasons such as adverse events (AEs) and inadequate response. Patients with RA and PS were followed up for 1 year, and patients with CD and UC for 2 years after the first administration of CT-P13. The PMS was conducted in accordance with the Good Post-Marketing Study Practice and Good Vigilance Practice Ordinances of the Ministry of Health, Labour and Welfare and their related rules.
2.2 Safety
All AEs including subjective/objective findings and laboratory test data were collected and the causal relationship of AEs to the study drug, CT-P13, was evaluated. Adverse events and ADRs, for which a causal relationship with CT-P13 was not ruled out, were coded in accordance with the System Organ Class and Preferred Term listed in the Medical Dictionary for Regulatory Activities (MedDRA/J; version 25.0). Adverse drug reactions of particular interest were IRs, infections, tuberculosis, ILD, and malignant neoplasms.
2.3 Efficacy Parameters
The efficacy of CT-P13 was assessed by using representative disease activity parameters for each disease. Disease Activity Score in 28 joints with C-reactive protein (DAS28-CRP) was used for RA, and its cut-off values for low, moderate, and high disease activity were 2.6, 3.2, and 5.1, respectively. Remission was defined as a DAS28-CRP score < 2.6 [11]. Similarly, the Crohn’s Disease Activity Index was used for CD with three cut-off values of 150, 220, and 450 dividing disease activity into four grades [12]. The partial Mayo score was used for UC, and the disease severity was classified into four levels with scores of 0–1, 2–4, 5–7, and 8–9 [13]. The absolute Psoriasis Area Severity Index was used to grade PS severity, which is divided into four categories: Psoriasis Area Severity Index ≤ 2, ≤ 5, ≤ 10, and > 10 [14, 15]. The data obtained on the nearest day to the scheduled administration timepoints (on weeks 2, 6, and every 8 weeks thereafter for naïve patients to IFX; and every 8 weeks for patients switched from IFX to CT-P13) were used as the representative values.
2.4 Efficacy Evaluation
The primary efficacy was assessed by the change in the proportion of patients at each disease activity level from baseline to 6 months after administration (the nearest day between weeks 10 and 32). The long-term efficacy was assessed by discontinuation of CT-P13 therapy because of insufficient efficacy. Cumulative hazards of drug discontinuation were plotted by the Nelson–Aalen method and differences between patient groups were analyzed by the log-rank test. The persistence of CT-P13 was also plotted by the Kaplan–Meier method with treatment discontinuation for any reason as an event. The duration of treatment was calculated as (date of last administration) − (date of first administration) + 1. Therefore, if a patient discontinued treatment any day after the first dosing, the treatment duration is 1 day.
2.5 Statistical Analysis
Multivariable analysis was performed using the Cox regression model to explore risk factors associated with the incidence of IRs and infections. A similar multivariable analysis was also applied for predictive factors associated with the discontinuation of CT-P13 administration because of insufficient efficacy. As explanatory variables, five categorized patient factors (target disease, sex, CRP values, history of allergy, comorbidities), three continuous patient factors (age, disease duration, body mass index), and two categorized therapeutic factors (prior biologics, combined steroid use for the target disease) were incorporated. Sex and age were forced into the multivariable Cox model, and the remaining variables with p < 0.2 were selected by the stepwise method. The Cox regression model was also used for the univariate analysis of risk factors for the occurrence of ILD. The hazard ratio (HR) and its two-sided Wald 95% confidence intervals (CIs) were estimated for each covariate. P values <0.05 were considered statistically significant.
3 Results
3.1 Patients
A total of 1915 patients were enrolled in the three PMSs, and 1816 patients (95%) who received at least one dose were included in this integrated analysis, as shown in Fig. 1. By disease, 987 patients (54%) had RA, 342 patients (19%) had CD, 322 patients (18%) had UC, and 165 patients (9%) had PS. Among patients with PS, 104 (63%) had psoriatic arthritis. Patients were classified into three groups according to their prior treatment status with biologics: 725 patients (40%) were naïve to biologics, 894 patients (49%) switched from the originator IFX to CT-P13, and 197 patients (11%) switched from other biologic treatment. Of these, 1150 patients (63%), who had a pre-treatment and post-treatment dataset of disease activity parameters used for the efficacy evaluation, were included in the analysis of primary efficacy. Excluding patients who discontinued treatment early, this population has slightly lower disease activity (Table S1 of the ESM), but is acceptable for the primary efficacy analyses performed separately for each disease and patient subgroup.
Flow diagram of disposition of patients included in the integrated analysis. aPatients without pre-treatment and post-treatment dataset on efficacy for rheumatoid arthritis (RA) [Disease Activity Score in 28 joints with C-reactive protein], Crohn’s disease (CD) [Crohn’s Disease Activity Index], ulcerative colitis (UC) [partial Mayo score], and psoriasis (PS) [Psoriasis Area and Severity Index]. IFX infliximab
The baseline characteristics of 1816 patients in the integrated analysis varied by disease (Table 1). The proportion of male individuals was low in patients with RA (24%) but around 70% in patients with other diseases. The mean age was 51.9 years, with patients with RA having the highest age at 58.8 years and patients with CD having the lowest at 37.9 years. Patients with PS had a longer disease duration of 15.8 years at the start of CT-P13 therapy, a higher body mass index, and a higher proportion of smokers. Comorbidities occurred in 45% of patients overall and at a higher incidence in patients with RA and PS. A history of ILD and other respiratory comorbidities were similarly skewed in RA and PS and rare in CD and UC.
Regarding the prior biologics therapy, the proportion of naïve patients was particularly high in UC (54%), while that of switched patients from IFX was high in CD and PS (both 64%). The proportion of patients who switched from other biologics was approximately 10% in all diseases. Steroid use for target diseases was 31% overall, and the proportion of steroid use for each disease corresponded to CRP values. Concomitant medications were disease specific, with high prescribing rates of topical steroids for PS, methotrexate for RA, and immunosuppressants and 5-aminosalicylic acid for CD and UC.
3.2 Drug Persistence
Of the 1816 patients, 1079 patients (59%) completed the study period of CT-P13 therapy (1 year for RA and PS, 2 years for CD and UC). Kaplan–Meier plots showed that drug survival was longest in CD and shortest in UC, with a 2-year persistence of 57% and 46% for CD and UC, respectively (Fig. 2a). The 2-year persistence rate in naïve patients was 53%, whereas the rate was higher in patients who switched from IFX (68%) and lower in patients switched from other biologics (42%) (Fig. 2b).
Kaplan–Meier plot of persistence of CT-P13 therapy in patients classified by a disease and b prior treatment status with biologics, and c reasons for drug discontinuation during the follow-up period of 1 year for rheumatoid arthritis (RA) and psoriasis (PS) and 2 years for Crohn’s disease (CD) and ulcerative colitis (UC). IFX infliximab, *p < 0.05; **p < 0.01; ***p < 0.001 by the log-rank test
Treatment was discontinued in 737 patients (41%) during the follow-up period for various reasons (Fig. 2c). Inefficacy was the most common reason for discontinuation in 267 patients (15%), while 36 patients (2%) achieved remission leading to drug discontinuation. Because of AEs, 184 patients (10%) had to discontinue treatment. A total of 84 patients (5%) discontinued treatment because of a patient’s decision or hospital policy. This high proportion is characteristic of patients taking biosimilars, along with an additional 64 patients (4%) who switched back to the originator IFX.
Discontinuations because of inefficacy were more common in UC and PS (19% for both) and discontinuations because of AEs were higher in UC (14%). Compared with naïve patients and patients who switched from other biologics, patients who switched from IFX had fewer discontinuations because of insufficient efficacy or AEs.
3.3 Safety
The incidence of AEs in all 1816 patients was 34.9%, including 7.6% with serious AEs (Table 2). Among them, ADRs and serious ADRs were observed in 24.2% and 4.6%, respectively. Adverse drug reactions leading to the discontinuation of CT-P13 were 9.3%, and four patients (0.2%) died of ADRs (two patients with interstitial pneumonia, one patient with pancytopenia followed by mesenteric hemorrhage and pneumonia, and 1 patient with an unknown cause of death).
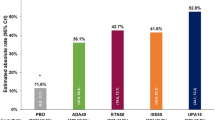
The incidence of ADRs by disease was 22.7% in RA, 24.0% in CD, 32.3% in UC, and 17.6% in PS. By prior treatment, the incidence of ADRs was 30.5% in naïve patients, lower in switched patients from IFX (17.0%), and higher in switched patients from other biologics (33.5%). All the reported ADRs and serious ADRs of CT-P13 by System Organ Class are listed in Table S2 in the ESM.
3.3.1 Infusion Reactions
Infusion reactions were the most frequent ADRs in 8.2% (149/1816) of patients and 1.5% of all infusions (232 times per 15,043 infusions). Most IRs were not serious except in 13 patients (0.7%), but 83 patients (4.6%) discontinued further infusions of CT-P13. A multivariable analysis for IRs was performed with baseline factors from an integrated analysis of 1816 patients using the Cox regression model (Table 3). The incidence of IRs was significantly lower in switched patients from IFX than in naïve patients (HR = 0.55, 95% CI 0.35–0.85, p = 0.007) and significantly higher in switched patients from other biologics (HR = 2.06, 95% CI 1.30–3.26 p = 0.002). Significant associations of IRs with UC (HR to RA = 2.10, 95% CI 1.30–3.37, p = 0.002) and a history of allergy (HR = 1.81, 95% CI 1.21–2.96, p = 0.004) were observed. High CRP levels (≥ 0.5 mg/dL) also tended to be associated with the occurrence of IRs (HR = 1.42, 95% CI 0.98–2.07, p = 0.066). Steroid use for the treatment of the target disease had no effect on the occurrence of first IRs (both 1.0% of infusions). Steroids (mainly injectables) were also used for the prevention of IRs in 46 patients (336 infusions), but surprisingly the incidence of IRs was increased, affecting up to 23.9% of the patients and 3.3% of infusions (odds ratio = 3.51, 95% CI 1.87–6.60, p < 0.001). However, in 81 patients who continued CT-P13 treatment after recovery from the first IRs, prophylactic use of steroids significantly suppressed second IRs to 8.1% of infusions (8/99 infusions in 25 patients) compared with 19.4% (26/134 infusions in 42 patients) without prophylaxis (odds ratio = 0.37, 95% CI 0.16–0.85, p = 0.019) (Table S3 of the ESM).
Regarding infusion time, 52% of all CT-P13 administrations were infused in 120 (± 15) min, and the incidence of IRs was 1.5% (232/15043 infusion) in this standard infusion time. Whereas by rapid infusion in 60 (± 15) min employed in 25% of administrations, IRs occurred with a similar incidence of 1.4% (108/7858 infusions). Additionally, among patients switched from IFX, 20% of patients received the first infusion of CT-P13 in 60 min, and the incidence of IRs was 2/176 patients (1.1%), which was not higher than 7/493 patients (1.4%) infused in 120 min.
3.3.2 Infection
The next most common ADR was infection, reported in 110 patients (6.1%). Serious infection occurred in 33 patients (1.8%), and 30 patients (1.7%) discontinued the administration of CT-P13. Frequently reported infections were pneumonia (22 patients, 1.2%), nasopharyngitis (21 patients, 1.2%), and herpes zoster (15 patients, 0.8%) (Table S4 of the ESM). The incidence of infection varied by disease and was lower in patients with CD and PS. In fact, in the Cox multivariable analysis, HRs of infection were significantly lower in patients with CD and PS compared with patients with RA (HR = 0.46, 95% CI 022–0.98, p = 0.044, and HR = 0.11, 95% CI 0.02–0.83, p = 0.032, respectively) (Table 3). Previous IFX therapy was also associated with a lower incidence of infection (HR = 0.57, 95% CI 0.36–0.89, p = 0.014). On the contrary, significant risk factors for the onset of infection were a history of allergy (HR = 1.68, 95% CI 1.05–2.66, p = 0.029), the presence of comorbidities (HR = 2.07, 95% CI 1.34–3.19, p = 0.001), and concomitant steroid use for the target disease (HR = 1.95, 95% CI 1.29–2.96, p = 0.002). Further investigation by type of comorbidity showed a significantly higher incidence of infections in patients with pre-existing infections, immune system disorders, anemia, and cardiac disorders (Table S5 of the ESM). Although no significant association was detected with age, which was included as a continuous variable in 1-year increments in the multivariable analysis, the incidence of infection increased sharply to 8.4% in elderly patients aged ≥ 65 years (41/486 patients).
3.3.3 Tuberculosis
Four cases (0.2%) of tuberculosis were among the reported infections. Two out of four patients had a history of tuberculosis and tested positive on either the tuberculin skin test or interferon-gamma release assay, but showed no abnormalities on chest imaging. The other two patients were negative on a prior examination. None received prophylactic anti-tuberculosis treatment (see Table S6 of the ESM for details).
A prior examination was performed in 92.4% of patients, and at an especially high rate (99.4%) in patients with PS (Table S7 of the ESM). The examination rate prior to CT-P13 therapy was high in biologic-naïve patients (98.1%), but only 86.7% in switched patients from IFX. There were 9.3% of patients who were positive by at least one of the examinations. In patients with PS, the positivity rate was as high as 17.1% (not only because of the high examination rate). Prophylactic administration of anti-tuberculosis drugs such as isoniazid was performed in 2.9% of patients and in 14.0% of high-risk patients who had positive examination results or a tuberculosis history.
3.3.4 Interstitial Lung Disease
Interstitial lung disease occurred in 16 patients at a 0.9% incidence. There were 11 (1.1%) and three (1.8%) patients with RA and PS, respectively, and only one (0.3%) each with CD and UC (Table 2). Of the 16 patients, four and two patients had a history of ILD and complications of other respiratory diseases, respectively. Their mean age was 63.8 years, and five patients (31%) had a smoking history. Methotrexate was used concomitantly in 12 patients (75%). A univariate Cox analysis showed a significantly higher HR of ILD in elderly patients with age ≥ 65 years, higher CRP ≥ 0.5 mg/dL, comorbidity, and a history of ILD (Table S8 of the ESM). Smoking history and methotrexate use also had a more than two-fold higher HR, although no statistical significance was observed (p = 0.105, 0.170). Of 16 patients, 13 patients were confirmed to have recovered, but two patients died of ILD.
3.4 Efficacy
The efficacy assessment was based on the change in the disease activity parameters for each disease, namely DAS28-CRP for RA, Crohn’s Disease Activity Index for CD, partial Mayo score for UC, and Psoriasis Area Severity Index for PS. In naïve patient groups, each parameter decreased rapidly after the administration of CT-P13, and clear efficacy for all four diseases was confirmed. In patients switched from IFX, the parameters that had been lowered with previous IFX treatment were maintained and further suppressed with continued administration of CT-P13. In patients switched from other biologics, the parameters also decreased after CT-P13 administration, although the decrease was slower than that observed in naïve patients (Fig. S1 of the ESM).
3.4.1 Primary Efficacy
In all four diseases, similar changes from before to 6 months after treatment were observed in the proportion of patients in four disease activity levels classified by disease parameter (Fig. 3). In naïve patient groups, CT-P13 showed distinct primary efficacy that sharply increased remissions and reduced the number of patients with moderate-to-high disease activity. In patient groups switched from IFX, the remission rate was more than two thirds at baseline with prior IFX treatment, and further increased 6 months after CT-P13 administration. The distribution of patients switched from other biologics changed similarly to that of the biologic-naïve patient groups, with an increased rate of remission. By employing the same analytical method, the primary efficacy of CT-P13 was confirmed in all diseases regardless of differences in disease and disease activity parameters.
Primary efficacy of CT-P13 assessed by changes in disease activity parameters from baseline to 6 months after the start of CT-P13 administration in three patient groups. Distribution of patients according to four disease activity levels based on a Disease Activity Score in 28 joints with C-Reactive Protein (DAS28-CRP) for rheumatoid arthritis (RA), b Crohn’s Disease Activity Index (CDAI) for Crohn’s disease (CD), c partial Mayo score for ulcerative colitis (UC), and d Psoriasis Area and Severity Index (PASI) for psoriasis (PS) [see methodological details described in the text]. IFX infliximab
3.4.2 Long-Term Efficacy
The cumulative hazards of drug discontinuation because of inefficacy (change of treatment) during the long-term follow-up period are plotted by the Nelson–Aalen method to examine the long-term efficacy of CT-P13. Discontinued hazards because of inefficacy increased gradually in all diseases. The increase was slower in CD, followed by RA, and higher in PS and UC (Fig. 4a). Treatment discontinuation because of inefficacy was the lowest in patients switched from IFX, followed by naïve patients, and most common in patients switched from other biologics (Fig. 4b). Additional data on the impact of differences in prior biologic therapy in patients switched from other biologics are presented in Fig. S2 of the ESM.
Nelson–Aalen plot of discontinuation of CT-P13 therapy because of inefficacy in each patient group classified by a disease and b prior treatment status, and c multivariable Cox regression analysis of baseline factors associated with discontinuation because of inefficacy. BMI body mass index, CD Crohn’s disease, CI confidence interval, CRP C-reactive protein, HR hazard ratio, IFX infliximab, PS psoriasis, RA rheumatoid arthritis, UC ulcerative colitis, *p < 0.05; **p < 0.01, ***p < 0.001
By assessing discontinuation hazards across four diseases, prognostic factors for the long-term efficacy of CT-P13 were detected by a multivariable analysis (Fig. 4c). In addition to disease and prior therapy, a higher CRP level was significantly associated with drug discontinuation (HR = 2.05, 95% CI 1.52–2.79, p < 0.001).
4 Discussion
In this report on the integrated analysis of three PMSs involving 1816 patients with four immune-mediated inflammatory diseases, we provided real-world evidence for the clinical issues that were left unresolved by the limited clinical trials with patients with RA and small post-marketing observational studies. First, the clear primary efficacy of CT-P 13 was confirmed in patients with CD, UC, and PS, as well as patients with RA. The decrease in disease activity parameters with CT-P13 therapy showed the same pattern across all four diseases, and the changes in the proportion of responding patients in the first 6 months of treatment were also similar. However, the long-term efficacy of CT-P13, assessed by treatment discontinuation because of inefficacy, varied by disease. This difference in discontinuation rate primarily reflects disease susceptibility to CT-P13, but is also influenced by baseline disease activity and available treatment options for each disease. In fact, baseline CRP, which corresponds to disease activity, was the strongest risk factor for treatment discontinuation in the multivariable analysis. The increase in the number of available treatment options might lower the barrier to changing CT-P13 to another therapy. Approved biologics are limited for CD, which is associated with a low discontinuation rate, and many novel biologics have been approved for UC and PS, which are associated with high discontinuation rates. This is consistent with reported findings that the persistence of biologics, including IFX, had declined over the years as new therapeutics were developed and treatment options increased [16, 17]. Aggressive changes in treatment to achieve treatment goals according to the treat-to-target strategy may accelerate the decline in drug survival [18,19,20].
Second, switching from IFX to CT-P13 did not cause any particular therapeutic difficulties and could be recommended in all diseases. Patients who switched from IFX had a significantly lower incidence of ADRs than naïve patients, probably because susceptible patients had already discontinued IFX before registration in this PMS. CT-P13 maintained the efficacy of previous treatment with IFX and further increased the proportion of responders, resulting in the highest persistence rate in all diseases. CT-P13 also showed primary efficacy to increase the remission rate in switched patients who had failed other biologic therapies, as well as initial therapy in biologic-naïve patients. However, during the follow-up period, discontinuation because of inefficacy and ADRs (especially IRs) occurred frequently, and the persistence rate was lower than that in naïve patients. Switching to other biologics is a common strategy when the first one fails, even though decreased efficacy among second-line treated patients was described [21]. CT-P13, which exhibited primary efficacy after switching, may be an option for treating patients with a biologic relapse, although expectations of long-term efficacy may be excessive. The finding that switching was more effective when using one prior therapy (than two or more biologics), or when the duration of prior biologic therapy was longer, has implications for treatment decisions and patient selection. In addition, when the reason for switching was intolerance, efficacy of CT-P13 was higher compared with failure because of inefficacy, although ADRs were more frequent. Moreover, surprisingly, there was no difference in the efficacy and safety whether the previous therapy was a TNF inhibitor or other molecular targeted biologics (Fig. S2 of the ESM).
Third, in this long-term PMS, no new safety signals were detected, and the safety profile and incidence of ADRs of CT-P13 were similar to those with the originator IFX described in previous Japanese PMSs [22, 23]. Among reported ADRs, IRs and infections were particularly frequent as observed in clinical trials and observational studies of CT-P13 [1, 2, 6, 22,23,24,25]. For IRs, a history of allergy was identified as a strong risk factor by a multivariable Cox regression analysis. An infusion reaction is a type of drug-induced allergy, and is associated with the induction of anti-IFX antibodies because of high antigenicity of the chimeric human-murine antibody. Monitoring anti-IFX antibody appearance improved the efficacy of IFX but is not routine in clinical practice [26], therefore checking the allergy history prior to an infusion may help manage IRs. Compared with naïve patients, patients switched from IFX had a lower HR for IR (0.55), likely owing to the exclusion of patients who developed IR to previous IFX. Conversely, patients switched from other biologics had a higher HR (2.06), possibly owing to a higher proportion of patients with a history of drug allergy in the course of previous biologic treatment (18.8% vs 5.7% of naïve patients). Steroid premedication was expected to prevent IRs but prior use of steroids was ineffective and even worse at preventing IRs. This unexpected result may be due to selection bias of high-risk patients for steroid use (especially in switched patients), and is in line with the results of a meta-analysis and a recent registry study indicating that premedication was not associated with a reduced risk of IRs [27, 28]. However, in patients who continued CT-P13 therapy after recovering from the first IRs, steroid premedication showed significant suppression of the subsequent second IRs, which occurred very frequently without any intervention (Table S3 of the ESM). Therefore, steroid premedication has a potential preventive efficacy and is useful for patients with a history of drug allergy. For infection, an allergy history, concomitant steroid use, and comorbidities were shown to be risk factors by the multivariable Cox regression analysis. Increased infection risk with allergy and steroid administration is well known, and a few reports showed a significant association of comorbidities with infection in patients treated with TNF inhibitors [29]. Older age was often reported to be a risk factor for infection, but age in 1-year increments was not associated with the onset of infection in a multivariable analysis. However, the incidence of infection was higher in elderly patients aged 65 years or older, consistent with previous reports [29]. Careful examination and continued observation are required for elderly patients having these risk factors for infection.
Fourth, this integrated analysis with a large number of patients allowed precise examination of the infrequent but important ADRs, tuberculosis, and ILD. The incidence of tuberculosis was 0.2%, which was lower than the incidence of 0.3% in the 6-month observation period with the originator IFX previously reported in 5000 patients with RA [22]. The incidence of tuberculosis in patients receiving TNF inhibitors has been reported to correlate only with the country-wide prevalence of latent tuberculosis, regardless of the patient’s sex, age, or disease [30]. The recent decline in tuberculosis incidence in Japan may have been reflected in the lower incidence of tuberculosis in patients with CT-P13. The incidence of tuberculosis with CT-P13 in this Japanese PMS (0.2%) was between the incidence in South Korea (0.71%) and the incidence in the European Union (0.04%) [24], corresponding to a high prevalence of tuberculosis in Asian countries, a low prevalence in Western countries, and an intermediate prevalence in Japan [31]. The rate of prior examination for tuberculosis in this PMS was high (Table S7 of the ESM), but tuberculosis developed in four patients, two of whom had been positive in the examination. Prophylactic administration of anti-tuberculosis drugs for high-risk patients and regular examinations are still necessary. Interstitial lung disease occurred in 16 (0.9%) patients, including 11 patients with RA (1.1%) and three patients with PS (1.8%). These incidences were higher than the incidences reported with the originator IFX in Japanese patients with RA (0.5%) and PS (0.26%) [22, 23]. The difference is probably owing to the longer observation period for our PMS (1 year vs 6 months) and older age of the enrolled patients (mean age: 58.8 vs 55.1 years in RA, and 53.8 vs 49.7 in PS), in addition to the recent increased awareness of drug-induced ILD. The RA-biased incidence may be explained by the fact that ILD is a frequent extra-articular manifestation of RA [32]. It has also been suggested that ILD is associated with PS, but less frequently [33]. A univariate Cox analysis showed a significantly higher incidence in patients with older age, higher CRP values, and a history of ILD. Smoking history and concomitant methotrexate use also tended to increase the incidence (Table S8 of the ESM). Of 16 patients who developed ILD, 12 patients had serious disease, and two patients (12.5%) died. A bibliographical search reported a higher mortality rate of 29% in 122 reported cases of ILD (89% with RA) from 39 studies on TNF inhibitors [34]. A higher risk of death was also reported in patients with older age (over 65 years), a history of ILD, and the use of immunosuppressants, which are in line with risk factors for ILD development in our analysis. Because of the poor prognosis of ILD, adequate attention and careful examination of respiratory symptoms should be exercised, especially in elderly patients with RA and PS with these potential risk factors.
Additionally, through the integrated analysis of all patients across four immune-mediated inflammatory diseases, we observed the following significant differences in responses to CT-P13 among diseases: (1) the incidence of IRs was relatively higher in UC; (2) infection was lower in CD and PS; (3) ILD occurred more frequently in RA and PS; (4) treatment discontinuation because of inefficacy was higher in PS; and (5) overall drug survival was highest in CD and lowest in UC. These differences among diseases may be related not only to baseline disease severity, patient age, and concomitant medications (such as steroids and immunosuppressants), but also to the involvement of TNF in the immunopathogenesis of each disease. Furthermore, differences in drug persistence are associated with available therapeutic options and treatment strategies for the disease [16, 17].
5 Limitations
The integrated analysis has some limitations. First, there are inherent differences in patient data because of disease differences. Efficacy parameters, in particular, were inevitably different depending on the disease, but we attempted to evaluate them in the same way across diseases for primary efficacy. In addition, long-term efficacy was evaluated by drug discontinuation as an event irrelevant to disease. Second, the PMSs for the integrated analysis had a single-arm design, and comparisons with the originator were made with previously reported data. Therefore, changes in patient backgrounds and treatment system during this period make comparative results less conclusive. Finally, data collected in real-world PMS are sparse compared with clinical trial data. Conversely, however, the collection of a sufficiently large amount of data from various types of patients has yielded valuable information reflective of real clinical practice.
6 Conclusions
In this integrated analysis, the incidence and types of ADRs were comparable to those of the originator IFX with no additional safety concerns. In naïve patients, CT-P13 had a sufficient effect on all four diseases, as shown in clinical trials in patients with RA. Switching from IFX to CT-P13 maintained efficacy, and discontinuations because of ADRs or inefficacy were few. Even in patients who relapsed on other biologics, CT-P13 also showed primary efficacy, although drug persistence was relatively low. In conclusion, as a cost-effective biosimilar, CT-P13 could be useful for the treatment of immune-mediate inflammatory diseases in biologic-naïve patients and switched patients from biologics.
References
Park W, Hrycaj P, Jeka S, Kovalenko V, Lysenko G, Miranda P, et al. A randomised, double-blind, multicentre, parallel-group, prospective study comparing the pharmacokinetics, safety, and efficacy of CT-P13 and innovator infliximab in patients with ankylosing spondylitis: the PLANETAS study. Ann Rheum Dis. 2013;72(10):1605–12. https://doi.org/10.1136/annrheumdis-2012-203091.
Takeuchi T, Yamanaka H, Tanaka Y, Sakurai T, Saito K, Ohtsubo H, et al. Evaluation of the pharmacokinetic equivalence and 54-week efficacy and safety of CT-P13 and innovator infliximab in Japanese patients with rheumatoid arthritis. Mod Rheumatol. 2015;25(6):817–24. https://doi.org/10.3109/14397595.2015.1022297.
Westhovens R, Wiland P, Zawadzki M, Ivanova D, Kasay AB, El-Khouri EC, et al. Efficacy, pharmacokinetics and safety of subcutaneous versus intravenous CT-P13 in rheumatoid arthritis: a randomized phase I/III trial. Rheumatology (Oxford). 2021;60(5):2277–87. https://doi.org/10.1093/rheumatology/keaa580.
Alten R, An Y, Kim DH, Yoon S, Peyrin-Biroulet L. Re-routing infliximab therapy: subcutaneous infliximab opens a path towards greater convenience and clinical benefit. Clin Drug Investig. 2022;42(6):477–89. https://doi.org/10.1007/s40261-022-01162-6.
Yoo DH, Hrycaj P, Miranda P, Ramiterre E, Piotrowski M, Shevchuk S, et al. A randomised, double-blind, parallel-group study to demonstrate equivalence in efficacy and safety of CT-P13 compared with innovator infliximab when coadministered with methotrexate in patients with active rheumatoid arthritis: the PLANETRA study. Ann Rheum Dis. 2013;72(10):1613–20. https://doi.org/10.1136/annrheumdis-2012-203090.
Tanaka Y, Yamanaka H, Takeuchi T, Inoue M, Saito K, Saeki Y, et al. Safety and efficacy of CT-P13 in Japanese patients with rheumatoid arthritis in an extension phase or after switching from infliximab. Mod Rheumatol. 2017;27(2):237–45. https://doi.org/10.1080/14397595.2016.1206244.
Nakagawa T, Kobayashi T, Nishikawa K, Yamada F, Asai S, Sameshima Y, et al. Infliximab biosimilar CT-P13 is interchangeable with its originator for patients with inflammatory bowel disease in real world practice. Intest Res. 2019;17(4):504–15. https://doi.org/10.5217/ir.2019.00030.
Sagami S, Nishikawa K, Yamada F, Suzuki Y, Watanabe M, Hibi T. Post-marketing analysis for biosimilar CT-P13 in inflammatory bowel disease compared with external data of originator infliximab in Japan. J Gastroenterol Hepatol. 2021;36(8):2091–100. https://doi.org/10.1111/jgh.15399.
Takeuchi T, Nishikawa K, Yamada F, Ohshima S, Inoue M, Yoshioka Y, et al. Real-world safety and efficacy of CT-P13, an infliximab biosimilar, in Japanese rheumatoid arthritis patients naïve to or switched from biologics. Mod Rheumatol. 2022;32(4):718–27. https://doi.org/10.1093/mr/roab068.
Morita A, Nishikawa K, Yamada F, Yamanaka K, Nakajima H, Ohtsuki M. Safety, efficacy, and drug survival of the infliximab biosimilar CT-P13 in post-marketing surveillance of Japanese patients with psoriasis. J Dermatol. 2022;49(10):957–69. https://doi.org/10.1111/1346-8138.16508.
Wells G, Becker JC, Teng J, Dougados M, Schiff M, Smolen J, et al. Validation of the 28-joint Disease Activity Score (DAS28) and European League Against Rheumatism response criteria based on C-reactive protein against disease progression in patients with rheumatoid arthritis, and comparison with the DAS28 based on erythrocyte sedimentation rate. Ann Rheum Dis. 2009;68(6):954–60. https://doi.org/10.1136/ard.2007.084459.
Best WR, Becktel JM, Singleton JW, Kern F. Development of a Crohn’s disease activity index. National Cooperative Crohn’s Disease Study. Gastroenterology. 1976;70(3):439–44.
Rutgeerts P, Sandborn WJ, Feagan BG, Reinisch W, Olson A, Johanns J, et al. Infliximab for induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2005;353(23):2462–76. https://doi.org/10.1056/NEJMoa050516.
Mahil SK, Wilson N, Dand N, Reynolds NJ, Griffiths CEM, Emsleyet R, et al. Psoriasis treat to target: defining outcomes in psoriasis using data from a real-world, population-based cohort study (the British Association of Dermatologists Biologics and Immunomodulators Register, BADBIR). Br J Dermatol. 2020;182(5):1158–66. https://doi.org/10.1111/bjd.18333.
Finlay AY. Current severe psoriasis and the rule of tens. Br J Dermatol. 2005;152(2):861–7. https://doi.org/10.1111/j.1365-2133.2005.06502.x.
Sánchez-Piedra C, Sueiro-Delgado D, García-González J, Ros-Vilamajo I, Prior-Español A, Moreno-Ramos MJ, et al. Changes in the use patterns of bDMARDs in patients with rheumatic diseases over the past 13 years. Sci Rep. 2021;11(1):15051. https://doi.org/10.1038/s41598-021-94504-x.
Shalom G, Cohen AD, Feldhamer I, Comaneshter D, Freud T, Pavlovsky L. Drug survival in patients with psoriasis is associated with the availability of biologic medications. J Eur Acad Dermatol Venereol. 2020;34(7):1524–8. https://doi.org/10.1111/jdv.16205.
Smolen JS, Aletaha D, Bijlsma JWJ, Breedveld FC, Boumpas D, Burmester G, et al. Treating rheumatoid arthritis to target: recommendations of an international task force. Ann Rheum Dis. 2010;69(4):631–7. https://doi.org/10.1136/ard.2009.123919.
Peyrin-Biroulet L, Sandborn W, Sands BE, Reinisch W, Bemelman W, Bryantet RV, et al. Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE): determining therapeutic goals for treat-to-target. Am J Gastroenterol. 2015;110(9):1324–38. https://doi.org/10.1038/ajg.2015.233.
Gisondi P, Talamonti M, Chiricozzi A, Piaserico S, Amerio P, Balato A, et al. Treat-to-target approach for the management of patients with moderate-to-severe plaque psoriasis: consensus recommendations. Dermatol Ther (Heidelb). 2021;11(1):235–52. https://doi.org/10.1007/s13555-020-00475-8.
Gisbert JP, Marín AC, McNicholl AG, Chaparro M. Systematic review with meta-analysis: the efficacy of a second anti-TNF in patients with inflammatory bowel disease whose previous anti-TNF treatment has failed. Aliment Pharmacol Ther. 2015;41(7):613–23. https://doi.org/10.1111/apt.13083.
Takeuchi T, Tatsuki Y, Nogami Y, Ishiguro N, Tanaka Y, Yamanaka H, et al. Postmarketing surveillance of the safety profile of infliximab in 5000 Japanese patients with rheumatoid arthritis. Ann Rheum Dis. 2008;67(2):189–94. https://doi.org/10.1136/ard.2007.072967.
Torii H, Terui T, Matsukawa M, Takesaki K, Ohtsuki M, Nakagawa H. Safety profiles and efficacy of infliximab therapy in Japanese patients with plaque psoriasis with or without psoriatic arthritis, pustular psoriasis or psoriatic erythroderma: results from the prospective post-marketing surveillance. J Dermatol. 2016;43(7):767–78. https://doi.org/10.1111/1346-8138.13214.
Lee SJ, Baek K, Lee S, Lee YJ, Park JE, Lee SG. Post-marketing pooled safety analysis for CT-P13 treatment of patients with immune-mediated inflammatory diseases in observational cohort studies. BioDrugs. 2020;34(4):513–28. https://doi.org/10.1007/s40259-020-00421-2.
Cheon JH, Nah S, Kang HW, Lim YJ, Lee SH, Lee SJ, et al. Infliximab biosimilar CT-P13 observational studies for rheumatoid arthritis, inflammatory bowel diseases, and ankylosing spondylitis: pooled analysis of long-term safety and effectiveness. Adv Ther. 2021;38(8):4366–87. https://doi.org/10.1007/s12325-021-01834-3.
Syversen SW, Jørgensen KK, Goll GL, Brun MK, Sandanger Ø, Bjørlykke KH, et al. Effect of therapeutic drug monitoring vs standard therapy during maintenance infliximab therapy on disease control in patients with immune-mediated inflammatory diseases: a randomized clinical trial. JAMA. 2021;326(23):2375–84. https://doi.org/10.1001/jama.2021.21316.
Fumery M, Tilmant M, Yzet C, Brazier F, Loreau J, Turpin J, et al. Premedication as primary prophylaxis does not influence the risk of acute infliximab infusion reactions in immune-mediated inflammatory diseases: a systematic review and meta-analysis. Dig Liver Dis. 2019;51(4):484–8. https://doi.org/10.1016/j.dld.2018.12.002.
Choquette D, Faraawi R, Chow A, Rodrigues J, Bensen WJ, Nantel F. Incidence and management of infusion reactions to infliximab in a prospective real-world community registry. J Rheumatol. 2015;42(7):1105–11. https://doi.org/10.3899/jrheum.140538.
Khan N, Vallarino C, Lissoos T, Darr U, Luo M. Risk of infection and types of infection among elderly patients with inflammatory bowel disease: a retrospective database analysis. Inflamm Bowel Dis. 2020;26(3):462–8. https://doi.org/10.1093/ibd/izz065.
Kedia S, Mouli VP, Kamat N, Sankar J, Ananthakrishnan A, Makharia G, et al. Risk of tuberculosis in patients with inflammatory bowel disease on infliximab or adalimumab is dependent on the local disease burden of tuberculosis: a systematic review and meta-analysis. Am J Gastroenterol. 2020;115(3):340–9. https://doi.org/10.14309/ajg.0000000000000527.
World Health Organization. Global tuberculosis report 2022 (issued 27 October 2022). https://www.who.int/publications/i/item/9789240061729. Accessed 9 Mar 2023.
England BR, Hershberger D. Management issues in rheumatoid arthritis-associated interstitial lung disease. Curr Opin Rheumatol. 2020;32(3):255–63. https://doi.org/10.1097/BOR.0000000000000703.
Ishikawa G, Dua S, Mathur A, Acquah SO, Salvatore M, Beasley MB, et al. Concomitant interstitial lung disease with psoriasis. Can Respir J. 2019;2019:5919304. https://doi.org/10.1155/2019/5919304.
Perez-Alvarez R, Perez-de-Lis M, Diaz-Lagares C, Pego-Reigosa JM, Retamozo S, Bove A, et al. Interstitial lung disease induced or exacerbated by TNF-targeted therapies: analysis of 122 cases. Semin Arthritis Rheum. 2011;41(2):256–64. https://doi.org/10.1016/j.semarthrit.2010.11.002.
Acknowledgements
The authors express their sincere gratitude to all the patients and physicians who participated in the Japanese Post-marketing Surveillance of Infliximab Biosimilar 1 ‘NK’ (CT-P13).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This integrated analysis and included three Japanese post-marketing surveillances were sponsored by Nippon Kayaku Co., Ltd. (Tokyo, Japan).
Conflict of interest
Tsutomu Takeuchi has received grants from AbbVie, Asahi Kasei Pharma, Ayumi Pharmaceutical, Boehringer-Ingelheim, Chugai Pharmaceutical, Daiichi Sankyo, DNA Chip Research, Eisai, Eli Lilly Japan, Mitsubishi Tanabe Pharma, Nippon Kayaku, Ono Pharmaceutical, and UCB Japan; consulting fees from AbbVie, Astellas Pharma, Chugai Pharmaceutical, Eli Lilly Japan, Gilead Sciences, GlaxoSmithKline, Janssen Pharmaceutical, Mitsubishi Tanabe Pharma, Novartis Pharma, Pfizer, and Taisho Pharmaceutical; and speaker fees from AbbVie, Astellas Pharma, AstraZeneca, Ayumi Pharmaceutical, Bristol Myers Squibb, Chugai Pharmaceutical, Daiichi Sankyo, Eisai, Eli Lilly Japan, Gilead Sciences, Janssen Pharmaceutical, Mitsubishi Tanabe Pharma, Novartis Pharma, Pfizer, Sanofi, Taisho Pharmaceutical, and UCB Japan. Kiyohiro Nishikawa was an employee of and holds stocks in Nippon Kayaku. Fumika Yamada is an employee of Nippon Kayaku. Akimichi Morita has received research grants from AbbVie, Eisai, Eli Lilly Japan, Janssen Pharmaceutical, Kyowa Kirin, LEO Pharma, Maruho, Mitsubishi Tanabe Pharma, Novartis, Sun Pharma Japan, Taiho Pharmaceutical, Torii Pharmaceutical, and Ushio; consulting fees from Amgen, Bristol Myers Squibb, Eli Lilly Japan, Janssen Pharmaceutical, Minophagen Pharmaceutical, Nippon Boehringer Ingelheim, Nippon Kayaku, and UCB Japan; and speaker fees from AbbVie, Amgen, Bristol Myers Squibb, Eisai, Eli Lilly Japan, Janssen Pharmaceutical, Kyowa Kirin, Maruho, Minophagen Pharmaceutical, Nippon Boehringer Ingelheim, Novartis, Sun Pharma Japan, Taiho Pharmaceutical, UCB Japan, and Ushio. Mamitaro Ohtsuki has received research grants from AbbVie, Eisai, Kyowa Kirin, Maruho, Mitsubishi Tanabe Pharma, Sun Pharma Japan, Taiho Pharmaceutical, and Torii Pharmaceutical; consulting fees from Bristol Myers Squibb, Eli Lilly Japan, LEO Pharma, Nippon Boehringer Ingelheim, Nippon Kayaku, Novartis, and UCB Japan; and speaker fees from AbbVie, Amgen, Bristol Myers Squibb, Eli Lilly Japan, Janssen Pharmaceutical, Kyowa Kirin, LEO Pharma, Maruho, Sanofi, Taiho Pharmaceutical, Torii Pharmaceutical, and UCB Japan. Yasuo Suzuki has received grants from AbbVie, EA Pharma, JIMRO, Kissei Pharmaceutical, Mitsubishi Tanabe Pharma, Mochida Pharmaceutical, and Nippon Kayaku; and speaker fees from AbbVie, EA Pharma, Janssen Pharmaceutical, Kyorin Pharmaceutical, Mitsubishi Tanabe Pharma, Mochida Pharmaceutical, Takeda Pharmaceutical, and Zeria Pharmaceutical. Mamoru Watanabe has received research funding from AbbVie, EA Pharma, Kissei Pharmaceutical, Kyorin Pharmaceutical, Mitsubishi Tanabe Pharma, Nippon Kayaku, Takeda Pharmaceutical, and Zeria Pharmaceutical; consultancy fees from AbbVie, EA Pharma, Eli Lilly Japan, Gilead Sciences, Kissei Pharmaceutical, Nichi-Iko Pharmaceutical, and Nippon Boehringer Ingelheim; and lecture fees from AbbVie, EA Pharma, Eli Lilly Japan, Gilead Sciences, Janssen Pharmaceutical, Kyorin Pharmaceutical, Mitsubishi Tanabe Pharma, Nippon Boehringer Ingelheim, Takeda Pharmaceutical, and Zeria Pharmaceutical. Hisashi Yamanaka has received consulting fees from AbbVie, Chugai Pharmaceutical, CorEvitas, Nippon Kayaku, Pfizer, Sato Pharmaceutical, Teijin Pharma, and YL Biologics; and speaker fees from AbbVie, Asahi Kasei Pharma, Ayumi Pharmaceutical, Bristol Meyers Squibb, Chugai Pharmaceutical, Eli Lilly Japan, Fuji Yakuhin, Mitsubishi Tanabe Pharma, Mochida Pharmaceutical, Pfizer, Teijin Pharma, and YL Biologics. Toshifumi Hibi has received research grants or scholarship contributions from AbbVie, Activaid, Alfresa Pharma, Bristol Myers Squibb, Eli Lilly Japan, Ferring Pharmaceuticals, Gilead Sciences, Janssen Pharmaceutical, JMDA, Mitsubishi Tanabe Pharma, Mochida Pharmaceutical, Nippon Kayaku, Pfizer, Takeda Pharmaceutical, and Zeria Pharmaceutical; advisory/consultancy fees from AbbVie, Celltrion, EA Pharma, Eli Lilly Japan, Gilead Sciences, Janssen Pharmaceutical, Mitsubishi Tanabe Pharma, Nichi-Iko Pharmaceutical, Nippon Kayaku, Takeda Pharmaceutical, and Zeria Pharmaceutical; and lecture fees from AbbVie, EA Pharma, Janssen Pharmaceutical, JIMRO, Mitsubishi Tanabe Pharma, Mochida Pharmaceutical, Pfizer, Sandoz, Takeda Pharmaceutical, and Zeria Pharmaceutical.
Ethics approval
The protocols of the PMSs were submitted to and approved by the Ministry of Health, Labour and Welfare. The protocol and ethical considerations of the PMS were reviewed and approved by internal review board members (approval numbers for PMSs in patients with rheumatoid arthritis, inflammatory bowel disease, and psoriasis are 20130717, 20140807, and 20150605, respectively) and/or independent ethics committee at each medical institutions participating in the PMS.
Consent to participate
The PMS was conducted in accordance with the Good Post-marketing Study Practice Ordinance of the Ministry of Health, Labour and Welfare, and informed consent from individual patients was not required.
Consent for publication
Not applicable.
Availability of data and material
The datasets generated during the current study are available from Nippon Kayaku on reasonable request (https://www.nipponkayaku.co.jp/english/contact.html).
Code availability
Not applicable.
Author contributions
All named authors meet the International Committee of Medical Journal Editors (ICMJE) authorship criteria for this article, and take responsibility for the integrity of the work. TT, KN, FY, AM, MO, and TH contributed to the study conceptualization and methodology. FY, AM, MO, YS, MW, HY, and TH collected data, which were analyzed and validated by KN and FY. The manuscript was drafted by KN, and critically reviewed by TT, FY, MW, HY, and TH. All authors read and approved the final manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Takeuchi, T., Nishikawa, K., Yamada, F. et al. Real-World Safety and Efficacy of Biosimilar CT-P13 in Patients with Immune-Mediated Inflammatory Diseases: Integrated Analysis of Three Japanese Prospective Observational Studies. Drug Saf 46, 991–1005 (2023). https://doi.org/10.1007/s40264-023-01340-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-023-01340-1