Abstract
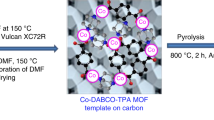
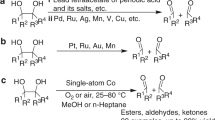
Reductive amination of aldehydes/ketones with aqueous NH3 and hydrogenation of nitriles to primary amines with Co catalysts were reported. Co@NC-700 exhibited remarkable activity and high selectivity for the reductive amination of aldehydes/ketones with aqueous NH3 and the hydrogenation of nitriles to primary amines. Several primary amines can be obtained with good to excellent yields via the reductive amination of aldehydes/ketones and the hydrogenation of nitriles. The nitrogen-doped carbon(C)-supported Co@NC-700 metal catalyst was prepared via the pyrolysis of bioMOF Co/adenine in activated C. Co@NC-700 can be reused five times without evident loss of activity.
Similar content being viewed by others
References
Artiukha E. A., Nuzhdin A. L., Bukhtiyarova G. A., Zaytsev S. Y., Plyusnin P. E., Shubin Y. V., Bukhtiyarov V. I., Catal. Sci. Technol., 2015, 5, 4741
Delidovich I., Hausoul P. J. C., Deng L., Pfützenreuter R., Rose M., Palkovits R., Chem. Rev., 2016, 116, 1540
Jagadeesh R. V., Murugesan K., Alshammari A. S., Neumann H., Pohl M. M., Radnik J., Beller M., Science, 2017, 358, 326
Tokmic K., Jackson B. J., Salazar A., Woods T. J., Fout A. R., J. Am. Chem. Soc., 2017, 139, 13554
Balaraman E., Srimani D., Diskin-Posner Y., Milstein D., Catal. Lett., 2015, 145, 139
Imm S., Bahn S., Zhang M., Neubert L., Neumann H., Klasovsky F., Pfeffer J., Haas T., Beller M., Angew. Chem. Int. Ed., 2011, 50, 7599
Muller T. E., Hultzsch K. C., Yus M., Foubelo F., Tada M., Chem. Rev., 2008, 108, 3795
Ahmed M., Seayad A. M., Jackstell R., Beller M., J. Am. Chem. Soc., 2003, 125, 10311
Buchwald S. L., Mauger C., Mignani G., Scholz U., Adv. Synth. Catal., 2006, 348, 23
Mukherjee A., Srimani D., Ben-David Y., Milstein D., ChemCatChem., 2017, 9, 559
Chusov D., List B., Angew. Chem. Int. Ed., 2014, 53, 5199
Zhang X. H., Ye W. J., Wang K. L., Tian Y. S., Xiao X., Chem. Res. Chinese Universities, 2015, 31(2), 203
Gross T., Seayad A. M., Ahmad M., Beller M., Org. Lett., 2002, 4, 2055
Gallardo-Donaire J., Ernst M., Trapp O., Schaub T., Adv. Synth. Catal., 2016, 358, 358
Pisiewicz S., Stemmler T., Surkus A. E., Junge K., Beller M., ChemCatChem., 2015, 7, 62
Ferraccioli R., Borovika D., Surkus A. E., Kreyenschulte C., Topf C., Beller M., Catal. Sci. Technol., 2018, 8, 499
Komanoya T., Kinemura T., Kita Y., Kamata K., Hara M., J. Am. Chem. Soc., 2017, 139, 11493
Klinkenberg J. L., Hartwig J. F., Angew. Chem., Int. Ed., 2011, 50, 86
Ramachandran P. V., Gagare P. D., Sakavuyi K., Clark P., Tetrahedron Lett., 2010, 51, 3167
Kadyrov R., Riermeier T. H., Angew. Chem. Int. Ed., 2003, 42, 5472
Talwar D., Salguero N. P., Robertson C. M., Xiao J., Chem. Eur. J., 2014, 20, 245
Yang H., Cui X., Deng Y., Shi F., Synth. Commun., 2014, 44, 1314
Ogo S., Uehara K., Abura T., Fukuzumi S., J. Am. Chem. Soc., 2004, 126, 3020
Liang G., Wang A., Li L., Xu G., Yan N., Zhang T., Angew. Chem. Int. Ed., 2017, 56, 3050
Alinezhad H., Yavari H., Salehian F., Curr. Org. Chem., 2015, 19, 1021
Gomez S., Peters J. A., Maschmeyer T., Adv. Synth. Catal., 2002, 344, 1037
Long J. L., Shen K., Li Y. W., ACS Catal., 2017, 7, 2754
Yano J. K., Denton T. T., Cerny M. A., Zhang X., Johnson E. F., Cashman J. R., J. Med. Chem., 2006, 49, 6987
Kise N., Ueda N., Tetrahedron Lett., 2001, 42, 2365
Adam R., Bheeter C. B., Jackstell R., Beller M., ChemCatChem., 2016, 8, 1329
Yasukawa T., Yang X., Kobayashi S., Org. Lett., 2018, 20, 5172
Lin X., Nie Z. Z., Zhang L. Y., Mei S. C., Chen Y., Zhang B. S., Zhu R. L., Liu Z. G., Green Chem., 2017, 19, 2164
Aijaz A., Masa J., Rosler C., Xia W., Weide P., Botz A. J. R., Fischer R. A., Schuhmann W., Muhler M., Angew. Chem. Int. Ed., 2016, 55, 4087
Murugesan K., Senthamarai T., Sohail M., Alshammari A. S., Pohl M. M., Beller M., Jagadeesh R. V., Chem. Sci., 2018, 9, 8553
Huang L., Wang Z., Geng L. F., Chen R. Z., Xing W. H., Wang Y., Huang J., RSC Adv., 2015, 5, 56936
Mao F., Sui D. J., Qi Z. L., Fan H. P., Chen R. Z., Huang J., RSC Adv., 2016, 6, 94068
Sui D. J., Mao F., Fan H. P., Qi Z. L., Huang J., Chinese J. Chem., 2017, 35, 1371
An J., Geib S. J., Rosi N. L., J. Am. Chem. Soc., 2010, 132, 38
Zhong W., Liu H. L., Bai C. H., Liao S. J., Li Y. W., ACS Catal., 2015, 5, 1850
Jaouen F., Herranz J., Lefevre M., Dodelet J. P., Kramm U. I., Herrmann I., Bogdanoff P., Maruyama J., Nagaoka T., Garsuch A., Dahn J. R., Olson T., Pylypenko S., Atanassov P., Ustinov E. A., ACS Appl. Mater. Interfaces, 2009, 1, 1623
Di M. X., Xiao G. Z., Huang P., Cao Y. H., Zhu Y., Chem. J. Chinese Universities, 2018, 39(2), 343
Ferraccioli R., Borovika D., Surkus A. E., Kreyenschulte C., Topf C., Beller M., Catal. Sci. Technol., 2018, 8, 499
Ji P. F., Manna K., Lin Z. K., Feng X. Y., Urban A., Song Y., Lin W. B., J. Am. Chem. Soc., 2017, 139, 7004
Muratsugu S., Kityakarn S., Wang F., Ishiguro N., Kamachi T., Yoshizawa K., Tada M., Phys. Chem. Chem. Phys., 2015, 17, 24791
Rezaei S. J. T., Khorramabadi H., Hesami A., Ramazani A., Amani R., Ahmadi R., Ind. Eng. Chem. Res., 2017, 56, 12256
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(No.21676140) and the Fund of the State Key Laboratory of Materials-oriented Chemical Engineering, China(No.ZK201711).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Liu, J., Guo, W., Sun, H. et al. Reductive Amination of Carbonyl Compounds with Ammonia and Hydrogenation of Nitriles to Primary Amines with Heterogeneous Cobalt Catalysts. Chem. Res. Chin. Univ. 35, 457–462 (2019). https://doi.org/10.1007/s40242-019-8390-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-019-8390-4