Abstract
Alcohol-related liver disease (ALD) is a major healthcare/economic burden and one of the leading causes of liver transplantation. New epidemiological studies that detail the course of the disease are needed since, despite its high prevalence, it is still a stigmatised condition with underlying pathology. Alcoholic hepatitis, as the highest expression of ALD, has high morbidity. Current treatments have suboptimal results with the exception of liver transplantation. Epidemiological studies must also be developed to improve prevention and implement early diagnosis policies. It is essential to develop multidisciplinary health models that allow the liver transplantation candidate to be approached in a holistic way, both for indication and follow up. The implementation of alcohol consumption biomarkers (ethyl glucuronide, phosphatidylethanol) can assist in diagnosing and supporting recovery. There are several initiatives with new therapies that must be validated to establish their effectiveness and indication.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Alcohol-related liver disease (ALD) is still a stigmatised condition, and epidemiological studies are needed to detail the course of the disease, improve prevention, and implement early diagnosis policies. |
The implementation of alcohol consumption biomarkers can assist in diagnosing and supporting recovery. |
Current treatments for severe alcoholic hepatitis have suboptimal results with the exception of liver transplantation. Therefore, it is essential to develop multidisciplinary health models that allow the liver transplantation candidate to be approached holistically in the clinical management of ALD complications. |
1 Introduction
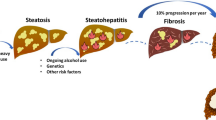
Alcohol-related liver disease (ALD) represents a spectrum of liver injury resulting from alcohol use, ranging from hepatic steatosis to more advanced forms such as alcoholic hepatitis (AH). ALD develops through several stages, beginning with hepatic steatosis and, in some individuals, gradually progresses to alcohol-related steatohepatitis culminating in cirrhosis (Fig. 1) [1, 2]. Progression through these stages is not linear. It depends on not only continued alcohol consumption but also other risk factors such as female sex, diet, genetic susceptibility and comorbid liver disease [3]. ALD continues to be a highly stigmatised condition to which insufficient attention has been paid [3]. Unfortunately, epidemiological data, such as alcohol consumption, are not very homogeneous [4].
Natural history of alcohol-related liver disease [1]. Open access article but not commercial. Reproduced with permission. AH alcoholic hepatitis, HCC hepatocellular carcinoma
This article provides an updated view on the prevalence, diagnosis, clinical spectrum and clinical management of ALD, taking into account the most recent publications and the recommendations of the leading scientific societies [4,5,6,7].
2 Alcohol-Related Liver Disease
2.1 Epidemiology
Accurate assessment of ALD prevalence is challenging, particularly given the difficulty with identifying early, asymptomatic stages of ALD, and the stigma of alcohol use and lack of candour in reporting [4]. The estimated prevalence of ALD in the US is 2–2.5%; in the European Union (EU) plus the UK it is estimated at 24–46 per million persons per year [8]. ALD, even as an advanced liver disease, is widely underestimated in the world compared with liver disease of viral or metabolic aetiology [9]. A substantial number of liver cirrhosis cases have been found during screening of the general population but in very few of these studies instruments for evaluation of alcohol consumption were used [10]. The prevalence of AH is estimated at 20% of alcoholics and, in 2007 in the US, it accounted for 0.71% of all hospital admissions [11]. Furthermore, the incidence of AH appears to be increasing. Specifically, in Denmark, the incidence of AH from 1999 to 2008 increased from 34 to 46 cases per million [12]. The burden of alcoholic cirrhosis has been increasing in recent years [13]; in relation to other liver diseases, its costs have increased the most [14]. In one-third of patients with significant liver fibrosis, alcohol consumption and metabolic features for liver diseases coexist, as demonstrated by a French cohort study carried out in 1358 subjects [15].
US reports of early liver transplants for AH have reported a notable increase in liver transplants for ALD and in the number of patients affected by AH, especially since 2014 [16].
2.2 Diagnosis
To make an adequate diagnosis of ALD, it is essential to carry out a correct assessment of alcohol use, taking into account the pattern of signs and symptoms of the patient, and to verify this by performing additional tests (analytical, radiological, elastography and liver biopsy) (Table 1) [17].
2.2.1 Alcohol Use Assessment: Alcohol Use Disorder
In ALD, the diagnosis goes beyond liver evaluation, and it is vital to evaluate alcohol consumption; psychiatric teams are key for evaluating patients. An adequate assessment of alcohol intake must consider the amount, the pattern of consumption and the chronology (Table 2) [18,19,20,21]. In this regard, a detailed medical history indicating the age of initiation of consumption, a registry of whether the consumption is daily or sporadic and, if possible, an interview with relatives can be useful. Efforts to uncover harmful alcohol use and dependence are aided by structured, validated screening tools such as the Alcohol Use Disorders Inventory Test (AUDIT) [22] (Recommendation Grade A1) or the CAGE Questionnaire [23]. Assessment of harmful alcohol consumption should be carried out in primary care facilities and emergency rooms (Recommendation Grade A2) [24].
2.2.2 Supplementary Tests
Non-invasive accurate alternatives, including several biochemical tests and a liver stiffness measurement, are increasingly being utilised to evaluate patients with suspected ALD (Recommendation Grade A1). In patients with high alcohol consumption, alteration of transaminases is observed with an aspartate aminotransferase (AST): alanine aminotransferase (ALT) ratio >2. Hyperbilirubinemia, coagulopathy, anaemia, ferritin elevation and leucocytosis with an elevated neutrophil count are common [17]. Radiological tests, such as abdominal ultrasound, computed tomography or magnetic resonance imaging, can be used to confirm data of chronic liver disease, steatosis and signs of portal hypertension, and rule out tumours or vascular complications. The estimation of liver fibrosis can be performed by transient elastography (Fibroscan®), observing a good correlation with liver biopsy for estimation. Although this method has not been approved as a standard for diagnosis of liver fibrosis in ALD, unlike in viral hepatitis, there are several studies that supports the use of Fibroscan® in ALD. This non-invasive elastography test is currently the most reliable noninvasive method for the diagnosis of advanced liver fibrosis and cirrhosis in ALD [25]. In a prospective, direct comparison of tests, Enhanced Liver Fibrosis test (ELF) and Fibroscan® identified advanced liver fibrosis in alcoholic patients from primary and secondary care with high diagnostic accuracy (AUROC values of 0.90 or higher using biopsy as reference) [26] and several studies have used transient elastography in patients with ALD [25, 27,28,29,30,31].
Finally, a liver biopsy can be carried out in patients who need confirmation of the aetiology or a complete diagnosis (Recommendation Grade A1).
2.2.3 Biomarkers of Alcohol Use
Alcohol use biomarkers are also of great importance in the diagnosis of ALD. These biomarkers can be used to aid in the diagnosis and to support recovery. Ethyl sulfate, phosphatidylethanol (PEth) or ethyl glucuronide (EtG) are not affected by liver disease and therefore are preferable [32]. PEth can be easily collected in dried blood spots—unfortunately it is not done routinely, nor is it widely available and it is expensive; EtG is measurable in urine and is cheaper than PEth.
2.3 Alcohol-Related Liver Disease (ALD) Management
Recent guidelines for ALD management have been released [4, 6, 24, 33], as summarized in the following section.
2.3.1 Importance of Abstinence
Alcohol consumption is the most important prognostic factor in ALD, and achieving abstinence is considered a fundamental aspect for the management of both this disease [34] and AH [35]. Because abstinence is the most important factor in improving survival from ALD, evaluation by the cessation team (psychiatrist, addiction specialists) is mandatory. In patients with moderate deprivation syndrome, treatment with baclofen could be considered; in those with severe deprivation syndrome, the use of benzodiazepines could be evaluated [4,5,6].
2.3.2 Assessment of Interaction with Other Pathologies
Co-existent heavy alcohol use by patients with other liver diseases (hepatitis C virus infection, metabolic-associated fatty liver disease, hemochromatosis, etc.) promotes the rapid development of advanced fibrosis and cirrhosis. Conversely, the different components of metabolic syndrome are important risk factors for alcohol-associated liver injury. High alcohol consumption associated with a high body mass index has been shown to have a supra-additive interaction, reporting an increase in mortality [36] and the risk of hepatocarcinoma [37]. Identification and management of these cofactors is highly encouraged (Recommendation Grade A1).
3 Complications of ALD: Alcoholic Hepatitis (AH)
In patients with cirrhosis or advanced fibrosis, AH is defined as an abrupt development of jaundice in those patients with recent very high alcohol consumption (Recommendation Grade A1) (>100 g/OH/day; binge drinking), a slight rise in transaminase levels (AST, ALT <300–400 IU/mL, ratio 2:1) and where other causes of liver disease have been ruled out. Mortality at 28 days is estimated between 25 and 40%. If a biopsy is available, a confirmed AH diagnosis can be made [4,5,6, 38].
In acute AH, the pattern of phenomena is vast—including the alteration of the intestinal flora, increased permeability in the digestive tract, decreased capacity for liver regeneration and cellular damage—leading to liver insufficiency and portal hypertension characteristic of AH [39]. A liver biopsy is recommended in case of doubt in the diagnosis. In these patients, by assessing fibrosis, polymorphonuclear infiltration, bilirubinostasis and megamitochondria, a score that will determine the prognosis can be established [40]. Recently, the possibility of confirming the diagnosis by determining cytokeratin 18 (M65) levels has been established, making it possible to avoid biopsy in a high percentage of patients [41].
3.1 Severity Assessment
Assessing the severity of the ALD allows us to establish adequate management (Recommendation Grade A1), and for this several scoring models have been developed. Currently, the most widely used scale is the model for end-stage liver disease (MELD), which considers severity when the score is >20 [4] (Table 3).
3.2 AH Management
The management of AH includes general measures to prevent infections (Recommendation Grade A1), avoid kidney function deterioration and improve nutrition. Infections are present in 20–25% of patients at admission. Between 20 and 60% of patients with AH present with systemic inflammatory response syndrome (SIRS), which is considered a significant prognostic factor. SIRS may be the initial trigger of cascade events leading to mortality in patients with AH. Identification of the critical element of SIRS associated with infection, such as procalcitonin values >0.45 ng/mL (positive predictive value [PPV] 83.3; negative predictive value [NPV] 71%), allows early detection and treatment, and improves the patient’s chances of survival. Empirical treatment is directed, taking into account that the pattern of infection usually shows spontaneous bacterial peritonitis upon admission; however, during admission, respiratory infections are the most prevalent. Proper treatment of infections allows for the initiation of corticosteroids, the cornerstone treatment for this disease [42, 43].
Acute kidney injury (AKI) is an early marker of mortality; it is present in 23% of patients at admission and significantly increases 90-day mortality (65% vs 7%). Therefore, in the management of these patients, it is vital to assess possible predictors of AKI, such as the presence of SIRS on admission, elevated bilirubin levels or an increase in the international normalised ratio (INR). When treating these patients, measures such as the avoidance of the use of nephrotoxic drugs (contrast agents) and the suspension of β-blockers must be taken into account [43]. Recently, the AKI risk score—which is useful in identifying patients at high risk for inpatient renal impairment and may be helpful to prevent it in patients with AH—has been developed. The MELD score at admission, hepatic encephalopathy and SIRS predicted inpatient AKI with odds ratios of 3.86, 2.24 and 1.14, respectively. The AKI risk score developed using these predictors stratified the risk of inpatient AKI as low (score <3), moderate (3–4), and high (>4) [44].
The specific management algorithm for AH is summarised in Fig 2 [5, 6, 45]. Specific measures in critically ill patients include the joint use of corticosteroids (Recommendation Grade A1) and N-acetyl cysteine (Recommendation Grade B2), which have been shown to reduce the development of AKI and improve survival compared with the use of a corticosteroid alone [46]. To evaluate the response to corticosteroid treatment, the Lille score, which is calculated from age, renal function, albumin, prothrombin time, serum bilirubin, and change of serum bilirubin at day 7, is used. The efficacy of the treatment is evaluated after 1 month; therefore, it can be interrupted if it is not effective. Currently, the Lille score is also recommended on day 4 to help reduce exposure to corticosteroids. The Lille score should be used to identify non-responders (Recommendation Grade A1), reassess prognosis and guide the treatment course after 7 days of corticosteroids.
In those patients for whom treatment is not effective, liver transplantation may be considered (Recommendation Grade A1). A meta-analysis that synthesised the available evidence on liver transplantation for AH to assess alcohol relapse and 6-month survival using stringent selection criteria showed that 14% of patients with clinically severe AH had alcohol relapse after liver transplantation. The percentage of alcohol relapse of AH transplanted patients and survival at 6 months was similar to that of patients who underwent elective liver transplantation [47]. Therefore, candidate selection for liver transplantation in ALD should not be based solely on a fixed interval of abstinence.
In patients with ALD or AH, proper management of alcohol abuse disorder is essential to improve the prognosis (Recommendation Grade A1). Abstinence is the primary long-term survival factor after an episode of AH. The absence of previous treatment for alcoholism and age (<48 years) [48] have been considered the main predictors of relapse. Finally, the creation of a multidisciplinary team that includes surgeons, gastroenterologists, toxicologists (addictions) and psychiatrists/psychologists, and the use of alcohol biomarkers have been proposed as measures to carry out the optimal management of alcohol abuse disorder [49].
3.3 New Targets
The treatment of AH is currently undergoing important advances, and new targets have been developed, such as anti-inflammatory and regenerative agents with colony-stimulating factors. For the regulation of the microbiome, studies are being developed that provide information regarding the efficacy of antibiotics or faecal transplantation treatments [50].
4 Conclusion
ALD represents an important economic and healthcare burden, but it continues to be a highly stigmatising condition. New epidemiological studies that detail the disease progression and novel tools to diagnose it at earlier stages are needed. Specifically, AH, as the highest expression of ALD, has high morbidity and mortality, and the current treatment has suboptimal results with the exception of liver transplantation, which should be a clear indication in selected patients. The implementation of alcohol consumption biomarkers can assist in diagnosing and supporting recovery, and the development of multidisciplinary health models that allow a holistic approach is mandatory. Numerous initiatives with new therapies are underway, which must validate their efficacy and provide precise treatment guidelines.
References
Mathurin P, Bataller R. Trends in the management and burden of alcoholic liver disease. J Hepatol. 2015;62(1 Suppl):S38-46.
Gao B, Bataller R. Alcoholic liver disease: pathogenesis and new therapeutic targets. Gastroenterology. 2011;141(5):1572–85.
Ndugga N, Lightbourne TG, Javaherian K, et al. Disparities between research attention and burden in liver diseases: implications on uneven advances in pharmacological therapies in Europe and the USA. BMJ Open. 2017;7(3):e013620.
Crabb DW, Im GY, Szabo G, Mellinger JL, Lucey MR. Diagnosis and treatment of alcohol-associated liver diseases: 2019 Practice Guidance From the American Association for the Study of Liver Diseases. Hepatology. 2020;71(1):306–33.
Bataller R, Cabezas J, Aller R, et al. Alcohol-related liver disease. Clinical practice guidelines. Consensus document sponsored by AEEH. Gastroenterol Hepatol. 2019;42(10):657–76.
Singal AK, Bataller R, Ahn J, Kamath PS, Shah VH. ACG Clinical Guideline: Alcoholic Liver Disease. Am J Gastroenterol. 2018;113(2):175–94.
Addolorato G, Abenavoli L, Dallio M, Federico A, Germani G, Gitto S, Leandro G, Loguercio C, Marra F, Stasi E. Alcohol associated liver disease 2020: a clinical practice guideline by the Italian Association for the Study of the Liver (AISF). Dig Liver Dis. 2020;52(4):374–91.
Esser MB, Hedden SL, Kanny D, Brewer RD, Gfroerer JC, Naimi TS. Prevalence of alcohol dependence among US adult drinkers, 2009–2011. Prev Chronic Dis. 2014;11:140329.
GBD 2017 Cirrhosis Collaborators. The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5(3):245–66.
Ginès P, Graupera I, Lammert F, Angeli P, Caballeria L, Krag A, Guha IN, Murad SD, Castera L. Screening for liver fibrosis in the general population: a call for action. Lancet Gastroenterol Hepatol. 2016;1(3):256–60.
Younossi Z, Henry L. Contribution of alcoholic and nonalcoholic fatty liver disease to the burden of liver-related morbidity and mortality. Gastroenterology. 2016;150(8):1778–85.
Sandahl TD, Jepsen P, Thomsen KL, Vilstrup H. Incidence and mortality of alcoholic hepatitis in Denmark 1999–2008: a nationwide population based cohort study. J Hepatol. 2011;54(4):760–4.
Mellinger JL, Shedden K, Winder GS, et al. The high burden of alcoholic cirrhosis in privately insured persons in the United States. Hepatology. 2018;68(3):872–82.
Hirode G, Wong RJ. Trends in the prevalence of metabolic syndrome in the United States, 2011–2016. JAMA. 2020;323(24):2526–8.
Roulot D, Roudot-Thoraval F, NKontchou G, et al. Concomitant screening for liver fibrosis and steatosis in French type 2 diabetic patients using Fibroscan. Liver Int. 2017;37(12):1897–906.
Lee BP, Vittinghoff E, Dodge JL, Cullaro G, Terrault NA. National Trends and Long-term Outcomes of Liver Transplant for Alcohol-Associated Liver Disease in the United States. JAMA Intern Med. 2019;179(3):340–8.
Dugum MF, McCullough AJ. Acute alcoholic hepatitis, the clinical aspects. Clin Liver Dis. 2016;20(3):499–508.
Galán I, González M, Valencia-Martín J. Patrones de consumo de alcohol en España: un país en transición. Rev Esp Salud Publica. 2014;88:529–40.
CDC/National Institute on Alcohol Abuse and Alcolism. Drinking levels defined. https://www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/moderate-binge-drinking. Accessed 11 Jul 2021.
World Health Organization. Global Status Report on Alcohol and Health 2014. Editors: Poznyak, Vladimir and Rekve, Dag. https://www.who.int/substance_abuse/publications/alcohol_2014/en/. Accessed 20 July 2021.
UK Chief Medical Officers’ Low Risk Drinking Guidelines 2016. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/545937/UK_CMOs__report.pdf. Accessed 15 Jul 2021.
Johnson JA, Lee A, Vinson D, Seale JP. Use of AUDIT-based measures to identify unhealthy alcohol use and alcohol dependence in primary care: a validation study. Alcohol Clin Exp Res. 2013;37(Suppl 1):E253–9.
Caballero Martínez L, Caballero Martínez F, Santodomingo CJ. Instrumentos de detección de alcoholismo: precisiones sobre el cuestionario CAGE [Instruments for detecting alcoholism: remarks on the CAGE questionnaire]. Med Clin (Barc). 1988;91(13):515 (Spanish).
European Association for the Study of the Liver. Electronic address: easloffice@easloffice.eu; European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of alcohol-related liver disease. J Hepatol. 2018;69(1):154-181.
Fernandez M, Trépo E, Degré D, Gustot T, Verset L, Demetter P, Devière J, Adler M, Moreno C. Transient elastography using Fibroscan is the most reliable noninvasive method for the diagnosis of advanced fibrosis and cirrhosis in alcoholic liver disease. Eur J Gastroenterol Hepatol. 2015;27(9):1074–9.
Thiele M, Madsen BS, Hansen JF, Detlefsen S, Antonsen S, Krag A. Accuracy of the enhanced liver fibrosis test vs FibroTest, elastography, and indirect markers in detection of advanced fibrosis in patients with alcoholic liver disease. Gastroenterology. 2018;154(5):1369–79.
Fuster D, García-Calvo X, Zuluaga P, Bolao F, Muga R. Assessment of liver disease in patients with chronic hepatitis C and unhealthy alcohol use. World J Gastroenterol. 2021;27(23):3223–37.
Mueller S, Millonig G, Sarovska L, Friedrich S, Reimann FM, Pritsch M, Eisele S, Stickel F, Longerich T, Schirmacher P, Seitz HK. Increased liver stiffness in alcoholic liver disease: differentiating fibrosis from steatohepatitis. World J Gastroenterol. 2010;16:966–72.
Nguyen-Khac E, Thiele M, Voican C, Nahon P, Moreno C, Boursier J, Mueller S, de Ledinghen V, Stärkel P, Gyune Kim S, Fernandez M, Madsen B, Naveau S, Krag A, Perlemuter G, Ziol M, Chatelain D, Diouf M. Non-invasive diagnosis of liver fibrosis in patients with alcohol-related liver disease by transient elastography: an individual patient data meta-analysis. Lancet Gastroenterol Hepatol. 2018;3:614–25.
Nahon P, Kettaneh A, Tengher-Barna I, Ziol M, de Lédinghen V, Douvin C, Marcellin P, Ganne-Carrié N, Trinchet JC, Beaugrand M. Assessment of liver fibrosis using transient elastography in patients with alcoholic liver disease. J Hepatol. 2008;49:1062–8.
Janssens F, de Suray N, Piessevaux H, Horsmans Y, de Timary P, Stärkel P. Can transient elastography replace liver histology for determination of advanced fibrosis in alcoholic patients: a real-life study. J Clin Gastroenterol. 2010;44:575–82.
Cabezas J, Lucey MR, Bataller R. Biomarkers for monitoring alcohol use. Clin Liver Dis (Hoboken). 2016;8(3):59–63.
Bataller R, Cabezas J, Aller R, Ventura-Cots M, Abad J, Albillos A, Altamirano J, Arias-Loste MT, Bañares R, Caballería J, Caballería L, Carrión JA, Diago M, Fernández Rodríguez C, Gallego R, García-Cortes M, García-Monzón C, Genescà J, Ginés P, Hernandez-Guerra M, Jorquera F, Lligoña A, Molina E, Pareja MJ, Planas R, Tomé S, Salmerón J, Romero-Gómez M. Alcohol-related liver disease. Clinical practice guidelines. Consensus document sponsored by AEEH. Gastroenterol Hepatol. 2019;42(10):657–76.
Verril C, Markham H, Templeton A, Carr NJ, Sheron N. Alcohol-related cirrhosis—early abstinence is a key factor in prognosis, even in the most severe cases. Addiction. 2009;104(5):768–74.
Louvet A, Labreuche J, Artru F, et al. Main drivers of outcome differ between short term and long term in severe alcoholic hepatitis: a prospective study. Hepatology. 2017;66(5):1464–73.
Hart CL, Morrison DS, Batty GD, Mitchell RJ, Davey SG. Effect of body mass index and alcohol consumption on liver disease: analysis of data from two prospective cohort studies. BMJ. 2010;340:c1240.
Ascha MS, Hanouneh IA, Lopez R, Tamimi TA, Feldstein AF, Zein NN. The incidence and risk factors of hepatocellular carcinoma in patients with nonalcoholic steatohepatitis. Hepatology. 2010;51(6):1972–8.
Crabb DW, Bataller R, Chalasani NP, et al. Standard definitions and common data elements for clinical trials in patients with alcoholic hepatitis: recommendation from the NIAAA alcoholic hepatitis consortia. Gastroenterology. 2016;150(4):785–90.
Mandrekar P, Bataller R, Tsukamoto H, Gao B. Alcoholic hepatitis: translational approaches to develop targeted therapies. Hepatology. 2016;64(4):1343–55.
Altamirano J, Miquel R, Katoonizadeh A, et al. A histologic scoring system for prognosis of patients with alcoholic hepatitis. Gastroenterology. 2014;146(5):1231-9.e1-6.
Bissonnette J, Altamirano J, Devue C, et al. A prospective study of the utility of plasma biomarkers to diagnose alcoholic hepatitis. Hepatology. 2017;66(2):555–63.
Louvet A, Wartel F, Castel H, et al. Infection in patients with severe alcoholic hepatitis treated with steroids: early response to therapy is the key factor. Gastroenterology. 2009;137(2):541–8.
Prado V, Caballería J, Vargas V, Bataller R, Altamirano J. Alcoholic hepatitis: How far are we and where are we going? Ann Hepatol. 2016;15(4):463–73.
Sujan R, Cruz-Lemini M, Altamirano J, et al. A validated score predicts acute kidney injury and survival in patients with alcoholic hepatitis. Liver Transpl. 2018;24(12):1655–64.
European Association for the Study of the Liver. Electronic address eee, European Association for the Study of the L. EASL Clinical Practice Guidelines: Management of alcohol-related liver disease. J Hepatol. 2018;69(1):154-81.
Nguyen-Khac E, Thevenot T, Piquet MA, et al. Glucocorticoids plus N-acetylcysteine in severe alcoholic hepatitis. N Engl J Med. 2011;365(19):1781–9.
Marot A, Dubois M, Trépo E, Moreno C, Deltenre P. Liver transplantation for alcoholic hepatitis: a systematic review with meta-analysis. PLoS ONE. 2018;13(1):e0190823.
Altamirano J, López-Pelayo H, Michelena J, et al. Alcohol abstinence in patients surviving an episode of alcoholic hepatitis: prediction and impact on long-term survival. Hepatology. 2017;66(6):1842–53.
Magistri P, Marzi L, Guerzoni S, et al. Impact of a multidisciplinary team on alcohol recidivism and survival after liver transplant for alcoholic disease. Transplant Proc. 2019;51(1):187–9.
Singal AK, Shah VH. Current trials and novel therapeutic targets for alcoholic hepatitis. J Hepatol. 2019;70(2):305–13.
Acknowledgements
The authors thank Ana Isabel Ortega for writing assistance on behalf of Springer Health care Iberica.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure statement
This article has been published as a part of a journal supplement wholly funded by Eisai.
Funding
No funding or sponsorship was received for this study. Support for editorial assistance was funded by Eisai.
Conflict of interest
The authors declare that there were no conflicts of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Not applicable.
Code availability (software application or custom code)
Not applicable.
Authors’ contribution
All authors have contributed to the conception and design, analysis and interpretation of data, drafting the article and revising it critically for important intellectual content, and to the final approval of the version to be published.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Cabezas, J. Management of Alcohol-Related Liver Disease and Its Complications. Clin Drug Investig 42 (Suppl 1), 47–53 (2022). https://doi.org/10.1007/s40261-022-01143-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-022-01143-9