Abstract
Dye-sensitized solar cells (DSSC) were fabricated using red bougainvilleaglabra flower dye extracts as natural dye sensitizers at three dye pH values of 1.23, 3.0, and 5.7. Water was used as dye-extracting solvent. DSSCs from dye extract of pH 3.0 had the highest photocurrent density Jsc of 3.72 mA/cm2 and fill factor FF of 0.59. While the DSSCs from dye sensitizer pHs of 1.23 and 5.7 had Jsc of 1.13 and 2.27 mA/cm2, and fill factors of 0.43 and 0.61, respectively. The maximum powers Pmax of the DSSCs were 0.50, 1.64, and 0.94 mW/cm2 for dye sensitizer pH of 1.2, 3.0, and 5.7, respectively.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dye-sensitized solar cell (DSSC) developed by Gratzel et al. [1] has attracted considerable attention due to its environmental friendliness and relative low cost of production [2]. Sensitizer dye in DSSC plays a key role in harvesting sunlight and transforming solar energy into electric energy and thus of paramount importance to photovoltaic cell performance and efficiency [3]. It attaches to the surface of a wide band-gap mesoporous semiconductor serving as electron transporter [4]. Ruthenium-based dye sensitizers are very expensive and hard to prepare, which restricts their large-scale applications in solar cells, stimulating the search for alternatives such as natural dyes.
Anthocyanins from natural pigments and carotenoids have been used as natural dye sensitizers in DSSC and have shown low to modest solar energy conversion efficiencies [2, 5–7]. These natural dyes are easy to prepare, cheap, non-toxic, environmentally friendly, and easily biodegradable.
Betalain is another interesting class of pigments, whose purified extracts from commercial sources have been subjected to photoelectrochemical study [8]. Betalains consisting of the yellow betaxanthins and red–violet betacyanins are a group of water-soluble nitrogen-containing alkaloid pigments characteristic of certain members of plant sub-order chenopodineae within caryophyllales and some higher fungi [9]. They absorb visible radiation over the range 476–600 nm, they are immonium derivatives of betalamic acid (the chromophore of all betalains) and divided into two structural groups: the red–violet betacyanins that have a maximum absorptivity at λ ≈ 535 nm and the yellow–orange betaxanthins with maximum absorptivity at λ ≈ 480 nm [10–13]. Betalains are characterized by high molar extinction coefficients in the visible region and pH-dependent redox properties [14, 15]. They are relatively stable over pH range 3–7, however, at pH less than 3.5 the absorption maximum shifts toward lower wavelength [16]. Acidic conditions are known to induce recondensation of betalamic acid with amine group of the addition residue [17] and favors betalain-sensitized photo-electrodes with high optical densities capable of complete absorption in the visible range of 400–600 nm [6]. Solar energy conversion efficiency being a function of photocurrent density Jsc, open-circuit voltage Voc, and fill factor FF [18], suggest their improvement is essential to raising conversion efficiency.
This work reports the use of water extracts of red Bougainvillea glabra flower a source of betalain as natural dye sensitizer and the influence of sensitizer dye pH on the photoelectric parameters of the DSSCs.
Bougainvillea is a member of the Nyctaginaceae family comprising of 18 shrubby species which though indigenous of South America are distributed widely in the world, with flowers ranging from white, yellow, orange, and various shades of red to purple and violet color [12–14]. The Betalain pigment in Bougainvillea is different from that in other sources due to the presence of saccharide type present in the betanidin (an aglycone of most betacyanins) which is produced 90 % from betacyanins, and the remaining 10 % comes from indicaxanthin a common betaxanthin [15]. Bougainvillea glabra has been found to have eleven violet–red pigments having maximum absorbance in wavelength range of 522–551 nm and is known to have two epimeric betacyanins; bougainvillein-r-1 and isobougainvillein-r-1 [15].
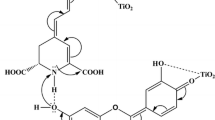
The general structure of Betalain is shown in Fig. 1 [10]; it contains carboxylic functional group which facilitates TiO2 surface binding.
Experimental
The red Bougainvillea glabra flower dye pigments were extracted by crushing 20 g of the flower in a porcelain mortar with a pestle and adding 50 ml of water to the crushed sample. This was filtered to get the raw dye extracts of the flower which was divided into three and used as sensitizing dye at three different pHs. The first, the as-extracted dye had a pH of 5.70, the pH of the second was adjusted to 3.0 by adding 4 drops of 20 % HCl to it, and the third after adding 8 drop of HCl had a pH value 1.23.
The transparent conducting oxide was fluorine-doped tin oxide (FTO) with a sheet resistance of 15 Ω/cm2 (SOLARONIX). The TiO2 film was prepared by blending commercial TiO2 powder (Degussa, P25) of Ca. 0.2 g, nitric solution (0.1 M) of 0.4 ml, polyethylene glycol (MW 10,0000) of ca. 0.08 g, and one drop of a non-ionic surfactant, Triton x-100. The mixture was well blended using an ultrasonic bath for 1 h. Squeegee was used to screen-print the resulting TiO2 paste on the conducting layer/FTO glass substrate. It was left for 30 min so that the paste could settle to reduce the irregularity of the surface. The substrates were then left to dry. This screen-printing procedure was repeated to obtain the TiO2 working electrode of appropriate thickness of 9 µm.
Prior to sensitisation, the TiO2 photo-anodes were pre-heated at 150 °C for 30 min, allowed to cool. It was then subjected to TiCl4 treatment by immersing in 40 mM of TiCl4 solution for 30 min at a temperature of 70 °C and subsequently sintered at 450 °C for 45 min. After cooling to 80 °C, the TiO2 electrodes were immersed into the three dye solutions of pH 1.23, 3.0, and 5.7 for 16 h. The photo-electrode were then rinsed with 99 % ethanol solution, to remove excess dye and thereafter left to dry. The counter electrode was made from platinum catalyst T/SP product by SOLARONIX which was screen-printed using a polyester mesh of 90 and dried. The substrates were sintered for 1 h at 45 °C. The DSSC was assembled from the various components following the procedure reported elsewhere [19].
Characterization
The absorption spectra of the dyes were determined using an AVASPEC 2048 UV–Visible spectrophotometer, while the morphology and composition characterization of the nanoporous TiO2 film was carried out using Carl Zeiss EVO MA-10 scanning electron microscope (SEM), with EDX model ISIS 300 Oxford. Simulated Solar irradiation was provided by a Solar Simulator, Model 4200-SCS Semiconductor Characterization System under the irradiation of AM 1.5 (100 mWcm−2), and the Current–Voltage curves were recorded by a digital Keithley multimeter Model 2400 coupled to a computer.
Results and discussion
Figure 2 shows the UV–Vis optical absorption spectra of the bougainvillea flower dye extracts at three different pHs. Absorption spectra of a dye reflect optical transition probability between the ground state, the excited state and the solar energy range absorbed by the dye. The three dyes’ extracts show absorption peaks centered between 310 and 340 nm in the UV-range, with maximum peaks at 330 nm for pH 5.7 and 327 nm for pH 3.0 and 1.2. The dye extracts at pH 1.23 and 3.0 have about the same absorption intensity in short wavelength range, with dye extract at a pH of 5.7 having the highest intensity in this range. The lowest absorption intensity in the long wavelength is observed for dye pH of 1.2, indicating a highly suppressed absorbance as a result of degradation of betanin in very strong acidic environment [20]. Dye extract at a pH of 3 displays broad absorption peak in the 480–560 nm range resulting from π − π* transitions due to the mixed contributions of the yellow–orange betaxanthins (480 nm) and of the red–purple betacyanin (540 nm) [6]. At a pH of 5.7, the extract has a highest but flattened absorption spectrum in the long-range wavelength indicating a suppressed absorption peak at this pH and a wider range of red, orange, yellow, and blue light can be absorbed. Absorbance peaks around 300 and 535 nm are characteristic absorptions for red–violet betalain group, betacyanin [14].
The betaxanthin and betanin concentration in the dye extracts in µM are estimated from the absorbance A at 482 and 536 nm from the absorbance spectra using [10],
[Bbetax] is the betaxanthin concentration and [Bbetan] the betanin concentration. The betaxanthin concentrations were 7.39, 8.79, and 15.17 µM for pH of 1.23, 3.0, and 5.7, respectively, while the betanin concentration at a pH of 1.2, 3.0 and 5.7 were 4.35, 8.15, and 11.26 µM, respectively.
Figure 3 shows the scanning electron micrograph of the TiO2 (anatase) film. The TiO2 film has thickness of 9 µm and a mean particle size of 20 nm. It shows a mesoporous surface of the spherical nanoparticles of TiO2 that forms nanopores across the surface. The EDX analysis of the TiO2 electrode in Fig. 4 shows the presence of Titanium, Oxygen, Nitrogen, and Carbon. The presence of chlorine in the compound was due to TiCl4 which was applied at a final treatment of the porous TiO2 electrodes in the anatase TiO2 compound contained.
The current–voltage curve of a solar cell yields important photoelectric parameters such as short-circuit current density Jsc, the open-circuit voltage Voc, the current Imp, and voltage Vmp at the maximum power point Pmax. The photoelectric characteristics of the DSSCs were inspected under the simulated sunlight source (AM1.5).
The combined J–V and P–V curves of the DSSCs at different pHs are shown in Fig. 5. The open-circuit voltage Voc of 0.44 V is obtained for dye pH of 1.2 and 3.0, respectively, while at a pH of 5.7 the Voc is 0.41. The DSSC prepared from a dye pH of 3.0 has the highest photocurrent density Jsc of 3.72 mA/cm2 and fill factor of and 0.59. At a dye sensitizer pH of 1.2, the DSSC has the lowest Photocurrent density of 1.13 mA/cm2 and lowest fill factor of 0.43 due to betanin degradation in very strong acidic environment [17, 20] with resultant inefficient light harvesting by the dye and inefficient charge injection into the TiO2 nanoparticles; also possible cell deterioration by acid leaching is expected as the pH goes lower.
The reduction in photoelectrical property observed at pH 5.7 compared to pH 3 results because at high pH betanin is poorly absorbed onto titania due to its inhibition by indicaxanthin (a common betaxanthin in red bougainvillea) competing with betanin for adsorption sites, but at lower pH protonation of the betalainic carboxylic acid group from its anionic form occurs promoting its adsorption onto the TiO2 [14].
The photoelectric parameters are listed in Table 1.
The obtained photoelectric parameters using bougainvilla glabra flower dye extract as sensitizer dye at pHs of 1.2, 3.0, and 5.7 are comparable to Jsc of 1.881–2.344 mA/cm2 though with lower Voc of 0.23–0.26 V from betalain pigments extracted from bracts of bougainvilla glabra and bougainvillaspectabilis at a pH of 5.7 [14].
Also although Jsc of up to 9.5 mA/cm2 have been reported at a dye pH of 1.0 from other sources of betalain, corresponding Voc was just between 0.23–0.3 V [6]. The obtained photoelectric parameters at dye pHs of 1.23 and 5.7 are much lower than that of black dye. The black dye has a certified Jsc of 20.5 mA/cm2 and a Voc of 0.72 V and is superior to all charge-transfer sensitizers on the basis of photovoltaic performance under AM 1.5 [21]. The Jsc 3.72 mA/cm2 at dye sensitizer pH of 3.0 is encouraging.
Conclusion
Betalain raw extracts of red bougainvilleaglabra flower were used as natural dye sensitizers for DSSCs at different dye sensitizer pH values. At a pH of 1.23, the DSSC had the lowest parameters as a result of betalain degradation in very acidic environment and leaching of the dye from the TiO2 surface. The DSSC from dye sensitizer pH of 3.0 offered the highest Jsc of 3.72 mA/cm2, a FF of 0.59, and highest Pmax of 1.64 mW/cm2. The poor absorption of betanin onto the titania at high pH lead to decreased photoelectrical parameter of the cell at pH of 5.7. The obtained photoelectric parameters are much lower than that of black dye which is superior to all charge-transfer sensitizers on the basis of photovoltaic performance under AM 1.5 presently, with a certified Jsc of 20.5 mA/cm2 and a Voc of 0.72 V [21]. Nevertheless, natural bougainvilleaglabra flower betalain raw extracts present promising low-cost, non-toxic, and environmentally friendly alternative photosensitizers in DSSCs.
References
O’Regan, B., Grätzel, M.: A low cost high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 353, 737–740 (1991)
Zhou, H., Wu, L., Gao, Y., Ma, T.: Dye-sensitized solar cells using 20 natural dyes as sensitizers. J. Photochem. Photobiol. A Chem 219, 188–194 (2011)
Chang, H., Wu, H.M., Chen, T.L., Huang, K.D., Jwo, C.S., Lo, Y.J.: Dye-sensitized solar cell using natural dyes extracted from spinach and ipomoea. J. Alloys Compd. 495(2), 606–610 (2010)
Erten-Ela, S.: Photovoltaic effect of nanostructured TiO2 layers in dye sensitized solar cells. J. Optoelectron. Adv. Mater. 14(9–10), 758–762 (2012)
Dai, Q., Rabani, J.: Photosensitization of nanocrystalline TiO2 films by anthocyanin dyes. J. Photochem. Photobiol. A Chem. 148, 17–24 (2002)
Calogero, G., Di Marco, G., Cazzanti, S., Caramori, S., Argazzi, R., Di Carlo, A., Alberto Bignozzi, C.: Efficient dye-sensitized solar cells using red turnip and purple wild Sicilian prickly pear fruits. Inter. J. Mol. Sci. 11, 254–267 (2010)
Gao, F.G., Bard, A.J., Kispert, L.D.: Photocurrent generated on a carotenoid-sensitized TiO2 nanocrystalline mesoporous electrode. J. Photochem. Photobiol. A Chem. 130, 49–56 (2000)
Zhang, D., Lanier, S.M., Downing, J.A., Avent, J.L., Lum, J., McHale, J.L.: Betalain, pigments for dye-sensitized solar cells. J. Photochem. Photobiol. A Chem. 195, 72–80 (2008)
Koboyashi, N., Schmidt, J., Wray, V., Schliemann, W.: Formation and occurrence of dopamine-derived betacyanins. Phytochem. 56, 429–436 (2001)
Strack, D., Vogt, T., Schliemann, W.: Recent advances in betalain research. Phytochem. 62, 247–269 (2003)
Azeredo, H.M.C.: Betalains, properties, sources, applications, and stability—a review. Inter. J. Food Sci. Technol. 44, 2365–2376 (2009)
Mabberley, D.F.: A Portable Dictionary of Vascular Plants. Cambridge University Press, UK (1997)
Impellizzeri, G., Piattelli, M., Sciuto, S.: Acylated betacyanins from Drosanthemumfloribundum. Phytochem. 12, 2295–2296 (1973)
Hernández-Martínez, A. R.,, Vargas S., Estevez, M., Rodríguez, R.: Dye-sensitized solar cells from extracted bracts Bougainvillea betalain pigments. In: 1st International Congress on Instrumentation and Applied Sciences, Cancun, Q. R., Mexico, 26–29 Oct 2010
Piattelli, M., Imperato, F.: Pigments of Bougainvillea glabra. Phytochem. 9, 2557–2560 (1970)
Jackman, R.L., Smith, J.L.: Anthocyanins and betalains. In: Hendry, G.F., Houghton, J.D. (eds.) Natural Food Colourants, pp. 244–309. Blackie Academic & Professional, London (1996)
Schwartz, S.J., von Elbe, J.H.: Identification of betanin degradation products. Eur. Food Res. Technol. 176, 448–453 (1983)
Sze, S.M., Lee, M.: Physics of Semiconductor Devices. Wiley, New York (1969)
Kimpa, M.I., Momoh, M., Isah, K.U., Yahya, H.N., Ndamitso, M.M.: Photoelectric characterization of dye sensitized solar cells using natural dye from pawpaw leaf and flame tree flower as sensitizers. Mater. Sci. Appl. 3, 281–286 (2012)
Pedreno, M.A., Escribano, J.: Correlation between antiradical activity and stability of betanine from Beta vulgaris L roots under different pH, temperature and light conditions. J. Sci. Food Agric. 81, 627–631 (2001)
Qin, Y., Peng, Q.: Ruthenium sensitizers and their applications in dye-sensitized solar cells. Inter. J. Photoenerg. 2012, 1–21 (2012)
Acknowledgments
This study was supported by grant from the University Board of Research (UBR) of the Federal University of Technology, Minna, Nigeria.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under license to BioMed Central Ltd. Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Isah, K.U., Ahmadu, U., Idris, A. et al. Betalain pigments as natural photosensitizers for dye-sensitized solar cells: the effect of dye pH on the photoelectric parameters. Mater Renew Sustain Energy 4, 39 (2015). https://doi.org/10.1007/s40243-014-0039-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40243-014-0039-0