Abstract
Aim
To compare safety and functional outcomes of intravenous thrombolysis (IVT) between females and males with acute ischaemic stroke (AIS) in relation to preadmission use of antiplatelets.
Methods
Multicentre cohort study of patients admitted from 1 January 2014 to 31 January 2020 to hospitals participating in the Swiss Stroke Registry, presenting with AIS and receiving IVT. Primary safety outcome was in-hospital symptomatic intracerebral haemorrhage (sICH). Primary functional outcome was functional independence at 3 months after discharge. Multivariable logistic regression models were fitted to assess the association between sex and each outcome according to preadmission use of antiplatelets.
Results
The study included 4996 patients (42.51 % females, older than males, median age 79 vs 71 years, p < 0.0001). Comparable proportions of females (39.92 %) and males (40.39 %) used antiplatelets before admission (p = 0.74). In total, 3.06 % females and 2.47 % males developed in-hospital sICH (p = 0.19), with similar odds (adjusted odds ratio, [AOR] 0.93, 95 % confidence interval, [CI] 0.63–1.39). No interaction was found between sex and preadmission use of either single or dual antiplatelets in relation to in-hospital sICH (p = 0.94 and p = 0.23). Males had higher odds of functional independence at 3 months (AOR 1.34, 95 % CI 1.09–1.65), regardless of preadmission use of antiplatelets (interaction between sex and preadmission use of either single or dual antiplatelets p = 0.41 and p = 0.58).
Conclusion
No sex differences were observed in the safety of IVT regarding preadmission use of antiplatelets. Males showed more favourable 3-month functional independence than females; however, this sex difference was apparently not explained by a sex-specific mechanism related to preadmission use of antiplatelets.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
As platelet count and reactivity are higher in females, sex differences in safety and functional outcomes of intravenous thrombolysis in acute ischaemic stroke patients could be influenced by preadmission use of antiplatelets. |
The present cohort study in the Swiss Stroke Registry showed that there is no interaction between patient’s sex and preadmission use of antiplatelets in relation to safety and functional outcomes of intravenous thrombolysis. |
1 Introduction
Intravenous thrombolysis (IVT) with recombinant tissue plasminogen activator (rtPA) is the only approved pharmacological reperfusion treatment for acute ischaemic stroke (AIS) [1]. Clinical outcomes of IVT with rtPA rely strictly on patient individuality, which encompasses biological sex among others [2, 3]. Sex differences have been investigated in relation to safety and functional outcomes of IVT with rtPA in patients with AIS. Concerning IVT safety, a sex relationship is debated, with only one large-scale study showing that male sex was independently associated with a higher risk of in-hospital symptomatic intracerebral haemorrhage (sICH) after IVT [4], while several other studies did not observe sex differences in this safety outcome measure [5,6,7]. Controversial findings have been reported on sex differences in functional outcomes of IVT: some studies documented a poorer functional outcome and quality of life in females compared to males following IVT [5, 7, 8], other studies reported the opposite [9, 10], while others observed no sex differences [4, 11, 12].
Although the use of antiplatelets (single or dual) before stroke onset was associated with an increased risk of haemorrhagic complications following IVT [13,14,15,16,17], preadmission use of antiplatelets is not considered a contraindication for IVT with rtPA in eligible patients with AIS [1]. Data on the association of preadmission use of antiplatelets with safety and functional outcomes after IVT with rtPA in patients with AIS are also contradictory. A few previous studies found no differences in the safety and functional outcomes of IVT with rtPA in patients with preadmission use of antiplatelets compared to naïve patients [18, 19], but in contrast, other studies reported that preadmission use of antiplatelets could compromise acute post-thrombolytic safety, although apparently without affecting long-term functional outcomes [14, 15, 20]. Finally, a higher risk of antiplatelet-associated in-hospital sICH was reported concomitant with an increased rate of favourable functional outcome after IVT in patients with preadmission use of antiplatelets [13, 16]. Data on dual antiplatelet therapy are equally controversial. In a few studies, preadmission use of dual antiplatelets was described as a predictor of increased risk of in-hospital sICH following IVT compared with naïve patients [16, 21, 22]. However, other studies showed comparable in-hospital sICH rates and functional outcomes following IVT regardless of pre-stroke preadmission use of dual antiplatelets [23,24,25].
Platelet count and reactivity are higher in females [26,27,28]. Among factors that could contribute to determine sex differences in platelet biology there are the expression of genetic polymorphisms affecting platelet responsiveness to agonists (with and without antiplatelet therapies), inflammatory marker levels that amplify platelet response and endothelial activation (thus influencing the thrombotic risk), and the role of specific hormones in mediating platelet activation and function [29, 30]. Moreover, in the response to antiplatelets, the propensity for increased bleeding among females has also been recognised although the link with possible sex differences in platelet biology is unknown [31,32,33,34].
Against this background, females with preadmission use of antiplatelets and AIS could respond differently from males to IVT. Studies specifically evaluating the interaction between patient’s sex and preadmission use of antiplatelets in relation to both safety and functional outcomes of IVT in patients with AIS are lacking. The aim of this cohort study was to compare safety and functional outcomes after IVT between females and males with regard to preadmission use of antiplatelets in a large multicentre Swiss cohort reflecting daily clinical practice of acute stroke care.
2 Methods
2.1 Swiss Stroke Registry
This is a multicentre cohort study from the Swiss Stroke Registry (SSR) [35]. The SSR is a national web-based registry established in 2014. It collects standardised data from all consecutive patients with acute stroke, cerebral haemorrhage and other cerebrovascular diseases, who have been hospitalised in certified stroke units and comprehensive stroke centres across Switzerland. Standardised data collection in the SSR includes patient demographics, medical history, medications used before admission (pre-stroke), pre-stroke functional status, diagnostic procedures, in-hospital treatments and outcomes (both during hospitalisation and at 3 months after discharge). Clinical evaluations, as well as National Institutes of Health Stroke Scale (NIHSS) and modified Rankin Scale (mRS) assessments, are performed by certified stroke neurologists as part of their clinical activity. If an in-person visit is not possible at 3 months, the mRS score is assessed by a phone interview with mRS-certified examiners. The Clinical Trial Unit of the University Hospital of Basel, Switzerland, manages the database and provides anonymised data for study purposes. The registry and the present study were both approved by the responsible ethics committee (CE Req-2020-01042). This study complies with the Declaration of Helsinki. The study followed STROBE recommendations [36].
2.2 Study Population
Consecutive patients admitted from 1 January 2014 to 31 January 31 2020, to hospitals participating in the SSR, presenting with AIS and receiving IVT with rtPA without endovascular treatment were included. Patients who used anticoagulants before admission and patients with missing data on age, sex and preadmission use of antiplatelets were excluded. The study population was divided into two groups based on patient’s biological sex and thereafter compared in terms of preadmission use of antiplatelets. Preadmission use of antiplatelets was distinguished as single (with either aspirin, clopidogrel, prasugrel, ticagrelor or dipyridamole) or dual (a combination of any of the aforementioned antiplatelets).
2.3 Outcomes
The primary safety outcome was in-hospital sICH defined as ICH with ≥4-point worsening of the NIHSS score (a measure of neurological deficits ranging from 0 to 42, with higher scores indicating greater stroke severity) [37] and occurring within 7 days of AIS onset. The primary functional outcome was functional independence defined as mRS score of 0 to 2, at 3 months after hospital discharge. The mRS is an ordinal scale that ranges from 0 (no symptoms) to 6 (death), which is widely used to measure functional outcomes [38]. Secondary functional outcomes at 3 months after discharge included favourable functional outcome (mRS score 0–1), global mRS score assessment evaluated by shift analysis [39], poor functional outcome (mRS score 3 to 6) and severe disability or death (mRS score 5–6).
2.4 Statistical Analysis
Medians with 25th and 75th percentiles were used to describe the distribution of continuous variables; counts and percentages described categorical variables. Baseline characteristics of the study population were compared between females and males using the Pearson χ2 test for categorical variables and the Wilcoxon rank sum test for continuous variables. Multivariable logistic regression models were fitted to assess the association between the patient’s sex and each outcome according to preadmission use of antiplatelets (single vs no use or dual vs no use). With this aim, interaction variables between patient’s sex and preadmission use of antiplatelets (either single or dual) were introduced. In the shift analysis, an ordinal logistic regression model was used and an overall point estimate provided as an odds ratio (OR) representing a shift in scores on the mRS. All models were adjusted for meaningful demographic and clinical patient variables potentially influencing outcome measures, including age, NIHSS score at admission, pre-stroke disability (defined by mRS score), medical history of hypertension, hyperlipidaemia, diabetes, coronary heart disease, smoking, atrial fibrillation, pre-stroke sICH and previous stroke, in-hospital acute treatment with anticoagulants. Supplementary Table 1 provides details of the variables included in the regression models. Analyses were performed on complete observations. Due to 12.15% and 11.63% of missing data for the pre-stroke disability variable in females and males, respectively, multiple imputation was used and multivariable regression models were thereafter repeated as sensitivity analyses. Owing to small sample sizes, separate analyses for individual antiplatelet regimens could not be performed. Analyses were performed using the Statistical Analysis System Software (version 9.4; SA Institute, Cary, NC). For all analyses, two-tailed p values less than 0.05 were considered significant.
3 Results
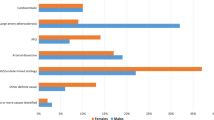
Of 5412 patients with AIS receiving IVT and admitted to Stroke Units or Stroke Centres from the SSR network between 1 January 2014 and 31 January 2020, 4996 patients met the inclusion criteria (Fig. 1). Of these, 2124 (42.51 %) were females and 2872 (57.49 %) were males. Table 1 summarizes the baseline characteristics of the study population according to the patient’s sex. Females were older than males (median 79 years, 25th–75th percentiles, 69–86 years, vs 71 years, 25th–75th percentiles 61–80 years, p < 0.0001). Comparable proportions of females (39.92 %) and males (40.39 %) used antiplatelets before admission (single, 37.99 % and 37.85 %, or dual, 1.93 % and 2.54 %, p = 0.7402). Females had a greater prevalence of hypertension (74.15 % vs 69.08 %, p < 0.0001) and atrial fibrillation (21.47 % vs 17.31 %, p < 0.0001) and were more likely to receive anti-hypertensive medications before admission (61.21 % vs 52.75 %, p < 0.0001), but had a lower prevalence of diabetes (16.76 % vs 20.19 %, p = 0.003) and hyperlipidaemia (59.84 % vs 65.08 %, p = 0.0004) and were less likely to receive lipid-lowering agents before admission (23.26 % vs 30.15 %, p < 0.0001). Before stroke onset, females displayed greater disability than males did (p < 0.0001), and presented higher NIHSS scores at admission (p < 0.0001). Concerning AIS aetiology, cardio-embolism was the most frequent pathogenic subtype among both female and male patients (593, 27.92 % and 688, 23.96 %, respectively).
3.1 Primary Safety Outcome: In-Hospital sICH
Overall, 136 (2.72 %) patients presented in-hospital sICH after IVT, with similar proportions of females and males, both as crude rates (3.06 % vs 2.47 %, p = 0.186, Table 2), and after risk adjustment (adjusted odds ratio, AOR 0.93, 95 % CI 0.63–1.39, Table 3). In the multivariable logistic regression analysis on the association of baseline patient characteristics with the probability of in-hospital sICH, preadmission use of a single antiplatelet agent did not significantly increase the odds of in-hospital sICH compared with no preadmission use of antiplatelets (AOR 1.40, 95 % CI 0.90–2.19) (Table 3). By contrast, preadmission use of dual antiplatelets was associated with higher odds of in-hospital sICH compared with no preadmission use of antiplatelets (AOR 2.72, 95 % CI 1.04–7.10) (Table 3), with a statistically significant trend from preadmission use of single to dual antiplatelets (p = 0.0392). No interaction was found between patient’s sex and preadmission use of either single or dual antiplatelets regarding in-hospital sICH (p = 0.94 and p = 0.23, respectively). The association between preadmission use of dual antiplatelets and in-hospital sICH was confirmed after multiple imputation (AOR 2.44, 95 % CI 1.06–5.61, Table 3).
3.2 Primary Functional Outcome: Functional Independence at 3 Months after Discharge
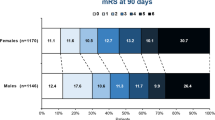
The proportion of patients who were independent at 3 months after discharge (mRS score 0–2) was 44.21 % (939/2124) of females and 54.70 % (1571/2872) of males (p < 0.0001). In the multivariable logistic regression analysis on the association of baseline patient characteristics with the probability of functional independence at 3 months after discharge, the odds of reaching functional independence at 3 months was higher in males than in females (AOR 1.34, 95 % CI 1. 09–1.65, Table 3). Preadmission use of either a single antiplatelet agent or dual antiplatelets increased the odds of functional independence at 3 months after discharge (AOR 0.88, 95 % CI 0.70–1.12, and AOR 1.62, 95 % CI 0.72–3.62, respectively, Table 3). No interaction was found between patient’s sex and preadmission use of either single or dual antiplatelets in relation to the primary functional outcome (p = 0.41 and p = 0.58, respectively). Sex differences in functional independence at 3 months after discharge were confirmed after multiple imputation of missing data (AOR 1.33, 95 % CI 1.10–1.60, Table 3).
3.3 Secondary Functional Outcomes at 3 Months after Discharge
Compared to females, males with AIS receiving IVT more frequently had a favourable functional outcome at 3 months after discharge (42.58 %, males vs 34.59 %, females, p < 0.0001), a better functional outcome across all levels of disability and mortality (mRS score ordinal, shift analysis, p < 0.0001), a lower rate of poor functional outcome (8.61 %, males vs 10.07 %, females, p < 0.0001) and a lower rate of severe disability or death (4.21 %, males vs 6.73 %, females, p < 0.0001) (Table 2).
In the multivariable logistic regression analyses on the association of baseline patient characteristics with the probability of secondary functional outcomes at 3 months after discharge, the odds of greater mRS scores were lower in males than in females (shift analysis, AOR 0.86, 95 % CI 0.75–0.99), as were the odds of poor functional outcome (AOR 0.75, 95 % CI 0.61– 0.92). No sex differences emerged from the multivariable logistic regression analyses for either the odds of favourable functional outcome or the odds of severe disability or death (both at 3 months after discharge) (Table 3).
Neither preadmission use of a single antiplatelet agent nor of dual antiplatelets increased the odds of any of the secondary functional outcomes assessed at 3 months after discharge (Table 3).
There was no interaction between patient’s sex and preadmission use of antiplatelets with regard to any of the secondary functional outcomes assessed (Table 4).
Multivariable logistic regression analyses after multiple imputation of missing data confirmed the lower odds of poor functional outcome in males than in females (AOR 0.75, 95 % CI 0.62–0.91, Table 3).
4 Discussion
We found no sex differences in the safety of IVT in AIS patients in relation to preadmission use of antiplatelets. Males showed higher odds of functional independence at 3 months than females after IVT; however, the sex difference was apparently not explained by a sex-specific mechanism related to preadmission use of antiplatelets.
In terms of safety, we confirmed that preadmission use of antiplatelets is per se independently associated with increased odds of in-hospital sICH (with a statistically significant trend from preadmission use of single to dual antiplatelets), but this relationship was not sex related. Overall, the rate of in-hospital sICH events occurring in our study population was comparable to that previously reported in populations from RCTs as well as from other registries [22, 40], with similar proportions for females and males as also highlighted in antecedent observational studies [4, 9]. Moreover, after adjustment, no sex differences were observed in the odds of in-hospital sICH following IVT, confirming a substantial part of the existing literature [5,6,7]. Since both antiplatelets and IVT with rtPA interfere with the haemostatic balance, the concern about an increased risk of haemorrhagic complications in AIS patients with preadmission use of antiplatelets when treated with IVT is high, all the more so because of the heterogeneous findings from previous studies [14, 15, 18,19,20]. Consistent with the only earlier study that assessed by subgroup analysis the effect of patient’s sex on the risk of in-hospital sICH in relation to preadmission use of antiplatelets [16], the present study confirmed that preadmission use of dual antiplatelets increased the odds of in-hospital sICH following IVT with a trend from single antiplatelet therapy to dual without sex differences.
In contrast, we observed better benefits of IVT in males compared to females. Males had more favourable functional outcomes at 3 months after discharge, confirming some previous observations [5, 7, 8]. Nevertheless, sex differences in IVT efficacy apparently were not explained by a sex-specific mechanism related to preadmission use of antiplatelets. We performed several analyses to address if our findings may be due in part to a bias towards more unfavourable characteristics contributing to sex differences in stroke functional outcomes among females in our population (older age, worse pre-stroke disability and higher NIHSS score at admission compared with males) [41]. Nevertheless, after correcting our analyses and adjusting the regression models for these variables, we still observed sex differences in functional outcomes. The reasons for sex disparities in IVT responses could be found in the living situation, family and caregiver support and the social background, but these assumptions are outside the goal of our study.
The results of this study are important in the era of precision- and gender-medicine, where biological sex is considered a health and disease modifier [42], and adds new data on the ongoing debate on sex differences on the safety and functional outcomes of IVT in females and males in relation to preadmission use of antiplatelets [2]. In the context of personalised medicine, optimisation of IVT therapy should not disregard the biological sex of AIS patients given that cellular, anatomical, hormonal and behavioural differences exist between the sexes and are associated with pharmacological responses and health [43, 44]. In order to consider a series of meaningful covariates expected to be confounders of the association between sex, preadmission use of antiplatelets and safety and functional outcomes, a multivariable approach was used here, similar to previous studies [16, 22, 25]. A better understanding of sex differences could lead to the development of optimized therapies for the prevention and treatment of acute ischaemic events while minimising bleeding risk.
This study has a number of strengths. First, it was based on a pre-specified methodological approach with a sex-specific design and an appropriate statistical analysis plan (multivariable analysis, formal interaction tests). Second, the study provides generalisability of the results because it refers to a stroke population treated with IVT from a large-scale registry that reflects real-world clinical practice. Third, the study performed a multiple imputation approach to overcome some missing data, which can be expected in large-scale registries like the SSR.
However, the study also has several limitations. Owing to small sample sizes, separate analyses for individual antiplatelet agent regimens could not be performed. Adherence to antiplatelets could have influenced stroke onset; however, this information is not recorded in SSR. In the SSR, the precise reason(s) why a patient was treated with IVT and not with mechanical thrombectomy as well as detailed radiological findings regarding the subtypes of haemorrhagic transformations are lacking. Moreover, in the SSR, hypertension following IVT, which is a known risk factor for the development of sICH [45], is not documented. Lastly, sex differences in the living situation, family support and social background were not included as covariates in the multivariable models of this study because they are not collected in the SSR. Further, due to the observational nature of the study, estimates may be affected by bias and unmeasured confounding.
5 Conclusion
The present study showed that there is no interaction between patient’s sex and preadmission use of antiplatelets in relation to IVT safety and functional outcomes. Therefore, when clinicians use IVT to treat AIS patients, worse safety and functional outcomes in females compared to males are not to be expected on the basis of the notion of preadmission use of antiplatelets. Future studies are needed that aim to discern the mechanism(s) that underpin the better functional outcomes observed in males with AIS following IVT.
References
Berge E, Whiteley W, Audebert H, et al. European stroke organisation (ESO) guidelines on intravenous thrombolysis for acute ischaemic stroke. Eur Stroke J. 2021;6(1):1–12.
Bushnell C, Howard VJ, Lisabeth L, et al. Sex differences in the evaluation and treatment of acute ischaemic stroke. Lancet Neurol. 2018;17(7):641–50.
Jiang M, Ma C, Li H, et al. Sex dimorphisms in ischemic stroke: from experimental studies to clinic. Front Neurol. 2020;11:504.
Lorenzano S, Ahmed N, Falcou A, et al. Does sex influence the response to intravenous thrombolysis in ischemic stroke?: answers from safe implementation of treatments in Stroke-International Stroke Thrombolysis Register. Stroke. 2013;44(12):3401–6.
Spaander FH, Zinkstok SM, Baharoglu IM, Thrombolysis in Ischemic Stroke Patients Collaborators (TrISP), et al. Sex differences and functional outcome after intravenous thrombolysis. Stroke. 2017;48(3):699–703.
Cai B, Li SD, Li H, Liu ZQ, Peng B. Sex differences of acute stroke treatment and in hospital outcomes after intravenous thrombolysis in patients with ischemic stroke. Front Neurol. 2020;11: 545860.
Carcel C, Wang X, Sandset EC, et al. Sex differences in treatment and outcome after stroke: pooled analysis including 19,000 participants. Neurology. 2019;93(24):e2170–80.
Elkind MS, Prabhakaran S, Pittman J, Koroshetz W, Jacoby M, Johnston KC, GAIN Americas Investigators. Sex as a predictor of outcomes in patients treated with thrombolysis for acute stroke. Neurology. 2007;68(11):842–8.
Lee S-J, Heo SH, Ambrosius WT, Bushnell CD. Factors mediating outcome after stroke: gender, thrombolysis, and their interaction. Transl Stroke Res. 2018;9(3):267–73.
Bonkhoff AK, Karch A, Weber R, Wellmann J, Berger K. Female stroke: sex differences in acute treatment and early outcomes of acute ischemic stroke. Stroke. 2021;52(2):406–15.
Emberson J, Lees KR, Lyden P, Stroke Thrombolysis Trialists’ Collaborative Group, et al. Effect of treatment delay, age, and stroke severity on the effects of intravenous thrombolysis with alteplase for acute ischaemic stroke: a meta-analysis of individual patient data from randomised trials. Lancet. 2014;384(9958):1929–35.
Hametner C, MacIsaac RL, Kellert L, Abdul-Rahim AH, Ringleb PA, Lees KR, VISTA Collaborators. Sex and stroke in thrombolyzed patients and controls. Stroke. 2017;48(2):367–74.
Uyttenboogaart M, Koch MW, Koopman K, Vroomen PC, De Keyser J, Luijckx GJ. Safety of antiplatelet therapy prior to intravenous thrombolysis in acute ischemic stroke. Arch Neurol. 2008;65(5):607–11.
Pan X, Zhu Y, Zheng D, Liu Y, Yu F, Yang J. Prior antiplatelet agent use and outcomes after intravenous thrombolysis with recombinant tissue plasminogen activator in acute ischemic stroke: a meta-analysis of cohort studies and randomized controlled trials. Int J Stroke. 2015;10(3):317–23.
Luo S, Zhuang M, Zeng W, Tao J. Intravenous thrombolysis for acute ischemic stroke in patients receiving antiplatelet therapy: a systematic review and meta-analysis of 19 studies. J Am Heart Assoc. 2016;5(5): e003242.
Xian Y, Federspiel JJ, Grau-Sepulveda M, et al. Risks and benefits associated with prestroke antiplatelet therapy among patients with acute ischemic stroke treated with intravenous tissue plasminogen activator. JAMA Neurol. 2016;73(1):50–9.
Robinson TG, Wang X, Arima H, ENCHANTED Investigators, et al. Low-versus standard-dose alteplase in patients on prior antiplatelet therapy: the ENCHANTED trial (enhanced control of hypertension and thrombolysis stroke study). Stroke. 2017;48:1877–83.
Tsivgoulis G, Katsanos AH, Zand R, et al. Antiplatelet pretreatment and outcomes in intravenous thrombolysis for stroke: a systematic review and meta-analysis. J Neurol. 2017;264(6):1227–35.
Frey BM, Boutitie F, Cheng B, WAKE-UP investigators, et al. Safety and efficacy of intravenous thrombolysis in stroke patients on prior antiplatelet therapy in the WAKE-UP trial. Neurol Res Pract. 2020;2:40.
Schellinger PD, Tsivgoulis G. Another enchantment from ENCHANTED (enhanced control of hypertension and thrombolysis stroke study): are savings and safety more salutary than efficacy? Stroke. 2017;48:1720–2.
Cucchiara B, Kasner SE, Tanne D, SAINT Investigators, et al. Factors associated with intracerebral hemorrhage after thrombolytic therapy for ischemic stroke: pooled analysis of placebo data from the Stroke-Acute Ischemic NXY Treatment (SAINT) I and SAINT II trials. Stroke. 2009;40(9):3067–72.
Diedler J, Ahmed N, Sykora M, et al. Safety of intravenous thrombolysis for acute ischemic stroke in patients receiving antiplatelet therapy at stroke onset. Stroke. 2010;41(2):288–94.
Tsivgoulis G, Katsanos AH, Mavridis D, et al. Intravenous thrombolysis for ischemic stroke patients on dual antiplatelets. Ann Neurol. 2018;84(1):89–97.
Malhotra K, Katsanos AH, Goyal N, et al. Safety and efficacy of dual antiplatelet pretreatment in patients with ischemic stroke treated with IV thrombolysis: a systematic review and meta-analysis. Neurology. 2020;94(7):e657–66.
Altersberger VL, Sturzenegger R, Räty S, Thrombolysis in Stroke Patients (TRISP) collaborators, et al. Prior dual antiplatelet therapy and thrombolysis in acute stroke. Ann Neurol. 2020;88(4):857–9.
Segal JB, Moliterno AR. Platelet counts differ by sex, ethnicity, and age in the United States. Ann Epidemiol. 2006;16(2):123–30.
Johnson M, Ramey E, Ramwell PW. Sex and age differences in human platelet aggregation. Nature. 1975;253(5490):355–7.
Faraday N, Goldschmidt-Clermont PJ, Bray PF. Gender differences in platelet GPIIb-IIIa activation. Thromb Haemost. 1997;77(4):748–54.
Wang TY, Angiolillo DJ, Cushman M, et al. Platelet biology and response to antiplatelet therapy in women: implications for the development and use of antiplatelet pharmacotherapies for cardiovascular disease. J Am Coll Cardiol. 2012;59(10):891–900.
Patti G, De Caterina R, Abbate R, Working Group on Thrombosis of the Italian Society of Cardiology, et al. Platelet function and long-term antiplatelet therapy in women: is there a gender-specificity? A “state-of-the-art” paper. Eur Heart J. 2014;35(33):2213–2223b.
Berger JS, Roncaglioni MC, Avanzini F, Pangrazzi I, Tognoni G, Brown DL. Aspirin for the primary prevention of cardiovascular events in women and men: a sex-specific meta-analysis of randomized controlled trials. JAMA. 2006;295:306–13.
Boersma E, Harrington RA, Moliterno DJ, et al. Platelet glycoprotein IIb/IIIa inhibitors in acute coronary syndromes: a meta-analysis of all major randomised clinical trials. Lancet. 2002;359:189–98.
Capodanno D, Angiolillo DJ. Impact of race and gender on antithrombotic therapy. Thromb Haemost. 2010;104:471–84.
Renda G, Patti G, Lang IM, Working Group on Thrombosis of the Italian Society of Cardiology, et al. Thrombotic and hemorrhagic burden in women: Gender-related issues in the response to antithrombotic therapies. Int J Cardiol. 2019;286:198–207.
Bonati L, Baumgartner RW, Bonvin C, et al. Ein Werkzeug für dieQualitätssicherung und Forschung. Swiss Med Forum. 2016;16:168–9. https://doi.org/10.4414/smf.2016.02576.
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370(9596):1453–7.
Goldstein LB, Bertels C, Davis JN. Interrater reliability of the NIH stroke scale. Arch Neurol. 1989;46(6):660–2.
Kasner SE. Clinical interpretation and use of stroke scales. Lancet Neurol. 2006;5(7):603–12.
Bath PM, Lees KR, Schellinger PD, European Stroke Organisation Outcomes Working Group, et al. Statistical analysis of the primary outcome in acute stroke trials. Stroke. 2012;43(4):1171–8.
National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med. 1995;333(24):1581–7.
Phan HT, Blizzard CL, Reeves MJ, Thrift AG, Cadilhac D, Sturm J, et al. Sex differences in long-term mortality after stroke in the INSTRUCT (INternational STRoke oUtComes sTudy): a meta-analysis of individual participant data. Circ Cardiovasc Qual Outcomes. 2017;10(2): e003436.
Mauvais-Jarvis F, Bairey Merz N, Barnes PJ, et al. Sex and gender: modifiers of health, disease, and medicine. Lancet. 2020;396(10250):565–82.
Franconi F, Campesi I. Sex and gender influences on pharmacological response: an overview. Expert Rev Clin Pharmacol. 2014;7(4):469–85.
Bartz D, Chitnis T, Kaiser UB, et al. Clinical advances in sex- and gender-informed medicine to improve the health of all: a review. JAMA Intern Med. 2020;180(4):574–83.
Waltimo T, Haapaniemi E, Surakka IL, Melkas S, Sairanen T, Sibolt G, et al. Post-thrombolytic blood pressure and symptomatic intracerebral hemorrhage. Eur J Neurol. 2016;23(12):1757–62.
Acknowledgements
Data used in preparation of this article were obtained from the Swiss Stroke Registry. We are grateful to the patients, families and clinical staff for their cooperation during investigation in this study. We thank Melanie Price Hirt for English language correction and editing. We thank Antonia Lenstra for the support. This work was presented at the 5th SFCNS Congress—Swiss Federation of Clinical Neuro-Societies Basel, Switzerland, September 28–30, 2022) and an abstract (P153) is available in Clin. Transl. Neurosci. 2022, 6(3): 22. https://doi.org/10.3390/ctn6030022.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Author contributions
RN and CWC researched literature and conceived the study. RN, FR, AP, PA, AC and CWC were involved in protocol development. RN, FR, AP and CWC were involved in data analysis. PA, GB, SS, DS, PM, KN, LB, GK, JN, TN, AL, SW, LS, FM, BR, NP, SR, MLM, EC, UF and CWC were involved in gaining ethical approval and patient recruitment. RN, FR and CWC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Open access funding provided by Università della Svizzera italiana.
Conflicts of interest
CWC. is a member of the iSchemaView Medical and Scientific Advisory Board. RN, FR, AP, PA, GB, SS, DS, PM, KN, LB, GK, JN, TN, AL, SW, LS, FM, BR, NP, SR, MLM, EC, UF, and AC declare that they have no potential conflicts of interest that might be relevant to this work.
Ethics approval
The registry and the present study were both approved by the responsible ethics committee (CE Req-2020-01042).
Consent to participate
Written informed consent was obtained from all subjects before the study.
Consent for publication
Not applicable.
Availability of data and material
The data that support the findings of this study are available from the Corresponding Author upon reasonable request.
Code availability
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Noseda, R., Rea, F., Pagnamenta, A. et al. Sex Differences in Outcomes of Intravenous Thrombolysis in Acute Ischemic Stroke Patients with Preadmission Use of Antiplatelets. CNS Drugs 37, 351–361 (2023). https://doi.org/10.1007/s40263-023-00997-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40263-023-00997-7