Abstract
Disseminated granuloma annulare (DGA) is an inflammatory skin disorder characterized by more than 10 erythematous, raised, ring-shaped plaques. Its treatment remains challenging, with conventional therapies showing variable efficacy. We report the case of a woman in her 50s with a 2-year history of DGA refractory to multiple treatments. Given the recent evidence of the role of the Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway in the granuloma annulare pathophysiology, treatment with upadacitinib 30 mg per day was started with rapid effectiveness and good tolerance. This case underscores the potential of JAK inhibitors as promising therapeutic options for recalcitrant granuloma annulare.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
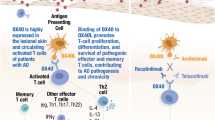
The role of the Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway in the pathophysiology of granulomatous skin diseases pathophysiology has recently been reported. |
Treatment with upadacitinib 30 mg per day was rapidly effective and well tolerated in a female patient in her 50s, with recalcitrant disseminated granuloma annulare. |
Safety profile was reassuring; induced lipid changes were easily managed. |
Introduction
Granuloma annulare (GA) is an inflammatory skin disease characterized by erythematous, raised, ring-shaped plaques, which may be localized or disseminated (DGA) when defined by at least 10 plaques [1]. Association with diseases such as diabetes, hyperlipidemia, autoimmune thyroiditis, or hepatitis C remain uncertain [2]. Genetic predisposition (e.g., HLA-B35) has been observed in rare cases of familial occurrences of GA [3]. Treatment can be difficult and challenging, particularly in diffuse and generalized forms. Various conventional treatments (systemic corticosteroids, phototherapy, antimalarials, dapsone, etc.) have been proposed on the basis of case reports or clinical experience, with variable effectiveness [4]. However, to date, there is no evidence-based treatment. The potential effect of biological agents on DGA is still unclear; remission and induction of GA have been reported with tumor necrosis factor alpha (TNFα) inhibitors or anti-interleukin (IL)-17 [5].
Case
We report the case of a woman in her 50s who presented to our dermatology consultation with a 2-year history of DGA. The patient was otherwise healthy, her past medical history included livedoid vasculopathy (currently in remission). Initial diagnosis of DGA had been confirmed by skin biopsy and previous treatments—including topical corticosteroids, hydroxychloroquine (200–400 mg/day for 1.5 years), doxycycline (200 mg/day for 2 months), methotrexate (12.5 mg/week for 2 months), and vitamin E—had all yielded poor response. The patient reported no systemic symptoms. Clinical examination revealed numerous large and extended ring-shaped plaques on both legs, upper limbs, hands, and feet (Fig. 1). Laboratory workup (including C-reactive protein, complete blood count, renal and hepatic functions, lactate dehydrogenase, fasting plasma glucose, hepatitis, and HIV serologies) was normal. Given the recalcitrant and extensive nature of the lesions, and the recent evidence of the role of the Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway in the GA pathophysiology [6], treatment with upadacitinib 30 mg per day was started (the patient provided written informed consent after discussion of the risk–benefit balance and possible adverse effects). At follow-up visit 2 months later, lesions had almost all disappeared (Fig. 2A, B) and complete remission was achieved after 6 months (Fig. 2C, D). Upadacitinib was well tolerated; an increase of total cholesterol (233 mg/dL; normal value < 190 mg/dL) and LDL-cholesterol (135 mg/dL; normal value < 115 mg/dL) was controlled with atorvastatin 20 mg daily. Treatment was stopped after a total of 10 months, with no disease recurrence at the 2-month follow-up visit.
Discussion
Evidence of the involvement of T helper (Th)1/Th2 cytokines and innate immunity (IL-1β, IL-6, TNFα) in the activation of the JAK-STAT pathway and thereby, in GA pathogenesis, has opened the way to new therapeutic options, especially for recalcitrant cases [6, 7]. Few cases of patients with DGA treated orally with tofacitinib (oral JAK1/3 inhibitor) [8], baricitinib (oral JAK1/2 inhibitor) [9, 10], and only two patients with upadacitinib (oral JAK1 inhibitor) have been reported [11, 12]. In the last two cases, a 15 mg daily dosage of upadacitinib led to an almost complete regression of the disease respectively after 1 month [11] and 4 months [12]. No information about safety/tolerance was provided. Despite having treated our patient with a higher dosage of 30 mg daily, the treatment was well tolerated inducing only easily controlled lipid changes. Although our clinical observation is limited to a single clinical case, it contributes to the growing literature in this area. The close therapeutic responses between our case and the two previous reported cases suggest that the use of the 15 mg dose could be just as effective, with fewer risks of side effects.
Conclusion
This observation, in line with other rare cases reported in the literature, is very encouraging for the rapid effectiveness and good tolerance of JAK inhibitors, and in particular upadacitinib, in the treatment of recalcitrant DGA. Larger cohort studies or clinical trials are warranted to confirm these findings.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Thornsberry LA, English JC 3rd. Etiology, diagnosis, and therapeutic management of granuloma annulare: an update. Am J Clin Dermatol. 2013;14(4):279–90. https://doi.org/10.1007/s40257-013-0029-5.
Fornons-Servent R, Bauer-Alonso A, Llobera-Ris C, Penín RM, Marcoval J. Granuloma annulare: a case–control study of possible associated diseases. Dermatol Pract Concept. 2022;12(4): e2022173. https://doi.org/10.5826/dpc.1204a173.
Rankin BD, Haber RM. Familial granuloma annulare: first report of occurrence in a father and daughter and updated review of the literature. JAAD Case Rep. 2021;17:61–4. https://doi.org/10.1016/j.jdcr.2021.09.023.
Lukács J, Schliemann S, Elsner P. Treatment of generalized granuloma annulare—a systematic review. J Eur Acad Dermatol Venereol. 2015;29(8):1467–80. https://doi.org/10.1111/jdv.12976.
Dopytalska K, Gabzdyl N, Szczerba M, Szymańska E, Walecka I. Is biologic therapy the future of granuloma annulare treatment? Dermatol Ther. 2022;35(1):e15188. https://doi.org/10.1111/dth.15188.
Min MS, Wu J, He H, et al. Granuloma annulare skin profile shows activation of T-helper cell type 1, T-helper cell type 2, and Janus kinase pathways. J Am Acad Dermatol. 2020;83(1):63–70. https://doi.org/10.1016/j.jaad.2019.12.028.
O’Reilly S, Ciechomska M, Cant R, Hügle T, van Laar JM. Interleukin-6, its role in fibrosing conditions. Cytokine Growth Factor Rev. 2012;23(3):99–107. https://doi.org/10.1016/j.cytogfr.2012.04.003.
Bosch-Amate X, Serra-García L, Alamon-Reig F, et al. Treatment of granuloma annulare with tofacitinib. Austral J Dermatol. 2022;63(3):400–3. https://doi.org/10.1111/ajd.13875.
Kim D, Kang HY. Rapid improvement of refractory generalized granuloma annulare with the Janus kinase inhibitor baricitinib in two patients. Clin Exp Dermatol. 2023;48(4):375–6. https://doi.org/10.1093/ced/llac110.
Ballová A, Kmečová Z, Péč J, Vorčáková K. Recalcitrant granuloma annulare induced by two different biologic agents resolved after Janus kinase inhibitor treatment. Dermatol Ther. 2022;35(8):e15641. https://doi.org/10.1111/dth.15641.
Slater KN, Valk B, Kartono F. A case of generalized granuloma annulare treated with upadacitinib. JAAD Case Rep. 2023;34:12–4. https://doi.org/10.1016/j.jdcr.2023.01.027.
Sondermann W, Hadaschik E, Specker C. Successful therapy of disseminated patch-type granuloma annulare with upadacitinib in a patient with rheumatoid arthritis. Dermatol Ther. 2022;35(1):e15211. https://doi.org/10.1111/dth.15211.
Acknowledgements
We thank the patient for participating in this study.
Medical Writing/Editorial Assistance.
We thank Dr. Mariana Andrade, M.D., (Andrade-Evrard SRL) who provided editorial assistance. Fees were funded by the authors.
Funding
No funding or sponsorship was received for this case report or publication of this article. The Rapid Service Fee was funded by the authors.
Author information
Authors and Affiliations
Contributions
Axel De Greef: Conceptualization, Methodology, Validation, Formal analysis, Investigation, Resources, Data Curation, Writing—Original Draft, Visualization; Ghita Benjelloun; Conceptualization, Methodology, Validation, Formal analysis, Investigation, Resources, Data Curation, Writing—Original Draft, Visualization; Evelyne Harkemanne: Conceptualization, Investigation; Marie Baeck: Conceptualization, Methodology, Validation, Formal analysis, Investigation, Resources, Data Curation, Writing—Review & Editing, Supervision.
Corresponding author
Ethics declarations
Conflict of Interest
Marie Baeck discloses her past participation on the upadacitinib advisory boards organized by AbbVie. Axel De Greef and Marie Baeck have previously participated as speakers in events sponsored by AbbVie. Ghita Benjelloun and Evelyne Harkemanne disclose no conflict of interest. All the authors declare that the current study was conducted in an independent manner.
Ethics Approval
This case report and data collection were conducted with the approval of the hospital and faculty institutional review board (Commission d’Ethique Biomédicale Hospitalo-Facultaire) of Université catholique de Louvain (UCLouvain), Belgium. All procedures in this study were in accordance with the 1964 Helsinki declaration (and its amendments). The patient in this manuscript has given written informed consent to participate and for publication of her case details.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
De Greef, A., Benjelloun, G., Harkemanne, E. et al. Successful Treatment of Disseminated Granuloma Annulare with Upadacitinib. Dermatol Ther (Heidelb) 14, 813–817 (2024). https://doi.org/10.1007/s13555-024-01117-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-024-01117-z