Abstract
Background
Muscle wasting is common and insidious in end-stage renal disease (ESRD). Loss of muscle quantity and quality reduces quality of life and increases mortality in ESRD patients. Additionally, secondary hyperparathyroidism (SHPT) causes muscle atrophy. Meanwhile, vitamin D, which is used for SHPT treatment, plays an essential role in muscle growth.
Objectives
We prospectively investigated the effect of active vitamin D administration on muscle mass.
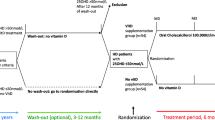
Methods
We measured muscle mass based on bioelectrical impedance analysis in 68 hemodialysis patients. Patients were divided into a control group (without active vitamin D administration) and a VitD group (with active vitamin D administration). We compared muscle mass at the beginning of treatment and 1 year later. We also investigated health-related quality of life (HR-QOL) using the Medical Outcome Study Short Form-36 (SF-36).
Results
The VitD group experienced a significant increase in the amount of change in total muscle mass and muscle mass percentage in men (p = 0.025) but not in women (p = 0.945). By multivariable logistic regression analysis, active vitamin D administration was independently associated with increased muscle mass percentage in men only. In the SF-36, the physical functioning (PF) scores were significantly decreased at the end of the study in the patients without active vitamin D treatment, especially in women.
Conclusion
Our results suggested that active vitamin D treatment was associated with increased muscle mass in men, and it might have a favorable effect on maintaining PF in HR-QOL in hemodialysis patients.



Similar content being viewed by others
References
Kouidi E, Albani M, Natsis K, et al. The effects of exercise training on muscle atrophy in haemodialysis patients. Nephrol Dial Transplant. 1998;13:685–99.
Lim PS, Cheng YM, Wei YH. Large-scale mitochondrial DNA deletions in skeletal muscle of patients with end-stage renal disease. Free Radic Biol Med. 2000;29:454–63.
McElroy A, Silver M, Morrow L, Heafner BK. Proximal and distal muscle weakness in patients receiving hemodialysis for chronic uremia. Phys Ther. 1970;50:1467–81.
Fahal IH, Bell GM, Bone JM, Edwards RH. Physiological abnormalities of skeletal muscle in dialysis patients. Nephrol Dial Transplant. 1997;12:119–27.
Frame B, Heinze EG Jr, Block MA, Manson GA. Myopathy in primary hyperparathyroidism. Observations in three patients. Ann Intern Med. 1968;68:1022–7.
Kopple JD, Cianciaruso B, Massry SG. Does parathyroid hormone cause protein wasting? Contrib Nephrol. 1980;20:138–48.
Patten BM, Bilezikian JP, Mallette LE, et al. Neuromuscular disease in primary hyperparathyroidism. Ann Intern Med. 1974;80:182–93.
Caso G, Garlick PJ. Control of muscle protein kinetics by acid-base balance. Curr Opin Clin Nutr Metab Care. 2005;8:73–6.
Diesel W, Emms M, Knight BK, et al. Morphologic features of the myopathy associated with chronic renal failure. Am J Kidney Dis. 1993;22:677–84.
Stannard SR, Johnson NA. Energy well spent fighting the diabetes epidemic. Diabetes Metab Res Rev. 2006;22:11–9.
Johansen KL, Shubert T, Doyle J, et al. Muscle atrophy in patients receiving hemodialysis: effects on muscle strength, muscle quality, and physical function. Kidney Int. 2003;63:291–7.
Desmeules S, Lévesque R, Jaussent I, et al. Creatinine index and lean body mass are excellent predictors of long-term survival in haemodiafiltration patients. Nephrol Dial Transplant. 2004;19:1182–9.
National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis. 2003;42(4 Suppl):S1–201.
Komrakova M, Krischek C, Wicke M, et al. Influence of intermittent administration of parathyroid hormone on muscle tissue and bone healing in orchiectomized rats or controls. J Endocrinol. 2011;209:9–19.
Endo I, Inoue D, Mitsui T, et al. Deletion of vitamin D receptor gene in mice results in abnormal skeletal muscle development with deregulated expression of myoregulatory transcription factors. Endocrinology. 2003;144:5138–44.
Vazquez G, de Boland AR, Boland R. Stimulation of Ca2+ release-activated Ca2+ channels as a potential mechanism involved in non-genomic 1,25(OH)2-vitamin D3-induced Ca2+ entry in skeletal muscle cells. Biochem Biophys Res Commun. 1997;239:562–5.
Marantes I, Achenbach SJ, Atkinson EJ, et al. Is vitamin D a determinant of muscle mass and strength? J Bone Miner Res. 2011;26:2860–71.
Holick MF. Environmental factors that influence the cutaneous production of vitamin D. Am J Clin Nutr. 1995;61(3 Suppl):638S–45S.
McKenna MJ. Differences in vitamin D status between countries in young adults and the elderly. Am J Med. 1992;93:69–77.
van der Wielen RP, Löwik MR, van den Berg H, et al. Serum vitamin D concentrations among elderly people in Europe. Lancet. 1995;346:207–10.
Mingrone G, Greco AV, Castagneto M, Gasbarrini G. A woman who left her wheelchair. Lancet. 1999;353:806.
Prabhala A, Garg R, Dandona P. Severe myopathy associated with vitamin D deficiency in western New York. Arch Intern Med. 2000;160:1199–203.
Janssen HC, Samson MM, Verhaar HJ. Vitamin D deficiency, muscle function, and falls in elderly people. Am J Clin Nutr. 2002;75:611–5.
Wanic-Kossowska M, Grzegorzewska A, Plotast H, Bombicki K. Does calcitriol therapy improve muscle function in uremic patients. Perit Dial Int. 1996;16(Suppl 1):S305–8.
Henderson RG, Russell RG, Ledingham JG, et al. Effects of 1,25-dihydroxycholecalciferol on calcium absorption, muscle weakness, and bone disease in chronic renal failure. Lancet. 1974;1:379–84.
Shinzato T, Nakai S, Fujita Y, et al. Determination of Kt/V and protein catabolic rate using pre- and post-dialysis blood urea nitrogen concentrations. Nephron. 1994;67:280–90.
Diamant MJ, Harwood L, Movva S, et al. A comparison of quality of life and travel-related factors between in-center and satellite-based hemodialysis patients. Clin J Am Soc Nephrol. 2010;5:268–74.
Muir SW, Montero-Odasso M. Effect of vitamin D supplementation on muscle strength, gait and balance in older adults: a systematic review and meta-analysis. J Am Geriatr Soc. 2011;59:2291–300.
Gordon PL, Sakkas GK, Doyle JW, Shubert T, Johansen KL. Relationship between vitamin D and muscle size and strength in patients on hemodialysis. J Ren Nutr. 2007;17:397–407.
Bertoli M, Luisetto G, Arcuti V, Urso M. Uremic myopathy and calcitriol therapy in CAPD patients. ASAIO Trans. 1991;37:M397–8.
Gómez-Fernández P. Sánchez Agudo L, Calatrava JM. [Chronic kidney insufficiency and respiratory muscle function. Changes induced by treatment with 1,25(OH)2D3]. Med Clin (Barc). 1990;94:204–7.
Kraemer M. A new model for the determination of fluid status and body composition from bioimpedance measurements. Physiol Meas. 2006;27:901–19.
Tengvall M, Ellegård L, Malmros V, et al. Body composition in the elderly: reference values and bioelectrical impedance spectroscopy to predict total body skeletal muscle mass. Clin Nutr. 2009;28:52–8.
Pietiläinen KH, Kaye S, Karmi A, et al. Agreement of bioelectrical impedance with dual-energy x-ray absorptiometry and MRI to estimate changes in body fat, skeletal muscle and visceral fat during a 12-month weight loss intervention. Br J Nutr. 2013;109:1910–6.
Basile C, Vernaglione L, Di Iorio B, et al. Development and validation of bioimpedance analysis prediction equations for dry weight in hemodialysis patients. Clin J Am Soc Nephrol. 2007;2:675–80.
Hazell TJ, DeGuire JR, Weiler HA. Vitamin D: an overview of its role in skeletal muscle physiology in children and adolescents. Nutr Rev. 2012;70:520–33.
Boland R, de Boland AR, Marinissen MJ, et al. Avian muscle cells as targets for the secosteroid hormone 1,25-dihydroxy-vitamin D3. Mol Cell Endocrinol. 1995;114:1–8.
Zanello SB, Boland RL, Norman AW. cDNA sequence identity of a vitamin D-dependent calcium-binding protein in the chick to calbindin D-9K. Endocrinology. 1995;136:2784–7.
Barton-Davis ER, Shoturma DI, Musaro A, Rosenthal N, Sweeney HL. Viral mediated expression of insulin-like growth factor I blocks the aging-related loss of skeletal muscle function. Proc Natl Acad Sci U S A. 1998;95:15603–7.
Brunner A, de Boland AR. 1,25-Dihydroxyvitamin D3 affects the synthesis, phosphorylation and in vitro calmodulin binding of myoblast cytoskeletal proteins. Z Naturforsch C. 1990;45:1156–60.
Drittanti L, de Boland AR, Boland R. Modulation of DNA synthesis in cultured muscle cells by 1,25-dihydroxyvitamin D-3. Biochim Biophys Acta. 1989;1014:112–9.
Araujo AB, Travison TG, Bhasin S, et al. Association between testosterone and estradiol and age-related decline in physical function in a diverse sample of men. J Am Geriatr Soc. 2008;56:2000–8.
Giannoulis MG, Sonksen PH, Umpleby M, et al. The effects of growth hormone and/or testosterone in healthy elderly men: a randomized controlled trial. J Clin Endocrinol Metab. 2006;91:477–84.
Tanaka M, Yamazaki S, Hayashino Y, et al. Hypercalcaemia is associated with poor mental health in haemodialysis patients: results from Japan DOPPS. Nephrol Dial Transplant. 2007;22:1658–64.
Dumler F, Falla P, Butler R, et al. Impact of dialysis modality and acidosis on nutritional status. ASAIO J. 1999;45:413–7.
Acknowledgments
This work was supported by research grant from the Japan Dialysis Outcome Research Group. All authors have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mori, A., Nishino, T., Obata, Y. et al. The Effect of Active Vitamin D Administration on Muscle Mass in Hemodialysis Patients. Clin Drug Investig 33, 837–846 (2013). https://doi.org/10.1007/s40261-013-0132-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-013-0132-7