Abstract
Introduction
To evaluate the impacts of phacoemulsification preoperative and intraoperative factors on postoperative subfoveal choroidal thickness (SFCT).
Methods
This prospective interventional study was conducted on patients undergoing uneventful phacoemulsification with posterior chamber intraocular lens (IOL) implantation at the private clinic Ophthalmica Eye Institute, in Thessaloniki, Greece. Forty-six eyes of 46 patients were included in the study. Operative time (OT), phacoemulsification time (PT), effective phacoemulsification time (EPT) and phacoemulsification power (PP) were recorded for each patient. All patients received dexamethasone and nepafenac drops for 4 weeks postoperatively. Choroidal thickness (CT) at five anatomical locations was measured preoperatively and at 1 week, 1 month and 3 months postoperatively.
Results
Subfoveal choroidal thickness significantly increased over baseline thickness at 1 week postoperatively, returned to preoperative levels at 1 month and increased again at 3 months. Changes at 1 week and 3 months postoperatively correlated to baseline choroidal thickness (p = 0.023 and p = 0.011, respectively). Spherical equivalent (SE) inversely correlated to SFCT throughout the entire follow-up period (baseline p = 0.044, 1-week p = 0.011, 1-month p = 0.013, 3-month p = 0.018). EPT was the most significant determinant of increased SFCT at all time points (1-week p = 0.011, 1-month p = 0.01, 3-month p = 0.015). PT and PP significantly correlated to the SFCT increase at 3 months postoperatively (p = 0.033 and p = 0.043, respectively). OT was not associated with a change in SFCT at any time point (p > 0.05).
Conclusion
Phacoemulsification can alter choroidal thickness until at least 3 months after surgery. Preoperative and intraoperative factors, notably SE, baseline SFCT, PT, PP and especially EPT, can influence postoperative CT increase.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
The study aimed to assess the effects of preoperative and intraoperative factors during phacoemulsification on postoperative subfoveal choroidal thickness (SFCT) and investigate a possible association between cataract surgery and the development or progression of age-related macular degeneration. |
What was learned from the study? |
SFCT significantly increased 1 week after surgery, returned to preoperative levels after 1 month and then increased again 3 months postoperatively. These changes were linked to the baseline choroidal thickness. |
Spherical equivalent (SE) was found to inversely correlate with SFCT throughout the entire follow-up period, and effective phacoemulsification time (EPT) was identified as the most significant determinant of increased SFCT at all time points. |
Phacoemulsification time (PT) and phacoemulsification power (PP) were significantly correlated with the increase in SFCT 3 months postoperatively, while operative time did not show any association with changes in SFCT. |
Based on the results, phacoemulsification can alter choroidal thickness for up to 3 months after surgery, and preoperative and intraoperative factors, particularly SE, baseline SFCT, PT, PP, and especially EPT, can influence the postoperative increase in choroidal thickness. |
Introduction
Phacoemulsification cataract surgery is currently the most widely performed intraocular procedure [1, 2]. Posterior segment complications, such as pseudophakic cystoid macular edema and worsening of diabetic retinopathy, may occur even after uneventful cataract surgery, thus compromising postoperative functional outcome. Moreover, a possible association between cataract surgery and the development or progression of age-related macular degeneration (AMD) has been suggested, even though many epidemiological trials have failed to demonstrate any such connection [3,4,5,6,7,8,9,10,11]. Factors that could potentially trigger macular injury following phacoemulsification include intraoperative light exposure, upregulation of free radicals, release of growth factors and prostaglandin and postoperative changes in choroidal thickness (CT) [12, 13].
With the advent of the enhanced-depth imaging (EDI) technique for spectral-domain optical coherence tomography (SD-OCT), which enables detailed examination of choroidal stromal and vascular structures in vivo, increasing interest has focused on the effects of cataract surgery on choroidal thickness and vascularity [2, 14, 15]. Many studies have demonstrated an increase in CT after phacoemulsification [1, 16,17,18]. The authors of a 2018 meta-analysis concluded that subfoveal choroidal thickness (SFCT) significantly increases from 1 week until at least 3 months postoperatively [14]. Similar studies have documented a sustained increase in SFCT for 6 months after surgery [19,20,21]. To date, only a few studies have comprehensively investigated the possible effects of preoperative and intraoperative parameters on postoperative CT. The aim of this study was to demonstrate the changes in SFCT after uneventful phacoemulsification and their correlation with clinical and surgical potential determinants.
Methods
Forty-six patients (46 eyes) undergoing elective phacoemulsification with intraocular lens implantation for senile cataracts at the Ophthalmica Eye Institute, Thessaloniki, Greece, were enrolled in this prospective interventional study over 3 months. Ethics approval was obtained from the Ophthalmica Eye Institute Ethics Committee, under reference number 102021/007, and the study protocol adhered to the Declaration of Helsinki. Written consent was obtained from all subjects participating in this survey. This study does not contain personal data of any individual person. Patients were screened for the following exclusion criteria: age < 50 years; spherical equivalent (SE) exceeding ± 5.00 diopters (D); axial length (AL) > 26.5 mm; previous intraocular surgery or laser treatment; any other intraocular disorder or systemic disease with a potential to affect the retina or choroid, including uveitis, macular degeneration, diabetes mellitus and hypertension; current anti-inflammatory drug intake; and low image quality in SD-OCT with Spectralis® (Heidelberg Engineering GmbH, Heidelberg, Germany) due to dense cataract or poor fixation. Patients having intraoperative complications, including posterior capsule rupture, vitreous loss and iris prolapse, or postoperative complications, such as macular edema, were also excluded from the study.
Spectral-Domain Optical Coherence Tomography
The Spectralis SD-OCT diagnostic imaging platform on EDI mode was used to determine SFCT after pupil dilation. Using the eye-track function, 100 frames were obtained and averaged. A 6-mm horizontal section running through the fovea was selected for measurement. The calipers in the Heidelberg Eye Explorer software v.1.10.2.0 (Heidelberg Engineering GmbH) were positioned perpendicularly to the outer surface of the retinal pigment epithelium (inner hyperreflective line) and the inner border of the sclera (outer hyperreflective line). Measurements were manually taken by two masked investigators at five anatomical locations: subfoveal and at a 1500-μm radius to the four cardinal directions: temporal, nasal, superior and inferior (Fig. 1). Values obtained by the two investigators were compared and averaged. In the event of discrepancies > 3 μm, a third masked investigator performed the decisive measurement.
Preoperative Evaluation
A thorough preoperative ophthalmic examination was performed for all recruited patients, comprising automated refractometry (KR-8900 autorefractor keratometer; Topcon Corp., Tokyo, Japan), logMAR best-corrected visual acuity (BCVA), slit-lamp examination, dilated fundoscopy, applanation tonometry and cataract grading according to the Lens Opacities Classification System III (LOCS III) [22]. Biometry for assessment of AL and anterior chamber depth was carried out using partial coherence interferometry (Allegro Wavelight BioGraph; Alcon Laboratories Inc., Fort Worth, TX, USA) and ultrasound (US-1800 Echoscan; NIDEK, Gamagori, Japan). Central corneal thickness was measured with the EM-3000 specular microscope (Tomey Corp., Nishi-ku, Nagoya, Japan) and considered in the intraocular pressure (IOP) evaluation.
Surgery
Preoperative mydriasis was obtained through the instillation of topical tropicamide 1% and phenylephrine hydrochloride 5% drops. Topical proparacaine hydrochloride 0.5% drops were used for anesthesia. Phacoemulsification was carried out by four experienced cataract surgeons (PT, MB, SA, AV) following a standardized technique through a 2.2-mm clear corneal incision. All operations were performed using the Stellaris PC platform (Bausch & Lomb, Rochester, NY, USA), and all patients received a one-piece acrylic intraocular lens implanted in the capsular bag and 0.1 ml of 1% intracameral cefuroxime at the end of the procedure. Total operative time (OT), phacoemulsification time (PT), effective phacoemulsification time (EPT) and average phacoemulsification power (PP) were recorded for each case.
The postoperative treatment regimen consisted of topical preservative-free dexamethasone 0.1% drops 4 times daily, tapered over 4 weeks; preservative-free ofloxacin 0.3% drops 4 times daily for 2 weeks; and nepafenac 0.1% drops 3 times daily for 4 weeks.
Postoperative Follow-up
Patients were examined at 1 week, 1 month and 3 months postoperatively. BCVA, anterior chamber cellular reaction and IOP were recorded on each visit, and Spectralis SD-OCT EDI was performed without mydriasis during the morning hours.
Statistics
The average CT values at the five anatomical locations (central, superior, inferior, nasal, temporal), defined as average SFCT (ASFCT), were calculated and analyzed. All values are expressed as the mean ± standard deviation (SD). Statistical analysis was performed with SPSS software for Windows (version 20.0; SPSS IBM Corp., Armonk, NY, USA). The distribution of variables was examined using frequency tables and descriptive statistics. Box and whisker plots and histograms were used to display the different data distributions. Parametric methods, including independent Student’s t test, analysis of variance and linear regression, were used for normally distributed variables. Non-parametric tests, such as Mann–Whitney U and Wilcoxon signed rank tests, were used for variables that did not show normality. The relationship between patients’ demographics, intraoperative phacoemulsification parameters and postoperative changes in CT at different time points was examined using the Spearman and Pearson rank correlation test. All tests of association were considered statistically significant if p ≤ 0.05.
Results
Preoperative Data
A total of 46 eyes of 46 Caucasian patients (25 males, 21 females) with a mean (± SD) age of 72 ± 9 years, who had undergone uncomplicated phacoemulsification, were included in the study. Baseline patient characteristics and preoperative SFCT at the five measurement sites are provided in Table 1.
Intraoperative Data
Surgical parameter values are shown in Table 2. There were no intraoperative complications, hence none of the 46 recruited subjects had to be excluded from the study.
Follow-up Data
Postoperative mean (± SD) IOP at 1 week was 14.9 ± 2.96 mmHg and did not correlate to SFCT (p > 0.05). Anterior chamber inflammation was minimal and resolved within the first month in all cases. Mean BCVA improved from 0.46 ± 0.33 at baseline to 0.02 ± 0.06 at the final follow-up examination. Figure 2 shows the change in CT over time for each of the five subfoveal locations measured. A similar trend is displayed in Fig. 3 for the ASFCT, with a significant increase of 8.3 ± 28.6 μm (p = 0.05) over baseline values at 1 week after surgery. A reduction towards preoperative levels (231.0 ± 70 vs. 227.5 ± 70 μm; p = 0.36) was observed at 1-month follow-up. However, a subsequent elevation in ASFCT occurred at 3 months postoperatively, raising it to 238.3 ± 66 μm (p = 0.077).
Correlation with Preoperative Factors
The mean change in ASFCT at 1 week and 3 months after surgery was significantly correlated to baseline ASFCT (p = 0.023 and p = 0.011, respectively). The mean ASFCT at baseline and throughout the whole postoperative period was found to be inversely correlated to the SE (p = 0.044 at baseline, p = 0.011 at 1 week, p = 0.013 at 1 month, p = 0.018 at 3 months).
Correlation with Intraoperative Factors
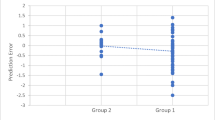
Of the four phacoemulsification parameters evaluated, EPT was found to be the most significant determinant of increased average SFCT at all time points (baseline p = 0.007, 1-week p = 0.011, 1-month p = 0.01 and 3-month p = 0.015). Statistical analysis also revealed a trend of correlation between PT and average SFCT, reaching statistically significant levels (p = 0.033) at 3 months following surgery. PP was significantly correlated to average SFCT increase at 3 months postoperatively (p = 0.05), whereas OT was not associated with a change in CT at any time point (p > 0.05). The correlation between each surgical parameter and mean CT at 3 months postoperatively is shown through scatterplots in Fig. 4.
Discussion
Cataract surgery with phacoemulsification is the most commonly performed surgical procedure in Western countries, featuring excellent results and a favorable safety profile [22]. Nonetheless, it has been linked with the deterioration of preexisting retinal disease, particularly diabetic retinopathy, while an ongoing discussion has been sparked regarding its effects on age-related macular degeneration [17, 23, 24]. The advent of EDI-OCT has enabled detailed observation of choroidal structure, revealing CT alterations in a variety of ocular conditions, including postoperative changes [1,2,3,4]. In this prospective interventional study, we monitored changes in CT for a total of 3 months following uncomplicated phacoemulsification. We also investigated the possible effect of non-modifiable—ocular—as well as modifiable—intrinsically surgical—parameters on CT increase.
SFCT significantly increased at 1 week, 1 month and 3 months after surgical intervention. This observation is consistent with findings reported in the available literature, which show an increase of SFCT following phacoemulsification, although not always reaching statistical significance [1, 3, 4, 7]. The 2018 meta-analysis by Zeng et al., which involved 13 studies with a total of 802 eyes, validated this observation, providing evidence of a statistically significant SFCT increase at least until the third postoperative month [4]. Furthermore, we noted a particular trend in SFCT variation, which decreased in the first postoperative month, compared to the first week, and increased in the third month. Most authors also report a SFCT peak during the third postoperative month [3, 25,26,27], although others detected a peak in SFCT earlier in follow-up [1, 4]. In our sample, the relative reduction in postoperative SFCT coincided with the withdrawal of topical nonsteroidal anti-inflammatory drugs (NSAIDs). According to the subgroup analysis by Zeng et al., patients who were not prescribed NSAIDs after phacoemulsification developed significant changes in SFCT, as opposed to patients who were treated with NSAIDs [4]. Therefore, we hypothesized that the two events might be related, although our study was not designed to answer this question.
The mechanism accounting for the postoperative change in CT is not clear. The release of proinflammatory cytokines and prostaglandins during cataract surgery and the subsequent disruption of the outer blood-retinal barrier that leads to anterior segment inflammation appear to involve the choroid [13]. However, this may not account for the sustained increase observed for months following phacoemulsification. Another proposed mechanism is light exposure during surgery, which induces a metabolic activation of the retinal pigment epithelium, potentially leading to angiogenesis [5, 6]. Whether the change in CT plays a role in postoperative macular complications has not been proved.
The influence of ocular parameters on SFCT changes, particularly IOP, has been repeatedly investigated [3, 8, 12]. Lower postoperative IOP has been linked with an SFCT incline, perhaps through positive ocular blood perfusion pressure [9]. In contrast, we did not detect a significant correlation between IOP and SFCT. Increased AL has also been shown to correlate with diminished SFCT changes [12, 28, 29]. In the present study, low SE and thicker baseline choroid were predictive of greater increases in postoperative SFCT, but not AL. We attribute the latter to the study design, which excluded eyes with AL longer than 26.5 mm, resulting in a sample with mostly average AL. This was aimed at minimizing confounding factors, enabling us to focus on the effects of intraoperative parameters on SFCT.
While there are existing reports on the impact of uncomplicated cataract surgery on CT, the present work provides a more detailed exploration of this relationship, contributing to the ongoing discussion in this area of research. This study demonstrated a strong correlation between increased EPT and elevated SFCT at all postoperative visits. Significant, though somewhat weaker associations between PT, PP and SFCT were also observed, but only in the third postoperative month. The significance of EPT was recently highlighted by Ma et al., who reported that this was the only predictive factor for increased SFCT at the third postoperative month in the classic phacoemulsification group [18]. Jiang et al. measured CT changes in a cohort of 100 eyes and reported no association with PT, but they did find a statistically significant effect of PP in three of the seven anatomical locations measured in their study [30]. Similarly to the 2014 study by Ohsugi et al., we found no correlation between CT and OT [10]. Intraoperative parameters are of particular interest, since the surgeon can control them. This may be especially important in cases where SFCT is already increased, such as in central serous chorioretinopathy, and in cases where the elevation in SFCT may correlate with disease progression, including severe diabetic retinopathy and age-related macular degeneration [17, 31,32,33]. In such cases, an approach that may reduce EPT would be the early indication of surgery, where minimal lens hardness could allow for the use of less ultrasound energy, thereby limiting postoperative inflammation. In cases where the early indication of surgery is no longer possible, adjustment of the postoperative drug regimen, keeping the use of anti-inflammatory drugs for a longer time, may be advisable.
We acknowledge certain limitations of our study. First, the relatively small sample size and short follow-up period may limit the generalizability of our findings. Second, the lack of a laser flare meter in our institution prevented us from obtaining more objective and quantifiable data on anterior chamber inflammation. Third, the surgeries were performed by multiple surgeons, which, despite their similar level of experience and surgical techniques, could introduce some degree of variability.
Furthermore, lifestyle factors, such as smoking and alcohol consumption, were not evaluated in this study. These factors could potentially influence CT and their inclusion might have added another layer of understanding to our results. Future studies could consider incorporating these factors to provide a more comprehensive analysis.
The use of NSAIDs in the postoperative period, which is a standard protocol in our institution, might have influenced the postoperative CT. Although this reflects the real-world clinical scenario, it might have masked the direct effects of the surgery on the choroid. Future studies could consider a design where the use of NSAIDs is controlled to better isolate the effects of the surgery itself.
Our study focused on CT as the primary outcome, and we did not evaluate the choroidal vascular index (CVI). The CVI could potentially provide more stable and insightful data. However, due to technical constraints and institutional IT policies, we were unable to use third-party software required for CVI measurements. We acknowledge this as a limitation and suggest that future studies could consider including CVI measurements provided they have the necessary resources and permissions to do so.
Despite these limitations, strict exclusion criteria were applied to prevent the influence of confounding factors. This included the exclusion of myopic eyes, which tend to have a high AL and hence a thinner choroid [25, 34]. The mean SE in our sample is low, which mitigates potential deviations in the analysis due to variations in AL. Furthermore, we excluded patients with diabetes based on recent reports of reduced CT after cataract surgery in diabetic patients, a fact attributed to perioperative hypoxia [16, 17, 29].
Moreover, we believe that a 5-point measurement of SFCT provides a reliable estimate of average SFCT, more representative than the solely central SFCT considered in other studies. Finally, this study expresses a comprehensive focus on surgical parameters as possible causative factors for increased CT, an aspect only partially covered by relevant studies.
Conclusion
Phacoemulsification is a widely performed procedure, with progressively increasing safety regarding both techniques and materials. However, specific surgical parameters, particularly ultrasound energy, carry an inherent potential for injury to sensitive intraocular tissues. The demonstration of possible correlations between surgical parameters and postoperative increase in CT may contribute to a deeper understanding of the pathophysiology of macular complications after phacoemulsification. Considering the potential association between SFCT increase and progression of retinal disease, keeping EPT as low as possible may be advisable. This could be achieved by offering early intervention to eyes considered to be at high risk for CT-associated complications. Further research is needed to clarify the possible correlations between choroidal changes and macular complications following phacoemulsification.
Data Availability
All data and material involved in this study are available upon request.
References
Abdellatif MK, Ebeid WM. Variations in choroidal and macular thickness maps after uneventful phacoemulsification. Semin Ophthalmol. 2018;33:719–25.
Roh HC, Park CY, Oh J-H. The variation of choroidal thickness and refractive error after cataract surgery. J Korean Ophthalmol Soc. 2016;57:924.
Yilmaz T, Karci AA, Yilmaz İ, et al. Long-term changes in subfoveal choroidal thickness after cataract surgery. Med Sci Monit. 2016;22:1566–70.
Noda Y, Ogawa A, Toyama T, et al. Long-term increase in subfoveal choroidal thickness after surgery for senile cataracts. Am J Ophthalmol. 2014;158:1362.
Arany Z, Foo SY, Ma Y, et al. HIF-independent regulation of VEGF and angiogenesis by the transcriptional coactivator PGC-1α. Nature. 2008;451:1008–12.
Ueta T, Inoue T, Yuda K, et al. Intense physiological light upregulates vascular endothelial growth factor and enhances choroidal neovascularization via peroxisome proliferator-activated receptor γ coactivator-1α in mice. Arterioscler Thromb Vasc Biol. 2012;32:1366–71.
Ibrahim AM, Elgouhary SM, Nassar MK, et al. Changes in choroidal thickness after cataract surgery. Semin Ophthalmol. 2018;33:664–70.
Bayhan SA, Bayhan HA, Muhafiz E, et al. Evaluation of choroidal thickness changes after phacoemulsification surgery. Clin Ophthalmol. 2016;10:961–7.
Mansberger SL, Gordon MO, Jampel H, et al. Ocular Hypertension Treatment Study Group. Reduction in intraocular pressure after cataract extraction: the ocular hypertension treatment study. Ophthalmology. 2012;119:1826–31.
Ohsugi H, Ikuno Y, Oshima K, et al. 3-D choroidal thickness maps from EDI-OCT in highly myopic eyes. Optom Vis Sci. 2013;90:599–606.
Abd El-Mawgoud S, Arfeen S, El-Gendy N, et al. Early changes of choroidal and macular thickness after uneventful phacoemulsification surgery. Delta J Ophthalmol. 2018;19:106.
Ohsugi H, Ikuno Y, Ohara Z, et al. Changes in choroidal thickness after cataract surgery. J Cataract Refract Surg. 2014;40:184–91.
Xu H, Chen M, Forrester JV, et al. Cataract surgery induces retinal proinflammatory gene expression and protein secretion. Investig Ophthalmol Vis Sci. 2011;52:249–55.
Zeng S, Liang C, He Y, et al. Changes of subfoveal choroidal thickness after cataract surgery: a meta-analysis. J Ophthalmol. 2018;2018:2501325. https://doi.org/10.1155/2018/2501325.
Der HJ, Xirasagar S, Kao LT, et al. Neovascular age-related macular degeneration is associated with cataract surgery. Acta Ophthalmol. 2018;96:e213–7.
Choudhury F, Varma R, McKean-Cowdin R, et al. Risk factors for four-year incidence and progression of age-related macular degeneration: the Los Angeles Latino eye study. Am J Ophthalmol. 2011;152:385–95.
Cugati S, Mitchell P, Rochtchina E, et al. Cataract surgery and the 10-year incidence of age-related maculopathy. The Blue Mountains Eye Study. Ophthalmology. 2006;113:2020–5.
Ma LY, Rong A, Jiang Y, et al. Effects of femtosecond laser-assisted cataract surgery on macular and choroidal thickness in diabetic patients. Ophthalmol Ther. 2021;10:137–50.
Wang JJ, Klein R, Smith W, et al. Cataract surgery and the 5-year incidence of late-stage age-related maculopathy: pooled findings from the Beaver Dam and Blue Mountains Eye Studies. Ophthalmology. 2003;110:1960–7.
Klein R, Klein BEK, Wong TY, et al. The association of cataract and cataract surgery with the long-term incidence of age-related maculopathy: the Beaver Dam Eye Study. Arch Ophthalmol. 2002;120:1551–8.
Klein R, Klein BEK, Jemen SC, et al. The relationship of ocular factors to the incidence and progression of age-related maculopathy. Arch Ophthalmol. 1998;116:506–13.
Micieli JA, Arshinoff SA. Cataract surgery. CMAJ. 2011;183:1621.
Feng Y, Zhu S, Skiadaresi E, et al. Phacoemulsification cataract surgery with prophylactic intravitreal bevacizumab for patients with coexisting diabetic retinopathy: a meta-Analysis. Retina. 2019;39:1720–31.
Chae JB, Joe SG, Yang SJ, et al. Effect of combined cataract surgery and ranibizumab injection in postoperative macular edema in nonproliferative diabetic retinopathy. Retina. 2014;34:149–56.
Jiang H, Li Z, Sun R, et al. Subfoveal choroidal and macular thickness changes after phacoemulsification using enhanced depth imaging optical coherence tomography. Ophthalmic Res. 2018;60:243–9.
Gudauskiene G, Matuleviciute I, Mockute R, et al. Changes in subfoveal choroidal thickness after uncomplicated cataract surgery. Biomed Pap. 2019;163:179–83.
Ikegami Y, Takahashi M, Amino K. Evaluation of choroidal thickness, macular thickness, and aqueous flare after cataract surgery in patients with and without diabetes: a prospective randomized study. BMC Ophthalmol. 2020;20:1–7.
Shahzad R, Siddiqui MAR, Zafar S, et al. Choroidal thickness changes following cataract surgery using swept source optical coherence tomography. Can J Ophthalmol. 2018;53:60–4.
Pierru A, Carles M, Gastaud P, et al. Measurement of subfoveal choroidal thickness after cataract surgery in enhanced depth imaging optical coherence tomography. Investig Ophthalmol Vis Sci. 2014;55:4967–74.
Chylack LT Jr, Wolfe JK, Singer DM, et al. The lens opacities classification system III. The Longitudinal Study of Cataract Study Group. Arch Ophthalmol. 1993;111:831–6.
Klein BEK, Howard KP, Lee KE, et al. The relationship of cataract and cataract extraction to age-related macular degeneration: the Beaver Dam Eye Study. Ophthalmology. 2012;119:1628–33.
Krepler K, Biowski R, Schrey S, et al. Cataract surgery in patients with diabetic retinopathy: visual outcome, progression of diabetic retinopathy, and incidence of diabetic macular oedema. Graefe’s Arch Clin Exp Ophthalmol. 2002;240:735–8.
Funatsu H, Yamashita H, Noma H, et al. Prediction of macular edema exacerbation after phacoemulsification in patients with nonproliferative diabetic retinopathy. J Cataract Refract Surg. 2002;28:1355–63.
Fujiwara T, Imamura Y, Margolis R, et al. Enhanced depth imaging optical coherence tomography of the choroid in highly myopic eyes. Am J Ophthalmol. 2009;148:445–50.
Funding
The authors have no public or private support to report. No funding or sponsorship was received for this study or publication of this article.
Author information
Authors and Affiliations
Contributions
Paris Tranos, Victoria Dimacali, Despoina Vasileiou, Spyridon Koronis and Achilleas Rasoglou contributed to the conception and design of the work, and to the acquisition, analysis or interpretation of the data. Penelope B de Politis, Renato Ambrosio Jr, Georgios D Panos and Zisis Gatzioufas contributed to revising the manuscript critically for important scholarly content. All authors are responsible for the final approval of the version to be published and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of Interest
The authors certify that they have no affiliations with or involvement in any organization or entity with any financial or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethical Approval
Ethics approval was obtained from the Ophthalmica Eye Institute Ethics Committee, under reference number 102021/007, and the study protocol adhered to the Declaration of Helsinki. Written consent was obtained from all subjects participating in this survey. This study does not contain personal data of any individual person.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Tranos, P., Dimacali, V., Vasileiou, D. et al. The Effects of Uneventful Phacoemulsification on Subfoveal Choroidal Thickness. Ophthalmol Ther 12, 3013–3023 (2023). https://doi.org/10.1007/s40123-023-00796-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-023-00796-9