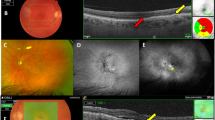
Abstract
Introduction
The aims of this work were to evaluate the real-world efficacy and safety of a loading dose of intravitreal faricimab in eyes with active neovascular age-related macular degeneration (n-AMD) or diabetic macular edema (DME) and to analyze the treatment outcome in relation to specific biomarkers.
Methods
Patients with active n-AMD or DME, treated with four monthly intravitreal injections of faricimab, were enrolled in this retrospective, uncontrolled study. Best-corrected visual acuity (BCVA), central subfield thickness (CST), presence of retinal fluid (RF) on optical coherence tomography (OCT), and adverse events were assessed at baseline and at weeks 4, 8, 12, and 16. Predefined biomarkers were evaluated at baseline (BL) and at last visit.
Results
Sixteen eyes of 15 patients with n-AMD (n-AMD group) and 15 eyes of 12 patients with DME (DME group) were included. Mean (± standard deviation) logarithm of minimum angle of resolution (logMAR) BL BCVA changed from 0.68 (± 0.43) to 0.53 (± 0.36; P = 0.13) and from 0.51 (± 0.34) to 0.32 (± 0.24; P: 0.048) at week 16 in n-AMD and DME group, respectively. A statistically significant mean CST reduction was reported in both groups at last visit (n-AMD: − 166.5 μm; P = 0.0009/DME: − 110.8 μm; P = 0.0086). Seventy-five and 33% of eyes with n-AMD and DME respectively achieved complete RF resolution at last visit. Subfoveal inner and outer retinal damage correlated with a lower final BCVA in n-AMD group. The presence of large (> 100 μm) juxtafoveal microaneurysms (MAs) was significantly correlated with a higher chance of residual fluid in eyes with DME.
Conclusions
Both n-AMD and DME groups achieved satisfactory anatomical results after a loading-dose of intravitreal faricimab. BCVA improvement might be hampered by pre-existing retinal damage in eyes with n-AMD. Large, juxtafoveal MAs might represent a hallmark of a slower anatomical response to the treatment in eyes with DME.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Why carry out this study? |
One of the major limitations of anti-vascular endothelial growth factor (anti-VEGF) drugs is their relatively short-lasting activity, which translates into the need for frequent retreatments over the long period. |
Faricimab, by neutralizing both vascular endothelial growth factor (VEGF) and angiopoietin 2 (ang-2), can offer a longer durability of action, with injection-free interval up to 16 weeks. |
The present study was planned to assess the early response to the treatment with faricimab in patients with neovascular age-related macular degeneration (n-AMD) and diabetic macular edema (DME) and to the investigate the potential role of predefined biomarkers as predictors of early response. |
What was learned from the study? |
Study results indicated that patients with n-AMD and DME can achieve satisfactory anatomical results together with visual acuity improvement or stabilization after a loading dose of faricimab. Complete retinal fluid resolution was reported in a fairly large number of eyes in both groups, as early as after the first two injections. The analysis of biomarkers disclosed that specific spectral domain optical coherence tomography (SD-OCT) features might serve as predictors of response in both study populations. |
The use of specific functional and structural biomarkers might help to predict the response to faricimab at a very early stage of the treatment. Further investigation is needed to define if these biomarkers can play a role at a later phase of the therapy. |
Introduction
Vascular endothelial growth factor (VEGF) inhibition represents the first-line treatment for retinal vascular disorders, including neovascular age-related macular degeneration (n-AMD), diabetic macular edema (DME), myopic choroidal neovascularization (CNV), and macular edema secondary to retinal vein occlusion (RVO).
Despite its well-established efficacy, anti-VEGF therapy does not come without limitations, though. The relatively short-term activity of anti-VEGF compounds translates into the need of frequent retreatments over a long period of time, imposing a significant burden on patients and the health-care system [1, 2]. Furthermore, treatment response is highly variable across different studies, with a rate of suboptimal response ranging between 30 and 50% [3, 4].
Many strategies have been proposed to optimize anti-VEGF treatment, without losing any of the benefits, including new treatment regimens, the use of biomarkers to guide the therapy, and the introduction of new drugs [5,6,7,8,9].
Intravitreal faricimab has emerged as a promising treatment for retinal diseases such as n-AMD and DME. Faricimab is a bispecific antibody that simultaneously binds and neutralizes both angiopoietin 2 (Ang-2) and VEGF-A, making it a unique therapeutic option [10].
Dual pathway inhibition might promote vascular stability and reduce neovascularization, hyperpermeability, and concurrent inflammation resulting in fibrosis and cell death leading to atrophy. This can translate into effects beyond anti-VEGF treatment alone, including improved durability compared with therapies targeting the VEGF pathway only [11].
Data from pivotal clinical trials have demonstrated sustained long-term efficacy and safety of intravitreal faricimab in patients with DME and n-AMD, with the possibility to extend the interval between two consecutive injections up to 16 weeks in almost 50% of the eyes [11, 12].
Preliminary data from real-world studies have also supported the efficacy of faricimab, with reports of improved retinal fluid control and vision stabilization in eyes with n-AMD and DME [13, 14].
Since the Italian Medicines Agency has approved the reimbursement of faricimab for the treatment of n-AMD and DME only in October 2023, clinical data on the efficacy of this drug in an Italian cohort of patients are still lacking.
Additionally, since the initial response to the loading phase has been advocated as a possible predictor of long-term outcome of anti-VEGF therapy, to define how do faricimab treated patients behave in the very early phase of their treatment, in a real-world setting, is paramount [15].
The aim of the present study was to investigate the efficacy and safety of a loading dose of intravitreal faricimab in patients with active n-AMD and DME, both naive and previously treated, and to correlate the anatomical and functional treatment response with structural and non-structural biomarkers.
Methods
This retrospective, uncontrolled study collected data from the Department of Ophthalmology, University of Foggia, between June 2023 and October 2023. The study followed the principles outlined in the Declaration of Helsinki. Informed consent for use of faricimab was obtained from all patients before treatment initiation. By signing it, patients also authorized their anonymized data could eventually be used for scientific purposes in the future. Patients were treated with approved diagnostic and therapeutic procedures according to generally accepted standards of care. A formal notification of the study protocol was sent to our local Ethics Committee (Policlinico Riuniti – University of Foggia). Ethical approval was waived for this study.
Patients with a diagnosis of active neovascular age-related macular degeneration (n-AMD) or diabetic macular edema (DME), who received four injections of faricimab every 4 weeks (Q4) as a loading dose, were included. Both naive and previously treated eyes could be enrolled. Inclusion criteria for patients affected by n-AMD were: age of 50 years or older; subfoveal CNV or juxtafoveal or extrafoveal CNV, with subfoveal component related to CNV activity, confirmed on fluorescein angiography; signs of exudation confirmed on spectral domain optical coherence tomography (SD-OCT); any type of CNV related to AMD: type 1 CNV (occult), type 2 CNV (classic), type 3 CNV (retinal angiomatous proliferation—RAP) and polypoidal choroidal vasculopathy (PCV). Concerning patients with DME, inclusion criteria were: age of 18 years or older, diagnosis of type 1 or 2 diabetes with good glycometabolic control, confirmed by glycated hemoglobin (HbA1c) level of 48 mmol/mol (6.5%) or below; macular edema involving the fovea center secondary to diabetic retinopathy (DR); central subfield thickness of 325 μm or more (average thickness between the internal limiting membrane and Bruch’s membrane in the central 1-mm diameter of the Early Treatment Diabetic Retinopathy Study [ETDRS] grid); a gradable DR (non-proliferative or proliferative) on color fundus photo.
Exclusion criteria were: CNV secondary to retinal diseases other than AMD, macular edema secondary to RVO, history of intraocular inflammation and/or uveitis, history of major ocular surgery and of intravitreal steroid injections in the previous 6 months, history of intravitreal anti-VEGF injection received less than 1 month before enrollment. Patients with media opacity, enough to prevent proper fundus visualization, were excluded as well.
To be enrolled, patients needed to have undergone a complete ophthalmologic evaluation including: best-corrected distance visual acuity (BCVA) measurement with ETDRS chart, anterior segment and fundus slit-lamp bio-microscopy with a non-contact fundus lens, intraocular pression (IOP) measurement with Goldman applanation tonometer, ultra-widefield color fundus photography (Optos 200Tx, Optos, Dunfermline, UK), fundus autofluorescence (FAF) and fluoresceine angiography (FA)—(Spectralis HRA + OCT; Heidelberg Engineering, Heidelberg, Germany), SD-OCT and OCT angiography (OCT-A) (Spectralis HRA + OCT; Heidelberg Engineering, Heidelberg, Germany).
For the OCT examination, the following scans were acquired in all eyes at each visit: a high-definition horizontal fovea-centered cross line B-scan at 30° at 100ART, a high-definition radial scan pattern centered on the foveal, a horizontal macula raster consisting of 49 B-scans 120 μm-spaced over an area of 20° and a 6 × 6-mm macular volume pattern. The ‘‘Thickness Map’’ function was used to automatically measure the mean CST, based on the mean retinal thickness within a circular area of 0.5-mm radius at the foveal center. OCT-A was performed with a 3 × 3-mm pattern.
The ophthalmologic assessment was performed at baseline, then every 4 weeks up to the end of the follow-up for each patient. FA was carried out at baseline.
Specific, predefined biomarkers were investigated (Table 1). Considering patients with n-AMD, the CNV type was defined according to the structural OCT and OCT-A features, together with the fluoresceine angiography pattern. Retinal fluid was classified as intraretinal fluid (IRF) and subretinal fluid (SRF) according to its location in respect to the neuroretina at SD-OCT. Ellipsoid zone (EZ) and external limiting membrane (ELM) irregularities were defined by the presence of multiple focal discontinuations or by the complete lack of the respective outer retinal bends at SD-OCT at the subfoveal location. Degenerative intraretinal cysts referred to large, irregular intraretinal cystoid spaces, spanning across different retinal layers, divided by very thin walls and not leaking on FA. Outer retinal tubulations (ORT) appeared as round or ovoid hyporeflective spaces with hyperreflective borders located at the outer nuclear layer at SD-OCT. The presence of well-demarcated, highly hyper-reflective material, in the subretinal space, with well-defined boundaries, configured a fibrotic scar. By contrast, sub-retinal hyper reflective material (SRHM) was identified as hyperreflective material with fuzzy borders, located beneath the neurosensory retina. Concerning eyes with DME, the main structural OCT biomarkers were: hyperreflective foci (HRF) identified as dot-like or round, regular lesions, less than 30 μm in size, with the same reflectivity of retinal nerve fiber layer; disorganization of inner retina layers (DRIL) defined by the complete loss of boundaries between the ganglion cell, inner plexiform, and outer nuclear plexiform layers at the foveal location.
Large juxtafoveal microaneurysms (MAs) were identified on SD-OCT scans as well-demarcated round or oval-shaped intraretinal structures with a hyper-reflective wall and an irregularly moderate, hyper o hypo-reflective lumen, with the smaller diameter exceeding 100 μm when manually measured from the outermost border. These lesions appeared as hyperfluorescent dots in the arteriovenous phase, associated with leakage in the late phase on FA. Only MAs surrounded by intraretinal cystoid spaces and located at the foveal center and/or in the parafoveal ring on a ETDRS grid were considered.
To reliably define each of these structural biomarkers, SD-OCT, OCT-A scans, and FA images were graded by two independent masked observers, expert in the field (A.G., G.C.). A third observer (E.G.) resolved any case of disagreement. Segmentation errors were manually corrected with built-in software whenever needed.
The main outcomes evaluated were mean BCVA and CST change during the follow-up, complete retinal fluid resolution, defined as absence of any retinal fluid (SRF and IRF) in each of the B-scans composing the horizontal macula raster pattern and the macular volume pattern on SD-OCT, and safety.
Secondary endpoints were: time to achieve complete retinal fluid resolution, proportion of eyes that gained vision (≥ 15, ≥ 10, ≥ 5 ETDRS letters) or avoided BCVA loss (≥ 15, ≥ 10, or ≥ 5 ETDRS letters) over time, eyes with final Snellen BCVA of 20/40 or better and correlations between treatment response and predefined biomarkers.
BCVA was reported in Snellen and then converted to logarithm of minimum angle of resolution (logMAR) unit for statistical purposes.
Incidence and severity of ocular and non-ocular adverse events was assessed at each follow-up visit and the day following each injection when patients were called back just for a safety evaluation.
Statistical Analysis
Descriptive statistics were performed. Categorical variables were expressed as counts and percentages in each category. Frequencies were expressed as percentages. Continuous variables were defined as means (standard deviation [SD]). Differences in continuous variables were assessed using Student’s t test for normally distributed data, while the Mann–Whitney U test was employed for variables demonstrating a non-normal distribution, as determined by the skewness and kurtosis test. Differences in rates were compared by the Chi-square test or Fisher’s exact test. Univariate survival analysis was carried out using the Kaplan–Meier method, with differences among groups (n-AMD vs. DME) being tested for significance using the log-rank test. Correlations were calculated using Spearman’s test. Significance was set at P < 0.05. Statistical analysis was carried out using STATA SE 15.
Results
Sixteen eyes of 15 patients with n-AMD and 15 eyes of 12 patients with DME were included. Each patient received a loading dose of four consecutive, every 4 weeks faricimab injections, together with a complete ophthalmologic assessment, up to week 12. Final evaluation was carried out at week 16.
Mean age was 78.87 ± 8.47 years (range, 60–93 years) in n-AMD group and 72.93 ± 13.74 (range 47–88) in DME group. Eyes with DME presented with a slightly better mean BCVA (0.51 ± 0.34) when compared to eyes with n-AMD (0.68 ± 0.43). Considering only non-naive eyes, the mean number of intravitreal injections received prior to entering the study was significantly greater in n-AMD group (15.5 ± 14.7) than in DME group (3.1 ± 1.6; P = 0.016). Other relevant demographic and baseline characteristics are listed in Table 2.
Functional Results
N-AMD Group
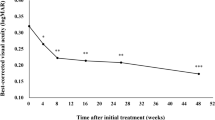
Final mean BCVA (0.53 ± 0.36 logMAR) did not show statistically significant variations when compared to BL values (0.68 ± 0.43 logMAR) in n-AMD group (P = 0.13), with seven eyes (44%) retaining their BL vision (gain or worsening < 5 ETDR letters). A gain in final vision of ≥ 15 letters, ≥ 10 letters but less than 15 and ≥ 5 letters but less than ten was reported in three (19%), one (6%) and four (25%) eyes, respectively. One (6%) eye lost more than five letters by the end of the follow-up (Fig. 1). A final BCVA of ≥ 20/40 was reported in six (37.5%) eyes. None of the eyes experienced a significant visual loss (≥ 15 ETDR letters loss) during the study period.
DME Group
Mean BL BCVA (0.51 ± 0.34 logMAR) significantly improved at month 3 (0.31 ± 0.23 logMAR; P = 0.042) and four (0.32 ± 0.24 logMAR; P = 0.048) in DME group. Six (40%) eyes gained 15 or more ETDRS letters, one (7%) gained at least ten ETDRS letters but less than 15 and three (20%) gained five or more ETDRS letters but less than ten. Vision remained stable in three (20%) eyes and decreased (≥ 5 ETDRS letters loss) in two (13%) (Fig. 2). Seven (47%) eyes presented with a final BCVA of 20/40 or better.
No cases of significant visual loss (≥ 15 ETDR letters loss) were reported.
Anatomical Results
N-AMD Group
Mean CST showed a statistically significant reduction from BL, starting from the very first month. Final mean CST decreased by 166.5 μm (P = 0.0009) when compared to BL value (Fig. 3A). Seventy-five percent of eyes achieved a complete fluid-free retina by the end of the follow-up in this group (Fig. 4).
Complete retinal fluid resolution was evident in six (37.5%) eyes after the first injection and in five (31.25%) eyes after the second injection. One eye (6.25%) achieved a complete dry retina after the third injection. No case of fluid recurrence during the follow-up were reported.
DME Group
Mean CST started to decrease significantly 1 month after the first injection in this population as well, with a reduction of 110.8 μm (P = 0.0086) at final visit, with respect to BL (Fig. 3B). Complete retinal fluid resolution was reported in 33% of diabetic eyes by the end of the follow-up (Fig. 4).
Two eyes (13.3%) achieved a fluid-free retina after the first injection, three eyes (20%) achieved the outcome after the second injection. A complete fluid-free retina was never reported in the remaining ten eyes (66.7) during the course of the study, even though a significant CST reduction was evident. Fluid recurrence and CST increasing were reported in none of the eyes from this group.
Biomarkers of Response
N-AMD Group
Looking at the role of structural and non-structural biomarkers on the functional response in n-AMD group, final BCVA was significantly associated with the presence of ELM irregularities (P = 0.0082, 95% CI 0.51–1.05), intraretinal chronic cysts and ORT (P = 0.03, 95% CI 1–1) and fibrotic scar (P = 0.03, 95% CI 1–1) at baseline SD-OCT and with the presence of ELM irregularities (P = 0.0012, 95% CI 0.63–1.09), EZ irregularities (P = 0.0065, 95% CI 0.47–0.90) and fibrotic scar (P = 0.042, 95% CI 0.26–1.38) at the final OCT. None of the remaining biomarkers evaluated was associated with the final BCVA.
The analysis of the association between biomarkers and the retinal fluid dynamic, intended as the rate of complete retinal fluid resolution and the time to achieve a complete dry retina, gave the following results: the presence of SRF at BL associated with a faster retinal fluid resorption in eyes with n-AMD (P = 0.033; 95% CI 0.35–1.46). None of the other predefined biomarkers showed a significant association with fluid dynamics. The complete analysis of biomarkers is listed in Table 3.
DME Group
None of the predefined biomarkers evaluated both at baseline and at last visit was significantly associated with final BCVA in this group.
The analysis of any possible association between structural and non-structural biomarkers with the chance of complete resolution of the retinal fluid by the end of the follow-up and the time to achieve a dry retina revealed that a greater number of large, parafoveal MAs correlated with a lower rate of complete fluid resolution in eyes with DME (mean no in eyes with resolved fluid: 1.6 ± 2.07; mean number in eyes with residual fluid: 6.8 ± 5.41).
The full analysis of biomarkers is outlined in Table 4.
Safety
No significant ocular and systemic adverse events have been reported.
Discussion
Intravitreal faricimab has emerged as a new promising treatment that can potentially overcome some of the issues related to conventional anti-VEGF drugs. The longer durability, with extended dosing up to every 16 weeks, together with a greater fluid control, may translate into a better management of vascular retinal disorders over a long period, without the need of frequent retreatments.
Increasing evidence derived from real-world clinical studies indicates that faricimab is an effective treatment for naive or previously treated n-AMD and DME patients who experience a significant anatomical improvement associated with visual acuity recovery or stabilization after therapy [16, 17].
The anatomical response was excellent in our study, with mean CST that significantly decreased after the first injection already, reaching the lowest values at months 3 and 4 in both study populations.
Central macular thickness variation may not be a reliable indicator of disease activity, especially in eyes with AMD where discrete amount of retinal fluid may persist even if a significant CST reduction is reported [18].
In the randomized Comparison of Age-Related Macular Degeneration Treatments Trials (CATT), macular fluid was still present in over half of patients receiving monthly ranibizumab therapy after 1 and 2 years of treatment (53.2 and 51.5%, respectively). In treatment-naive patients receiving aflibercept every 8 and 4 weeks, approximately 32.3 and 35.2%, respectively, had residual fluid after 52 weeks of therapy [3, 4].
Rates of persistent DME in clinical studies (based on CSFT ≥ 250 µm) can be in the range of 20–60% after 2 years of treatment [19, 20].
Inadequate suppression of retinal fluid, particularly IRF, has resulted in poor visual outcomes despite continuous anti-VEGF treatment [18].
Complete retinal fluid resolution, indicated by the lack of any detectable fluid (IRF and SRF) in each of the B-scan composing the horizontal macula raster pattern and the macular volume pattern at SD-OCT, was set as one of the major indicators of response to the treatment in our study. The number of eyes that achieved complete retinal fluid resolution was significantly greater in n-AMD population (75% of the eyes reaching the outcome), when compared to DME group (33% of the eyes achieving a complete dry retina). We are currently unable to fully justify this difference in anatomical response. It might be related to a slower response in diabetic eyes that might need more injections to achieve a complete regression of the fluid.
Another possible explanation is that DME group might include more eyes resistant to faricimab that will never achieve a dry retina despite prolonged treatments. Nevertheless, four injections are too few to define a proper response to a given anti-VEGF treatment. Several studies have demonstrated that some patients may need six or even more injection to reach the outcomes [21,22,23].
We cannot exclude that eyes with DME included in our study will achieve a complete anatomical response on the longer run after additional injections.
An interesting finding came from the analysis of anatomical biomarkers. Eyes with DME with residual IRF at the end of the follow-up tended to have larger parafoveal MAs at BL. These MAs appeared as round or oval lesions with an outer hype reflective ring at SD-OCT and did not change in shape, size, and number after the loading phase. Moreover, these vascular lesions were constantly surrounded by pockets of IRF.
Makoto Hatano et al. demonstrated that detection of MAs in the nasal macula using pretreatment indocyanine green angiography may indicate resistance to anti-VEGF therapy in DME patients. The presence of large MAs in the nasal macula indicates that the DME condition is physiologically and anatomically severe, and such cases are likely to resist anti-VEGF therapy [24].
Masahiko Sugimoto et al., found a significant correlation between the CRT and number of MAs at both BL and after treatment with aflibercept in eyes with DME and that the improvement of DME by anti-VEGF therapy correlated with the size of Mas [25].
Furthermore, Yoshihiro Takamura et al. demonstrated a significant reduction of size and number of MAs in faricimab-treated DME patients, with a lower rate of formation of new lesions [26].
We speculated that the presence of large MAs that do not regress after treatment may indicate a more severe condition, associated with more mature vascular lesions, eventually resistant to the dual inhibition [27].
Another relevant point that emerged from our study is the response to the treatment in terms of complete retinal fluid regression, which occurred after the first or the second injection in most eyes.
Looking at the data from RCTs and real-life study as well, it is possible to appreciate that anatomical response to faricimab is surprisingly quick, with a significant CST reduction after the first injection already [17]. This is further supported by other reports from the literature where eyes with AMD or DME, unresponsive to a given treatment and then switched to faricimab, achieve a significant CST reduction or a complete retinal fluid regression as soon as after the first 3 or 4 injections [13, 28,29,30].
Very short-term response to an anti-VEGF treatment has been advocated as an indicator of long-term outcome, since a patient who does not show even a small degree of improvement during the loading phase will likely continue not to respond to that treatment in the future. Several studies indicate that the long-term response can be predicted even after the first injections in some cases [15, 31,32,33].
The present study was not designed to assess the long-term response to faricimab, rather to define the short-term outcome of a loading-dose, so early response cannot be correlated to longer-term outcomes in our patients. A study extension is needed to elucidate this point.
The analysis of biomarkers in respect to fluid dynamics disclosed that the presence of SRF might correlate with a faster retinal fluid resolution in eyes with n-AMD. To the best of our knowledge, this is the first evidence of a potential role of SRF as a biomarker of anatomical response in patients affected by n-AMD undergoing treatment with faricimab.
Despite the excellent anatomical response, with most eyes experiencing retinal fluid resolution, visual acuity stayed stable in AMD group, without significant improvement. This may be, at least in part, justified by the presence of preexisting damage to the retina induced by chronicity in non-naive eyes. A significant correlation between disruption of outer retinal layers (ELM and EZ), intraretinal chronic degeneration, fibrotic scar and final BCVA was demonstrated in eyes with AMD. The subset of previously treated eyes in AMD group received significantly more injection (mean 15) than those in DME group (mean 3), indicating a longer lasting pathology with maybe poor fluid control and more frequent reactivation of the neovascular lesion over the previous months.
Our results are in line with those emerged from other studies on faricimab in AMD patients resistant to previous anti-VEGF treatment, where mean BCVA tended to remain stable, without relevant improvement, despite a good anatomical response and proper fluid control [28, 29].
In contrast, eyes with DME exhibited a significant BCVA improvement, with almost 50% of eyes gaining ten or more ETDR letters. None of the investigated biomarkers correlate with final BCVA, even though a tendency toward a worst final visual outcome was evident in those eyes with HRF at baseline and with subfoveal EZ alterations at final OCT. We cannot rule out completely that the lack of a statistically significant correlation could be attributable to the small sample size. Previously treated eyes in DME group received a lower number of injections before being switched to faricimab, indicating a shorter duration of the macular edema and maybe a lower rate of chronic retina damage.
Intravitreal faricimab was well tolerated with no evidence of systemic and ocular adverse events so far. Longer follow-up and a wider cohort of patients are needed to further validate this finding.
Several limitations need to be acknowledged in the present study. First, the small sample size might reduce the reliability of our results, especially regarding the potential role of biomarkers as predictor of response. We could not perform a multivariate analysis due to a limited number of eyes.
The short follow-up makes it impossible to reach conclusions on the behavior of our patients in the long run. We cannot know for example if eyes with DME that display a partial response after four injections will continue to improve after additional treatments. Conversely, it is not possible to know if eyes that achieved a prompt and complete retinal fluid resolution will be able to maintain the result even under extended treatment intervals. The lack of a control group stands as an additional major limitation.
Nevertheless, this is one of the first real-life Italian experiences with faricimab in a mixed population. Although being retrospective in nature, the present study involved patients who received a strict, according to the protocol treatment, a comprehensive ophthalmologic evaluation together with an instrument-based multi-modal imaging assessment with standardized acquisition protocols at baseline and at every follow-up visit, and the record of all relevant clinical information and eventual adverse events. The analysis of predefined biomarkers adds extra value to our findings.
Conclusions
In conclusion, intravitreal faricimab appeared to be safe and effective in eyes with n-AMD and DME, both naive and previously treated, with substantial short-term anatomical improvement and visual acuity stabilization. Predefined biomarkers might be used to predict the degree of anatomical response to the loading phase and to identify those patients who will experience a suboptimal BCVA recovery. Longer follow-up and a larger cohort of patients are needed to further validate our results.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ferro Desideri L, Traverso CE, Nicolò M, Munk MR. Faricimab for the treatment of diabetic macular edema and neovascular age-related macular degeneration. Pharmaceutics. 2023;15:1413.
Spooner KL, Fraser-Bell S, Hong T, Wong JG, Chang AA. Long-term outcomes of anti-VEGF treatment of retinal vein occlusion. Eye. 2022;36:1194–201.
Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group, et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology. 2012;119:1388–98.
Ashraf M, Souka AAR. Aflibercept in age-related macular degeneration: evaluating its role as a primary therapeutic option. Eye Lond Engl. 2017;31:1523–36.
Ohji M, et al. Efficacy and safety of intravitreal aflibercept treat-and-extend regimens in exudative age-related macular degeneration: 52- and 96-week findings from ALTAIR: a randomized controlled trial. Adv Ther. 2020;37:1173–87.
Sarohia GS, et al. Treat-and-extend versus alternate dosing strategies with anti-vascular endothelial growth factor agents to treat center involving diabetic macular edema: a systematic review and meta-analysis of 2346 eyes. Surv Ophthalmol. 2022;67:1346–63.
Schmidt-Erfurth U, Waldstein SM. A paradigm shift in imaging biomarkers in neovascular age-related macular degeneration. Prog Retin Eye Res. 2016;50:1–24.
Sorour OA, et al. Persistent diabetic macular edema: definition, incidence, biomarkers, and treatment methods. Surv Ophthalmol. 2023;68:147–74.
Garweg JG, et al. Brolucizumab in neovascular age-related macular degeneration and diabetic macular edema: ophthalmology and diabetology treatment aspects. Ophthalmol Ther. 2023;12:639–55.
Khan M, Aziz AA, Shafi NA, Abbas T, Khanani AM. Targeting angiopoietin in retinal vascular diseases: a literature review and summary of clinical trials involving faricimab. Cells. 2020;9:1869.
Heier JS, et al. Efficacy, durability, and safety of intravitreal faricimab up to every 16 weeks for neovascular age-related macular degeneration (TENAYA and LUCERNE): two randomised, double-masked, phase 3, non-inferiority trials. Lancet Lond Engl. 2022;399:729–40.
Wykoff CC, et al. Efficacy, durability, and safety of intravitreal faricimab with extended dosing up to every 16 weeks in patients with diabetic macular oedema (YOSEMITE and RHINE): two randomised, double-masked, phase 3 trials. Lancet Lond Engl. 2022;399:741–55.
Rush RB, Rush SW. Faricimab for treatment-resistant diabetic macular edema. Clin Ophthalmol Auckl NZ. 2022;16:2797–801.
Leung EH, et al. Initial real-world experience with faricimab in treatment-resistant neovascular age-related macular degeneration. Clin Ophthalmol Auckl NZ. 2023;17:1287–93.
Dugel PU. Can long-term response to anti-VEGF therapy be predicted after three injections in patients with DME? Retina Today. https://retinatoday.com/articles/2016-mar/can-long-term-response-to-anti-vegf-therapy-be-predicted-after-three-injections-in-patients-with-dme. Accessed March 2016.
Cheng AM, Joshi S, Banoub RG, Saddemi J, Chalam KV. Faricimab effectively resolves intraretinal fluid and preserves vision in refractory, recalcitrant, and nonresponsive neovascular age-related macular degeneration. Cureus. 2023;15: e40100.
Khanani AM, et al. The real-world efficacy and safety of faricimab in neovascular age-related macular degeneration: the TRUCKEE study—6 month results. Eye Lond Engl. 2023;37:3574–81.
Kaiser PK, et al. Retinal fluid and thickness as measures of disease activity in neovascular age-related macular degeneration. Retina. 2021;41:1579.
Khoramnia R, et al. Exploring the role of retinal fluid as a biomarker for the management of diabetic macular oedema. Eye Lond Engl. 2023. https://doi.org/10.1038/s41433-023-02637-2.
Sadda SR, et al. Relationship between duration and extent of oedema and visual acuity outcome with ranibizumab in diabetic macular oedema: a post hoc analysis of Protocol I data. Eye Lond Engl. 2020;34:480–90.
Gonzalez VH, et al. Early and long-term responses to anti-vascular endothelial growth factor therapy in diabetic macular edema: analysis of protocol I data. Am J Ophthalmol. 2016;172:72–9.
Santos AR, et al. Optical coherence tomography baseline predictors for initial best-corrected visual acuity response to intravitreal anti-vascular endothelial growth factor treatment in eyes with diabetic macular edema: the CHARTRES study. Retina Phila Pa. 2018;38:1110–9.
Busch C, et al. Real-world outcomes of non-responding diabetic macular edema treated with continued anti-VEGF therapy versus early switch to dexamethasone implant: 2-year results. Acta Diabetol. 2019;56:1341–50.
Hatano M, et al. Evaluation of microaneurysms as predictors of therapeutic response to anti-VEGF therapy in patients with DME. PLoS One. 2022;17: e0277920.
Sugimoto M, et al. Multiple effects of intravitreal aflibercept on microvascular regression in eyes with diabetic macular edema. Ophthalmol Retina. 2019;3:1067–75.
Takamura Y, et al. Turnover of microaneurysms after intravitreal injections of faricimab for diabetic macular edema. Investig Ophthalmol Vis Sci. 2023;64:31.
Lammer J, et al. Association of microaneurysms on adaptive optics scanning laser ophthalmoscopy with surrounding neuroretinal pathology and visual function in diabetes. Investig Ophthalmol Vis Sci. 2018;59:5633–40.
Szigiato A, et al. Short-term outcomes of faricimab in patients with neovascular age-related macular degeneration on prior anti-VEGF therapy. Ophthalmol Retina. 2023. https://doi.org/10.1016/j.oret.2023.08.018.
Pandit SA, et al. Clinical outcomes of faricimab in patients with previously treated neovascular age-related macular degeneration. Ophthalmol Retina. 2023. https://doi.org/10.1016/j.oret.2023.10.018.
Raimondi R, et al. Real-world outcomes of treatment resistant neovascular-age related macular degeneration switched from aflibercept to faricimab. Ophthalmol Retina. 2023. https://doi.org/10.1016/j.oret.2023.11.015.
Koyanagi Y, et al. Visual outcomes based on early response to anti-vascular endothelial growth factor treatment for diabetic macular edema. Ophthalmol J Int Ophtalmol Int J Ophthalmol Z Augenheilkd. 2018;239:94–102.
Maeda S, et al. Response to initial anti-vascular endothelial growth factor for diabetic macular edema is significantly correlated with response to third consecutive monthly injection. J Clin Med. 2022;11:6416.
Xue K, et al. Prediction for 2-year vision outcomes using early morphologic and functional responses in the comparison of age-related macular degeneration treatments trials. Ophthalmol Retina. 2023;7:564–72.
Acknowledgements
We thank the participants of the study.
Funding
No funding or sponsorship was received for this study. The journal’s Rapid Service Fee was funded by the institution (Department of Ophthalmology, University of Foggia, Foggia, Italy).
Author information
Authors and Affiliations
Contributions
Ermete Giancipoli: conceptualization and design of the study, drafting the manuscript; Antonella Guglielmi: conceptualization and design of the study, final revision of the manuscript, corresponding author; Anna Valeria Bux: conceptualization and design of the study, data collection; Giulia Clima: conceptualization and design of the study, data collection; Francesco Pignatelli: data analysis, final revision of the manuscript; Francesco Boscia: supervision, final revision of the manuscript; Pasquale Viggiano: data analysis; Giacomo Boscia: data analysis; Francesca Fortunato: statistical analysis; Gianluca Besozzi: data analysis; Alfredo Niro: data analysis, final revision of the manuscript; Stefano Dore: data analysis; Cristiana Iaculli: supervision, conceptualization and design of the study, critical revision of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Ermete Giancipoli and Francesco Boscia are consultants for Roche. Antonella Guglielmi, Anna Valeria Bux, Giulia Clima, Francesco Pignatelli, Pasquale Viggiano, Giacomo Boscia, Francesca Fortunato, Gianluca Besozzi, Alfredo Niro, and Stefano Dore have nothing to disclose. Cristiana Iaculli had a financial arrangement with Roche within the past 12 months. Anna Valeria Bux has changed her affiliation to “Department of Ophthalmology, Ospedale di Venere, Carbonara di Bari, Via Ospedale di Venere 1, 70131, Bari (BA)—Italy”.
Ethical approval
This study followed the principles outlined in the Declaration of Helsinki. Informed consent for use of faricimab was obtained from all patients before treatment initiation. By signing it, patients also authorized their anonymized data could eventually be used for scientific purposes in the future. Patients were treated with approved diagnostic and therapeutic procedures according to generally accepted standards of care. A formal notification of the study protocol was sent to our local Ethics Committee (Policlinico Riuniti—University of Foggia). Ethical approval was waived for this study.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Giancipoli, E., Guglielmi, A., Bux, A.V. et al. Real-World Outcomes of a Loading Phase with Intravitreal Faricimab in Neovascular Age-Related Macular Degeneration (n-AMD) and Diabetic Macular Edema (DME). Ophthalmol Ther 13, 2163–2184 (2024). https://doi.org/10.1007/s40123-024-00980-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-024-00980-5