Abstract
Background
Dextromethorphan hydrobromide (DM) is a widely used antitussive. This study determined, for the first time, the basic pharmacokinetic profile of DM and its active metabolite, dextrorphan (DP) in children and adolescents.
Methods
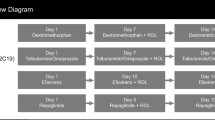
Thirty-eight male and female subjects at risk for developing an upper respiratory tract infection (URTI), or symptomatic with cough due to URTI, were enrolled in this single-dose, open-label study: ages 2–5 years (Group A, n = 8), 6–11 years (Group B, n = 17), 12–17 years (Group C, n = 13). Subjects were genotyped for cytochrome P450 (CYP) 2D6 polymorphisms and characterized as poor (PM) or non-poor metabolizers (non-PM). Groups A and B were dosed using an age–weight dosing schedule (DM range 7.5–24.75 mg); a 30-mg dose was used for Group C.
Results
Average exposures to total DP increased as age group increased, and average exposure to DM was highest in the adolescent group. One subject in that group was a PM. The terminal half-life (t ½) values were longer in the adolescent group due in part to the single PM subject. No relationship between body weight and pharmacokinetic parameters was noted.
Conclusions
This is the first evaluation of the pharmacokinetic characteristics of DM in children and adolescents. A single dose of DM in this population was safe, and well tolerated at all doses tested. The data are used to model and compare pediatric DM exposures with those of adults.

Similar content being viewed by others
References
AHFS Drug Information. Dextromethorphan hydrobromide. 2006.
Ohi Y, Tsunekawa S, Haji A. Dextromethorphan inhibits the glutamatergic synaptic transmission in the Nucleus Tractus Solitarius of guinea pigs. J Pharmacol Sci. 2011;116:54–62.
Wills RJ, Martin KS. Dextromethorphan/dextrorphan disposition in rat plasma and brain. Pharmacol Res. 1988;5:193.
Dextromethorphan. In: Martindale—the complete drug reference. Available from: http://www.medicinescomplete.com/mc/martindale/current. Accessed 12 Mar 2014.
Pert CB, Snyder SH. Properties of opiate-receptor binding in rat brain. Proc Natl Acad Sci U S A. 1973;70(8):2243–7.
Moghadamnia AA, Rostami-Hodjegan A, Abdul-Manap R, Wright CE, Morice AH, Tucker GT. Physiologically based modelling of inhibition of metabolism and assessment of the relative potency of drug and metabolite: dextromethorphan vs dextrorphan using quinidine inhibition. Br J Clin Pharmacol. 2003;56(1):57–67.
Pope LE, Khalil MH, Berg JE, et al. Pharmacokinetics of dextromethorphan after single or multiple dosing in combination with quinidine in extensive and poor metabolizers. J Clin Pharm. 2004;44(10):1132–42.
Barnhart JW. The urinary excretion of dextromethorphan and three metabolites in dogs and humans. Toxicol Appl Pharmacol. 1980;55:43–8.
Gorski JC, Jones DR, Wrighton SA, et al. Characterisation of dextromethorphan N-demethylation by human liver microsomes: contribution of the cytochrome P450 3A (CYP3A) subfamily. Biochem Pharmacol. 1994;48:173–82.
Schmider J, Greenblatt DJ, Fogelman SM, et al. Metabolism of dextromethorphan in vitro: involvement of cytochromes P450 2D6 and 3A314, with a possible role of 2E1. Biopharm Drug Dispos. 1997;18:227–40.
Von Moltke LL, Greenblatt DJ, Grassi JG, et al. Multiple human chromosomes contribute to biotransformation of dextromethorphan in vitro: role of CYP2C9, CYP2C19, CYP2D6, and CYP3A. J Pharm Pharmacol. 1998;50:997–1004.
Jacq-Aigran E, Funck-Brentano C, Cresteil T. CYP2D6- and CYP3A-dependent metabolism of dextromethorphan in humans. Pharmacogenetics. 1993;3:197–204.
Brinn R, Brosen K, Gram LF, et al. Sparteine oxidation is practically abolished in quinidine treated patients. Br J Clin Pharmacol. 1986;22:194–7.
Nielsen MD, Brosen K, Gram LF. A dose–effect study of the in vivo inhibitory effect of quinidine on sparteine oxidation in man. Br J Clin Pharmacol. 1990;29:299–304.
Zhang Y, Britto MR, Valderhaug KL, et al. Dextromethorphan: enhancing its systemic bioavailability by way of low-dose quinidine-mediated inhibition of cytochrome P4502D6. Clin Pharmacol Ther. 1992;51:647–55.
Schadel M, Wu D, Otton SV, et al. Pharmacokinetics of dextromethorphan and metabolites in humans: influence of the CYP 2146 phenotype and quinidine inhibition. J Clin Psychopharmacol. 1995;15:263–9.
Samer CF, Lorenzini KI, Rollason V, Daali Y, Desmeules JA. Applications of CYP450 testing in the clinical setting. Mol Diagn Ther. 2013;17:165–84.
Capon DA, Bochner F, Kerry N, et al. The influence of CYP2D6 polymorphism and quinidine on the disposition and antitussive effect of dextromethorphan in humans. Clin Pharmacol Ther. 1996;60:295–307.
Churchill JA. Posology in infants and children. Dev Med Child Neurol. 1964;6:610–3.
Munzenberger PJ, McKercher P. Pediatric dosing: the pharmacist’s dilemma. Contemp Pharm Pract. 1980;3:11–4.
Sharfstein JM, North M, Serwint JR. Over the counter but no longer under the radar: pediatric cough and cold medications. New Engl J Med. 2007;357(23):2321–4.
Food and Drug Administration. For consumers. Using over-the-counter cough and cold products in children. Available from: http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm048515.htm. Accessed 16 Dec 2013.
AmpliChip Package Insert, v5. Roche, 2009. Available from: http://www.amplichip.us/documents/CYP450_P.I._US-IVD.pdf.
Howie SH. Blood sample volumes in child health research: review of safe limits. Bull World Health Organ. 2011;89:46–53. doi:10.2471/BLT.10.080010.
US Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research, Center for Veterinary Medicine Guidance for industry: bioanalytical method validation. May 2001.
Ramsay MA, Savege TM, Simpson BR, Goodwin R. Controlled sedation with alphaxalone–alphadolone. BMJ. 1974;2:656–9.
Rebsamen MC, Desmeules J, Daali Y, et al. The Amplichip CYP450 test: cytochrome P450 2D6 genotype assessment and phenotype prediction. Pharmacogenomic J. 2009;9:34–41.
Silvasti M, Karttunen P, Tuktiainen H, Kokkonen P, Hanninen U, Sykanen S. Pharmacokinetics of dextromethorphan and dextrorphan: a single dose comparison of three preparations in human volunteers. Int J Clin Pharmacol Ther Toxicol. 1987;25(9):493–7.
Blake MJ, Gaedigk A, Pearce RE, et al. Ontogeny of dextromethorphan O- and N-demethylation in the first year of life. Clin Pharmacol Ther. 2007;81(4):510–6.
Department of Health and Human Services, Food and Drug Administration. Cough, cold, allergy, bronchodilator, and antihistaminic drug products for over-the-counter human use. Final monograph for OTC antitussive drug products. Fed Regist. 1987;52(155):30042–57. Available from: http://www.fda.gov/downloads/Drugs/DevelopmentApprovalProcess/DevelopmentResources/OvertheCounterOTCDrugs/StatusofOTCRulemakings/ucm114742.pdf. Accessed 24 Feb 2014.
American Academy of Pediatrics Committee on Drugs. Use of codeine- and dextromethorphan-containing cough remedies in children. Pediatrics. 1997;99:918–20.
Acknowledgments
We thank Advanced Biomedical Research, Inc. who performed the data management, statistical and pharmacokinetic analysis, and report writing for this study. We thank Vincent Fabien who was the study statistician employed at Novartis Consumer Health, S.A. We also appreciate the assistance with manuscript preparation provided by Mark Calcamuggio (The Write Company, LLC) and further technical review and editorial assistance by Dr. Judith Rosen Farrar (Life Sciences Press, LLC).
Financial Disclosure
This study was sponsored by Novartis Consumer Health, Inc., which also provided support for manuscript preparation. The latter consisted solely of editing and formatting for journal submission with no contribution to content.
The material in this manuscript represents original material and has not been submitted or published elsewhere. Eric Guenin was an employee of Novartis Consumer Health, Inc. at the time of the study and writing of the manuscript; he is now employed at Reckitt Benckiser. Marianna Armogida is an employee of Novartis Consumer Health, S.A. Dennis Riff was the principal investigator and is an employee of Advanced Clinical Research Institute.
Authorship Statement
All authors were fully involved in developing and conducting the study, evaluating the data and writing/editing this manuscript. The paper meets the authorship requirements of the International Committee of Medical Journal Editors.
Author information
Authors and Affiliations
Corresponding author
Additional information
At the time the study was conducted, Dr. Guenin held the position of Director, Clinical Research, Novartis Consumer Health, Inc., Parsippany, NJ, USA.
Rights and permissions
About this article
Cite this article
Guenin, E., Armogida, M. & Riff, D. Pharmacokinetic Profile of Dextromethorphan Hydrobromide in a Syrup Formulation in Children and Adolescents. Clin Drug Investig 34, 609–616 (2014). https://doi.org/10.1007/s40261-014-0210-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-014-0210-5