Abstract
Introduction
Various vaccines for protection against COVID-19 were provided emergency approval in late 2020 to early 2021. There is a scarcity of long-term safety data for many of these.
Objective
The main aim of this study is to provide the one-year safety results of the ChAdOx1-nCoV-19/AZD1222 vaccine and determine the risk factors of adverse events of special interest (AESIs) and persistent AESIs.
Methods
This was a prospective observational study conducted from February 2021 to April 2022 in a tertiary hospital in North India and its two associated centers. Health care workers, other frontline workers, and the elderly vaccinated with the ChAdOx1-nCoV-19 vaccine constituted the study population. Individuals were contacted telephonically at pre-decided intervals for one year and health issues of significant concern were recorded. Atypical adverse events developing after a booster dose of the COVID-19 vaccine were assessed. Regression analysis was conducted to determine risk factors of AESI occurrence and determinants of AESIs persisting for at least one month at the time of final telephonic contact.
Results
Of 1650 individuals enrolled, 1520 could be assessed at one-year post-vaccination. COVID-19 occurred in 44.1% of participants. Dengue occurred in 8% of participants. The majority of the AESIs belonged to the MedDRA® SOC of musculoskeletal disorders (3.7% of 1520). Arthropathy (knee joint involvement) was the most common individual AESI (1.7%). Endocrinal disorders such as thyroid abnormalities and metabolic disorders such as newly diagnosed diabetes developed in 0.4% and 0.3% of individuals, respectively. Regression analysis showed females, individuals with a pre-vaccination history of COVID-19, diabetes, hypothyroidism, and arthropathy had 1.78-, 1.55-, 1.82-, 2.47- and 3.9-times higher odds of AESI development. Females and individuals with hypothyroidism were at 1.66- and 2.23-times higher risk of persistent AESIs. Individuals receiving the vaccine after COVID-19 were at 2.85- and 1.94 times higher risk of persistent AESIs compared, respectively, to individuals with no history of COVID-19 and individuals developing COVID-19 after the vaccine. Among participants receiving a booster dose of the COVID-19 vaccine (n = 185), 9.7% developed atypical adverse events of which urticaria and new-onset arthropathy were common.
Conclusion
Nearly half of the ChAdOx1-nCoV-19 vaccine recipients developed COVID-19 over one year. Vigilance is warranted for AESIs such as musculoskeletal disorders. Females, individuals with hypothyroidism, diabetes, and pre-vaccination history of COVID-19 are at higher risk of adverse events. Vaccines received after natural SARS-CoV-2 infection may increase the risk of persistence of adverse events. Sex and endocrinal differences and timing of the COVID-19 vaccine with respect to natural infection should be explored as determinants of AESIs in the future. Pathogenetic mechanisms of vaccine-related adverse events should be investigated along with comparisons with an unvaccinated arm to delineate the overall safety profile of COVID-19 vaccines.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Musculoskeletal disorders are commonly observed adverse events of special interest (AESIs) after 1 year of ChAdOx1-nCoV-19 vaccination. |
Females and those with hypothyroidism are at greater risk of both AESIs and persistent AESIs following ChAdOx1-nCoV-19 vaccination. |
Vaccination received after recovery from COVID-19 is an independent risk factor of AESIs and persistent AESIs. |
1 Introduction
In the fight against COVID-19, various vaccines based on novel and pre-existing platforms were developed at an unprecedented scale and granted emergency use authorization (EUA) in late December 2020 to early January 2021. Among these, mRNA-based COVID-19 vaccines, and adenoviral vectored vaccines coding for the Spike protein of SARS-CoV-2 were largely employed in the USA and European nations [1, 2]. In India, ChAdOx1-nCoV-19 (COVISHIELD, Serum Institute of India) based on the chimpanzee adenoviral platform of Oxford-AstraZeneca’s vaccine and the inactivated SARS-CoV-2 based vaccine, COVAXIN (Bharat Biotech) were the first to receive approval from the drug regulatory body for stepwise mass rollout [3]. The controlled setting results of more than desired protection rates against COVID-19 and an acceptable short-term safety profile were the basis of approval of these vaccines for public use. The performance of vaccines in the real world often varies from controlled settings and comprehensive assessment of vaccine safety and performance can be ascertained through active monitoring of vaccinated individuals in the post-approval phase. The claims of excellent protection rates against symptomatic COVID-19 have been challenged in the post-marketing period [4,5,6]. In addition, an upsurge of conditions such as autoimmune diseases, atypical thrombotic events, and cardiac complications in the post-vaccination period, is alerting researchers to conduct a thorough assessment of the safety profile of COVID-19 vaccines [7,8,9]. Despite more than 18 months since emergency approval and close to 5 billion people vaccinated worldwide, long-term safety-specific data on most COVID-19 vaccines are not available in the public domain [10]. Here, in continuation of our first short-term real-world safety study on the ChAdOx1-nCoV-19 vaccine, we provide the long-term safety data in vaccinated individuals over one year of follow-up.
2 Methods
2.1 Study Design and Participants
This was a prospective observational study conducted from February 2021 to April 2022 in a tertiary hospital in North India. In accordance with the Indian government’s policies of the mass rollout of COVID-19 vaccination, health care workers, other frontline workers, and those aged ≥ 60 years were the groups vaccinated on a priority basis in the first phase. Accordingly, the present study predominantly recruited health care and other frontline workers in the initial phase followed by the elderly during the last few days of enrollment. The details of the enrollment procedure have been already published [11]. The enrolled participants were contacted at pre-planned time periods, with a final follow-up at 12 months following vaccination. A support telephone number was provided to participants to report any atypical health issues or COVID-19-related complaints. The preliminary findings of the study pertaining to adverse events following immunization (AEFIs) and the occurrence of breakthrough COVID-19 during the first six months of follow-up have already been published [11, 12].
2.2 Safety Analysis
Adverse events following immunization (AEFIs) were the primary outcome, and COVID-19-specific details as well as long-term safety analysis in terms of adverse events of special interest (AESIs) were the secondary outcomes of interest. The preliminary results of AEFIs have already been published and the present study aims to provide insight into the AESIs [11]. The format for AESIs used in the study was based on the guiding document on AESIs by the CEPI-SPEAC-Brighton Collaboration as well as the studies available in PubMed/Medline on atypical adverse events following COVID-19 vaccination of any type from the period of January 2021 to January 2022 [13]. In addition, atypical adverse events notified by the study participants to the study staff at any time during the study period were also incorporated in the AESI format for final follow-up at one year. Apart from this, persistent adverse events defined by us as any health issue persisting for at least one month during the final follow-up were also recorded. The MedDRA® terminology was used for labelling system organ class (SOC) of AESIs and severity categorization of each AESI was done using the Food and Drug Administration (FDA) scale of severity of vaccine-related adverse events. Supplementary Table 1 shows the general guidance format used for obtaining information. Apart from general information, any specific medical diagnosis made was enquired about, and medical records were requested for all cases of new diagnoses, deaths, or serious AESIs. However, providing records was at the discretion of participants.
2.3 Outcome Measures
The institute was affected by the pandemic of COVID-19 during the second wave in India (February 2021–June 2021, with a peak in April 2021) and the third wave (December 2021–March 2022). Data pertaining to COVID-19 during this period as well as during the intermediate time period were enquired about telephonically in accordance with the study protocol. The booster dose of the COVID-19 vaccine was initiated in the study center in the month of January 2022. Information about the same was also enquired about as deemed suitable at the time of 1-year follow-up. COVID-19 was categorized as ‘Confirm’ and ‘Suspect’ and the severity of COVID-19 was rated as per the investigators’ assessment and in accordance with the recommendations of the MoHFW (Ministry of Health and Family Welfare, Government of India). As defined previously, we also labeled cases as ‘RT-PCR negative suspects’ with symptomatology resembling COVID-19 who tested negative on a single RT-PCR-based test and in whom the RT-PCR test was not repeated [11, 12]. In view of a dengue epidemic that affected the region in the months of July–October 2021, the history of laboratory confirmed dengue was also enquired separately during the final follow-up. Any dengue that required hospitalization with or without the need for intravenous fluids and with or without platelet transfusion was categorized as ‘serious’. Among AESIs, we included new-onset rheumatologic disorders, new-onset endocrinal disorders such as thyroid abnormalities, new-onset diabetes, cardiac disorders such as heart failure, myocardial infarction or myocarditis, nervous system disorders such as headaches and weakness in limbs, blood disorders such as thrombocytopenia, reproductive system disorders such as menstrual abnormalities and flares of underlying diseases such as diabetes, hypertension and arthropathy among others. To validate the findings of our previously published study highlighting the importance of the timing of the COVID-19 vaccine with respect to the occurrence of COVID-19 on persistent adverse events, individuals were categorized into three groups defined as follows [4].
Group A: Individuals with no history of COVID-19 until the last follow-up.
Group B: Individuals who received any dose of vaccine (first, second, or booster) after any episode of COVID-19.
Group C: Individuals with a history of COVID-19 after the vaccine but who did not receive any vaccine thereafter.
Individuals whose booster dose information was not available, and in whom this information was likely to change the categorization of patients in the above groups, were excluded from the analysis of the risk of persistent AESIs.
2.4 Sample Size
The sample size for the present study was based on the primary outcome of rates of AEFIs following COVID-19 vaccination. Details of the study methodology, sample size, and preliminary results of safety analysis have already been published [11].
2.5 Statistical Analysis
Data were recorded as frequencies and percentages for categorical variables such as the occurrence of COVID-19 and dengue and the development of AESIs. Descriptive analysis of each AESI was performed. Bivariate analysis was done for the association between the occurrence of AESIs and co-variates such as demographics, pre-existing co-morbidities, history of COVID-19 in 2020 (before vaccination), and history of COVID-19 in 2021 or 2022 (after vaccination). A separate association analysis was conducted to explore the differences in incidence and patterns of AESIs between three age subgroups (18–44 years, 45–59 years, and ≥ 60 years) as well as their relationship with dengue.
The chi-square test was also applied to ascertain the relationship between risk of persistent AESIs and co-variates including the timing of vaccine with respect to COVID-19 among others. Variables with p < 0.05 or those deemed clinically relevant were incorporated in binary logistic regression analysis. The SPSS version 16 was used for performing statistical analysis.
3 Results
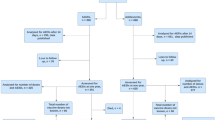
A total of 1650 individuals were enrolled in the study (Fig. 1). The baseline characteristics of the participants are provided in Supplementary Table 2. Median (Q1, Q3), age of participants was 35 (29, 47) years. Elderly patients aged ≥ 60 years constituted around 8% (n = 133) of the study group. Hypertension and diabetes were the most common comorbidities, observed in 11.5% and 9.4% of participants respectively. COVID-19 before vaccination was reported by 13.5%. As expected, a statistically significant, higher representation of co-morbidities such as hypertension, diabetes, and heart disease (p < 0.05) was observed in the elderly compared to individuals aged < 60 years (Supplementary Table 2). After excluding 125 participants who were lost to follow-up at the time of final follow-up, and 5 who died before June 2021, the remaining 1520 participants (including 115 elderly) were assessed for the occurrence of AESIs (Fig. 1). Of the total 1520, booster dose information that was likely to change the categorization of participants into three groups as defined earlier was missing for 48 participants. After removing these 48, the remaining 1472 persons were analyzed for risk of persisting adverse outcomes (health events persisting for a minimum period of 4 weeks at the time of final follow-up at one year).
The STROBE flow diagram of the study. Of 1650 vaccinees enrolled, data for 730 (adverse events following immunization [AEFIs]) and 1500 (occurrence of COVID-19) have been already published (#). 125 participants were lost to follow-up at one year, and 5 participants died. 1520 participants were analyzed for risk of AESIs. Among these, booster dose information likely to change classification into groups A, B, C were unavailable for 48 participants (*). AESIs persistent for at least one month at the time of one year follow-up were assessed in 1472 individuals
The occurrence of AEFIs and COVID-19 during the second wave of the pandemic has been already described and published [11, 12]. To briefly mention here, AEFIs occurred in 40% of individuals after the first dose with systemic involvement (such as fever, headache, etc.) in 31%. Corresponding rates after the second dose were reduced to 15.7% and 13.6%, respectively. Around 0.5% of individuals developed AEFIs of FDA grade 3 severity and 0.1% of individuals developed ‘serious’ AEFIs [11]. Nearly 27–46% of individuals receiving one or two doses of the ChAdOx1-nCoV-19 developed COVID-19 during the second wave of the pandemic in India, which peaked in the month of April 2021 in the study region [12].
Supplementary Table 3 describes in detail the occurrence of COVID-19 during the third wave of the pandemic, the occurrence of dengue, and the SOC-wise distribution of all AESIs. COVID-19 during the third wave of the pandemic occurred in 259 participants (17.0%) and was of ‘mild’ severity in the majority (95.4% of affected). COVID-19 at any time post-vaccination occurred in 671 out of 1520 (44.1%) participants. A more than 2 times higher risk of COVID-19 was observed in younger adults (aged 18–44 years) compared to the elderly (aged ≥ 60 years) (Supplementary Table 4). Interestingly, the occurrence of COVID-19 during the 3rd wave of the pandemic was statistically more common in vaccinated individuals with a history of COVID-19 in the 2nd wave of the pandemic (Table 1c) (p = 0.001). Dengue occurred in 8% of individuals and of those affected, a serious form of dengue occurred in 19.7% individuals. Adverse events of special interest of any kind occurred in 210 participants (13.8%). Musculoskeletal and connective tissue disorders (MCTDs) were commonly reported AESIs (57, 3.7% of all participants) predominated by complaints of arthropathy (54, 3.5%). The knee joint (n = 26, 1.7% of participants) and lower back (n = 12, 0.8%) were the commonly affected sites of arthropathy. Outcome-wise, the majority of the vaccinees with new-onset arthropathy reported persisting joint pain at one year of follow-up (48, 3.1% of participants) with a median (Q1, Q3) duration of symptoms of 120 days (86.2, 180). Next to MCTDs, were general disorders (32, 2.1%) and infections and infestations (32, 2.1%). New-onset hypertension, thyroid abnormalities, and newly diagnosed diabetes developed in 13 (0.9%), 6 (0.4%), and 5 (0.3%) individuals, respectively. Serious AESIs other than mortality belonged to diverse SOCs and occurred in 14 individuals (0.9%), as shown in Supplementary Table 3. Among the serious AESIs, three miscarriages and five serious cardiac events were reported. Details of the remaining individual serious AESIs are mentioned in Supplementary Table 3. A total of 5 persons died during the first four months of follow-up (Supplementary Table 5).
3.1 Determinants of Individuals at Risk of AESIs
In unadjusted analysis (Table 1a), AESIs occurred commonly in individuals aged ≥ 40 years, females, and individuals with diabetes, hypertension, arthropathy, heart disease, or hypothyroidism. Pre-vaccination history of COVID-19 (in the first wave of 2020) also shared a positive association with the occurrence of AESIs although the statistical significance was marginal (p = 0.057). No significant association was observed between AESIs and a history of post-vaccination COVID-19 (p = 0.29).
After adjusting for potential confounders, regression analysis showed a statistically significant 1.78-, 1.82- 2.47-, and 3.9-times higher odds of occurrence of AESIs in females, individuals with diabetes, hypothyroidism, and arthropathy, respectively, with respect to comparators (Table 1b). Vaccines received after having had an episode of COVID-19 in the year 2020 were associated with 1.55-times higher odds of AESI development compared to individuals with no pre-vaccination history of COVID-19. Post-vaccination COVID-19 (in the years 2021 and 2022) was not associated with any risk of AESI development.
3.2 Association Between AESIs, Age Subgroups, and Dengue
A separate association analysis was performed for AESIs and a history of dengue. Although a significant association existed between the occurrence of any AESI and dengue, except for flares of underlying disease, no other AESI shared a statistically significant association with dengue (Supplementary Table 4). Owing to the lack of information about the onset of AESIs with respect to dengue, dengue could not be incorporated further in the regression model of determinants of AESIs.
No statistically significant difference was observed in the incidence of AESIs in age subgroups of 18–44 years, 45–59 years, and ≥ 60 years. Individually, flares of underlying diseases were more than 3-times more common in the elderly (aged ≥ 60 years) compared to young adults (aged 18–44 years) with statistical significance (Supplementary Table 4). No significant difference was observed for the incidence of remaining AESIs in different age subgroups.
3.3 Determinants of Individuals at Risk of Persistent AESIs
Of 210 individuals developing AESIs, 153 complained of persistent AESIs. Median (Q1, Q3) duration of persistence of AESIs was 120 days (60, 180). In unadjusted analysis, a statistically significant association of persistent AESIs was noticed with age, sex, body mass index, diabetes, hypertension, heart disease, prior arthropathy, and hypothyroidism. Timing of vaccine with respect to COVID-19 also shared a statistically significant association (Table 2a). After adjusting for covariates, regression analysis highlighted a statistically significant 1.66- and 2.23-times higher odds of persistence of AESIs in females and individuals with hypothyroidism. With marginal statistical significance, 2.32- and 3.41-times higher odds of persistence of AESIs were observed in individuals with pre-existing heart disease and arthropathy. An interesting association was observed with the timing of the vaccine received with respect to COVID-19. Receiving any dose of the COVID-19 vaccine after any prior history of COVID-19 was associated with 1.94-times higher odds of persistence of AESIs compared to COVID-19 occurring after receipt of any dose of the COVID-19 vaccine. Compared to vaccinees who did not develop COVID-19, individuals receiving the COVID-19 vaccine after any COVID-19 of the past were observed to be at 2.85-times higher odds of persistence of AESIs with statistical significance (Table 2b). No association was seen between persistence of AESIs and dengue.
3.4 Adverse Events After Booster Dose of COVID-19 Vaccine
Until the last follow-up, 185 participants had received the booster dose of COVID-19 vaccines. Of these, 18 vaccinees reported significant adverse events, giving an adverse event rate of 9.7%. Urticaria and new-onset arthropathy were the common adverse events in this subset (details in Supplementary Table 6).
4 Discussion
Post-approval, the claims of high effectiveness of COVID-19 vaccines against infection have been challenged by some studies with modest protection against the delta variant and limited protection against the omicron variant of SARS-CoV-2 being observed in the results of various real-world studies [4, 5, 14,15,16]. Likewise, negative vaccine efficacy rates in select groups at certain intervals have been observed in exploratory outcomes of some controlled studies as well as in the findings of some real-world studies [4, 6, 17, 18]. The post-approval phase also witnessed reports of atypical adverse events and AESIs. These included cases of vaccine-induced thrombosis and thrombocytopenia (VITT), new-onset cardiac events, flares, and new-onset of autoimmune phenomena such as rheumatoid arthritis, diabetes mellitus, Guillain Barre syndrome, and encephalitis [8, 9, 19,20,21]. Though passive reporting of AESIs is recommended by drug regulatory bodies to strengthen the safety profile of vaccines, spontaneous reporting rates are low mainly because of scanty awareness of AESIs and deficient knowledge of reporting, even among medical professionals. Actual estimates of AESIs can be calculated by active surveillance methods. A prospective cohort-based study involving vaccinated individuals is one of the preferred approaches. To fill the information gap, the present study was conducted to underline the long-term safety profile of the ChAdOx1 nCoV-19 vaccine. Specifically, the study addressed the occurrence of COVID-19, dengue, and AESIs over one year of active monitoring of vaccinees.
Nearly one-third of our study participants developed COVID-19 during the 2nd wave and around 17% were affected during the 3rd wave of the pandemic. Close to 44% of individuals developed COVID-19 at any time since vaccination and over one year of follow-up. That occurrence of COVID-19 in the third wave was common in participants with a history of COVID-19 in the second wave, suggests a need for understanding the genetic basis of predilection to SARS-CoV-2, as well as raises questions on the concept of longer hybrid immunity gained after natural infection in vaccinated individuals. The protective effect of primary or booster doses of the COVID-19 vaccine against subsequent viral strains also needs a comprehensive assessment. Apart from COVID-19, dengue occurred in 8% of participants. With nearly 20% of the affected individuals developing dengue requiring hospitalization, future research involving an unvaccinated arm is needed for comparison and for generating pathogenetic hypotheses.
To our knowledge, no study has investigated the long-term incidence of AESIs with COVID-19 vaccines. In the present study, disorders involving the musculoskeletal system largely predominated by arthropathy were the most common AESIs and occurred in close to 4% of participants. Pain in the knee joint followed by new-onset lower back pain were the most commonly observed arthropathies. The joint problems were persistent in most participants at the time of the last follow-up, with a median duration of persistence of four months. Flares of rheumatologic conditions post-COVID-19 vaccination have been explored in various studies and a pooled rate of 7% has been documented with COVID-19 vaccines with rates as high as 14.4% with the ChAdOx1-nCoV-19 vaccine [22, 23]. The majority of the flares have been reported within 1–2 weeks of receiving the vaccine and the long-term effect of vaccines in patients with underlying musculoskeletal disorders still remains unexplored. Events belonging to the SOC of ‘general disorders and administration site conditions’ and ‘infections and infestations’ were the other commonly reported health issues. Weakness and persistent fatigability were the most common events reported among the general disorders, and typhoid and recurrent viral upper respiratory tract infections were commonly reported non-COVID-19, non-dengue infections. Among other rare and atypical adverse events, new-onset hypertension, thyroid function abnormalities, and new-onset diabetes were reported. In the absence of data on baseline incidence rates in the study population, the possibility of many new-onset conditions being chance events cannot be ruled out. However, considering a probable autoimmune etiology of many of the AESIs, especially rheumatological and thyroid-related conditions, further research may be warranted into whether the ChAdOx1 vaccine may act as a potential trigger [24]. To our knowledge, studies assessing the long-term safety of COVID-19 vaccines in an active surveillance design are lacking in the public domain. However, case series and reports of flares and new-onset musculoskeletal, neurological, and endocrinal abnormalities have been reported post-COVID-19 vaccination, commonly after mRNA-based and adenoviral vectored vaccines [7, 25, 26]. The pathogenesis of these adverse events needs to be elucidated but has been hypothesized to be related to molecular mimicry. The S protein of SARS-CoV-2 and anti-Spike antibodies generated in response to vaccination have been shown to cross-react with multiple host tissue antigens such as mitochondrial protein M2, actin, and thyroid peroxidase, among others [24]. Bystander activation of dormant T cells to active T cells and stimulation of innate immunity by the cytokines produced in response to adenoviral entry into host cells are other proposed mechanisms of immune-mediated inflammatory conditions post-COVID-19 vaccination [27].
Fourteen participants in our study (0.9 %) developed adverse events classified as ‘serious’, some of which have been published in the preliminary analyses of the study, and as part of other ongoing work [4, 11, 12]. A total of 5 deaths were reported, one of which occurred in an adult male with diabetes because of sepsis and encephalopathy (both doses received), and the remaining deaths occurred in the elderly. Of the latter, one elderly male had aggravation of pre-existing congestive heart failure within hours of vaccination, followed by a stroke event and weeks later by COVID-19 (single dose received). One death occurred because of cardiac arrest in an individual with pre-existing diabetes and hypertension (both doses received), one because of cardiac arrest in an individual with underlying coronary artery disease after recovery from COVID-19 (single dose received), and one possibly because of COVID-19 itself (single dose received). The causality of death events could not be assessed due to the design of the study, nor could much inference be drawn due to the lack of comparator arm and background mortality statistics.
After adjusting for covariates, regression analysis showed close to 2 times higher odds of occurrence of AESIs in females and individuals with diabetes and nearly 2.5 times higher odds of AESIs in individuals with hypothyroidism. In our previously published work, female sex and history of hypothyroidism were projected as risk factors of adverse events following immunization (AEFIs) with the ChAdOx1-nCoV-19 vaccine [11]. Understanding the hormonal and sex-related differences in the occurrence of adverse events following COVID-19 vaccines is of interest considering common trends between AEFIs and AESIs. Another interesting positive association of AESIs was observed with the pre-vaccination history of COVID-19. As we previously suggested, guidelines need to be revisited with regard to vaccinating individuals who have recovered from COVID-19 [4].
An adjusted analysis was also performed to determine individuals at risk of persistent AESIs. Close to 1.6-times and 2.2-times higher risk was observed in females and individuals with hypothyroidism. Though pre-existing heart disease and arthropathy were also associated with a higher risk of persistent AESIs, relevant conclusions cannot be drawn at present because of the poor overall representation of these co-morbidities in the study sample. Validating the findings of our recently published study, an interesting link was observed between individuals with persistent AESIs and the timing of the COVID-19 vaccine. Any dose of the vaccine received after recovery from COVID-19 was associated with higher odds of persistent AESIs compared to both COVID-19 occurring after vaccination, and not developing any COVID-19 until the last follow-up.
Close to 10% of individuals receiving the booster dose developed adverse events of concern. Cutaneous conditions and new-onset arthropathy were commonly reported in this subset. Although the number of individuals receiving the booster dose until the last follow-up was small, detailed safety studies should be conducted to delineate the safety profile of booster doses.
4.1 Limitations
The study was based on vaccinated individuals and data on events such as third wave of COVID-19, dengue, and AESIs need to be compared with an unvaccinated population, which may be difficult to obtain considering that most of the Indian population is vaccinated. Individuals with co-morbidities represented a small sample and results may not be generalized to these groups. With this being a telephonic study, a detailed evaluation of many AESIs was not possible. Also, the study being observational with the last two follow-ups conducted at six-month intervals, information such as the exact onset of AESIs without laboratory workup, is expected to be affected by recall bias. However, medical records were obtained electronically for the majority of the diagnosed and serious cases of AESIs. Although the exact time of onset of AESIs could not be verified due to recall bias, the occurrence of each AESI was enquired about with respect to the time of COVID-19 post-vaccination. However, the onset of AESIs was not assessed with respect to the time of dengue as the majority of the AESIs occurred in the overlap period with dengue. Although a statistically significant association existed between dengue and AESIs, a lack of information about the exact onset of AESIs with respect to dengue resulted in the failure to include dengue as an independent predictor in the regression model. These shortcomings should be addressed in the future to understand the possible causal role of dengue in the onset of post-vaccination AESIs. However, except for flares of underlying disease, no other individual AESI shared a statistically significant association with dengue. Another limitation of most long-term safety studies is the inability to perform causality assessment in the absence of a comparator arm. Further, the safety-related data are specific to ChAdOx1-nCoV-19 and cannot be generalized to mRNA vaccines and inactivated vaccines. Only a small number of individuals had received booster doses until the last contact and a larger population needs to be recruited to understand the safety profile of boosters. Further, among participants receiving booster doses, AESIs were usually reported within 2–3 weeks of the booster dose. As such, safety-related information might be inadequate or underreported for individuals contacted within days of receiving the boosters.
5 Conclusion
COVID-19 occurred in close to 44% individuals monitored for one year post-vaccination with the ChAdOx1-nCoV-19 vaccine. Adverse events of special interest occurred commonly in females, individuals with hypothyroidism, diabetes, or a pre-vaccination history of COVID-19. Females and individuals with hypothyroidism were also at high risk of persistent adverse events. Although the effect of COVID-19 on the persistence of AESIs cannot be underestimated, vaccines received after COVID-19 were associated with two times the risk of persistence of adverse events compared to vaccine received before COVID-19. Among AESIs, caution is advised particularly for arthropathy, recurrent viral infections, and severe forms of dengue. The sex-related and hormonal differences in the occurrence of COVID-19 vaccine-related adverse events should be explored in the future. With considerable rates of breakthrough COVID-19 and non-modest rates of AESIs in the vaccinated, blanket recommendations on mass vaccination need strategic reconsideration. Females, individuals with co-morbidities such as hypothyroidism, and those with a history of COVID-19 should be informed about the benefit of protection offered by vaccines as well as the risk of post-vaccination adverse events. An individualized vaccination strategy rather than a further mass rollout may be a better alternative for public health safety.
References
U.S. Food and Drug Administration. COVID-19 vaccines. [Internet]. 2021. https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/covid-19-vaccines. Accessed 5 Jul 2021.
European Medicines Agency. COVID-19 Vaccines. 2021.
Press Information Bureau. Government of India. Press Statement by the Drugs Controller General of India (DCGI) on Restricted Emergency approval of COVID-19 virus vaccine. 2021.
Kaur U, Bala S, Joshi A, Reddy NTS, Japur C, Chauhan M, et al. Persistent Health Issues, Adverse Events, and Effectiveness of Vaccines during the Second Wave of COVID-19: A Cohort Study from a Tertiary Hospital in North India. Vaccines; 2022;10:1153. https://www.mdpi.com/2076-393X/10/7/1153. Accessed 24 Jul 2022.
Behera P, Singh AK, Subba SH, Mc A, Sahu DP, Chandanshive PD, et al. Effectiveness of COVID-19 vaccine (Covaxin) against breakthrough SARS-CoV-2 infection in India. Hum Vaccin Immunother. 2022. https://doi.org/10.1080/21645515.2022.2034456.
Desai D, Khan AR, Soneja M, Mittal A, Naik S, Kodan P, et al. Effectiveness of an inactivated virus-based SARS-CoV-2 vaccine, BBV152, in India: a test-negative, case-control study. Lancet Infect Dis. 2022;22:349–56.
Chen Y, Xu Z, Wang P, Li X, Shuai Z, Ye D, et al. New-onset autoimmune phenomena post-COVID-19 vaccination. Immunology. 2022;165:386–401. https://doi.org/10.1111/imm.13443.
Schultz NH, Sørvoll IH, Michelsen AE, Munthe LA, Lund-Johansen F, Ahlen MT, et al. Thrombosis and thrombocytopenia after ChAdOx1 nCoV-19 vaccination. N Engl J Med. 2021;384:2124–30. https://doi.org/10.1056/NEJMoa2104882.
Singh R, Chakrabarti SS, Gambhir IS, Verma A, Kumar I, Ghosh S, et al. Acute Cardiac Events After ChAdOx1 nCoV-19 Corona Virus Vaccine: Report of Three Cases. Am J Ther. 2022;29:e579-e585. https://doi.org/10.1097/MJT.0000000000001472.
Our World in Data. Coronavirus (COVID-19) Vaccinations [Internet]. https://ourworldindata.org/covid-vaccinations?country=OWID_WRL. Accessed 29 Sep 2022.
Kaur U, Ojha B, Pathak BK, Singh A, Giri KR, Singh A, et al. A prospective observational safety study on ChAdOx1 nCoV-19 corona virus vaccine (recombinant) use in healthcare workers- first results from India. EClinicalMedicine. 2021;38: 101038.
Kaur U, Bala S, Ojha B, Jaiswal S, Kansal S, Chakrabarti SS. Occurrence of COVID-19 in priority groups receiving ChAdOx1 nCoV-19 coronavirus vaccine (recombinant): a preliminary analysis from north India. J Med Virol. 2022;94:407–12.
SPEAC. SO2-D2.1.2 Priority List of COVID-19 Adverse events of special interest: Quarterly update. December 2020.
Lopez Bernal J, Andrews N, Gower C, Gallagher E, Simmons R, Thelwall S, et al. Effectiveness of Covid-19 Vaccines against the B.1.617.2 (Delta) Variant. N Engl J Med. 2021;385:585–94. https://doi.org/10.1056/NEJMoa2108891.
Tsang NNY, So HC, Cowling BJ, Leung GM, Ip DKM. Effectiveness of BNT162b2 and CoronaVac COVID-19 vaccination against asymptomatic and symptomatic infection of SARS-CoV-2 omicron BA.2 in Hong Kong: a prospective cohort study. Lancet Infect Dis. 2022; https://linkinghub.elsevier.com/retrieve/pii/S1473309922007320
Andrews N, Stowe J, Kirsebom F, Toffa S, Rickeard T, Gallagher E, et al. Covid-19 Vaccine effectiveness against the omicron (B.1.1.529) variant. N Engl J Med. 2022;386:1532–46. https://doi.org/10.1056/NEJMoa2119451.
Voysey M, Costa Clemens SA, Madhi SA, Weckx LY, Folegatti PM, Aley PK, et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials. Lancet (London, England). 2021;397:881–91.
Lopez Bernal J, Andrews N, Gower C, Robertson C, Stowe J, Tessier E, et al. Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on covid-19 related symptoms, hospital admissions, and mortality in older adults in England: test negative case-control study. BMJ. 2021;373: n1088.
Maramattom BV, Krishnan P, Paul R, Padmanabhan S, CherukudalVishnuNampoothiri S, Syed AA, et al. Guillain-Barré syndrome following ChAdOx1-S/nCoV-19 vaccine. Ann Neurol. 2021;90:312–4.
Singh R, Kaur U, Singh A, Chakrabarti SS. Refractory hypereosinophilia associated with newly diagnosed rheumatoid arthritis following inactivated BBV152 COVID-19 vaccine. J Med Virol. 2022;94:3482–7. https://doi.org/10.1002/jmv.27742.
Chakrabarti SS, Tiwari A, Jaiswal S, Kaur U, Kumar I, Mittal A, et al. Rapidly progressive dementia with asymmetric rigidity following ChAdOx1 nCoV-19 vaccination. Aging Dis. 2022;13:633–6. https://doi.org/10.14336/AD.2021.1102.
Xie Y, Liu Y, Liu Y. The flare of rheumatic disease after SARS-CoV-2 vaccination: a review. Front Immunol. 2022. https://doi.org/10.3389/fimmu.2022.919979/full.
Sattui SE, Liew JW, Kennedy K, Sirotich E, Putman M, Moni TT, et al. Early experience of COVID-19 vaccination in adults with systemic rheumatic diseases: results from the COVID-19 Global Rheumatology Alliance Vaccine Survey. RMD Open. 2021;7: e001814. https://doi.org/10.1136/rmdopen-2021-001814.
Vojdani A, Vojdani E, Kharrazian D. Reaction of Human Monoclonal Antibodies to SARS-CoV-2 Proteins With Tissue Antigens: Implications for Autoimmune Diseases. Front Immunol. 2021. https://doi.org/10.3389/fimmu.2020.617089/full.
Ursini F, Ruscitti P, Raimondo V, De Angelis R, Cacciapaglia F, Pigatto E, et al. Spectrum of short-term inflammatory musculoskeletal manifestations after COVID-19 vaccine administration: a report of 66 cases. Ann Rheum Dis. 2022;81:440–1. https://doi.org/10.1136/annrheumdis-2021-221587.
Sriwastava S, Sharma K, Khalid S, Bhansali S, Shrestha A, Elkhooly M, et al. COVID-19 vaccination and neurological manifestations: a review of case reports and case series. Brain Sci. 2022;12:407. https://www.mdpi.com/2076-3425/12/3/407
Liu Q, Muruve DA. Molecular basis of the inflammatory response to adenovirus vectors. Gene Ther. 2003;10:935–40.
Acknowledgements
The authors UK and SSC thank the Institutions of Eminence Scheme in the Banaras Hindu University for research support. SSC thanks the Indian Council of Medical Research for research support. UK thanks Mr. Gaurav Kumar Gupta for project assistance.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
No funding support.
Conflicts of interests
The authors have no conflicts of interest to declare.
Ethics approval
The study was conducted after permission from the Institute Ethical Committee of the Institute of Medical Sciences, Banaras Hindu University (Approval number Dean/2021/EC/2526). No human experimentation was performed. All procedures were performed as per the Declaration of Helsinki and its subsequent modifications.
Consent to participate
Written informed consent was taken from each participant for participating in the study
Consent for publication
Not applicable
Availability of data and material
All data produced in the present study are available upon reasonable request to the authors, as per institutional and national legal norms and procedures.
Code availability
Not applicable
Author contributions
Conceptualization: UK, SSC, SK, VJ. Methodology: UK, SSC, VJ. Formal analysis and investigation: UK, VJ, ZF, KM, VS, AD, KLA, AKY. Writing—original draft preparation: UK, ZF. Writing—review and editing: UK, SSC. Supervision: UK, SSC, SK. All authors read and approved the final version.
Supplementary Information
Below is the link to the electronic supplementary material.
40264_2023_1301_MOESM1_ESM.pdf
The article has previously been published as a pre-print (Qeios ID: 457MET) at https://doi.org/10.32388/457MET. (PDF 390 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaur, U., Fatima, Z., Maheshwari, K. et al. Long-Term Safety Analysis of the ChAdOx1-nCoV-19 Corona Virus Vaccine: Results from a Prospective Observational Study in Priority Vaccinated Groups in North India. Drug Saf 46, 553–563 (2023). https://doi.org/10.1007/s40264-023-01301-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-023-01301-8