Abstract
Background
Although safe and effective anti-retrovirals (ARVs) are readily available, non-adherence to ARVs is highly prevalent among people living with human immunodeficiency virus/acquired immunodeficiency syndrome (PLWHA). Different adherence-improving interventions have been developed and examined through decision analytic model-based health technology assessments. This systematic review aimed to review and appraise the decision analytical economic models developed to assess ARV adherence-improvement interventions.
Methods
The review protocol was registered on PROSPERO (CRD42022270039), and reporting followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist. Relevant studies were identified through searches in six generic and specialized bibliographic databases, i.e. PubMed, Embase, NHS Economic Evaluation Database, PsycINFO, Health Economic Evaluations Database, tufts CEA registry and EconLit, from their inception to 23 October 2022. The cost-effectiveness of adherence interventions is represented by the incremental cost-effectiveness ratio (ICER). The quality of studies was assessed using the quality of the health economics studies (QHES) instrument. Data were narratively synthesized in the form of tables and texts. Due to the heterogeneity of the data, a permutation matrix was used for quantitative data synthesis rather than a meta-analysis.
Results
Fifteen studies, mostly conducted in North America (8/15 studies), were included in the review. The time horizon ranged from a year to a lifetime. Ten out of 15 studies used a micro-simulation, 4/15 studies employed Markov and 1/15 employed a dynamic model. The most commonly used interventions reported include technology based (5/15), nurse involved (2/15), directly observed therapy (2/15), case manager involved (1/15) and others that involved multi-component interventions (5/15). In 1/15 studies, interventions gained higher quality-adjusted life years (QALYs) with cost savings. The interventions in 14/15 studies were more effective but at a higher cost, and the overall ICER was well below the acceptable threshold mentioned in each study, indicating the interventions could potentially be implemented after careful interpretation. The studies were graded as high quality (13/15) or fair quality (2/15), with some methodological inconsistencies reported.
Conclusion
Counselling and smartphone-based interventions are cost-effective, and they have the potential to reduce the chronic adherence problem significantly. The quality of decision models can be improved by addressing inconsistencies in model selection, data inputs incorporated into models and uncertainty assessment methods.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1. To achieve the World Health Organization goal to end the HIV epidemic by 2030, adherence to anti-retrovirals is crucial. |
2. Smartphone, nurse and case manager interventions are cost-effective and have a very good scope to achieve the targeted adherence levels in people living with HIV/AIDS. |
3. Future decision models can be improved by improving the methods, especially in reporting of model selection, validation, data inputs and uncertainty assessment methods. |
1 Introduction
The introduction of anti-retroviral (ARV) therapy has revolutionized the treatment of people living with human immunodeficiency virus (HIV)/acquired immunodeficiency syndrome (AIDS) [1, 2]. ARVs have significantly reduced the morbidity and mortality associated with HIV/AIDS [3,4,5]. Adherence to ARV therapy is closely associated with the suppression of the plasma HIV viral load (VL) [1]. Reducing plasma VL helps to restore the immune system, reduces HIV transmission, and improves the quality of life (QoL) of people living with HIV/AIDS (PLWHA) [6, 7]. Although the optimal level of adherence to achieve viral suppression is unknown, a recent meta-analysis found that minimal > 80% adherence to ARVs resulted in a high level of viral suppression [6, 8]. PLWHA often have poor adherence to ARVs due to factors such as forgetfulness, stigma, discrimination, poor living conditions, medication side effects, religious beliefs and practices, lack of social support, inclination towards alternative therapies and financial constraints [9,10,11,12,13]. Suboptimal adherence to ARV therapy can lead to insufficient viral suppression and the emergence of drug-resistant viral strains, resulting in regimen failure, progression to AIDS, and mortality [14, 15].
To improve medication adherence, a variety of interventions, including educational, behavioural, pharmaceutical care, smartphone-based reminders and psychosocial, as well as combinations of these, are used [16,17,18,19,20,21]. Furthermore, simplifying the complexity of the treatment regimen is likely to improve adherence [22]. There are reports that adherence interventions can reduce healthcare resource utilization, leading to cost offsets and cost savings for the healthcare system [20]. Although the negative impact of non-adherence on clinical outcomes has been well studied, less is known about its economic consequences and whether interventions to improve adherence are cost-effective or not. In the USA, it has been estimated that nonadherence to medications is responsible for more than US$100 billion in avoidable direct healthcare costs annually [23].
Adherence interventions are critical for improving the clinical outcomes of PLWHA; however, evaluating the health economic outcomes is equally important for clinicians, third-party payers and PLWHA in deciding the use of interventions in HIV/AIDS management [24, 25]. The cost-effectiveness impacts of different types of interventions are compared using findings from corresponding clinical trials in model-based health technology assessment (HTA). The implementation cost of adherence interventions is usually substantial, and HTA is, therefore, essential to inform the decision makers on the potential impact of the adherence interventions on both clinical and health economic outcomes. The cost-effectiveness of adherence intervention is subject to the balance of three elements: clinical and economic benefits of intervention, cost of intervention and payer’s willingness-to-pay (WTP) threshold [24, 26]. Various clinical trials have been conducted to assess the effect of adherence interventions in PLWHA, as a result, there have been numerous economic models developed in HIV/AIDS care, to estimate the effects of intensive interventions on costs and benefits [27,28,29,30,31]. Several systematic reviews have been performed on the economic trials in HIV care; however, none of these review studies has focused on the decision analytic models [6, 16, 32, 33]. Therefore, this systematic review aims to review and appraise the decision analytic models developed to assess ARV adherence-improvement interventions in PLWHA.
2 Methods
The protocol for this review was registered on the PROSPERO database (registration number: CRD42022270039). We followed Cochrane handbook guidance and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to conduct and report this review [34, 35]. The PRISMA checklist is attached in the Supplementary File Table S1.
2.1 Literature Search
The following general and subject-specific bibliographic databases were searched from inception to 23 October 2022: EMBASE, PubMed, PsycINFO, NHS Economic Evaluation Database, Health Economic Evaluations Database (HEED), Tufts CEA registry and EconLit through Ebsco. The search strategy was adapted for each database and comprised diverse expressions, including medical subject headings, synonyms and acronyms for the topic of HIV/AIDS, adherence and analytical decision models. We used Google Scholar as a supplementary citation tracking resource to search for any further studies not identified from a systematic database search. The full search strategies for the individual databases can be found in the Supplementary File Table S2. The bibliographic references of all the included studies were searched to find other potentially eligible studies. No language restrictions were applied.
2.2 Studies Inclusion Criteria
A study was included in the review if it involved: (i) PLWHA on current treatment with ARV therapy, (ii) any adherence interventions (counselling, lifestyle behaviour change, smartphone-based reminders, etc.), (iii) comparison of intervention with the standard of care or other adherence enhancing interventions, (iv) reporting incremental cost-effectiveness, (v) analyses using decision analytical models, (vi) published in a peer-reviewed journal. Clinical trials without decision analytic modelling, return on investment studies and conference abstracts not available as full texts were excluded from the review. In addition, letters to the editor, case reports, expert opinions, commentaries, perspectives and qualitative studies were excluded from the study.
2.3 Data Screening and Extraction
The primary search was conducted by one of the investigators (AA1). All citations from databases were imported into Endnote version X9.3.3. in which duplicates were removed. AA1 and JD independently screened the titles and abstracts of the identified studies and shortlisted the articles for full-text review. A third reviewer (SK) resolved the discrepancies in shortlisting of studies to reach a consensus. The full text of potentially eligible articles was then reviewed to confirm the eligibility. At the final stage of the full-text review, the included articles that met all the predefined criteria were read by all the investigators to confirm the inclusion of the articles.
The characteristics of the selected studies were extracted using a standardized table adapted from the Joanna Briggs Institute’s economic evaluation guidelines and previous economic evaluation reviews [24, 36,37,38]. Information such as authors, study publication year, country, characteristics of the population, intervention, usual care (UC) or control or comparator group, perspective, time horizon, nature of evaluation [i.e. cost-effectiveness analysis (CEA) or cost-utility analysis (CUA)], study design (i.e. model type), outcome measures (e.g. QALYs, life months/years saved, HIV transmission prevention or other outcomes) and cost-effectiveness results [the incremental cost-effectiveness ratio (ICER)] were extracted from the studies. All costs were converted into 2022 US dollars to make a meaningful comparison of the ICERs across the identified studies. The CCEMG-EPPI-Centre Cost Converter version 1.4, which employs the purchasing power parity approach derived from the IMF, World Economic Outlook database, was employed to convert all non-US dollar currencies to US dollars [39]. For studies that did not provide a reference year, the base year was assumed to be 2 years before the publication date. Data extraction was undertaken by AA1, and JD and double-checked by SK. Disagreements were resolved through mutual discussions among the authors.
2.4 Data Synthesis
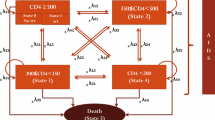
The number of studies included and excluded during the selection process was presented in a PRISMA flowchart. The format and scope of reported economic evaluation contexts, targeted population, intervention nature and conditions, health outcomes and costs differed significantly between studies, making quantitative data aggregation such as meta-analysis impossible. Consequently, the dominance ranking framework (permutation matrix) was used for qualitatively synthesizing the included studies as advised by Joanna Briggs Institute [36, 40]. Colour coding was used in the dominance ranking framework to indicate implications for decision makers. A ‘blue’ code indicates the situation in which the intervention is favoured, i.e. has better health outcomes and lower costs. A ‘red’ code shows the case in which costs are higher, and the intervention is less effective. A ‘yellow’ code shows that there is no obvious decision that the intervention is more effective and more costly or less effective and less costly. A cost-effectiveness league table was created, which includes a list of healthcare interventions in ascending order (from low to high) of their ICER [41]. Further, economic findings were synthesized and presented as a narrative summary alongside a tabular summary.
2.5 Quality Assessment
To assess the quality of the included studies, the Quality of Health Economic Studies (QHES) tool was used [42]. The QHES checklist has 16 questions that must be answered with a yes or no. The quality score was calculated by adding all of the points for ‘yes’ answers. Each question has a different weighting based on importance, and the umulative score of all questions is 100. The following cut-off points were used to determine the quality: extremely poor quality (0–24), poor quality (25–49), fair quality (50–74) and excellent quality (75–100). Quality assessment was undertaken by AA1 and JD and double-checked by SK. Disagreements were resolved by discussion between review authors (AA1, JD, SK).
3 Results
The steps involved in the study selection are presented in the PRISMA flow chart (Fig. 1). Database searches and additional sources identified a total of 2070 relevant citations. Following the removal of 334 duplicates, the title, abstract and keywords for 1736 papers were screened. A total of 67 articles were chosen for full-text screening. Fifteen studies were included [43,44,45,46,47,48,49,50,51,52,53,54,55,56,57], with the remainder excluded due to the reasons listed in Fig. 1.
3.1 Characteristics of the Included Models
Table 1 summarizes the included studies’ characteristics and main health economic outcomes. Eight studies were conducted in the USA [43, 50,51,52, 54,55,56,57], one in the Netherlands [46], two in Kenya [45, 48], one in Côte d’Ivoire [53], one in Eswatini [44], one in Italy [49], and one in Zimbabwe [47]. Ten studies followed the micro-simulation modelling [43,44,45, 47, 48, 51,52,53, 55, 57], four studies followed Markov analytical model [46, 49, 50, 56], and one followed the dynamic modelling [54]. Three studies followed the specific micro-simulation model known as the Cost-Effectiveness of Preventing AIDS Complications (CEPAC) model [43, 53, 55]. Stevens et al. employed a combined stochastic deterministic approach [44]. Seven studies conducted both CEA and CUA [43, 46, 50,51,52, 54, 55], the CEA technique alone was used in six studies [44, 47,48,49, 53, 57] and the CUA approach alone was used in two studies [45, 56]. Seven studies were conducted from a societal perspective [46, 50,51,52, 55,56,57], five from healthcare [43,44,45, 47, 49], two from payers [48, 53] and one study did not state any perspective [54]. In ten studies, the time horizon was a lifetime [45, 46, 48, 50,51,52,53, 55,56,57], in three studies it was 20 years [44, 47, 54], in one study it was 10 years [49] and in one study, it was 1 year [43]. One study looks at the impact of intervention on the vertical HIV transmission prevention [52], while the other studies look at the horizontal transmission [43,44,45,46,47,48,49,50,51, 53,54,55,56,57]. The selection of a model is an important step and only six studies justified the selection of a model [44, 45, 47, 48, 52, 56]. Two studies provided partial justifications for the model selection [46, 51]. Calibration and internal consistency assessment of the model is an important step and only six studies reported this [44, 45, 47, 48, 52, 56]. In Markov models, cycle lengths ranged from 6 month [46] to 1 year [49]. Only three studies reported the details of software they used used for simulation [49, 50, 54]. The majority of the studies were funded by the government (12/15) [29, 43,44,45,46, 48, 50, 52,53,54,55, 57], followed by industry (2/15) [49, 56].
3.2 Input Parameters and Sensitivity Analysis
Data sources were consistently under-reported, particularly how data sources were identified. In the majority of studies, healthcare resource use was reported from country-specific sources, hospital databases, resource utilization studies, systematic reviews and expert panels. Eleven studies took effectiveness data from one or more observational cohorts after expert opinion without mentioning the details of experts [43, 47,48,49,50,51,52,53,54, 56, 57]. Three studies took data from corresponding trials and cohorts to plug in the model [44, 46, 55]. Only one study by Patel et al. used the health utilities data from the systematic review [45]. To calculate health state utilities, three studies used the SF-6D [45, 50, 55], two used the EQ-5D [47, 49] and one used the MOS-HIV [54]. De Bruin et al. and McCabe employed standard gamble in addition to SF-6D to calculate the health state utilities [46, 52]. Munakata et al. employed a medical outcome survey (MOS-HIV) and standard gamble [56] while Stevens et al. used SF-6D, EQ-5D, visual analogue scale (VAS) and HIV/AIDS target-QoL tools to calculate the health state utilities [44]. Neilan et al. were unable to locate the health states for young people living with HIV (YWH), so they used adult utilities after adjustments [43]. Cost inputs employed in models were country specific, including claims databases and a list of national tariffs and drug prices.
All the papers performed some form of parameter uncertainty analysis. One-way sensitivity analysis was performed by 12 studies [43,44,45,46, 48, 50,51,52,53,54,55,56] and two way or multi-way sensitivity analysis was performed by 11 studies [43,44,45, 48,49,50,51,52, 54,55,56]. A scenario sensitivity analysis was performed for the base case, worse-case or best-case scenarios in four studies to apprise the ideal scenario for cost-effective use of the adherence interventions [46, 47, 53, 57]. Colombo et al. performed a probabilistic sensitivity analysis (PSA) [49].
3.3 Methodological Quality of Included Studies
The quality scores ranged from 58 to 100. Only two studies [47, 50] were classified as fair quality as they did not report the transparent model structure, model choices, assumptions, incremental analysis and uncertainty. The majority of the studies achieved high quality [43,44,45,46, 48, 49, 51,52,53,54,55,56,57]. The overall average quality score for the studies was 91%. The details of the quality scores for each study are presented in Table 2.
3.4 Summary of Adherence Interventions
Interventions were categorized based on the intervention target population. Broadly, interventions were classified in four categories: viral rebound, pregnant, young and adult PLWHA. Further interventions were classified based on the intervention nature. The most commonly used interventions reported include technology based in 5/15 [43, 45, 47, 50, 54], nurse involved in 2/15 [46, 55], directly observed therapy (DOT) in 2/15 [52, 57], prevention of loss to follow-up in 2/15 [48, 53], case manager involved in 1/15 [51], simplification of regimen in 1/15 [49], Link4Health in 1/15 [44] and ideal adherence (observed in clinical trials) in 1/15 [56]. Key details of adherence interventions are given in the Table, and a brief context of interventions is given below.
3.5 Adherence Interventions for Viral Rebound PLWHA
3.5.1 Nurse-Involved Intervention
Adherence-improving self-management strategy (AIMS) is a nurse-led, one-on-one self-management intervention incorporating feedback from electronic medication monitors, delivered during routine clinical visits [46]. The study by De Bruin et al. (the Netherlands) included patients with HIV who were either treatment experienced or treatment naive and at risk of viral rebound. In AIMS, the intervention was tailored to the needs and abilities of individual patients [46]. The nurse explained to each patient through a simple graph explaining how drug concentrations vary with adherence patterns and affect treatment outcomes. AIMS was cost-effective (i.e. less expensive and more effective) because it reduced lifetime societal costs by $977 per patient while increasing QALYs by 0.034 per patient with an ICER of $ 17,411, which is less than the WTP threshold of the Netherlands [46]. Overall, AIMS was feasible to deliver in routine care, reduce viral load, increase QALYs, save resources and was ready to be implemented in at least high-income settings.
3.6 Adherence Interventions for Pregnant PLWHA
3.6.1 Directly Observed Antiretroviral Therapy (DOT) Intervention
McCabe et al. (USA) predicted that DOT would reduce vertical HIV transmission in HIV-infected women receiving treatment by the third trimester of pregnancy [52]. Relative to usual care (UC), DOT was associated with a relative risk of HIV transmission of 0.39, was cost-effective in the cohort as a whole (ICER $18,038 per QALY, i.e. less than the USA commonly reported WTP threshold of $50,000) and led to cost-savings in women whose viral loads (VLs) on self-administered antiretroviral therapy (ART) would have exceeded 1000 copies/ml. DOT optimizes adherence to ART in pregnancy and has the potential to diminish mother-to-child HIV transmission in a highly cost-effective manner [52].
3.7 Adherence Interventions for Young PLWHA
3.7.1 Smartphone-Based Interactive Interventions
Neilan et al. (USA) assessed a hypothetical 12 month adherence intervention comprising two-way interactive smartphone-based reminders for adolescents that increased cohort-level viral suppression in young with HIV (YWH) in care by an absolute 10% for $100/month/person [43]. At 12 months, adherence intervention would decrease HIV transmissions by 15% and deaths by 12% when compared with UC. Adherence intervention would increase discounted life expectancy/person by 8 months at an added lifetime cost/person of $5737, resulting in an ICER of $8552/QALY [43]. The study found that adherence interventions aimed at YWH would improve virologic suppression and could have a significant impact on HIV transmissions, life expectancy, deaths and costs.
3.8 Adherence Interventions for Adult PLWHA
3.8.1 Smartphone-Based Interactive Interventions
In the intervention study by Patel et al. (Kenya), weekly interactive short message service (SMS) reminders were sent to PLWHA in addition to UC [45]. Patients were asked to reply within 2 days if they face any problems. If they did not reply, the clinician called them to inquire about their health. The intervention was followed for 12 months, and VL and self-reported adherence were measured at months 6 and 12. The intervention increased the survival time of PLWHA from 22.11 to 22.64 years. Moreover, the SMS-based intervention was cost-effective by the World Health Organization (WHO) standards, with an ICER of $1169.77/QALY [45]. The intervention was effective in improving adherence and increasing health outcomes and communication between patient and care provider without the need for a visit to the health facility.
Philips et al. (Zimbabwe) reported adherence monitoring interventions (AMI), including a recording of on-time drug pick-up and electronic monitors of adherence in real time using mobile phone technology [47]. AMIs resulted in a 6% increase in the proportion of ART-experienced people with VL 1000 copies/ml, and it was cost-effective if it cost up to $57 per person per year on ART, owing primarily to the cost savings from care differentiation. The cost thresholds identified suggest that there is a clear scope for adherence monitoring-based interventions to provide net population health gain, with potential cost-effective use in situations where VL monitoring is or is not available [47]. AIMS provide net population health gains in low-income settings in Sub-Saharan Africa, with the potential for cost-effective use in situations where viral load monitoring is either available or not.
3.8.2 Nurse-Involved Interventions
Freedberg et al. (USA) evaluated the nurse intervention including two, 1 hour home visits per week for the first 6 weeks of therapy and extrapolated the benefits to the lifetime of PLWHA through a societal perspective [55]. The intervention increased the expected survival (from 94.5 to 100.9 quality-adjusted life months) and estimated discounted direct lifetime medical costs (from $357,729 to $368,301). Nurse intervention was cost-effective by the most commonly reported US threshold value-for-money of $50,000, with an ICER of $19,873.88/QALY [55]. The intervention provided long-term survival benefits to PLWHA.
3.8.3 Nurse and Smartphone Combined Intervention
Zaric et al. (USA) developed a model to evaluate the cost-effectiveness of counselling interventions to improve adherence to ART among MSMs [54]. The intervention was comprised of counselling by an experienced nurse given before the initiation of ART combined with phone support. If the impact of HIV transmission is ignored, the counselling intervention has a cost-effectiveness ratio of $35,942 per QALY gained. When HIV transmission is included, the ICER is much lower: $10,430 and $12,262 per QALY gained in the moderate- and high-prevalence populations, respectively [54]. Counselling to improve adherence to ART increased length of life, modestly reduced HIV transmission and cost substantially less than $50,000 per QALY gained over a wide range of assumptions, but did not reduce the proportion of drug-resistant strains. Such counselling provides only modest benefits as a tool for HIV prevention but can provide significant benefits for individual patients at an affordable cost.
3.8.4 Link4Health Intervention
Steven et al. (Eswatini) evaluated the cost-effectiveness of a scale-up of the Link4Health strategy in Eswatini [44]. The Link4Health intervention comprised (1) faster ART initiation, (2) point-of-care CD4 lymphocyte count testing, (3) cell phone session reminders, (4) provision of informational pamphlets and a care and prevention package including commodities, and (5) mobile phone credits. Modelling reported that uptake of Link4Health intervention would decrease new HIV proliferation over 20 years by 11,059 infections, a 7% decrease from the projected 169,019 cases, and prevent 5313 deaths, an 11% decrease from the projected 49,582 deaths. From the perspective of the health sector, Link4Health led to an incremental cost per infection prevention of $14,409 and an incremental cost per QALY gain of $4059/QALY [44]. The scale-up of the Link4Health strategy would substantially reduce HIV-related deaths and avoid new HIV infections.
3.8.5 DOT Intervention
The intervention by Goldie et al. (USA) comprised DOT, automatic medication dispensers, beepers and portable alarms [57]. For relatively healthy patients with early disease, interventions reduced virologic failure rates by 10% and increased quality-adjusted life expectancy by 3.2 months. For patients with advanced disease and those from an urban cohort, adherence interventions costing about $737 per month (e.g. DOT) reduced failure by about 25% to have ICER below $50,000 per QALY [57]. According to the findings, interventions that improve adherence to combination antiretroviral therapy by at least 10–20% will result in quality-adjusted life expectancy gains comparable to opportunistic infection prophylaxis.
3.8.6 Computer-Delivered Intervention (CDI)
Ownby et al. (USA) developed the lifetime CDI through the internet to improve adherence [50]. The authors found the CDI was cost-effective as judged by the conventional benchmark of $50,000 per QALY ($39,229 cost/QALY). The intervention’s cost-effectiveness depends largely on the number of persons using it and the duration of its effectiveness [50]. The intervention was associated with net cost savings for intermediate and larger assumed effects and longer durations of intervention effectiveness.
3.8.7 Simplification of Complex Regimen
Colombo et al. (Italy) reported that a single-tablet regimen (STR) (0.755 QALYs/year) resulted in better patient quality of life, with a higher number of QALYs than for the multi-pill regimen (0.72 QALYs/year) [49]. The STR was the most cost-effective treatment strategy, with an ICER of $34,875 versus $42,068 for the multi-pill regimen. The STR resulted in better adherence, and therefore better quality of life as perceived by patients, corresponding to a $6544 lower ICER per QALY versus the multiple regimens, with a 17% lower cost in favour of the STR [49].
3.8.8 Case Manager Involved Intervention
Gopalappa et al. (USA) simulated an intervention that included up to five sessions with a case manager over 90 days [51]. Within 6 months, approximately 78% of people who received the intervention visited an HIV clinic at least once, compared with 60% of UC participants who received informational pamphlets and passive referrals to HIV care providers. Modelling results predicted that the linkage-to-care goal increased life expectancy by 0.4 years and delayed the onset of AIDS by 1.2 years on average for every HIV-diagnosed person. Increasing early linkage-to-care costs an extra $78,195 per QALY gained, considering only benefits to index person [51]. Early linkage-to-care and treatment initiation provides significantly improved life expectancy for HIV-infected persons and is likely to play an important role in HIV prevention and care services.
3.8.9 Prevention of Loss to Follow-Up (LTFU) Interventions
Losina et al. (Côte d'Ivoire) projected the clinical benefits and cost-effectiveness of the LTFU prevention intervention programme [53]. Interventions prevented LTFU in resource-limited settings substantially improved survival and were cost-effective by international criteria with efficacy of at least 12–41%, depending on the cost of intervention, based on a reported 18% cumulative incidence of LTFU at 1 year after ART initiation. An intervention costing $102/person/year (inclusive of all the components described above) would be cost-effective [53]. The findings suggest that moderately effective strategies for preventing LTFU in resource-limited settings would improve survival, provide good value for money and should be used to improve HIV treatment programmes.
Kessler et al. (Kenya) evaluated interventions to enhance HIV-infected patients’ retention in care (RIC) [48]. The cost-effectiveness ratio of ensuring full access to ART for ART-eligible patients ($1300 versus $3700) was higher than that of reducing attrition by 40% at an average cost of $10 per person. The authors reported that an outreach intervention had limited clinical benefit. However, if intervention costs are $10 per person, an intervention that can reduce attrition by 40% (or more) may be a cost-effective next implementation option after the start of earlier ART treatment [48]. The results suggest that programmes should consider retention-focused programmes once they have already achieved high degrees of ART coverage among eligible patients.
3.8.10 Ideal Adherence
The ideal adherence intervention of Munakata et al. (USA) (adherence observed in clinical trials) was cost-effective at $43,345 QALY gained [56]. Ideal adherence interventions can increase adherence to ideal levels and can cost up to $2358/year per patient, but the incremental cost-effectiveness is still less than $50,000/QALY gained. Ideal adherence to ART increased quality-adjusted life expectancy by 12% compared with typical adherence (adherence observed in observational studies) [56].
3.9 Findings Synthesis
Table 3 shows that in five studies, ICERs cost less than 25% of the WTP threshold; in three studies, ICERs cost 25–50% of the WTP threshold; in three studies, ICERs cost 50–75% of the WTP threshold and in four studies, ICERs cost 75–100% of the WTP threshold limit. Figure 2 shows the synthesis of findings by the dominance ranking framework. One study was cost-effective (blue box) [46]. In 14 studies, the intervention had high effectiveness but the cost was also higher; therefore, policymakers have to consider the factors such as incremental cost-effectiveness measures and priorities/willingness to pay [43,44,45,46,47,48,49,50,51,52,53,54,55,56,57] The reported ICERs for all interventions were within the commonly accepted WTP thresholds of the respective countries. Most adherence interventions had a higher implementation cost than standard care, but the benefits accrued over the period of used time horizons (1 year to lifetime) were substantially higher in the intervention group. Interventions, for example, would increase PLWHA survival, reduce the proportion of ARV drug resistance, reduce HIV transmission and prevent new HIV infections and deaths associated with AIDS. Furthermore, studies have shown that interventions will improve the quality of life of PLWHA and, as a result, reduce disability-related productivity loss.
4 Discussion
This systematic review examined the cost-effectiveness of ARV therapy adherence interventions for PLWHA in studies that utilized decision-based analytic models. Although a few (15 studies) decision analytic models are available, we still found a significant evidence gap in this area, which can be better filled by incorporating the suggestions from this systematic review in future decision analytic models. The findings suggest that cost of interventions were below the WTP threshold and are ready to implement after careful interpretation. However, there is little evidence for low- and middle-income countries, especially countries in the Asia Pacific region. Most studies were conducted in high-income countries and some African countries, suggesting that adherence interventions were more ready to be applied in developed regions and some African countries where these studies were conducted [43, 46, 49,50,51,52, 54,55,56,57]. The studies were funded by government and industry, demonstrating that both the public and private sectors have a strong interest in implementing cost-effective adherence interventions for PLWHA in the healthcare system.
The included studies demonstrated scientific rigour in their modelling approaches, as indicated by adherence to the QHES checklist requirements regarding methodology items (e.g. time horizon, model assumptions, uncertainty, comparators, health outcome measures, sensitivity analysis and subgroup analysis). However, the data inputs used in the models were diverse and predominantly sourced from observational studies and expert opinions, lacking in-depth information. Consequently, the implementation of these findings requires consideration of factors beyond cost-effectiveness, such as the study’s time horizon, data inputs, perspectives and modelling assumptions. [58]. To assess the acceptability of the PLWHA intervention, examining model cohorts/trials for dropout rates, as well as methodological limitations studies, can help to reach a implementation decision [59]. Different settings face diverse challenges in implementing adherence programmes due to stakeholder priorities and funding limitations. Stronger evidence is needed to justify investment in adherence promotion for marginalized populations, including pregnant women, MSM, FSWs, adolescents and transgender PLWHA, given limited resources and delivery capacity [60].
Smartphone-based and nurse- and case-manager-involved interventions can be implemented in resource-limited settings considering their low implementation cost. To further implement, the respective settings need to consider the training of nurses and case managers that can influence and educate PLWHA [61]. For smartphone-based interventions, the availability of smartphones to PLWHA and their literacy level to use the smartphone applications are crucial before implementation [62, 63]. A recent randomized-controlled trial (RCT) reported that smartphone-based adherence intervention was convenient to implement and easy to use for the overwhelming majority of participants, and PLWHA found the intervention useful and highly acceptable overall [62]. Smartphone-based interventions improved the PLWHA’s understanding of their own care and they felt empowered to ask their providers questions about their care with less stigma and discrimination. Smartphone interventions have nationwide reach and this approach eliminates provider burden and lack of expertise barriers [62]. Moreover, a recent mHealth intervention has been developed that can help to reduce long waits, stigma, multiple visits, negative experiences with healthcare professionals and discrimination [64]. Technology-driven interventions are ideal for marginalized communities that are facing huge stigma and discrimination such as MSM, female sex workers (FSWs), transgender and LGBTQ+. mHealth intervention development at the start are mostly costly but once developed they are highly cost-effective in terms of prevention of HIV transmission and increasing life expectancy in PLWHA [43, 50].
The review found limited evidence of interventions designed to improve adherence in key PLWHA populations such as young, pregnant women therefore, in future research, we recommend the development of more focused interventions and their HTA in young, women and marginalized communities such as LGBTQ+, FSWs, transgender, drug users and MSMs [65, 66]. Other interventions that have been found to be effective, such as pharmacist interventions that have been found to be effective in improving PLWHA adherence and other health outcomes such as anti-retroviral resistance and quality of life [67], should be further considered for economic modelling, as well as Suubi+Adherence interventions that have adequately engaged adolescents living with HIV [30, 68].
To further improve the decision modelling, future modelling research could include leveraging routinely gathered clinical data to inform parameters rather than depending solely on retrospective or prospective cohorts or a single RCT and expert opinion. If this is not possible, it is critical to include adequate detail on the techniques of soliciting expert opinion, including who the experts are and how their opinion was elicited. Modelling studies can follow the International Society of Pharmacoeconomics and outcome research (ISPOR) guidelines to identify the health states utilities [69]. The time horizon of future modelling interventions should be extended and an effort should be made to evaluate a greater range of adherence interventions. Future modelling studies should also focus on assessing the methodological, structural and heterogeneity components along with parameter uncertainty assessment during sensitivity analysis.
5 Limitations
We included only studies that were published in peer-reviewed journals; therefore, some guidance or policy documents relevant to this review may have been missed. It is widely acknowledged that strict journal word limits make it difficult for authors to convey all significant aspects of their modelling technique. This barrier, however, is expected to be reduced in the future with an improved ability to disseminate supplementary material. We also did not search the grey literature, although we did search conference abstracts and looked for the availability of peer-reviewed full texts.
6 Conclusions
Technology-based interventions and counselling interventions are cost-effective and can be implemented after careful interpretation to address the chronic adherence issue of PLWHA. Although there are some methodological issues, identified decision analytical modelling studies reported that counselling interventions and smartphone-based interventions have a significant potential to improve adherence in PLWHA with an acceptable ICER. These interventions are ready for implementation in the countries where they are studied, and other countries can follow suit by taking into account study methodological flaws, available funding and cultural acceptability factors. However, more evidence is needed to assess the impact of adherence interventions in marginalized HIV/AIDS communities.
References
Ahmed A, et al. Health-related quality of life and its predictors among adults living with HIV/AIDS and receiving antiretroviral therapy in Pakistan. Qual Life Res. 2021;30(6):1653–64.
Padilla M, et al. The boundary of HIV care: Barriers and facilitators to care engagement among people with HIV in the United States. AIDS Patient Care STDS. 2022;36(8):321–31.
Rudolph AE, et al. Perceived HIV treatment norms modify the association between HIV-related stigma and adherence to antiretroviral therapy among persons living with HIV in Baltimore, Maryland. AIDS Behav. 2022;26(2):537–48.
Manosuthi W, Charoenpong L, Santiwarangkana C. A retrospective study of survival and risk factors for mortality among people living with HIV who received antiretroviral treatment in a resource-limited setting. AIDS Res Ther. 2021;18(1):71.
Ahmed A, et al. Translation and cross-cultural adaptation of WHOQOL-HIV Bref among people living with HIV/AIDS in Pakistan. Health Qual Life Outcomes. 2021;19(1):1–11.
Ahmed A, et al. Effect of pharmacist care on clinical outcomes among people living with HIV/AIDS: a systematic review and meta-analysis. Res Social Adm Pharm. 2022;18(6):2962–80.
Hung C-C, et al. Impact of COVID-19 on the HIV care continuum in Asia: insights from people living with HIV, key populations, and HIV healthcare providers. PLoS ONE. 2022;17(7): e0270831.
Bezabhe WM, et al. Adherence to antiretroviral therapy and virologic failure: a meta-analysis. Medicine. 2016;95(15):e3361.
Ahmed A, et al. Stigma, social support, illicit drug use, and other predictors of anxiety and depression among HIV/AIDS patients in Pakistan: a cross-sectional study. Front Public Health. 2021;9:1428.
Agegnehu CD, et al. Burden and associated factors of virological failure among people living with HIV in sub-Saharan Africa: a systematic review and meta-analysis. AIDS Behav. 2022;26:1–10.
Ahmed A, et al. Barriers and enablers for adherence to antiretroviral therapy among people living with HIV/AIDS in the era of COVID-19: a qualitative study From Pakistan. Front Pharmacol. 2021;12:3968.
Bomfim IGDO, Santos SDS, Napoleão AA. Adherence to antiretroviral therapy in people living with HIV/AIDS: a cross-sectional study. AIDS Patient Care STDs. 2022;36(7):278–84.
Ahmed A et al. Concerns of HIV-positive migrant workers in COVID-19 pandemic: a call for action. J Glob Health. 2020;10(2).
Larrea-Schiavon S, et al. Increased rate of protease inhibitor-resistance associated mutations in human immunodeficiency viruses infecting mexicans who had been living abroad. Arch Med Res. 2022;53(3):296–303.
Yuan D, et al. Prevalence and determinants of virological failure, genetic diversity and drug resistance among people living with HIV in a minority area in China: a population-based study. BMC Infect Dis. 2020;20(1):1–10.
Whiteley LB, et al. A review of interventions to enhance HIV medication adherence. Curr HIV/AIDS Rep. 2021;18(5):443–57.
Duthely LM, Sanchez-Covarrubias AP. Digitized HIV/AIDS treatment adherence interventions: a review of recent SMS/texting mobile health applications and implications for theory and practice. Front Commun. 2020;5: 530164.
Pellowski JA, et al. A systematic review and meta-analysis of antiretroviral therapy (ART) adherence interventions for women living with HIV. AIDS Behav. 2019;23(8):1998–2013.
Ekstrand ML, et al. A behavioral adherence intervention improves rates of viral suppression among adherence-challenged people living with HIV in South India. AIDS Behav. 2020;24(7):2195–205.
Ahmed A, et al. The impact of clinical pharmacist services on patient health outcomes in Pakistan: a systematic review. BMC Health Serv Res. 2021;21(1):1–14.
Ahmed A, et al. Pharmacist-involved antiretroviral stewardship programs in people living with HIV/AIDS: a systematic review. AIDS Patient Care STDS. 2023;37(1):31–52.
Elnaem MH, et al. Impact of medication regimen simplification on medication adherence and clinical outcomes in patients with long-term medical conditions. Patient Prefer Adher. 2020;14:2135.
Cutler RL, et al. Economic impact of medication non-adherence by disease groups: a systematic review. BMJ Open. 2018;8(1): e016982.
Jiang X, Ming W-K, You JH. The cost-effectiveness of digital health interventions on the management of cardiovascular diseases: systematic review. J Med Internet Res. 2019;21(6): e13166.
Petrou S, Gray A. Economic evaluation using decision analytical modelling: design, conduct, analysis, and reporting. BMJ. 2011;342:d1766.
Rossi JG, et al. Cost-effectiveness of artificial intelligence as a decision-support system applied to the detection and grading of melanoma, dental caries, and diabetic retinopathy. JAMA Netw Open. 2022;5(3):e220269–e220269.
Magidson JF, et al. Act healthy: a randomized clinical trial evaluating a behavioral activation intervention to address substance use and medication adherence among low-income, Black/African American individuals living with HIV/AIDS. AIDS Behav. 2022;26(1):102–15.
Cavazos-Rehg P, et al. The impact of a family-based economic intervention on the mental health of HIV-infected adolescents in Uganda: results from Suubi+ Adherence. J Adolesc Health. 2021;68(4):742–9.
Wijnen BFM, et al. Cost-effectiveness and cost-utility of the adherence improving self-management strategy in human immunodeficiency virus care: a trial-based economic evaluation. Clin Infect Dis. 2019;68(4):658–67.
Ssewamala FM, et al. The long-term effects of a family based economic empowerment intervention (Suubi+ Adherence) on suppression of HIV viral loads among adolescents living with HIV in southern Uganda: Findings from 5-year cluster randomized trial. PLoS ONE. 2020;15(2): e0228370.
Kranzer K, et al. Economic incentives for HIV testing by adolescents in Zimbabwe: a randomised controlled trial. Lancet HIV. 2018;5(2):e79–86.
Swann M. Economic strengthening for retention in HIV care and adherence to antiretroviral therapy: a review of the evidence. AIDS Care. 2018;30(sup3):99–125.
Dang M, et al. Barriers and facilitators to HIV pre-exposure prophylaxis uptake, adherence, and persistence among transgender populations in the United States: a systematic review. AIDS Patient Care STDS. 2022;36(6):236–48.
Page MJ, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Syst Rev. 2021;10(1):1–11.
Cumpston M, et al. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev. 2019;10(10.1002):14651858.
Gomersall JS, et al. Conducting systematic reviews of economic evaluations. JBI Evid Implement. 2015;13(3):170–8.
Le LK-D, et al. Cost-effectiveness evidence of mental health prevention and promotion interventions: a systematic review of economic evaluations. PLoS Med. 2021;18(5): e1003606.
García-Pérez L, et al. Interventions to improve medication adherence in mental health: the update of a systematic review of cost-effectiveness. Int J Psychiatry Clin Pract. 2020;24(4):416–27.
Morciano M, Thomas J, Shemilt I. A web-based tool for adjusting costs to a specific target currency and price year. Evid Policy A J Res Debate. 2010;6(1):51.
Nixon J, Khan KS, Kleijnen J. Summarising economic evaluations in systematic reviews: a new approach. BMJ. 2001;322(7302):1596–8.
Mauskopf J, Rutten F, Schonfeld W. Cost-effectiveness league tables. Pharmacoeconomics. 2003;21(14):991–1000.
Ofman JJ, et al. Examining the value and quality of health economic analyses: implications of utilizing the QHES. J Manag Care Pharm. 2003;9(1):53–61.
Neilan AM, et al. Modeling adherence interventions among youth with HIV in the United States: clinical and economic projections. AIDS Behav. 2021;25(9):2973–84.
Stevens ER, et al. Cost-effectiveness of a combination strategy to enhance the HIV care continuum in Swaziland: Link4Health. PLoS ONE. 2018;13(9): e0204245.
Patel AR, et al. Economic evaluation of mobile phone text message interventions to improve adherence to HIV therapy in Kenya. Medicine. 2017;96(7): e6078.
de Bruin M, et al. Effectiveness and cost-effectiveness of a nurse-delivered intervention to improve adherence to treatment for HIV: a pragmatic, multicentre, open-label, randomised clinical trial. Lancet Infect Dis. 2017;17(6):595–604.
Phillips AN, et al. Cost effectiveness of potential ART adherence monitoring interventions in Sub-Saharan Africa. PLoS ONE. 2016;11(12): e0167654.
Kessler J, et al. Impact and cost-effectiveness of hypothetical strategies to enhance retention in care within HIV treatment programs in East Africa. Value Health. 2015;18(8):946–55.
Colombo GL, Di Matteo S, Maggiolo F. Antiretroviral therapy in HIV-infected patients: a proposal to assess the economic value of the single-tablet regimen. Clinicoecon Outcomes Res. 2013;5:59–68.
Ownby RL, et al. Cost effectiveness of a computer-delivered intervention to improve HIV medication adherence. BMC Med Inform Decis Mak. 2013;13:29.
Gopalappa C, et al. Cost effectiveness of the National HIV/AIDS Strategy goal of increasing linkage to care for HIV-infected persons. J Acquir Immune Defic Syndr. 2012;61(1):99–105.
McCabe CJ, Goldie SJ, Fisman DN. The cost-effectiveness of directly observed highly-active antiretroviral therapy in the third trimester in HIV-infected pregnant women. PLoS ONE. 2010;5(4): e10154.
Losina E, et al. Cost-effectiveness of preventing loss to follow-up in HIV treatment programs: a Côte d’Ivoire appraisal. PLoS Med. 2009;6(10): e1000173.
Zaric GS, et al. The cost-effectiveness of counseling strategies to improve adherence to highly active antiretroviral therapy among men who have sex with men. Med Decis Making. 2008;28(3):359–76.
Freedberg KA, et al. Cost-effectiveness of an intervention to improve adherence to antiretroviral therapy in HIV-infected patients. J Acquir Immune Defic Syndr. 2006;43(Suppl 1):S113–8.
Munakata J, et al. Clinical and economic outcomes of nonadherence to highly active antiretroviral therapy in patients with human immunodeficiency virus. Med Care. 2006;44(10):893–9.
Goldie SJ, et al. Projecting the cost-effectiveness of adherence interventions in persons with human immunodeficiency virus infection. Am J Med. 2003;115(8):632–41.
Van Soest HL, et al. Analysing interactions among sustainable development goals with integrated assessment models. Glob Trans. 2019;1:210–25.
Mubiana-Mbewe M, et al. Effect of enhanced adherence package on early ART uptake among HIV-positive pregnant women in Zambia: an individual randomized controlled trial. AIDS Behav. 2021;25(3):992–1000.
Wulandari LPL, et al. Preferences for pre-exposure prophylaxis for HIV: a systematic review of discrete choice experiments. EClinicalMedicine. 2022;51: 101507.
Crowley T, Mokoka E, Geyer N. Ten years of nurse-initiated antiretroviral treatment in South Africa: a narrative review of enablers and barriers. South Afr J HIV Med. 2021;22(1):1–13.
DeFulio A, et al. Smartphone-based incentives for promoting adherence to antiretroviral therapy: a randomized controlled trial. Prev Med Rep. 2021;21: 101318.
Bezabih AM, et al. Behavioral theories and motivational features underlying eHealth interventions for adolescent antiretroviral adherence: systematic review. JMIR Mhealth Uhealth. 2021;9(12): e25129.
Shrestha R, et al. Clinic-integrated mHealth intervention (“JomPrEP” App) to improve uptake of HIV testing and pre-exposure prophylaxis among men who have sex with men in Malaysia: protocol for intervention development and multi-phase trial. JMIR Research Protocols, 2022. 17/11/2022:43318.
Zhang C, et al. An integrated conceptual framework of behavioral intervention technologies to promote healthcare utilization among socially-marginalized populations in the United States. J Nurs Stud Patient Care. 2022;3(1):12.
Giovenco D, et al. Adapting technology-based HIV prevention and care interventions for youth: Lessons learned across five US Adolescent Trials Network studies. MHealth. 2021;7.
Ahmed A, et al. The economic impact of pharmacist care for people living with HIV/AIDS: a systematic review. Explor Res Clin Soc Pharm. 2021;3: 100066.
Brathwaite R, et al. Predicting the individualized risk of poor adherence to ART medication among adolescents living with HIV in Uganda: the Suubi+ Adherence study. J Int AIDS Soc. 2021;24(6): e25756.
Brazier J, et al. Identification, review, and use of health state utilities in cost-effectiveness models: an ISPOR good practices for outcomes research task force report. Value Health. 2019;22(3):267–75.
Acknowledgements
We would like to thank the chief editor and reviewers of the Applied Health Economics and Health Policy journal for their thorough review and suggestions to improve this review.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Funding
None.
Availability of data and material
All data generated or analysed during this study are included in this published article.
Code availability
Not applicable.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication (from patients/participants)
Not applicable.
Authors contributions
Ali Ahmed (AA1): Conceptualization, methodology, software, validation, analysis, investigation, resources, data curation, writing an original draft, editing, visualization, project administration; Jaman Abdulelah Dujaili (JD): Methodology, data curation, validation; Lay Hong Chuah (LHC): Supervision, methodology, resources; Furqan Khurshid Hashmi (FKH): Supervision, data curation, resources; Long Khanh-Dao Le (LL): Supervision, validation; Saval Khanal (SK): Data curation, supervision; Ahmed Awaisu (AA2): Supervision, review, validation; Nathorn Chaiyakunapruk (NC): Conceptualization, methods, supervision. All authors approved the final version of the manuscript for submission.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Ahmed, A., Dujaili, J.A., Chuah, L.H. et al. Cost-Effectiveness of Anti-retroviral Adherence Interventions for People Living with HIV: A Systematic Review of Decision Analytical Models. Appl Health Econ Health Policy 21, 731–750 (2023). https://doi.org/10.1007/s40258-023-00818-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40258-023-00818-4