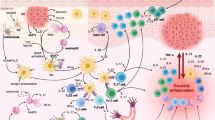
Abstract
Cutaneous immune-related adverse events encompass a spectrum of dermatological manifestations, including lichenoid reactions, psoriasiform eruptions, eczematous dermatitis, immunobullous disorders, granulomatous reactions, pruritus, vitiligo, and severe cutaneous adverse reactions such as Stevens–Johnson syndrome. The conventional approach to treating high-grade or refractory cutaneous immune-related adverse events has involved high-dose systemic corticosteroids. However, their use is limited owing to the potential disruption of antitumor responses and associated complications. To address this, corticosteroid-sparing targeted immunomodulators have been explored as therapeutic alternatives. Biologic agents, commonly employed for non-cutaneous immune-related adverse events such as colitis, are increasingly recognized for their efficacy in treating various patterns of cutaneous immune-related adverse events, including psoriasiform, immunobullous, and Stevens–Johnson syndrome-like reactions. This review consolidates findings from the English-language literature, highlighting the use of biologic agents in managing diverse cutaneous immune-related adverse event patterns, also encompassing maculopapular, eczematous, and lichenoid eruptions, pruritus, and transient acantholytic dermatosis (Grover disease). Despite the established efficacy of these agents, further research is necessary to explore their long-term effects on antitumor responses.
Similar content being viewed by others
References
Tattersall IW, Leventhal JS. Cutaneous toxicities of immune checkpoint inhibitors: the role of the dermatologist. Yale J Biol Med. 2020;93(1):123–32.
Lacouture M, Sibaud V. Toxic side effects of targeted therapies and immunotherapies affecting the skin, oral mucosa, hair, and nails. Am J Clin Dermatol. 2018;19(Suppl. 1):31–9. https://doi.org/10.1007/s40257-018-0384-3.
Geisler AN, Phillips GS, Barrios DM, et al. Immune checkpoint inhibitor-related dermatologic adverse events. J Am Acad Dermatol. 2020;83(5):1255–68. https://doi.org/10.1016/j.jaad.2020.03.132.
Fay CJ, Jakuboski S, McLellan B, et al. Diagnosis and management of dermatologic adverse events from systemic melanoma therapies. Am J Clin Dermatol. 2023;24(5):765–85. https://doi.org/10.1007/s40257-023-00790-8.
Apalla Z, Sibaud V. Immunotherapy-mediated dermatological adverse events: the urgent need for a common, clinically meaningful, management strategy. Support Care Cancer. 2020;28(12):5597–9. https://doi.org/10.1007/s00520-020-05701-9.
Ferreira MN, Ramseier JY, Leventhal JS. Dermatologic conditions in women receiving systemic cancer therapy. Int J Womens Dermatol. 2019;5(5):285–307. https://doi.org/10.1016/j.ijwd.2019.10.003.
Friedman CF, Proverbs-Singh TA, Postow MA. Treatment of the immune-related adverse effects of immune checkpoint inhibitors: a review. JAMA Oncol. 2016;2(10):1346–53. https://doi.org/10.1001/jamaoncol.2016.1051.
Phillips GS, Wu J, Hellmann MD, et al. Treatment outcomes of immune-related cutaneous adverse events. J Clin Oncol. 2019;37(30):2746–58. https://doi.org/10.1200/JCO.18.02141.
Arbour KC, Mezquita L, Long N, et al. Impact of baseline steroids on efficacy of programmed cell death-1 and programmed death-ligand 1 blockade in patients with non-small-cell lung cancer. J Clin Oncol. 2018;36(28):2872–8. https://doi.org/10.1200/JCO.2018.79.0006.
Faje AT, Lawrence D, Flaherty K, et al. High-dose glucocorticoids for the treatment of ipilimumab-induced hypophysitis is associated with reduced survival in patients with melanoma. Cancer. 2018;124(18):3706–14. https://doi.org/10.1002/cncr.31629.
Montfort A, Filleron T, Virazels M, et al. Combining nivolumab and ipilimumab with infliximab or certolizumab in patients with advanced melanoma: first results of a phase Ib clinical trial. Clin Cancer Res. 2021;27(4):1037–47. https://doi.org/10.1158/1078-0432.CCR-20-3449.
Lesage C, Longvert C, Prey S, et al. Incidence and clinical impact of anti-TNFalpha treatment of severe immune checkpoint inhibitor-induced colitis in advanced melanoma: the Mecolit Survey. J Immunother. 2019;42(5):175–9. https://doi.org/10.1097/CJI.0000000000000268.
Badran YR, Cohen JV, Brastianos PK, Parikh AR, Hong TS, Dougan M. Concurrent therapy with immune checkpoint inhibitors and TNFalpha blockade in patients with gastrointestinal immune-related adverse events. J Immunother Cancer. 2019;7(1):226. https://doi.org/10.1186/s40425-019-0711-0.
Barrios DM, Phillips GS, Geisler AN, et al. IgE blockade with omalizumab reduces pruritus related to immune checkpoint inhibitors and anti-HER2 therapies. Ann Oncol. 2021;32(6):736–45. https://doi.org/10.1016/j.annonc.2021.02.016.
Apalla Z, Nikolaou V, Fattore D, et al. European recommendations for management of immune checkpoint inhibitors-derived dermatologic adverse events: the EADV Task Force “Dermatology for cancer patients” position statement. J Eur Acad Dermatol Venereol. 2022;36(3):332–50. https://doi.org/10.1111/jdv.17855.
Monsour EP, Pothen J, Balaraman R. A novel approach to the treatment of pembrolizumab-induced psoriasis exacerbation: a case report. Cureus. 2019;11(10): e5824. https://doi.org/10.7759/cureus.5824.
Johnson D, Patel AB, Uemura MI, et al. IL17A blockade successfully treated psoriasiform dermatologic toxicity from immunotherapy. Cancer Immunol Res. 2019;7(6):860–5. https://doi.org/10.1158/2326-6066.CIR-18-0682.
Lo JJ, Heberton MM, Pacha O, Huen AO, Patel AB. Biologic therapies for checkpoint inhibitor-induced cutaneous toxicities: a single-institution study of 17 consecutively treated patients. Support Care Cancer. 2022;30(2):989–94. https://doi.org/10.1007/s00520-021-06548-4.
Esfahani K, Miller WH Jr. Reversal of autoimmune toxicity and loss of tumor response by interleukin-17 blockade. N Engl J Med. 2017;376(20):1989–91. https://doi.org/10.1056/NEJMc1703047.
Kochi Y, Miyachi H, Tagashira R, et al. Simultaneous development of generalized pustular psoriasis and pemphigoid with multiple autoantibodies in a complete responder of pembrolizumab for lung cancer. J Dermatol. 2023. https://doi.org/10.1111/1346-8138.16832.
Hoa S, Laaouad L, Roberts J, et al. Preexisting autoimmune disease and immune-related adverse events associated with anti-PD-1 cancer immunotherapy: a national case series from the Canadian Research Group of Rheumatology in Immuno-Oncology. Cancer Immunol Immunother. 2021;70(8):2197–207. https://doi.org/10.1007/s00262-021-02851-5.
Vinaixa Aranzazu A, Morillas-Lahuerta V, De Tord MB, Carrascosa Carrillo JM. Ixekizumab for the treatment of erythrodermic psoriasis triggered by durvalumab-tremelimumab in a cancer patient. Eur J Dermatol. 2021. https://doi.org/10.1684/ejd.2021.4087.
Gleason L, Hunter E, Cohen A, Suriano J, Nikbakht N. Atezolizumab-induced psoriasiform drug eruption successfully treated with ixekizumab: a case report and literature review. Dermatol Online J. 2023. https://doi.org/10.5070/D329160215.
Kost Y, Mattis D, Muskat A, Amin B, McLellan B. Immune checkpoint inhibitor-induced psoriasiform, spongiotic, and lichenoid dermatitis: a novel clinicopathological pattern. Cureus. 2022;14(8): e28010. https://doi.org/10.7759/cureus.28010.
Seervai RNH, Heberton M, Cho WC, et al. Severe de novo pustular psoriasiform immune-related adverse event associated with nivolumab treatment for metastatic esophageal adenocarcinoma. J Cutan Pathol. 2022;49(5):472–81. https://doi.org/10.1111/cup.14185.
Nikolaou V, Sibaud V, Fattore D, et al. Immune checkpoint-mediated psoriasis: a multicenter European study of 115 patients from the European Network for Cutaneous Adverse Event to Oncologic Drugs (ENCADO) Group. J Am Acad Dermatol. 2021;84(5):1310–20. https://doi.org/10.1016/j.jaad.2020.08.137.
Glinos GD, Fisher WS, Morr CS, Seminario-Vidal L. Nivolumab-induced psoriasis successfully treated with risankizumab-rzaa in a patient with stage III melanoma. JAAD Case Rep. 2021;11:74–7. https://doi.org/10.1016/j.jdcr.2021.03.029.
Gargiulo L, Ibba L, Valenti M, Costanzo A, Narcisi A. Pembrolizumab-induced plaque psoriasis successfully treated with risankizumab in a patient with stage IV cutaneous melanoma. Melanoma Res. 2023;33(2):152–4. https://doi.org/10.1097/CMR.000000000000087.
Mital R, Otto TS, Savu A, et al. Detection of novel therapies using a multi-national, multi-institutional registry of cutaneous immune-related adverse events and management. Int J Dermatol. 2023;62(8):1020–5. https://doi.org/10.1111/ijd.16714.
Hansen I, Heidrich I, Abeck F, et al. Successful treatment of PD-1 inhibitor-induced psoriasis with infliximab. J Eur Acad Dermatol Venereol. 2023;37(5):e621–3. https://doi.org/10.1111/jdv.18780.
Kim YE, Kim TM, Jo SJ. Histologically-diagnosed psoriasiform dermatitis induced by nivolumab successfully controlled by etanercept: a case report. J Dermatol. 2019;46(12):e464–6. https://doi.org/10.1111/1346-8138.15091.
Lopez AT, Khanna T, Antonov N, Audrey-Bayan C, Geskin L. A review of bullous pemphigoid associated with PD-1 and PD-L1 inhibitors. Int J Dermatol. 2018;57(6):664–9. https://doi.org/10.1111/ijd.13984.
Siegel J, Totonchy M, Damsky W, et al. Bullous disorders associated with anti-PD-1 and anti-PD-L1 therapy: a retrospective analysis evaluating the clinical and histopathologic features, frequency, and impact on cancer therapy. J Am Acad Dermatol. 2018;79(6):1081–8. https://doi.org/10.1016/j.jaad.2018.07.008.
Asdourian MS, Shah N, Jacoby TV, Reynolds KL, Chen ST. Association of bullous pemphigoid with immune checkpoint inhibitor therapy in patients with cancer: a systematic review. JAMA Dermatol. 2022;58(8):933–41. https://doi.org/10.1001/jamadermatol.2022.1624.
Said JT, Talia J, Wei E, et al. Impact of biologic therapy on cancer outcomes in patients with immune checkpoint inhibitor-induced bullous pemphigoid. J Am Acad Dermatol. 2023;88(3):670–1. https://doi.org/10.1016/j.jaad.2022.06.1186.
Ridpath AV, Rzepka PV, Shearer SM, Scrape SR, Olencki TE, Kaffenberger BH. Novel use of combination therapeutic plasma exchange and rituximab in the treatment of nivolumab-induced bullous pemphigoid. Int J Dermatol. 2018;57(11):1372–4. https://doi.org/10.1111/ijd.13970.
Singer S, Nelson CA, Lian CG, Dewan AK, LeBoeuf NR. Nonbullous pemphigoid secondary to PD-1 inhibition. JAAD Case Rep. 2019;5(10):898–903. https://doi.org/10.1016/j.jdcr.2019.07.015.
Sharma P, Barnes M, Nabeel S, LiPera W. Pembrolizumab-induced bullous pemphigoid treated with rituximab. JCO Oncol Pract. 2020;16(11):764–6. https://doi.org/10.1200/JOP.19.00751.
Virgen CA, Nguyen TA, Di Raimondo C, et al. Bullous pemphigoid associated with cemiplimab therapy in a patient with locally advanced cutaneous squamous cell carcinoma. JAAD Case Rep. 2020;6(3):195–7. https://doi.org/10.1016/j.jdcr.2020.01.008.
Povilaityte E, Gellrich FF, Beissert S, Abraham S, Meier F, Gunther C. Treatment-resistant bullous pemphigoid developing during therapy with immune checkpoint inhibitors. J Eur Acad Dermatol Venereol. 2021;35(9):e591–3. https://doi.org/10.1111/jdv.17321.
Sowerby L, Dewan AK, Granter S, Gandhi L, LeBoeuf NR. Rituximab treatment of nivolumab-induced bullous pemphigoid. JAMA Dermatol. 2017;153(6):603–5. https://doi.org/10.1001/jamadermatol.2017.0091.
Grimaux X, Delva R, Jadaud E, Croue A. Nivolumab-induced bullous pemphigoid after radiotherapy and abscopal effect. Australas J Dermatol. 2019;60(3):e235–6. https://doi.org/10.1111/ajd.12987.
Kaul S, Wang A, Grushchak S, Albrecht J. Pembrolizumab-induced reactivation of bullous pemphigoid. Int J Dermatol. 2021;60(6):757–8. https://doi.org/10.1111/ijd.15366.
Morris LM, Lewis HA, Cornelius LA, Chen DY, Rosman IS. Neutrophil-predominant bullous pemphigoid induced by checkpoint inhibitors: a case series. J Cutan Pathol. 2020;47(8):742–6. https://doi.org/10.1111/cup.13687.
Wesolow JT, Jalali S, Clark LD. A rare case of bullous pemphigoid secondary to checkpoint inhibitor immunotherapy: a tense situation. Cureus. 2021;13(7): e16169. https://doi.org/10.7759/cureus.16169.
Sadik CD, Langan EA, Gutzmer R, et al. Retrospective analysis of checkpoint inhibitor therapy-associated cases of bullous pemphigoid from six German dermatology centers. Front Immunol. 2020;11: 588582. https://doi.org/10.3389/fimmu.2020.588582.
Schauer F, Rafei-Shamsabadi D, Mai S, et al. Hemidesmosomal reactivity and treatment recommendations in immune checkpoint inhibitor-induced bullous pemphigoid: a retrospective, monocentric study. Front Immunol. 2022;13: 953546. https://doi.org/10.3389/fimmu.2022.953546.
Brown AM, Masterson W, Lo J, Patel AB. Systemic treatment of cutaneous adverse events after immune checkpoint inhibitor therapy: a review. Dermatitis. 2021. https://doi.org/10.1097/DER.0000000000000776.
Yun JSW, Chan OB, Goh M, McCormack CJ. Bullous pemphigoid associated with anti-programmed cell death protein 1 and anti-programmed cell death ligand 1 therapy: a case series of 13 patients. Australas J Dermatol. 2023;64(1):131–7. https://doi.org/10.1111/ajd.13960.
Damsky W, Kole L, Tomayko MM. Development of bullous pemphigoid during nivolumab therapy. JAAD Case Rep. 2016;2(6):442–4. https://doi.org/10.1016/j.jdcr.2016.05.009.
Choi SH, Chung KB, Kim DY. Inhibition of transforming growth factor beta and immune checkpoints induces a distinctively distributed, severe bullous pemphigoid. Acta Derm Venereol. 2021;101(6): adv00489. https://doi.org/10.2340/00015555-3853.
Klepper EM, Robinson HN. Dupilumab for the treatment of nivolumab-induced bullous pemphigoid: a case report and review of the literature. Dermatol Online J. 2021;27(9). doi: https://doi.org/10.5070/D327955136.
Bruni M, Moar A, Schena D, Girolomoni G. A case of nivolumab-induced bullous pemphigoid successfully treated with dupilumab. Dermatol Online J. 2022. https://doi.org/10.5070/D328257396.
Pop SR, Strock D, Smith RJ. Dupilumab for the treatment of pembrolizumab-induced bullous pemphigoid: a case report. Dermatol Ther. 2022;35(8): e15623. https://doi.org/10.1111/dth.15623.
Khazaeli M, Grover R, Pei S. Concomitant nivolumab-associated Grover disease and bullous pemphigoid in a patient with metastatic renal cell carcinoma. J Cutan Pathol. 2023;50(6):520–3. https://doi.org/10.1111/cup.14383.
Shipman WD, Singh K, Cohen JM, Leventhal JS, Damsky WD, Tomayko MM. Immune checkpoint inhibitor-induced bullous pemphigoid skin has elevated interleukin-4 and interleukin-13 expression and responds to IL-4R inhibitionImmune checkpoint inhibitor-induced bullous pemphigoid skin has elevated interleukin-4 and interleukin-13 expression and responds to IL-4R inhibition. J Invest Dermatol. 2022;142(8):S5.
Mihailescu ML, Brockstein BE, Desai N, Waldinger J. Successful reintroduction and continuation of nivolumab in a patient with immune checkpoint inhibitor-induced bullous pemphigoid. CPC Case Rep. 2020;2:100031. https://doi.org/10.1016/j.cpccr.2020.100031.
Cardona AF, Ruiz-Patino A, Zatarain-Barron ZL, et al. Refractory bullous pemphigoid in a patient with metastatic lung adenocarcinoma treated with pembrolizumab. Case Rep Oncol. 2021;14(1):386–90. https://doi.org/10.1159/000514144.
Molina GE, Yu Z, Foreman RK, Reynolds KL, Chen ST. Generalized bullous mucocutaneous eruption mimicking Stevens-Johnson syndrome in the setting of immune checkpoint inhibition: a multicenter case series. J Am Acad Dermatol. 2020;83(5):1475–7. https://doi.org/10.1016/j.jaad.2020.03.029.
Cai ZR, Lecours J, Adam JP, et al. Toxic epidermal necrolysis associated with pembrolizumab. J Oncol Pharm Pract. 2020;26(5):1259–65. https://doi.org/10.1177/1078155219890659.
Coleman E, Ko C, Dai F, Tomayko MM, Kluger H, Leventhal JS. Inflammatory eruptions associated with immune checkpoint inhibitor therapy: a single-institution retrospective analysis with stratification of reactions by toxicity and implications for management. J Am Acad Dermatol. 2019;80(4):990–7. https://doi.org/10.1016/j.jaad.2018.10.062.
Zhang L, Wu Z. Adalimumab for sintilimab-induced toxic epidermal necrolysis in a patient with metastatic gastric malignancy: a case report and literature review. Clin Cosmet Investig Dermatol. 2023;16:457–61. https://doi.org/10.2147/CCID.S401286.
Chen PY, Li ZY, Cai SQ. Case report: cadonilimab-related toxic epidermal necrolysis-like reactions successfully treated with supplemental adalimumab. Front Immunol. 2023;14:1188523. https://doi.org/10.3389/fimmu.2023.1188523.
Meledathu S, Gordon M, Thornton M, Ashinoff R. Management of Stevens-Johnson syndrome/toxic epidermal necrolysis: a case report and literature review. J Drugs Dermatol. 2023;22(11):e24–8. https://doi.org/10.36849/JDD.6999.
Kian W, Zemel M, Elobra F, et al. Intravenous immunoglobulin efficacy on pembrolizumab induced severe toxic epidermal necrolysis. Anticancer Drugs. 2022;33(1):e738–40. https://doi.org/10.1097/CAD.0000000000001162.
Sandhu M, Kc B, Bhandari J, Gambhir HS, Farah R. Pembrolizumab-associated Stevens-Johnson syndrome in a patient with metastatic non-small cell lung cancer: a case report. Cureus. 2023;15(7): e41439. https://doi.org/10.7759/cureus.41439.
Borg L, Buhagiar M, La Ferla E, Pisani D, Said J, Boffa MJ. Pembrolizumab-induced toxic epidermal necrolysis. Case Rep Oncol. 2022;15(3):887–93. https://doi.org/10.1159/000526931.
Ingen-Housz-Oro S, Milpied B, Badrignans M, et al. Severe blistering eruptions induced by immune checkpoint inhibitors: a multicentre international study of 32 cases. Melanoma Res. 2022;32(3):205–10. https://doi.org/10.1097/CMR.0000000000000819.
Yang Y, Li J, Till BG, et al. Toxic epidermal necrolysis-like reaction following combination therapy with camrelizumab and apatinib for advanced gallbladder carcinoma. Front Oncol 2021;11:728523. https://doi.org/10.3389/fonc.2021.728523.
Nayar N, Briscoe K, Fernandez PP. Toxic epidermal necrolysis-like reaction with severe satellite cell necrosis associated with nivolumab in a patient with opilimumab refractory metastatic melanoma. J Immunother. 2016;39(3):149–52. https://doi.org/10.1097/CJI.0000000000000112.
Vivar KL, Deschaine M, Messina J, et al. Epidermal programmed cell death-ligand 1 expression in TEN associated with nivolumab therapy. J Cutan Pathol. 2017;44(4):381–4. https://doi.org/10.1111/cup.12876.
Ahmad M, Murphy MJ, Damsky W, Leventhal J. Dupilumab-induced psoriasis in the setting of pembrolizumab therapy: an analysis of cytokine expression. Int J Dermatol. 2022;62(8):e42–6. https://doi.org/10.1111/ijd.16538.
Kuo AM, Gu S, Stoll J, et al. Management of immune-related cutaneous adverse events with dupilumab. J Immunother Cancer. 2023;11(6): e007324. https://doi.org/10.1136/jitc-2023-007324.
Hibler BP, Markova A. Treatment of severe cutaneous adverse reaction with tocilizumab. Br J Dermatol. 2020;183(4):785–7. https://doi.org/10.1111/bjd.19129.
Park JJ, Park E, Damsky WE, Vesely MD. Pembrolizumab-induced lichenoid dermatitis treated with dupilumab. JAAD Case Rep. 2023;37:13–5. https://doi.org/10.1016/j.jdcr.2023.05.004.
Alamon-Reig F, Bosch-Amate X, Giavedoni P, et al. Use of omalizumab is associated with improvement of pruritic skin disorders induced by immune checkpoint inhibitors: a retrospective cohort from the European Task Force of Dermatology for Cancer patients. J Am Acad Dermatol. 2024;90(2):429–32. https://doi.org/10.1016/j.jaad.2023.10.012.
Blaise M, Cardot-Leccia N, Seitz-Polski B, et al. Tocilizumab for corticosteroid-refractory immune checkpoint inhibitor-induced generalized morphea. JAMA Dermatol. 2023;159(1):112–4. https://doi.org/10.1001/jamadermatol.2022.5146].
Herrscher H, Tomasic G, Castro GA. Generalised morphea induced by pembrolizumab. Eur J Cancer. 2019;116:178–81. https://doi.org/10.1016/j.ejca.2019.05.018.
DeMaio A, Hashemi KB, Avery A, Metcalf JS, Winterfield LS. A case of nivolumab-induced scleroderma-like syndrome successfully treated with intravenous immunoglobulin. JAAD Case Rep. 2023;31:76–9. https://doi.org/10.1016/j.jdcr.2022.06.007.
Barbosa NS, Wetter DA, Wieland CN, Shenoy NK, Markovic SN, Thanarajasingam U. Scleroderma induced by pembrolizumab: a case series. Mayo Clin Proc. 2017;92(7):1158–63. https://doi.org/10.1016/j.mayocp.2017.03.016.
Shelton E, Doolittle C, Shinohara MM, Thompson JA, Moshiri AS. Can’t handle the itch? Refractory immunotherapy-related transient acantholytic dermatosis: prompt resolution with dupilumab. JAAD Case Rep. 2022;22:31–3. https://doi.org/10.1016/j.jdcr.2022.01.029.
Panou E, Stavridi F, Nikolaou C, Stratigos A, Nikolaou V. Dupilumab for the treatment of immune checkpoint blockers’ induced pruritus. J Eur Acad Dermatol Venereol. 2023;37(12):e1475–6. https://doi.org/10.1111/jdv.19369.
Utsunomiya A, Oyama N, Iino S, et al. A case of erythema multiforme major developed after sequential use of two immune checkpoint inhibitors, nivolumab and ipilimumab, for advanced melanoma: possible implication of synergistic and/or complementary immunomodulatory effects. Case Rep Dermatol. 2018;10(1):1–6. https://doi.org/10.1159/000485910.
Belzer A, Pach JJ, Valido K, Leventhal JS. The impact of dermatologic adverse events on the quality of life of oncology patients: a review of the literature. Am J Clin Dermatol. 2024. https://doi.org/10.1007/s40257-024-00847-2.
Schulz TU, Zierold S, Sachse MM, et al. Persistent immune-related adverse events after cessation of checkpoint inhibitor therapy: prevalence and impact on patients’ health-related quality of life. Eur J Cancer. 2022;176:88–99. https://doi.org/10.1016/j.ejca.2022.08.029.
Vigarios E, Epstein JB, Sibaud V. Oral mucosal changes induced by anticancer targeted therapies and immune checkpoint inhibitors. Support Care Cancer. 2017;25(5):1713–39. https://doi.org/10.1007/s00520-017-3629-4.
Phillips GS, Freites-Martinez A, Wu J, et al. Clinical characterization of immunotherapy-related pruritus among patients seen in 2 oncodermatology clinics. JAMA Dermatol. 2019;155(2):249–51. https://doi.org/10.1001/jamadermatol.2018.4560.
Kamata M, Tada Y. Safety of biologics in psoriasis. J Dermatol. 2018;45(3):279–86. https://doi.org/10.1111/1346-8138.14096.
Kamata M, Tada Y. Efficacy and safety of biologics for psoriasis and psoriatic arthritis and their impact on comorbidities: a literature review. Int J Mol Sci. 2020;21(5):1690. https://doi.org/10.3390/ijms21051690.
Davis JS, Ferreira D, Paige E, Gedye C, Boyle M. Infectious complications of biological and small molecule targeted immunomodulatory therapies. Clin Microbiol Rev. 2020;33(3):e00035-e119. https://doi.org/10.1128/CMR.00035-19.
Kang JH, Bluestone JA, Young A. Predicting and preventing immune checkpoint inhibitor toxicity: targeting cytokines. Trends Immunol. 2021;42(4):293–311. https://doi.org/10.1016/j.it.2021.02.006.
Shipman WD, Singh K, Cohen JM, Leventhal J, Damsky W, Tomayko MM. Immune checkpoint inhibitor-induced bullous pemphigoid is characterized by interleukin (IL)-4 and IL-13 expression and responds to dupilumab treatment. Br J Dermatol. 2023;189(3):339–41. https://doi.org/10.1093/bjd/ljad149.
Shi J, Song X, Traub B, Luxenhofer M, Kornmann M. Involvement of IL-4, IL-13 and their receptors in pancreatic cancer. Int J Mol Sci. 2021. https://doi.org/10.3390/ijms22062998.
Verheijden RJ, May AM, Blank CU, et al. Association of anti-TNF with decreased survival in steroid refractory ipilimumab and anti-PD1-treated patients in the Dutch Melanoma Treatment Registry. Clin Cancer Res. 2020;26(9):2268–74. https://doi.org/10.1158/1078-0432.CCR-19-3322.
Fattore D, Annunziata MC, Panariello L, Marasca C, Fabbrocini G. Successful treatment of psoriasis induced by immune checkpoint inhibitors with apremilast. Eur J Cancer. 2019;110:107–9. https://doi.org/10.1016/j.ejca.2019.01.010.
Apalla Z, Psarakis E, Lallas A, Koukouthaki A, Fassas A, Smaragdi M. Psoriasis in patients with active lung cancer: is apremilast a safe option? Dermatol Pract Concept. 2019;9(4):300–1. https://doi.org/10.5826/dpc.0904a11.
Gresham LM, Kirchhof MG. A case of drug-induced bullous pemphigoid secondary to immunotherapy treated with upadacitinib: a case report. SAGE Open Med Case Rep. 2023. https://doi.org/10.1177/2050313X231160926.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the preparation of this article.
Conflict of interest
Jolanta Pach, Kailyn Valido, Annika Belzer, and Jonathan S. Leventhal have no conflicts of interest that are directly relevant to the content of this article. Jonathan S. Leventhal serves on the advisory boards of La Roche-Posay and Sanofi and Regeneron Pharmaceuticals and receives clinical trial funding from Azitra, Inc. and OnQuality.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
No datasets were generated or analyzed during the current study.
Code availability
Not applicable.
Author contributions
All authors contributed to the study conception and design. Material preparation and the literature review were performed by JP, KV, and AB. The first draft of the manuscript was written by JP, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pach, J., Valido, K., Belzer, A. et al. The Use of Biologic Agents for the Treatment of Cutaneous Immune-Related Adverse Events from Immune Checkpoint Inhibitors: A Review of Reported Cases. Am J Clin Dermatol (2024). https://doi.org/10.1007/s40257-024-00866-z
Accepted:
Published:
DOI: https://doi.org/10.1007/s40257-024-00866-z