Abstract
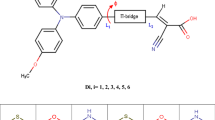
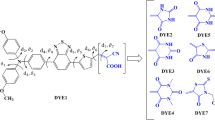
With density functional theory(DFT) method, the optimization of molecular configurations and the calculation of frontier molecular orbitals were achieved for triphenylamine(TPA)-based dye-sensitized solar cell materials at the B3LYP/6-31G(d, p) level. Time-dependent density functional theory(TD-DFT) was applied to calculating the probability of the transition from the ground state to the excited state. And UV-Vis absorption spectra were derived with Franck-Condon approximation. The conjugation length, substitution groups and spatial effects show a slight influence on the dihedral angle of the TPA group. The increase of conjugation length may cause a smaller energy gap as well as a higher highest occupied molecular orbital(HOMO) and a lower lowest unoccupied molecular orbital (LUMO). The introduction of methoxyl group and TPA group could lower the energy gap while the HOMO and LUMO were elevated in energy.
Similar content being viewed by others
References
Hagfeldt A., Boschloo G., Sun L. C., Kloo L., Pettersson H., Chem. Rev., 2010, 110, 6595
Tian H. N., Yang X. C., Pan J. X., Chen R. K., Liu M., Zhang Q. Y., Hagfeldt A., Sun L. C., Adv. Funct. Mater., 2008, 18, 3461
Choi H., Baik C., Kang S. O., Ko J., Kang M. S., Nazeeruddin M. K., Grätzel M., Angew. Chem. Int. Ed., 2008, 47, 327
Qin H., Wenger S., Xu M., Gao F., Jing X., Wang P., Zakeeruddin S. M., Grätzel M., J. Am. Chem. Soc., 2008, 130, 9202
Wang M. K., Xu M. F., Shi D., Li R. Z., Gao F. F., Zhang G. L., Yi Z. H., Humphry-Baker R., Wang P., Akeeruddin S. M., Grätzel M., Adv. Mater., 2008, 20, 4460
Hagberg D. P., Yum J. H., Lee H., de Angelis F., Marinado T., Karlsson K. M., Humphry-Baker R., Sun L. C., Hagfeldt A., Grätzel M., Nazeeruddin M. K., J. Am. Chem. Soc., 2008, 130, 6259
Yum J. H., Hagberg D. P., Moon S. J., Karlsson K. M., Marinado T., Sun L. C., Hagfeldt A., Nazeeruddin M. K., Grätzel M., Angew. Chem. Int. Ed., 2009, 48, 1576
Kim S., Lee J. K., Kang S. O., Ko J., Yum J. H., Fantacci S., De A. F., Di C. D., Nazeeruddin M. K., Grätzel M., J. Am. Chem. Soc., 2006, 128, 16701
El-Nahass M. M., Kamel M. A., El-Deeb A. F., Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2011, 79, 1499
Daniel P. H., Tannia M., Karl M. K., Kazuteru N., Peng Q., Gerrit B., Tore B., Anders H., Sun L. C., J. Org. Chem., 2007, 72, 9550
Li L., Lin X. Y., Li Z. H., Chem. Res. Chinese Universities, 2011, 27(6), 1006
Wang L. M., Zhang J. P., Wang R. S., Chem. Res. Chinese Universities, 2003, 19(4), 508
Chen J., Wang J., Bai F. Q., Zheng Q. C., Zhang H. X., Chem. Res. Chinese Universities, 2012, 28(4), 696
Cohen A. J., Mori-Sánchez P., Yang W., Chem. Rev., 2012, 112(1), 289
Martsinovich N., Troisi A., Energy Environ. Sci., 2011, 4, 4473
Jacquemin D., Perpète E. A., Ciofini I., Adamo C., Acc. Chem. Res., 2009, 42(2), 326
Milián-Medina B., Gierschner J., Org. Electron., 2012, 13, 985
Dienes Y., Eggenstein M., Kárpáti T., Sutherland T. C., Nyulászi L., Baumgartner T., Chem. Eur. J., 2008, 14, 9878
Fleischhauer J., Zahn S., Beckert R., Grummt U. W., Birckner E., Görls H., Chem. Eur. J., 2008, 18, 4549
Risko C., Kushto G. P., Kafati Z. H., Brédas J. L., J. Chem. Phys., 2004, 121, 9031
Becke A. D., J. Chem. Phys., 1993, 98, 5648
Ditchfield R., Herhe W. J., Pople J. A., J. Chem. Phys., 1971, 54, 724
Qing W., Wayne M. C., Edia E. B., Kenneth W. J., David L. O., Penny J. W., Keith G., Robin H. B., Mohammad K. N., Michael G., J. Phys. Chem. B, 2005, 109, 15397
Tomas E., Chen L., Neil P., Jan S., Felix E., Rüdiger S., Gerrit B., Andreas H., Klaus M., Anders H., J. Phys. Chem. C, 2007, 111, 15137
Franck J., Trans. Faraday Soc., 1925, 21, 536
Condon E. U., Phys. Rev., 1928, 32, 858
Parr R. G.,, Yang W., Density Functional Theory of Atoms and Molecules, Oxford Science Publication, New York, 1989
Dreizler R. M., Gross E. K. U., Density Functional Theory, Springer-Verlag, Heidelberg, 1995
Runge E., Gross E. K. U., Phy. Rev. Lett., 1984, 52, 997
Gross E. K. U., Kotm W., Phys. Rev. Lett., 1985, 55, 285
Serrano-Andres L., Merchan M., J. Mol. Struct.(Theochem.), 2005, 729, 99
Frisch M. J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A. Jr., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam N. J., Klene M., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Salvador P., Dannenberg J. J., Dapprich S., Daniels A. D., Farkas Ö., Foresman J. B., Ortiz J. V., Cioslowski J., Fox D. J., Gaussian 09, Revision A.1, Gaussian Inc., Wallingford CT, 2009
Kitamura T., Ikeda M., Shigaki K., Inoue T., Anderson N. A., Ai X., Lian T. Q., Yanagida S., Chem. Mater., 2004, 16, 1806
Hagberg D. P., Edvinsson T., Marinado T., Boschloo G., Hagfeldt A., Sun L. C., Chem. Commun., 2006, 2245
Hagberg D. P., Yum J. H., Lee H., de Angelis F., Marinado T., Karlsson K. M., Humphry-Baker R., Sun L. C., Hagfeldt A., Grätzel M., Nazeeruddin M. K., J. Am. Chem. Soc., 2008, 130, 6259
Zhang J., Li H. B., Sun S. L., Geng Y., Wu Y., Su Z. M., J. Mater. Chem., 2012, 22(2), 568
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, J., Wang, M. Optical and electrical properties of triphenylamine derivatives for dye-sensitized solar cells and designing of novel molecule. Chem. Res. Chin. Univ. 29, 584–588 (2013). https://doi.org/10.1007/s40242-013-2213-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-013-2213-9