Abstract
Introduction
Neurotrophic keratopathy (NK) is a rare degenerative ocular disease that can be difficult to treat. There were no effective resolutive treatments for severe NK caused by ocular graft-versus-host disease (oGVHD) along with virus infection. To address this question, we designed a prospective cohort study to evaluate the efficacy and safety of topical recombinant human nerve growth factor (rhNGF) in patients with recalcitrant NK of oGVHD and viral infection.
Methods
This prospective cohort study enrolled patients with recalcitrant NK diagnosed with oGVHD and treated with rhNGF. Clinical evaluations included the range of epithelial defects, best corrected visual acuity, intraocular pressure, slit-lamp examination, and corneal fluorescein staining. Examinations of the central corneal thickness, corneal sensitivity, and nerve fiber regeneration were performed at each visit at 4, 8, 12, 20 weeks and 6 months, respectively, after initiating rhNGF treatment.
Results
All enrolled patients were diagnosed with NK at stage 2 (7 eyes, 63.6%) or stage 3 (4 eyes, 36.4%) and responded to rhNGF treatment. Five of 11 (45.5%) and 9 of 11 eyes (81.8%) achieved complete corneal epithelial healing after 4 and 8 weeks, respectively. All 11 eyes (100%) achieved complete corneal healing after 12 weeks. There was also a significant reduction in the corneal ulcer area during each visit (P < 0.001), as well as in the corneal fluorescein staining score (P < 0.010). There was a significant improvement in corneal sensation when compared to the baseline (P < 0.050).
Conclusion
Topical treatment with rhNGF effectively promoted the complete corneal healing of persistent epithelial defects and corneal ulcers in patients with recalcitrant NK in oGVHD and viral infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
The study addresses a significant clinical problem by investigating a novel treatment approach for a rare and challenging condition—neurotrophic keratopathy (NK)—in patients with ocular graft-versus-host disease (oGVHD) and viral infections. |
What was learned from the study? |
Patients with severe NK at stage 2 and 3 experienced a remarkable recovery in corneal health with recombinant human nerve growth factor (rhNGF) treatment, and the improvement was sustained over 6 months. The observations of corneal healing and regeneration of corneal nerve fibers are particularly notable. |
rhNGF treatment was well tolerated, providing important information regarding the safety of this novel therapy. |
Introduction
Neurotrophic keratopathy (NK), also known as neurotrophic keratitis, is a rare degenerative ocular disease. When the trigeminal innervation of the cornea is damaged, morphological and metabolic epithelial abnormalities progress, leading to NK [1, 2]. Severe NK may cause corneal thinning, scarring, neovascularization, or even corneal melting and perforation with irreversible visual loss [1, 3]. Various conditions, such as ocular herpetic infection, ocular or neurologic surgery, trauma, diabetes, and dry eye disease are associated with NK [4].
Graft-versus-host disease (GVHD) is a major complication following allogeneic hematopoietic stem cell transplantation, which occurs in many organs. It has been reported that about 60–90% of patients with chronic GVHD develop ocular complications [5,6,7]. Most patients with ocular graft-versus-host disease (oGVHD) experience severe dry eye [7], which may cause punctate epithelial keratopathy and painful epithelial erosions. Furthermore, this condition is often associated with recalcitrant NK, and may also lead to further corneal complications such as secondary infections, non-healing corneal ulcers, stromal thinning, necrosis, limbal stem cell deficiency, neovascularization, and corneal perforation [6].
Prior to 2018, there was no effective and specialized therapy available for NK, which has been identified as an orphan disease; specifically, there were no effective resolutive treatments for severe NK caused by GVHD along with virus infection. In the early stages of NK, discontinuation of toxic topical and systemic therapies and application of preservative-free lubricants are recommended. Moreover, bandage contact lenses, matrix-regenerating agents, and autologous serum may be used for treatment. Surgery is carefully considered in severe cases where amniotic membrane transplantation or tarsorrhaphy is performed. However, these treatments cannot improve the condition but only maintain the condition at a steady state for a short time. Topical recombinant human nerve growth factor (rhNGF, cenegermin, Oxervate, Dompè Farmaceutici, Milan, IT) was approved as a first-line treatment in 2018 by the European Medicines Agency and the US Food and Drug Administration [4] and was approved in 2020 in China.
A few clinical trials that reported the efficacy of rhNGF in promoting corneal healing in patients with NK [4, 8]. However, rhNGF had never been used in a specific group of people such as patients with oGVHD. In this study, we aimed to evaluate the efficacy and safety of rhNGF treatment for recalcitrant NK in patients with oGVHD.
Methods
Ethics
This prospective study included patients with recalcitrant NK of oGVHD. The patients were treated with rhNGF from January 2021 (the first patient enrolled) to December 2022 (the last follow-up) at the Peking University Third Hospital Ophthalmology Center. Written informed consent was obtained from each patient before participation in the study. The procedures were reviewed and approved by the Institutional Review Board of Peking University Third Hospital (IRB00006761-M2021101) and were compliant with the tenets of the Declaration of Helsinki.
Study Participants
The diagnosis of NK is mostly based on the clinical interpretation of corneal signs. It had been traditionally classified into three stages as described by Mackie (1995) and modified by Dua et al. [2] as follows: Stage 1 (mild) is characterized by the presence of corneal punctate keratopathy, which is frequently associated with epithelial hyperplasia, mild degrees of stromal edema, corneal neovascularization, and reduction of tear film breakup time. Stage 2 (moderate) is distinguished by the evidence of persistent epithelial defect (PED), which usually shows an oval or circular shape, involving most frequently the paracentral cornea. The epithelium surrounding the epithelial ulceration often appears edematous, cloudy, and poorly adherent with its consequent spontaneous detachment and progression of the extension of the epithelial defect. Stage 3 (severe) is characterized by stromal involvement and clinically presents as a frank corneal ulcer with stromal thinning that may progress to melting and perforation [9].
In this study, all the enrolled patients were diagnosed with NK at stage 2 (seven eyes, 63.6%) and stage 3 (four eyes, 36.4%); all had a history of chronic oGVHD. In addition, all of them were suspected to have a viral infection, while some were positive for the virus with laboratory tests. Before the study, all patients were treated with topical antibiotics, lubricants, therapeutic contact lenses, or even amniotic membrane transplantation for at least 4 weeks without clinical success, and therefore we classified the enrolled patients as having recalcitrant NK.
Examination and Treatment Procedures
The enrolled patients were treated with cenegermin 20 μg/mL (Dompé Farmaceutici Spa, Milan, Italy) one drop six times a day in the affected eye for 8 weeks. Therapeutic contact lens usage or amniotic membrane transplantation was discontinued during the study, along with the matrix-regenerating agents and autologous serum. The application of preservative-free lubricants (sodium hyaluronate, Santen Pharmaceutical Co., Ltd.) is still recommended, and antiviral medications (ganciclovir for systemic or local administration) or topical antibiotic eye drops (levofloxacin eye drops, Santen Pharmaceutical Co., Ltd.) are allowed to continue.
All the patients underwent a complete ophthalmic examination at each visit. It included measures of the best corrected visual acuity (BCVA), intraocular pressure (IOP) measured by Icare tonometer, slit-lamp examination, corneal fluorescein staining, and central corneal thickness (CCT) measured by anterior segment optical coherence tomography (AS-OCT). Corneal sensitivity was measured using a Cochet-Bonnet esthesiometer (Luneau Ophthalmologie, Chartres, France) as the cornea was contacted with a 6-cm-length monofilament nylon thread. The filaments were applied to superior, inferior, nasal, and temporal quadrants as well as the center. The thread was gradually shortened by 5 mm until a positive response was recorded. The longest filament length that induced a positive response was recorded as the threshold for corneal sensation. Patients were evaluated at baseline (before starting Cenegermin), and then at 4 weeks, 8 weeks, 12 weeks, 20 weeks, and 6 months after the initiation of rhNGF treatment. The safety and tolerability of the drug were estimated in this study using a questionnaire followed by their medical records.
Photo Processing
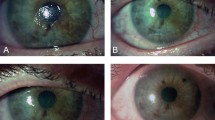
Anterior segment photos were analyzed using the semi-automated tracing program ImageJ, a freely available image processing and analysis platform distributed by the National Institutes of Health. We marked the persistent epithelial defect (PED) section and the cornea edge, and the software automatically calculated the area (Fig. 1).
To get the percentage of the persistent epithelial defect (PED) area in the cornea graft, we divided the PED area (manually circled in B, 754,820) by the cornea graft area (circled in A, 1,270,235), which allows us to determine the corneal epithelial healing precisely (percentage area of PED was 59.4%, 754,820/1,270,235)
Statistical Analysis
Statistical analysis was performed using IBM SPSS version 25.0 for Mac (IBM Corp., Armonk, NY, USA). Visual acuity data were collected from medical records and converted to a logarithm of the minimal angle of resolution (logMAR). Quantitative data were analyzed using Student’s t test or paired t tests. Paired t tests were used to analyze the area of corneal epithelial defect between baseline and each visit. Statistical significance was set at P value < 0.050 was considered significant.
Results
Characteristics of Patients
A total of nine eligible patients (11 eyes) were included in the study from January 24, 2021 (enrollment of the first patient) to July 11, 2022 (the date of the last person’s follow-up visit). The demographic features of the patients at baseline are presented in Table 1. The mean age was 35.6 ± 11 years old and 55.6% of patients were men. Eye involvement was bilateral in two patients.
Treatment Response
All nine patients (11 eyes) responded to rhNGF therapy. Specifically, complete corneal epithelial healing was achieved in 5 of 11 eyes (45.5%) after 4 weeks of rhNGF treatment and 9 of 11 (81.8%) after 8 weeks. All 11 eyes (100%) achieved complete corneal epithelial healing at 12 weeks. Even though the patients were only treated with rhNGF for 8 weeks, notably, significant reductions in the ulcer area were observed of each visit (P < 0.001, respectively) after the completion of rhNGF treatment (Fig. 2). During the study, we did not observe any eye that experienced recurrence of the epithelial defect within 6 months.
The corneal fluorescein staining score at baseline and the changes over time are shown in Fig. 3. The mean score decreased from 13.3 ± 2.7 at baseline to 10.4 ± 3.7 (P < 0.010), 7.9 ± 4.6 (P < 0.010), 4.6 ± 2.9 (P < 0.001), 0.7 ± 1.2 (P < 0.001), and 1.4 ± 1.9 (P < 0.001) at 1 week, 2 weeks, 4 weeks, 8 weeks, 12 weeks, and 6 months after treatment, respectively.
BCVA is one of the variables used to evaluate efficacy outcomes. We observed a slight decrease from logMAR 1.2 ± 0.6 at baseline to logMAR 0.9 ± 0.7 (P < 0.050) at the 8-week visit (Fig. 4A). However, there was no statistically significant difference in BCVA between the baseline and later follow-up visits. No difference in IOP was observed between baseline and follow-up visits (Fig. 4B). Central cornea thickness (CCT) is also an observational target, but there is no statistical significance in CCT between baseline and follow-up visits (Fig. 5). There was a significant increase in corneal sensation in the center of the cornea and average sensation in the superior, inferior, nasal, and temporal quadrants as well as the center compared to the baseline (Fig. 6).
Corneal sensitivity measured by Cochet-Bonnet esthesiometer at baseline and the changes over time. A Central corneal sensitivity; B the average corneal sensitivity of superior, inferior, nasal, and temporal quadrants as well as the center. Paired t test was used for comparisons of each visit. *P < 0.050; **P < 0.010; ***P < 0.001
Safety
The tolerance of rhNGF was satisfactory. No treatment discontinuation was required because of the side effects and no serious adverse drug effects were observed. Adverse events in patients were tolerable and reversible after discontinuation of rhNGF, including eye irritation (7/11, 63.6%), corneal leukoma (3/11, 27.3%), neovascularization (2/11, 18.2%), headache (5/11, 45.5%), and sleepiness (3/11, 27.2%).
Discussion
Patients with GVHD are a particular population suffering from severe dry eye, which may be associated with recalcitrant NK [6]. Previous studies have shown that the clinical manifestations of dry eyes due to GVHD were more severe than those with dry eyes of other etiologies. One study demonstrated that nerve fiber density was decreased in the oGVHD group whereas there was no effect in the dry eye syndrome group [5, 7]. In our study, all the patients had GVHD combined with ocular symptoms. In addition, all the patients were considered to have virus infection with positive PCR results for 64.6% eyes in anterior chamber aqueous humor, making these cases very difficult to treat and requiring more effort for complete cornea epithelial recovery. Previous studies have demonstrated that NGF plays a key role in the modulation of immune reaction, trophic support, healing of ocular surface, corneal sensitivity, and tear film function [10,11,12].
This study evaluated the efficacy of rhNGF treatment in patients with GVHD and NK. After 8 weeks of treatment and during the follow-up evaluations up to 6 months after the end of therapy, a notable increase in corneal epithelium recovery was observed in all patients (Fig. 7). However, not all patients recovered within 8 weeks (81.8%) of treatment with rhNGF, which was inconsistent with earlier studies [1, 3, 8] which reported that all patients could achieve complete corneal healing within 8 weeks of rhNGF treatment protocol. The two cases that did not recover within 8 weeks were classified as stage 3 NK with deep ulceration and stromal melting. One of the cases was a notable ulcer in the central cornea and accompanying cornea melting, with the central corneal thickness of (CCT) 370 μm.
A representative patient with GVHD, a 45-year-old man, underwent penetrating keratoplasty (PKP) surgery and suffered from NK with persistent epithelial defect (PED). After a month of traditional treatment with preservative-free lubricants and matrix-regenerating agents, there was still a large area of PED, so he began treatment with rhNGF. Anterior photo of the patient (A) and fluorescein stain (B) showed an 8 × 8 mm PED; (C) in vivo confocal microscopy (IVCM) showed no subbasal nerve fibers; 4 weeks after the treatment of rhNGF, the anterior segment photo (D) with fluorescein stain (E) showed a significant reduction of the PED area. (F) IVCM showed some slender subbasal nerve fibers. Corneal epithelium recovered within 8 weeks of treatment in this case. 6 months later, the anterior segment photo (G) showed the integrity of corneal epithelium with no fluorescein stain (H), while the IVCM (I) showed a total restoration of the subbasal nerve fibers
Patients with oGVHD usually underwent severe dry eye, and corneal epithelial defects were almost impossible to resolve [6, 13]. The epithelial defect healed in all patients and no patients experienced recurrence during the 6 months follow-up after completion of rhNGF treatment. However, we found that some patients had increased corneal fluorescein scores after 6 months without statistical significance. Dry eye conditions and chronic ocular surface inflammation may cause the increased score of fluorescein staining, which we attributed it to abnormal connections of corneal epithelial cells. Therefore, attention should be paid to the ocular surface of patients with GVHD accompanied with recalcitrant NK throughout the therapy and focus on controlling dry eye and ocular surface inflammation [5, 14, 15], even after the corneal epithelium has been recovered.
We observed that three eyes developed corneal leukoma at the end of the study and found that the process of corneal epithelial healing followed three steps: first, recovery of stroma with fibrous tissue hyperplasia and increase in corneal thickness; second, the corneal epithelium began to grow but was accompanied by neovascularization; third, the cornea healed with reduced inflammation on the ocular surface but was followed by corneal leukoma.
One patient was diagnosed with GVHD and CMV infection in both eyes and suffered from PED and stromal melting. The patient was then treated with rhNGF. Finally, his corneal defect recovered and he avoided corneal perforations or even emergency surgery. However, the developed keratoleukoma after the treatment; therefore, in order to improve his visual acuity, the patient underwent penetrating keratoplasty (PKP). We were concerned that he might develop PED again after the PKP surgery; however, his epithelium grew rapidly and recovered in only 2 weeks without using rhNGF (Fig. 8). This case provides objective evidence that nerve growth continues to increase beyond the end of the therapy.
Representative images of a case of NK and GVHD, a 27-year-old male subject with CMV(+) in both eyes. (A) Anterior segment photo with fluorescein stain showed an 8 × 9 mm ulcer in the center of the cornea (B). Reduction of epithelial defect area and complete healing at week 8 with the treatment of rhNGF. (C) 6 months after treatment, he suffered from keratoleukoma. (D) 2 weeks after PKP surgery, the corneal epithelium was completely healed without using rhNGF
Although three of the study eyes developed keratoleukoma and corneal neovascularization after 6 months of treatment with rhNGF, it has a protective effect to some extent. Persistent epithelial defect is a serious complication which should be rapidly managed to avoid corneal perforation [16]. When the corneal ulcer is close to perforation, rhNGF plays a certain role in accelerating corneal stroma repair. In addition, it can stabilize and reduce the inflammation of the ocular surface, which provides advantageous conditions for future corneal transplantation surgery. However, no cases in stage 2 were observed of neovascularization or corneal leukoma. Therefore, we believe that the application of rhNGF in patients with GVHD is safe and should be performed as early as possible.
However, on the basis of the current study, we still do not know whether the combination of rhNGF and steroid therapy can prevent keratoleukoma in these patients. More cases are required to confirm the effectiveness of this combination therapy.
BCVA is one of the variables used to evaluate efficacy outcomes. As we mentioned above, a slight decrease was observed from logMAR 1.2 ± 0.6 at baseline to logMAR 0.9 ± 0.7 (P < 0.05) at the 8-week visit, indicating an improvement in visual acuity, but no statistically significant differences in BCVA were observed between the baseline and later follow-up visits. The potential reasons were that some patients developed corneal leukoma which seriously affects their vision or that the poor baseline visual acuity of patients and limited sample size affected the statistical results.
In the relevant clinical studies of the application of rhNGF to treat neurotrophic keratitis, persistent epithelial defect repair and the improvement of the patient’s corneal sensation were observed [17]. Corneal nerve fibers were also significantly increased after rhNGF treatment [18]. This finding was associated with improved corneal sensitivity and regeneration of the subbasal nerve plexus. This phenomenon has also been observed in our study. Even though the patient with GVHD underwent PKP surgery and all subbasal nerves had been cut off, we found regeneration of corneal nerve fibers 6 months after stopping treatment of rhNGF (Figs. 7,8).
GVHD injury to the corneal surface is caused by physical or chemical insults that induce damaged cells and locally infiltrating immune cells to release large numbers of signal molecules. These released molecules provoke sustained firing of nociceptors, a lowered stimulatory threshold, and stronger impulse transmission following suprathreshold stimulation. These combined factors collectively contribute to peripheral sensitization experienced as persistent irritation or pain, and the pain is exaggerated or felt more severely than it should be [18]. This likely explained why we observed a higher rate of eye irritation (7/11, 63.6%) in patients with GVHD than in patients with other types of NK. Eye irritation is mild, and transient and acceptable, without requiring other treatment. In our study, eye irritation mainly appeared in the second week after using rhNGF, it gradually disappeared after 2–3 weeks. No discontinuation of treatment was required owing to side effects. The tolerance of rhNGF was satisfactory.
However, our study had some limitations. First and most importantly, the sample size was relatively small and inadequate for more detailed analyses and comparisons. However, patients with severe NK caused by oGVHD and along with virus infection are challenging to treat. Patients suffered a lot with poor prognosis. This novel treatment will greatly benefit them. Second, we did not set a control group as a result of the rare nature of GVHD combined with recalcitrant NK and the lack of an effective comparator treatment.
Conclusion
This prospective clinical trial provides multiple lines of evidence suggesting that topical treatment with rhNGF was effective in promoting complete corneal healing of persistent epithelial defects and corneal ulcers in patients with recalcitrant NK. Treatment with rhNGF was well tolerated and can help the recovery of corneal epithelium and subbasal corneal nerve regeneration. Although some patients developed keratoleukoma after 6 months, it provides suitable conditions for future corneal transplantation.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Di Zazzo A, Varacalli G, Mori T, Coassin M. Long-term restoration of corneal sensitivity in neurotrophic keratopathy after rhNGF treatment. Eur J Ophthalmol. 2022;32(1):Np15-np18.
Dua HS, Said DG, Messmer EM, et al. Neurotrophic keratopathy. Prog Retin Eye Res. 2018;66:107–31.
Deeks ED, Lamb YN. Cenegermin: a review in neurotrophic keratitis. Drugs. 2020;80(5):489–94.
Pflugfelder SC, Massaro-Giordano M, Perez VL, et al. Topical recombinant human nerve growth factor (cenegermin) for neurotrophic keratopathy: a multicenter randomized vehicle-controlled pivotal trial. Ophthalmology. 2020;127(1):14–26.
Giannaccare G, Pellegrini M, Bernabei F, Scorcia V, Campos E. Ocular surface system alterations in ocular graft-versus-host disease: all the pieces of the complex puzzle. Graefes Arch Clin Exp Ophthalmol. 2019;257(7):1341–51.
Munir SZ, Aylward J. A review of ocular graft-versus-host disease. Optom Vis Sci. 2017;94(5):545–55.
Tepelus TC, Chiu GB, Maram J, et al. Corneal features in ocular graft-versus-host disease by in vivo confocal microscopy. Graefes Arch Clin Exp Ophthalmol. 2017;255(12):2389–97.
Mastropasqua L, Lanzini M, Dua HS, et al. In vivo evaluation of corneal nerves and epithelial healing after treatment with recombinant nerve growth factor for neurotrophic keratopathy. Am J Ophthalmol. 2020;217:278–86.
Mastropasqua L, Nubile M, Lanzini M, Calienno R, Dua HS. In vivo microscopic and optical coherence tomography classification of neurotrophic keratopathy. J Cell Physiol. 2019;234(5):6108–15.
Lambiase A, Sacchetti M, Bonini S. Nerve growth factor therapy for corneal disease. Curr Opin Ophthalmol. 2012;23(4):296–302.
Labetoulle M, Baudouin C, Calonge M, et al. Role of corneal nerves in ocular surface homeostasis and disease. Acta Ophthalmol. 2019;97(2):137–45.
Pan Y, Liu F, Qi X, Hu Y, Xu F, Jia H. Nerve growth factor changes and corneal nerve repair after keratoplasty. Optom Vis Sci. 2018;95(1):27–31.
Belmonte C, Acosta MC, Merayo-Lloves J, Gallar J. What causes eye pain? Curr Ophthalmol Rep. 2015;3(2):111–21.
Nair S, Vanathi M, Mukhija R, Tandon R, Jain S, Ogawa Y. Update on ocular graft-versus-host disease. Indian J Ophthalmol. 2021;69(5):1038–50.
Ogawa Y, Kawakami Y, Tsubota K. Cascade of inflammatory, fibrotic processes, and stress-induced senescence in chronic GVHD-related dry eye disease. Int J Mol Sci. 2021;22(11):6114.
Trinh T, Santaella G, Mimouni M, et al. Assessment of response to multimodal management of neurotrophic corneal disease. Ocul Surf. 2021;19:330–5.
Ahuja AS, Bowden FW 3rd, Robben JL. A novel treatment for neurotrophic corneal ulcer using topical cenegermin (OXERVATE™) containing recombinant human nerve growth factor. Cureus. 2020;12(11):e11724.
Pedrotti E, Bonacci E, Chierego C, et al. Eight months follow-up of corneal nerves and sensitivity after treatment with cenegermin for neurotrophic keratopathy. Orphanet J Rare Dis. 2022;17(1):63.
Funding
This study was supported by fund of Investigator Initiated Research 2021–1211 of Dompé farmaceutici S.p.A. The journal’s Rapid Service fee was funded by Peking University Third Hospital.
Author information
Authors and Affiliations
Contributions
Yi Qu designed the study. Yi Qu, Rongmei Peng, Bohao Hu and Siyi Ma collected the data. Shaofeng Gu and Zheng Liu helped check the data. Yi Qu, Rongmei Peng, and Jing Hong contributed to the interpretation of the results and critical revision of the manuscript. All the authors have read and approved the final manuscript. Jing Hong is the study guarantor.
Corresponding author
Ethics declarations
Conflict of Interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Ethical Approval
The study was approved by the Peking University Third Hospital Medical Science Research Ethics Committee (IRB00006761-M2021101) and was conducted in compliance with the principles of the Declaration of Helsinki. Written informed consent was obtained from all patients.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Qu, Y., Peng, R., Hu, B. et al. A New Treatment for Recalcitrant Neurotrophic Keratopathy of Ocular Graft-Versus-Host Disease with Virus Infection. Ophthalmol Ther 13, 469–479 (2024). https://doi.org/10.1007/s40123-023-00857-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-023-00857-z