Abstract
Introduction
Diabetic macular oedema (DMO) is a complication of diabetic retinopathy that can result in vision loss. The disease can impact different spheres of a patient’s life, including physical and psychological health, work, and activities of daily living, entailing an important use of healthcare and non-healthcare resources. This study aimed to estimate the socio-economic burden of DMO in Spain.
Methods
The burden of DMO was estimated from a societal perspective, per patient, year of treatment since diagnosis, and type of treatment. Four categories were considered: direct healthcare costs (DHC), direct non-healthcare costs (DNHC), labour productivity losses (LPL), and intangible costs (IC) associated with loss of quality of life. Average annual costs were calculated by multiplying the resources used per patient by their corresponding unit price (or financial proxy). For a more accurate estimation, differences in resource use between treatments (intravitreal anti-vascular endothelial growth factor injections of ranibizumab or aflibercept, and intravitreal dexamethasone implants) and year since diagnosis (first, second, and third year or beyond) were considered and presented separately. The reference year for costs was 2021.
Results
The average annual costs of DMO in the first year of treatment after diagnosis was estimated at €18,774, €17,512, and €16,188 per patient treated with ranibizumab, aflibercept, and dexamethasone, respectively. This burden would be reduced to €15,783, €15,701, and €12,233 in the second year, and to €15,119, €15,043, and €12,790 in the third year, respectively. Diagnosis of DMO entails an additional one-off cost of €485. DHC accounted for the greatest proportion of total annual costs per patient, independent of the year, with LPL also making an important contribution to total costs.
Conclusions
The socio-economic impact of DMO on patients, the healthcare system, and society at large is substantial. The constant increase in its prevalence accentuates the need for planning and implementation of healthcare strategies to prevent vision loss and reduce the socio-economic burden of the disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Diabetic macular oedema (DMO) is a highly prevalent disease that imposes a substantial burden on patients, the healthcare system, and society at large. |
5.7% of patients with diabetes mellitus (DM) type 1 and 6.4% of patients with DM type 2 present DMO. |
This study aimed to estimate the socio-economic burden of DMO in Spain. |
The average annual costs of DMO in year 1 was estimated at €18,774, €17,512, and €16,188 per patient treated with ranibizumab, aflibercept, and dexamethasone, respectively. |
There are potential benefits of prompt effective treatments that prevent disease progression. |
This burden would be reduced to €15,783, €15,701, and €12,233 in year 2, and to €15,119, €15,043, and €12,790 in year 3+, respectively. |
Introduction
Diabetic retinopathy is among the leading causes of blindness (5.6%) and moderate–severe vision impairment (3.0%) worldwide [1]. Diabetic macular oedema (DMO) is a complication of diabetic retinopathy whereby a failure of the blood–retinal barrier results in the accumulation of fluid in the centre of the retina [2, 3]. This build-up causes blurred and distorted central vision [3]. Treatment of DMO consists of intravitreal injections of anti-vascular endothelial growth factor (anti-VEGF), established as the treatment of choice alongside corticosteroids [4]. Other available treatments include laser photocoagulation and surgical intervention [3].
The disease and its associated treatments can lead to complications and/or adverse events (AEs) such as infectious endophthalmitis [5], increased intraocular pressure [5], retinal detachment [6], vitreous haemorrhage [7], and rubeosis [7], among others. In 2014, the average annual cost of diagnosis, treatment, and follow-up was estimated at €4199.4 per patient in Spain [8]. Patients with DMO also often perceive a negative impact of the disease on their functionality and quality of life, mainly due to vision quality loss [9]. As such, DMO has been associated with a poor physical health status and impaired work productivity [10], as well as reduced quality of life given loss of visual acuity [11]. Taken together, the effects of DMO impose a considerable burden on both the healthcare system and society.
In Spain, 12.5% of the population 18 years or older have diabetes mellitus (DM) (1.0% DM type 1 and 11.5% DM type 2) [12]. Moreover, 5.7% of patients with DM type 1 and 6.4% of patients with DM type 2 present with DMO [13]. Studies suggest that the global number of adults with DMO will increase by 51.9% in the next 20 years, in relation to the increase in patients with diabetes [14]. Early diagnosis and early treatment could prevent progressive vision loss in patients with DMO [8].
Projections of increased DMO prevalence highlight the need for planning and implementing healthcare strategies that prevent vision loss. It is therefore important to understand the economic and social burden of DMO, and thus the potential benefits of treating the disease more effectively and preventing vision loss. Therefore, the aim of this study was to estimate the burden per patient with DMO in Spain from a societal perspective, according to the type of treatment and time since diagnosis.
Methods
Study Design
The burden of DMO was estimated using a prevalence approach, from a societal perspective, per patient, year of treatment since diagnosis, and type of treatment. Four cost categories were considered, including direct healthcare costs (DHC), direct non-healthcare costs (DNHC), labour productivity losses (LPL), and intangible costs (IC) associated with loss of quality of life (Fig. 1). The cost of diagnosis was further included as a one-off cost.
Cost items associated with DMO (e.g., resources used) were obtained through a narrative literature review carried out in PubMed®. Subsequently, an online meeting was held with an advisory committee of four well-known experts in DMO in Spain (three retina-vitreous specialised ophthalmologists and one hospital pharmacist) to discuss and validate the information extracted from the literature review.
Given differences in resource consumption over time since diagnosis, three groups of patients were considered: (a) patients in their first year of treatment after diagnosis (year 1), (b) patients in their second year of treatment after diagnosis (year 2), and (c) patients in their third year of treatment after diagnosis or later (year 3+). In addition, three types of treatment were considered: (a) treatment with intravitreal injections of ranibizumab, (b) treatment with intravitreal injections of aflibercept, and (c) treatment with intravitreal dexamethasone implants in patients refractory to three loading doses of anti-VEGF (ranibizumab or aflibercept). The latter would account for 40.0% of patients [15] that would receive 3 months (months 1–3) of treatment with intravitreal anti-VEGF injections, followed by 9 months (months 4–12) of treatment with intravitreal dexamethasone implants in their first year of treatment.
Moreover, the number of eyes affected by DMO was accounted for in resource consumption estimates. Accordingly, the proportion of patients with unilateral and bilateral DMO at the time of diagnosis and over the first 3 years of treatment was estimated (see the Supplementary Material—Table S1).
Cost Items
Items Associated with Diagnostic One-Off Costs
The cost of diagnosis includes medical visits and specific tests which are presented in Table 1.
Items Associated with Direct Healthcare Costs
DHC include treatment and follow-up costs associated with medical visits, tests, intravitreal injections and/or implants, and AEs. The number of intravitreal anti-VEGF injections by year since diagnosis was based on a treat-and-extend regimen, which was discussed and validated by the advisory committee of this study. A detailed description of cost items used to estimate DHC are presented in Table 2. For patients treated with dexamethasone, year 1 includes cost items associated with anti-VEGF intravitreal injections until dexamethasone switch due to refractoriness (Fig. 2).
Items Associated with Direct Non-healthcare Costs
DNHC include informal care, formal care, commuting for medical visits, and use of non-visual technical aids (Table 3). Informal care was defined as unpaid personal care related to DMO, provided by the patient’s affective environment (e.g., relatives, friends). Accompanying the patient to medical visits was the only item considered as informal care, given the lack of further information on the matter. On the other hand, formal care was defined as paid personal care related to DMO, provided by professionals. Due to the lack of published information on formal care for patients with DMO, the probability of a patient with neovascular age-related macular degeneration (nAMD) needing formal care (0.139) [24] was taken as a reference and assumed it would be 25.0% lower for a patient with DMO as it is less disabling than nAMD. Therefore, the probability of a patient with DMO needing formal care was estimated to be 0.104, irrespective of treatment type and year since diagnosis. Moreover, non-visual technical aids included the use of items such as a magnifying glass, special lamp, computer application, or smoke sensors. Due to the lack of DMO information on this subject, the probability of such patient needing non-visual technical aids was assumed to be 25.0% lower than in patients with nAMD (0.370) [24], as DMO is less disabling in comparison to nAMD. Therefore, the probability of a patient with DMO needing non-visual technical aids was estimated at 0.278, irrespective of treatment and year since diagnosis.
Items Associated with the Cost of Labour Productivity Losses
LPL was defined as the impact of the disease on a patient’s work activity and included (a) working hours lost to DMO medical visits (based on the percentage of patients who work, the number of specialist visits, and an average of 3 h spent on each visit), per treatment type and year since diagnosis (Table 4), and (b) working hours lost associated with changes in work status (reduction in working hours, loss or abandonment of employment, or being granted an incapacity for work) per year since diagnosis (Table 5).
Items Associated with Intangible Costs
IC included the impact of DMO on patients’ activities of daily living and quality of life, regardless of treatment type and year since diagnosis. The annual impact of DMO on the patient’s activities of daily living is presented in Table 6. The annual impact of DMO on patients’ quality of life was estimated as the average loss of utility (measured by quality-adjusted life years or QALYs) relative to the general population. Accordingly, the average utility value of patients with DMO (0.823) [26] was subtracted from the average utility value of the Spanish population aged ≥ 18 years (0.910) [27], yielding an average utility loss of 0.087. The mean utility of patients in the first year of treatment with ranibizumab [26] was assumed for the other treatments, as the mean utility associated with other treatments was not available.
Cost Estimation
The burden per patient with DMO by treatment type and year since diagnosis was calculated by multiplying cost items by their unit prices or financial proxies. The latter can be found in the Supplementary Material—Tables S19 to S23. Unless otherwise specified, they were applied irrespective of treatment type and year since diagnosis.
DHC were estimated indirectly by multiplying cost items by their unit prices, with the exception of the cost of medication that was estimated from the laboratory price of an intravitreal injection with ranibizumab (Lucentis®, Novartis Farmacéutica S.A., 10 mg/mL–1 prefilled syringe of 0.165 mL or one vial of 0.23 mL) or aflibercept (Eylea®, Bayer Hispania S.L., 40 mg/mL–1 prefilled syringe of 90 μL or one vial of 100 μL) which was €742.0 for both medications, and of an intravitreal dexamethasone implant (Ozurdex®, Allergan S.A., one intravitreal implant of 700 μg) which was €950.0. Regarding DNHC, the cost of informal care was estimated using the proxy good method, whereby the time spent by the informal caregiver in caring for the patient due to the disease is valued at the cost of replacing that person with a non-qualified worker. Therefore, the hourly minimum wage for domestic workers (€7.6) [30] was multiplied by the number of hours an informal caregiver spends in accompanying the patient with DMO to medical visits. To estimate the cost of formal care and given the lack of information for DMO, 75% [31] of the annual cost in formal care for patients with AMD (€1421.6) [24] was assumed, as DMO is less disabling []. Therefore, the annual cost of formal care per patient with DMO was estimated at €1066.2. The cost associated with commuting to attend medical visits was estimated by multiplying the number of commutes between home and the health centre by the cost per commute (€8.0). This cost was calculated as the average cost per commute by car (€5.3), bus (€6.0), and taxi (€12.0) [24]. The cost per commute by car and bus was calculated by multiplying the number of kilometres per commute (27.9 km and 31.8 km, respectively) and the cost financed per kilometre travelled (€0.2) [24]. To estimate the cost of non-visual technical aids, and given the lack of information for DMO, 75.0% [31] of the annual expenditure per nAMD patient on non-visual technical aids was assumed (€28.5) [24], as DMO is less disabling []. Therefore, the annual expenditure per patient with DMO on non-visual technical aids was estimated at €21.4.
To estimate the LPL, the human capital method was used. According to this method, work time lost to illness (i.e., due to medical visits, reductions in working hours, employment loss or abandonment, or being granted an incapacity for work) is valued at the market wage [32]. Finally, the average utility lost was monetised by multiplying this loss by the incremental cost-effectiveness threshold per QALY gained (€21,000) [33].
Total costs (TC) per DMO patient were calculated as the sum of the four cost categories (DHC, DNHC, LPL, and IC) according to the year since diagnosis. Given the prevalence approach used in the present study, the cost of diagnosis was not included in the TC [34]. Moreover, it is a varying one-off cost that cannot be ascribed to a specific time range [24].
The reference year for all the costs is 2021, with the exception of the cost of medication, obtained from the drug database of the General Council of Official Colleges of Pharmacists (Consejo General de Colegios Oficiales de Farmacéuticos) [35,36,37,38,39], and the cost of health services, for which the median of the health tariffs from the official bulletins of all the autonomous communities of Spain (previously updated as indicated in each bulletin) was estimated. Other costs were updated to the year 2021 using the Consumer Price Index [40].
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. The four members of the advisory committee are authors of this manuscript and were informed that their advice would be used in the development of this article.
Sensitivity Analysis
When two different data points associated with same resource were identified in the literature, the lowest value was used in both the baseline and lower scenarios, and the highest value was used in the upper scenario. This criterion follows a conservative approach to prevent overestimating the burden of disease, as both the baseline and lower scenarios represent the minimum burden, while the upper scenario suggests how far the cost can potentially increase. Resource consumption and costs presented in this article refer to the baseline scenario, and those of the lower and upper scenarios can be found in the Supplementary Material.
Results
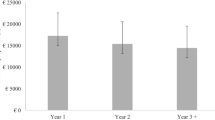
The TC for year 1 of DMO treatment after diagnosis was estimated at €18,773.6 per patient treated with ranibizumab, €17,512.1 per patient treated with aflibercept, and €16,187.7 per patient refractory to anti-VEGF treatment who switched to dexamethasone. In year 2, TC per patient would be reduced by 15.9%, 10.3%, and 24.4% from year 1, respectively. Thereafter, in year 3+, TC per patient treated with anti-VEGF would decrease by 4.2% from year 2, and the burden per patient treated with dexamethasone would increase by 4.6% with respect to year 2. A detailed description of the costs associated with DMO and the distribution of the total cost among cost categories by treatment type and year since diagnosis are shown in Tables 7 and 8, respectively. Figure 3 includes the results of the sensitivity analysis for TC represented as error bars.
Regarding DHC, the burden for year 1 was estimated at €11,986.6 per patient treated with ranibizumab, €10,725.2 per patient treated with aflibercept, and €9400.8 per patient refractory to anti-VEGF therapy who switched to dexamethasone. In year 2, the DHC per patient would be reduced by 29.1%, 21.5%, and 46.8% from year 1, respectively. In year 3+, DHC per patient would be reduced by 13.0%, 13.1%, and 2.3% from year 2, respectively. In addition, diagnosis would generate an estimated one-time additional cost of €484.5 per diagnosed DMO patient.
Regarding DNHC, the burden for year 1 was estimated at €1358.0 per patient, irrespective of treatment type. In year 2, the DNHC per patient treated with anti-VEGF would be reduced by 8.5% compared to year 1, while it would be reduced by 11.4% in patients treated with dexamethasone. In year 3+, the DNHC per patient would remain the same as in the previous year regardless of treatment type.
Regarding LPL, the burden for year 1, due to medical visits and the impact of the disease on their work, was estimated at €3317.3 per patient, regardless of treatment type. In year 2, LPL per patient treated with anti-VEGF would increase by 18.4% compared to year 1, while it would increase by 18.0% in patients treated with dexamethasone. In year 3+, LPL per patient would increase by 11.3% compared to year 2, irrespective of treatment type. Finally, IC were estimated at €2111.7 per patient per year, irrespective of treatment type and year since diagnosis.
Discussion
This study estimated the burden of DMO per patient per year since diagnosis and by type of treatment from a societal perspective. The average annual costs of DMO in year 1 was estimated at €18,773.6, €17,512.1, and €16,187.7 per patient treated with ranibizumab, aflibercept, and dexamethasone, respectively. This burden would be reduced to €15,783.1, €15,700.9, and €12,232.9 in year 2, and to €15,119.5, €15,043.0, and €12,789.8 in year 3+, respectively. In addition, a one-off cost of €484.5 associated with the diagnosis of DMO should be considered. DHC accounted for the greatest proportion of TC, independent of the year since diagnosis. In addition, LPL also contributed largely to TC.
There are hardly any comprehensive studies on the burden of DMO in Spain [8, 19]. Romero-Aroca et al. (2016) explored the direct costs of treatment of patients with DMO managed with anti-VEGF intravitreal injections, including medical visits, examinations, and interventions carried out for each patient during an 8-year follow-up [8]. The authors estimated a mean annual cost of €7153.6 per patient, which is in line with the DHC found in this study for patients treated with ranibizumab and aflibercept. However, in their study, only the first treated eye was considered in cost calculations, which may explain the slightly higher DHC found in the present study. In another study, Abraldes et al. (2016) estimated DHC and LPL associated with DMO [19]. In their study, DHC were estimated at €6271 per patient and year, but the costs of anti-VEGF intravitreal injections were not included. This can explain the somewhat lower DHC compared to the present study. Moreover, the cost of temporary and permanent work disability reported in Abraldes et al. (2016) was €393 and €7051 per patient per year, respectively—that is, almost double than the LPL costs found in our study. However, we considered different types of LPL (i.e., not only temporary and permanent work disability, which has high associated costs) including reduced working hours.
Some limitations should be considered when interpreting the findings of this study. Regarding the cost of diagnosis, it may have been underestimated, as only a single visit to specialised care for diagnostic tests was considered. Accordingly, the possibility of diagnostic delays which would increase the cost of diagnosis was not considered. Nevertheless, the cost of DMO diagnosis reported in the present study (€484.5) builds on previous reports which estimated the cost of DMO diagnosis in Spain at €132.2 per patient [8], as the present study accounted for additional diagnostic tests. Regarding DNHC associated with informal care, the only item considered was the time spent on accompanying the patient to medical visits, as no other data were found in the published scientific literature. Therefore, the actual costs of informal care may be higher than those reported here. Finally, the only information on IC identified from the scientific literature was the loss of utility for patients in their first year of treatment with ranibizumab, which was applied regardless of treatment type and year since diagnosis. Given disease progression, utility would be expected to decrease with time; however, treatment efficacy may compensate such loss.
This study provided a comprehensive estimate of the burden of DMO for the healthcare system, patients, and society at large. First of all, in addition to the commonly reported DHC, the present study included DNHC (e.g., costs associated with formal and informal care), LPL (e.g., earnings lost due to the impact of the disease), and IC (e.g., costs associated with the loss of quality of life). Second of all, differences in resource consumption between treatments, year since diagnosis, and disease progression since diagnosis were considered. Regarding disease progression, calculations incorporated the patient's level of eye involvement at diagnosis (unilateral or bilateral) and its expected evolution over the first 3 years of treatment. Consequently, some costs were expected to increase considerably from year 1 to year 3+. This was the case for LPL. However, in addition, a decrease in DHC was observed, which could be associated with a reduction in the number of visits, tests, and intravitreal injections, and hence AEs.
Conclusion
DMO is a highly prevalent disease that imposes a substantial burden on patients, the healthcare system, and society at large. Increasing DMO prevalence accentuates the need to develop healthcare strategies that prevent vision loss through early diagnosis and efficient treatment. This study provides a comprehensive analysis of the socio-economic burden of DMO, revealing potential benefits of prompt effective treatments that prevent disease progression.
Data Availability
The datasets may be available from the corresponding author upon reasonable request.
References
GBD 2019 Blindness and Vision Impairment Collaborators, Vision Loss Expert Group of the Global Burden of Disease Study. Causes of blindness and vision impairment in 2020 and trends over 30 years, and prevalence of avoidable blindness in relation to VISION 2020: the Right to Sight: an analysis for the Global Burden of Disease Study. Lancet Glob Health. 2021;9(2):e144–60.
Udaondo P, Parravano M, Vujosevic S, Zur D, Chakravarthy U. Update on current and future management for diabetic maculopathy. Ophthalmol Ther. 2022;11(2):489–502.
Schmidt-Erfurth U, Garcia-Arumi J, Bandello F, Berg K, Chakravarthy U, Gerendas BS, et al. Guidelines for the management of diabetic macular edema by the European Society of Retina Specialists (EURETINA). Ophthalmologica. 2017;237(4):185–222.
Figueira J, Henriques J, Carneiro Â, Marques-Neves C, Flores R, Castro-Sousa JP, et al. Guidelines for the management of center-involving diabetic macular edema: treatment options and patient monitorization. OPTH. 2021;15:3221–30.
Bhandari S, Gabrielle PH, Nguyen V, Daien V, Viola F, Bougamha W, et al. Dexamethasone implant for diabetic macular oedema: 1-year treatment outcomes from the fight retinal blindness! Registry Ophthalmol Ther. 2022;11(2):797–810.
Ehlers JP, Yeh S, Maguire MG, Smith JR, Mruthyunjaya P, Jain N, et al. Intravitreal pharmacotherapies for diabetic macular edema. Ophthalmology. 2022;129(1):88–99.
Gabrielle PH, Nguyen V, Creuzot-Garcher C, Arnold JJ, Mehta H, Duran MA, et al. Three-year treatment outcomes of aflibercept versus ranibizumab for diabetic macular edema data from the Fight Retinal Blindness! registry. Retina. 2022;42(6):1085–94.
Romero-Aroca P, de la Riva-Fernandez S, Valls-Mateu A, Sagarra-Alamo R, Moreno-Ribas A, Soler N, et al. Cost of diabetic retinopathy and macular oedema in a population, an eight year follow up. BMC Ophthalmol. 2016;4(16):136.
Chen E, Looman M, Laouri M, Gallagher M, Van Nuys K, Lakdawalla D, et al. Burden of illness of diabetic macular edema: literature review. Curr Med Res Opin. 2010;26(7):1587–97.
Pignot M, DiBonaventura M. The relationship between macular edema and health outcomes among patients with diabetes in Western Europe. Value in Health. 2014;17(7):A353–4.
Gonder JR, Walker VM, Barbeau M, Zaour N, Zachau BH, Hartje JR, et al. Costs and quality of life in diabetic macular edema: Canadian Burden of Diabetic Macular Edema Observational Study (C-REALITY). Journal of Ophthalmology. 2014;26(2014): e939315.
Ruiz-García A, Arranz-Martínez E, García-Álvarez JC, García-Fernández ME, Palacios-Martínez D, Montero-Costa A, et al. Prevalence of diabetes mellitus in Spanish primary care setting and its association with cardiovascular risk factors and cardiovascular diseases, SIMETAP-DM study. Clin Investig Arterioscler. 2020;32(1):15–26.
Romero-Aroca P, Sagarra-Alamo R, Baget-Bernaldiz M, Fernández-Ballart J, Méndez-Marin I. Prevalence and relationship between diabetic retinopathy and nephropathy, and its risk factors in the North-East of Spain, a Population-Based Study. Ophthalmic Epidemiol. 2010;17(4):251–65.
Teo ZL, Tham YC, Yu M, Chee ML, Rim TH, Cheung N, et al. Global prevalence of diabetic retinopathy and projection of burden through 2045. Ophthalmology. 2021;128(11):1580–91.
Abdallah W, Barakat M, Goldenberg D, Itty S, Dugel PU, Jamal K, et al. Assessing the efficacy and safety of intravitreal dexamethazone implant in treating macular edema in eyes with an incomplete response to anti-vascular endothelial growth factor agents. Invest Ophthalmol Vis Sci. 2019;60(9):98.
Advisory Committee. Burden of Disease Study of patients with Diabetic Macular Edema and patients with neovascular Age-Related Macular Degeneration in Spain. 2022.
Cervera E, De Andrés-Nogales F, Armadá F, Arias L, Oyagüez I, Martínez C. Budget impact analysis of dexamethasone intravitreal implant for the treatment of diabetic macular oedema. Farm Hosp. 2018;42(6):244–50.
Brown GC, Brown MM, Turpcu A, Rajput Y. The cost-effectiveness of ranibizumab for the treatment of diabetic macular edema. Ophthalmology. 2015;122(7):1416–25.
Abraldes MJ, Pareja A, Roura M, en representación del grupo del estudio OBSERVAR. Analysis of costs associated with the management and morbidity of diabetic macular oedema and macular oedema secondary to retinal vein occlusion. Arch Soc Esp Oftalmol. 2016;91(6):273–80.
Ruiz-Adame Reina M. P-01. CALIDAD DE VIDA EN PERSONAS CON EDEMA MACULAR DIABÉTICO. In: XXXVIII Jornadas de Economía de la Salud. Las Palmas de Gran Canaria: Asociación de Economía de la Salud; 2018.
Menezo M, Roca M, Menezo V, Pascual I. Intravitreal dexamethasone implant Ozurdex in the treatment of diabetic macular edema in patients not previously treated with any intravitreal drug: a prospective 12-month follow-up study. Curr Med Res Opin. 2019;35(12):2111–6.
Iglicki M, Busch C, Lanzetta P, Sarao V, Veritti D, Rassu N, et al. Vitrectomized vs non-vitrectomized eyes in DEX implant treatment for DMO—Is there any difference? the VITDEX study. Eye [Internet]. 2022 Jan 18 [cited 2022 May 27]; https://www.nature.com/articles/s41433-022-01931-9.
Rosenblatt A, Udaondo P, Cunha-Vaz J, Sivaprasad S, Bandello F, Lanzetta P, et al. A collaborative retrospective study on the efficacy and safety of intravitreal dexamethasone implant (Ozurdex) in patients with diabetic macular edema. Ophthalmology. 2020;127(3):377–93.
Arias Barquet L, Esteban Vega AI, García Arumí J, Hidalgo Vega Á, Ruiz Moreno JM, Sánchez Fierro J, et al. Objetivo DMAE. Situación actual y propuestas de mejora para la atención sociosanitaria de la DMAE [Internet]. Barcelona: Nephila Health Partnership, S.L.; 2020 [cited 2020 Oct 14]. https://www.novartis.es/sites/www.novartis.es/files/Objetivo-DMAE-Informe%20interactivo%20final%20280920.pdf. Accessed 14 Oct 2020.
Giocanti-Aurégan A, García-Layana A, Peto T, Gentile B, Chi GC, Mirt M, et al. Drivers of and barriers to adherence to neovascular age-related macular degeneration and diabetic macular edema treatment management plans: a multi-national qualitative study. PPA. 2022;16:587–604.
Mitchell P, Annemans L, Gallagher M, Hasan R, Thomas S, Gairy K, et al. Cost-effectiveness of ranibizumab in treatment of diabetic macular oedema (DME) causing visual impairment: evidence from the RESTORE trial. Br J Ophthalmol. 2012;96(5):688–93.
Instituto Nacional de Estadística, Ministerio de Sanidad, Servicios Sociales e Igualdad. Encuesta Nacional de Salud 2012 [Internet]. España: Ministerio de Sanidad, Servicios Sociales e Igualdad; 2012 [cited 2018 Nov 30]. http://www.mscbs.gob.es/estadEstudios/estadisticas/encuestaNacional/encuestaNac2011/MetodologiaENSE2011_12.pdf. Accessed 30 Nov 2018.
International Federation on Ageing, International Agency for the Prevention of Blindness, International Diabetes Federation. The Diabetic Retinopathy Barometer Report Global Findings [Internet]. Brussels, Belgium: International Diabetes Federation; 2020. Available from: DRBarometer.com. Accessed 5 May 2022.
Gamboa Moreno E, Mateo-Abad M, Ochoa de Retana García L, Vrotsou K, Del Campo PE, Sánchez Perez Á, et al. Efficacy of a self-management education programme on patients with type 2 diabetes in primary care: a randomised controlled trial. Prim Care Diabetes. 2019;13(2):122–33.
Ministerio de Trabajo y Economía Social. Real Decreto 817/2021, de 28 de septiembre, por el que se fija el salario mínimo interprofesional para 2021. [Internet]. 2021. https://www.boe.es/buscar/act.php?id=BOE-A-2021-15770. Accessed 10 Aug 2022.
Assumption. Burden of Disease Study of patients with Diabetic Macular Edema and patients with neovascular Age-Related Macular Degeneration in Spain. 2022.
Instituto Nacional de Estadística. Encuesta Anual de Estructura Salarial Año 2019 [Internet]. 2021 [cited 2022 Feb 16]. https://www.ine.es/dyngs/INEbase/operacion.htm?c=Estadistica_C&cid=1254736177025&menu=resultados&secc=1254736061996&idp=1254735976596#!tabs-1254736061996. Accessed 16 Feb 2022.
Ortega Eslava A, Marín Gil R, Fraga Fuentes MD, López-Briz E, Puigventós Latorre F. Guía de evaluación económica e impacto presupuestario en los informes de evaluación de medicamentos [Internet]. SEFH. Sociedad Española de Farmacia Hospitalaria; 2016 [cited 2019 Mar 18]. https://gruposdetrabajo.sefh.es/genesis/genesis/Documents/GUIA_EE_IP_GENESIS-SEFH_19_01_2017.pdf. Accessed 18 Mar 2019.
Byford S. Economic note: cost of illness studies. BMJ. 2000;320(7245):1335–1335.
Consejo General de Colegios Farmacéuticos. EYLEA 40 mg/ml 1 JERINGA PRECARGADASOLUCION INYECTABLE 90 microlitros [Internet]. BotPlus Web. 2022 [cited 2022 Apr 22]. https://botplusweb.portalfarma.com/botplus.aspx. Accessed 22 Apr 2022.
Consejo General de Colegios Farmacéuticos. EYLEA 40 MG/ML 1 VIAL SOLUCIÓN INYECTABLE 100 microlitros [Internet]. BotPlus Web. 2022 [cited 2022 May 20]. https://botplusweb.portalfarma.com/botplus.aspx. Accessed 22 May 2022.
Consejo General de Colegios Farmacéuticos. LUCENTIS 10 mg/ml 1 JERINGA PRECARGADA SOLUCION INYECTABLE 0,165 ml [Internet]. BotPlus Web. 2022 [cited 2022 Apr 22]. https://botplusweb.portalfarma.com/botplus.aspx. Accessed 22 Apr 2022.
Consejo General de Colegios Farmacéuticos. LUCENTIS 10 MG/ML 1 VIAL SOLUCIÓN INYECTABLE 0.23 ML + 1 AGUJA CON FILTRO [Internet]. 2022 [cited 2022 May 20]. https://botplusweb.portalfarma.com/botplus.aspx. Accessed 22 May 2022.
Consejo General de Colegios Farmacéuticos. OZURDEX 700 microgramos 1 IMPLANTE INTRAVITREO EN APLICADOR [Internet]. BotPlus Web. 2022 [cited 2022 Apr 7]. https://botplusweb.portalfarma.com/botplus.aspx. Accessed 7 Apr 2022.
Instituto Nacional de Estadística. Índices nacionales: general y de grupos ECOICOP [Internet]. INE. 2022 [cited 2022 Sep 16]. https://www.ine.es/jaxiT3/Tabla.htm?t=22553&L=0. Accessed 16 Sept 2022.
Acknowledgements
Medical Writing/Editorial Assistance
Angélica López-Angarita supported medical writing as an employee of Weber, a company that has received payments from Roche to conduct this study.
Funding
This study and the journal’s Rapid Service Fee were funded by Roche Farma S.A. The funder was not involved in the study design, data collection, analysis, interpretation of data, or the writing of this article.
Author information
Authors and Affiliations
Contributions
María Merino, Teresa Martín Lorenzo, Paulina Maravilla-Herrera, and Beatriz Gil Jiménez contributed to the study conception and design. María Merino, Teresa Martín Lorenzo, and Paulina Maravilla-Herrera acquired the data. José M. Ruiz-Moreno, María Gámez Lechuga, Pilar Calvo, María Merino, Teresa Martín Lorenzo, Paulina Maravilla-Herrera, Beatriz Gil Jiménez, and Maximino J. Abraldes analysed and interpreted the data. María Merino and Teresa Martín Lorenzo drafted the manuscript. José M. Ruiz-Moreno, María Gámez Lechuga, Pilar Calvo, María Merino, Teresa Martín Lorenzo, Paulina Maravilla-Herrera, Beatriz Gil Jiménez, and Maximino J. Abraldes critically revised and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
José M. Ruiz-Moreno, María Gámez Lechuga, Pilar Calvo, and Maximino J. Abraldes declare having received payments from Roche for participating as members of the advisory committee for this study. María Merino, Teresa Martín Lorenzo, and Paulina Maravilla-Herrera declare that they are employees of Weber, a company that has received payments from Roche to conduct this study. Beatriz Gil Jiménez declares to be an employee of Roche Farma, a company that has funded this study.
Ethical Approval
Due to the type of study, no clinical research ethics committee approval was required. Nevertheless, the present study conforms with the ethical principles of the Declaration of Helsinki. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. The four members of the advisory committee are authors of this manuscript and were informed that their advice would be used in the development of this article.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Ruiz-Moreno, J.M., Gámez Lechuga, M., Calvo, P. et al. Burden of Disease Study of Patients with Diabetic Macular Oedema in Spain. Ophthalmol Ther 13, 1937–1953 (2024). https://doi.org/10.1007/s40123-024-00959-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-024-00959-2