Abstract
Introduction
Timely anti-vascular endothelial growth factor (VEGF) treatment is crucial for visual function in neovascular age-related macular degeneration (nAMD). The aim of this study was to assess the reasons for anti-VEGF treatment delay during the COVID-19 lockdown and its clinical impact in patients with nAMD.
Methods
A retrospective, observational, multicentre study in patients with nAMD treated with anti-VEGF therapy was performed in 16 centres nationwide. Data were retrieved from the FRB Spain registry, patient medical records and administrative databases. Patients were divided into two groups based on whether they received or missed intravitreal injections during the COVID-19 lockdown.
Results
A total of 302 eyes from 245 patients were included (timely treated group [TTG] 126 eyes; delayed treatment group [DTG] 176 eyes). Visual acuity (VA; ETDRS letters) decreased from baseline to post-lockdown visit in the DTG (mean [standard deviation] 59.1 (20.8) vs. 57.1 (19.7); p = 0.020) and was maintained in the TTG (64.2 [16.5] vs. 63.6 [17.5]; p = 0.806). VA worsened by an average of − 2.0 letters in the DTG and by − 0.6 in the TTG (p = 0.016). A higher proportion of visits were cancelled due to hospital overload in the TTG (76.5%) than in the DTG (47%), and a higher proportion of patients missed visits in the DTG (53%) than in the TTG (23.5%, p = 0.021), with fear of COVID-19 infection being the main reason for missed visits (60%/50%).
Conclusions
Treatment delays were caused by both hospital saturation and patients’ decision; the latter being mainly driven by fear of COVID-19 infection. These delays had a detrimental effect on the visual outcomes in nAMD patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Treatment delays suffered during COVID-19 lockdown had a detrimental effect on the delivery of intravitreal injections with a significant impact in visual outcomes, particularly in neovascular age-related macular degeneration (nAMD) eyes. |
The reasons for these treatment delays have not been specifically explored in the literature. This study aims to identify and assess the main reasons for cancelled or missed visits during the COVID-19 pandemic lockdown among nAMD patients included in a nationwide, multicenter series. |
What was learned from the study? |
In our series, half of the visits were cancelled by the patients and half by healthcare providers due to capacity issues. Fear of infection with COVID-19 was the reason most frequently provided by patients for missing visits. |
Knowing the specific reasons for treatment delays is key to prepare strategies to mitigate vision loss in future pandemic scenarios. |
Introduction
Only a few weeks after the first case of coronavirus disease 2019 (COVID-19) was reported in Wuhan, the number of cases and related deaths grew exponentially worldwide. During the first COVID-19 wave, Spain was one of the epicentres of the pandemic, with one of the highest number of cases and deaths in Europe [1]. In an attempt to contain COVID-19 spread, the Spanish government declared a state of emergency and imposed a strict nationwide lockdown on 14 March 2020 [2]. The healthcare system had to be adapted to treat the largest number of COVID-19 patients as possible, jeopardizing the management of patients with other diseases, such as neovascular age-related macular degeneration (nAMD).
Injections of intravitreal anti-vascular endothelial growth factor (anti-VEGF) agents are widely used as first-line treatment for patients with nAMD. Compliance with intravitreal injections is crucial since even slight deviations may result in vision loss [3,4,5,6]. During the pandemic, recommendations for ophthalmic care were published by ophthalmologists in Spain [7,8,9]. Regarding nAMD patients, these recommendations stated that urgent injections and injections that could not be delayed should continue to be administered during the lockdown; they also stated that potential COVID-19-related health risks for the general population had to be weighed against possible progression of eye diseases, such as AMD [9]. The recommendations also mentioned that a progression of these eye diseases had to be assumed due to the impossibility of adequate patient monitoring [9].
Optimal ophthalmologic care was hampered under these unprecedented circumstances [10]. Patients likely suffered delays in receiving anti-VEGF treatment due to an overwhelmed healthcare system. Injection delays may also have occurred due to reasons intrinsic to the patient. Patients with AMD are usually aged > 65 years and have comorbidities [11]—i.e. the population more likely to succumb to COVID-19 [12]. Thus, fear to attend treatment visits and of contracting the virus might have also prevented these patients from receiving anti-VEGF injections. A number of studies have been published on the effect of delaying anti-VEGF treatment in the context of the COVID-19 pandemic [13,14,15,16,17,18], but the underlying reasons for this delay remain unknown. The present study aimed to further investigate the main reasons underlying delays in anti-VEGF treatment in patients with nAMD in Spain, as well as to assess the impact of such delays on visual acuity (VA) during the lockdown.
Methods
A retrospective, observational, multicentre study was performed in patients with nAMD treated with anti-VEGF therapy in Ophthalmology Departments of 16 centres in Spain. Data from a cohort of patients participating in the FRB Spain registry who fulfilled the inclusion criteria were collected. Study data that were not available in the FRB registry were specifically collected from patients’ medical records and the administrative database of each centre.
Inclusion criteria were nAMD patients aged ≥ 50 years who had initiated treatment with an anti-VEGF agent before 15 March 2020, were included in the FRB registry and had made at least one visit during the previous 3 months immediately preceding the lockdown start date. Patients who participated in a clinical trial during the study period were excluded. Patients were recruited consecutively, following a chronological order backwards in time. Data from a total of 18 months were collected for each patient, if available (from a maximum of 10 months before the declaration of a state of emergency in Spain and for 8 months afterwards).
The study was approved by the Independent Ethics Committee of Clinical Research of Hospital Clinic de Barcelona on 30 November 2020 and adhered to the Declaration of Helsinki and its later amendments. All patients had signed the informed consent form before their inclusion in the FRB registry.
The “lockdown” was defined as the time during which the most restrictive measures were imposed in Spain, starting from the declaration of the state of alarm on 15 March 2020 and ending on 4 May 2020. The “baseline visit” was defined as the last monitoring visit before lockdown, which may have occurred up to 3 months before actual lockdown (from 14 December 2019 to 14 March 2020). The “pre-baseline visit period” was defined as the period from May 2019 to the last monitoring visit before lockdown (period of up to 10 months). The “post-lockdown visit” was defined as the first monitoring visit immediately after the end of lockdown. The “study period” was from 14 May 2019 to 30 November 2020. “Monitoring visits” were defined as those visits during which the patient was clinically evaluated; if only treatment was received, visits were considered to be “treatment visits”.
Two pre-specified subgroups were considered. The “delayed treatment group” (DTG) was defined as those patients who missed ≥ 1 intravitreal injection during lockdown. The “timely treatment group” (TTG) was defined as those patients who received ≥ 1 injections and/or patients who did not miss any preplanned visit during lockdown.
Data obtained from the FRB registry included: demographics (age and sex); date of diagnosis of nAMD; VA (converted to Early Treatment of Diabetic Retinopathy Study [ETDRS] letters); anti-VEGF therapy (drug, number and date of injections); medical history; smoking habit; comorbidities; and baseline structural evaluations. Data collected specifically from the medical records included: reasons for cancelling/missing and maintaining/attending scheduled visits. Information on the number, date and type of visits (monitoring and/or treatment) scheduled was extracted from the administrative database of the participating centre. Data from medical records and administrative database of each centre were introduced in an electronic case report form (eCRF).
Statistical calculations were performed using the Statistical Package for Social Sciences (SPSS) version 22.0 (SPSS IBM Corp., Armonk, NY, USA). Mean (standard deviation [SD], minimum [min] and maximum [max] or confidence intervals [CI]) for quantitative variables and absolute and relative frequency distributions for qualitative variables were determined. Student’s t-test was used for variables that followed a normal distribution; for variables that did not follow a normal distribution, the Mann–Whitney (independent samples) or Wilcoxon (paired samples) tests were used. Multivariate analyses were performed to detect whether the factor variable (patient group) influenced the dependent variable (difference in VA) once the potential influence of the covariates was eliminated. A significance level of 0.05 was chosen for all analyses.
Results
Baseline Demographic and Clinical Characteristics
A total of 302 eyes from 245 patients meeting the selection criteria were included in the study (126 eyes from 97 patients in the TTG and 176 eyes from 148 patients in the DTG). The mean age (min–max) was 78 years (52–97 years) in the TTG and 81.7 years (59–96 years) in the DTG (p = 0.001). There was no significant difference between the groups in any of the other demographic and clinical characteristics, except for a younger average age at diagnosis (p = 0.002) and a lower percentage of patients with hypercholesterolemia (p = 0.034) in the TTG. Structural assessments were also similar among groups, with the exception of the frequency of type of choroidal neovascularization (CNV) classified by optical coherence tomography/optical coherence tomography angiography (OCT/OCTA) (p = 0.016) or not classified (p = 0.008) and the presence of active (only subretinal fluid [SRF]) lesion activity CNV (p = 0.026), which were more prevalent in the DTG (Table 1).
VA Changes Caused by Anti-VEGF Treatment delay
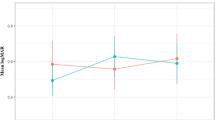
The mean change in VA (ETDRS letters) was measured between the baseline visit and the post-lockdown visit. VA worsened by an average of −0.6 in the TTG and by −2.0 in the DTG (p = 0.016); this corresponds to a difference of approximately 1.4 ETDRS letters between the TTG and DTG (Fig. 1a).
As shown in Fig. 1b, in the TTG, the mean (SD) VA at baseline and the post-lockdown visit were 64.2 (16.5) and 63.6 (17.5) ETDRS letters, respectively. In the DTG, the mean (SD) VA at baseline and the post-lockdown visit were 59.1 (20.8) and 57.1 (19.7) ETDRS letters, respectively. The difference between the VA at baseline and that at the post-lockdown visit were not significant in the TTG (p = 0.806); in contrast; a worsening of VA from baseline to the post-lockdown visit was observed in the DTG, with the difference being significant (p = 0.020). At baseline, there were no significant differences between the VA of the DTG and TTG (59.1 vs. 64.2 ETDRS letters, respectively) (p = 0.063); in contrast, at the post-lockdown visit, a significantly lower mean VA was observed in the DTG than in the TTG (57.1 vs. 63.6 ETDRS letters, respectively; p = 0.004).
VA differences between groups were assessed taking into account the sociodemographic and clinical characteristics, treatment frequency and VA before lockdown that had been found to be significantly different between the groups. No significant differences were detected by group with respect to post-lockdown VA while adjusting for these covariates (p = 0.087).
Reasons for Cancelling/Missing and Maintaining/Attending Scheduled Visits
A total of 309 visits were registered for the 245 patients enrolled in the study. The mean (SD) number of scheduled visits during lockdown was 1.4 (0.6) in the TTG and 1.2 (0.4) in the DTG (p = 0.005). No differences in the type of visits were observed between groups (monitoring visit: TTG, 15.7%; DTG, 17.7%; treatment visit: TTG, 53.7%; DTG, 45.7%; monitoring and treatment visit: TTG, 30.6%; DTG, 36.6%; p = 0.372). Reasons for cancelling/missing visits were different between groups (p = 0.021), with a higher proportion of visits cancelled due to hospitial/clinic saturation caused by the COVID-19 in the TTG (76.5%) than in the DTG (47%) (Fig. 2a). Among the most reported reasons for missing visits from patient´s perspective, fear of COVID-19 infection was the most common in both groups (Fig. 2b). The specific reasons for maintaining/attending visits are presented in Fig. 2c.
Frequency of Eyes with VA Change
The frequency of eyes with VA change from baseline visit to post-lockdown visit, according to the specified categories (Fig. 3a), did not differ between groups (p = 0.348). Nevertheless, the proportion of patients with 5–9 ETDRS letters gained was threefold higher in the TTG (11%) than in the DTG (3.6%).
a Change in VA per group: ≤ − 15, loss of ≥ 15 (ETDRS)letters; − 15, − 10,], loss of 10 to 14 letters; (− 10, − 5], loss of between 5 and 9 letters; (− 5, 5), loss of between 1 and 4 letters or improvement up to 4 letters; [5, 10), improvement of between 5 and 9 letters; [10, 15), improvement of between 10 and 14 letters; ≥ 15: improvement of ≥ 15. b Change in the number of injections and visits per group: Before baseline visit, from 14 May 2019 up to the last visit before lockdown (15 March 2020); After baseline visit, from the last visit before lockdown (15 March 2020, including baseline visit) up to 30 November 2020. BCVA Best-corrected visual acuity
Number of Injections and Visits During Study Period
During the study period, the number of injections and visits was significantly higher in the TTG than in the DTG (p < 0.001) (Fig. 3b). During this period, 104 patient eyes received a combination of anti-VEGF treatments and 197 patient eyes received monotherapy (24, bevacizumab; 91, aflibercept; 82, ranibizumab). No differences between groups were observed regarding type of anti-VEGF treatment during the study period (bevacizumab: 17.5% [TTG], 17.7% [DTG]; aflibercept: 64.3% [TTG], 60% [DTG]; ranibizumab: 61.9% [TTG], 53.7% [DTG]).
Association Between Injection Delay and VA Change
The mean (SD) number of days without receiving anti-VEGF treatment was higher in the DTG than in the TTG when both time from original visit within lockdown to actual visit (time 1: 48.2 [4.8] DTG; 27.3 [14.8] TTG) and time from baseline visit to first injection visit (time 2: 140.5 [81.0] DTG; 43.6 [28.4] TTG) were considered (p < 0.001). The analysis yielded a significant negative correlation between the number of days without receiving anti-VEGF treatment and change in VA (ETDRS letters) when time 1 (rs = – 0.165; p = 0.010) and time 2 (rs = – 0.171; p = 0.007) were considered (Fig. 4).
Correlation between injection delay and VA change in each group. Time 1: number of days between scheduled visits during lockdown (from the start of lockdown to the next attended visit). If a patient did not attend any scheduled visit, the length of the lockdown was assumed. Time 2: number of days between baseline visit to next visit when patients received treatment (if the visit occurred), regardless of whether the visit occurred during lockdown or not. In the case of loss of follow-up, 30 November 2020 was considered as the end date
Timely Re-treatment and Treatment Discontinuation
Mean (SD; 95% CI) VA (ETDRS letters) change was 0.8 (10.2; CI − 0.89 to 2.54) from first monitoring visit after the lockdown until the last monitoring visit available in the group of patients with delayed treatment who received timely re-treatment.
A total of 55 (22.4%) patients were lost to follow-up after lockdown. This number was significantly higher among patients in the DTG (n = 41, 27.7%) than in the TTG (n = 14, 14.4%) (p = 0.015).
Discussion
The specific aims of this study were to investigate further the reasons that drove treatment delays on intravitreal therapy during the first COVID-19 wave in Spain, as well as to describe their negative impact on VA outcomes. The results show that patients with delayed treatment (DTG) had a higher decrease in VA than those patients timely treated (TTG). Despite patients differing in a number of demographic and clinical characteristics, a greater visual loss in the DTG post-lockdown was still observed after these characteristics were considered as confounding factors. The results reported in this study may help physicians to take measures to increase service capacities, patient compliance and adherence to treatments to prevent visual loss in future pandemic scenarios.
Our results are in line with those of prior national [19] and international [13, 17, 18, 20,21,22,23] studies. Table 2 presents a literature review of recent studies. A retrospective single-centre study conducted in Madrid (270 nAMD eyes) by Valverde-Megías et al. [19] compared outcomes before versus after the lockdown with at least 12 weeks between visits. In that study, patients with delayed treatment had a lower mean number of injections in the year preceding the lockdown (5.3) compared to our DTG (11.6) and had a longer lockdown period (considering the end of the state of alarm on 21 June as the end of the lockdown, instead of the start of phase 0 on 4 May). This might have affected the slightly higher decrease in ETDRS letters observed by Valverde-Megías et al. (from 60.2 to 55.9) compared to our study (from 59.1 to 57.1). The difference in the number of injections could be, in turn, due to our study including the clinical practice of 16 centres in Spain whereas the study by Valverde-Megías et al. [19] only included one centre. Their study showed that VA decreased from 60.2 to 55.9 (mean change: − 4.1) ETDRS letters and 29% of the eyes lost > 5 letters in nAMD patients with delayed treatment, which is similar to the percentage observed in our study (24%) [19]. Geographical differences may also have affected these outcomes, as our multicentre series included areas where the severity of the first wave of the pandemic was different, in contrast with the single-centre series, which may have either over- or underestimated these results either way. Nevertheless, we do believe that our results may represent a more accurate national estimate of the clinical findings reported and better reflect the situation nationwide over the first pandemic wave.
A retrospective study conducted in two practices in Minnesota during the first COVID-19 wave (93 nAMD eyes) [13] showed that nAMD patients with delayed visits by more than 2 weeks had worse visual outcomes than those who attended their scheduled visits. VA (Logarithm of the Minimum Angle of Resolution [logMAR]) worsened in the DTG by an average of 0.125 and in the TTG by an average of 0.028 (p = 0.07). In line with our observations, the TTG had better baseline VA (0.391) than the DTG (0.729). In the UK, another retrospective study (585 nAMD eyes) [22] also showed that delayed treatment (by ≥ 8 weeks) decreased VA (DTG: 60.1 before vs. 55.2 after [mean change − 5.18); TTG: 61.4 before vs. 69.9 after [– 2.47]).
A prospective study in Italy (112 nAMD eyes) [14] additionally demonstrated that treatment delays during the pandemic were associated with worse short-term outcomes. In the Italian study, VA (logMAR) was worse during and immediately after the lockdown (0.45) than before (0.50). This difference in VA was associated with the time within visits. In our study, we observed a correlation between the number of days without receiving anti-VEGF treatment and change in VA (a longer period without treatment was associated with higher VA decreases in the DTG). This association was also observed in the Italian study [14] even after accounting for confounding factors, such as age and gender. Other studies that also controlled for covariates (including age, sex, baseline VA, anti-VEGF agent and number of injections and dosing assignment) yielded similar results [24], suggesting that treatment delay impairs VA, independently of patients’ characteristics at baseline, with longer delays resulting in worse visual outcomes. The proportion of nAMD eyes with delayed treatment during the COVID-19 first wave in our study (58.2%) was slightly higher compared to that reported in Italy (53.6%) [15], Israel (50%) [25], France (47.1%) [26] or the UK [22] (36.6%) studies. An international study of 5782 eyes using data of the FRB registry (from Australia, France, Ireland, Italy, Netherlands, New Zealand, Spain and Switzerland) reported that Spain was the country where patients received the fewest number of injections after the lockdown [23]. Differences in the proportion of patients who did not receive intravitreal injections due to the pandemic among studies can be expected, since the measures aimed at reducing the transmission of the virus, such as regulations regarding minimal care provided in outpatient visits and mobility restrictions, varied widely among countries. However, results from the above-mentioned international study [23] showed that despite differences between countries, injections and visits decreased during the lockdowns and eyes lost vision in proportion to the reduced number of injections in all countries [23].
Overall, our study confirmed prior findings that delays in anti-VEGF injections, due to the COVID-19 pandemic [13, 14, 17, 19,20,21,22] or any other cause [3,4,5, 27,28,29,30] negatively impacts VA. A recent study that also used data from the FRB registry showed a significant loss in VA in eyes in which clinicians temporarily extended treatment intervals to > 12 weeks compared to those with no extensions in treatment interval, suggesting that treatment delays greater than this threshold may be detrimental for VA and should be considered cautiously [31].
The main strength and novelty of the present study is that it reported the reasons for cancelling or missing visits during the COVID-19 pandemic in nAMD patients treated at several centres in a country highly affected by the first COVID-19 wave. Prior studies discussed potential reasons for anti-VEGF treatment delays but did not collect these reasons as part of the study objectives [13, 15, 26]. Here, we showed that one-half of the visits were cancelled by the patients. Fear of infection with COVID-19 was the reason most frequently provided by our patients for missing visits, as previously suggested in a single-centre study conducted in Egypt [32].
Patients with delayed treatment in our study were older than those receiving timely treatment. Older patients might have preferred to avoid visits due to a higher risk in this population of morbidity and mortality from COVID-19 [33]. In line with this, older adults were more likely to miss ophthalmology clinic visits during COVID-19 in California [34]. Also, the average age of patients undergoing visits and intravitreal injections during the lockdown in Italy was lower than that during the same period in 2019 [15]. The measures taken during the COVID-19 pandemic by the authorities, directed to reduce the transmission of COVID-19, drastically modified healthcare practices. Despite the centres trying to maintain scheduled intravitreal injections during the lockdown, our results confirm that half of the visits during the lockdown were cancelled by the centres.
The optimal treatment regimen for nAMD remains still under debate. While monthly injections used in clinical trials improve and stabilize VA [35, 36], they pose a great burden for patients, caregivers and physicians in real-world practice. To reduce this burden, other on-demand treatment regimens, such as pro re nata (PRN), or pro-active regimens, such as treat and extend (T&E) and fixed bimonthly regimens, are being progressively more used in routine clinical care [37,38,39]. Pro-active regimens have resulted in better visual outcomes in routine clinical care than those observed in the PRN era [38]. Indeed, retina specialists in Spain prefer the T&E regimen [37]. However the success of this strategy depends on the ability to perform the preplanned visits and maintain an effective treatment injection schedule, which is very challenging due to limitations in healthcare resources and logistical difficulties [37], especially highlighted during the pandemic. To overcome these challenges, a ‘single act’ visit at which both monitoring and treatment are conducted has recently been proposed [40]; such an approach would allow the use of healthcare resources more efficiently and, critically, effective clinical care to continue to be delivered in restricted capacity scenarios, such as the pandemic wave. On the other hand, undertreatment of nAMD patients in the real-world has been proposed as one of the main causes for vision loss in the long-term [41]. Different series using proactive treatment regimens have consistently showed greater visual gains to be correlated to a higher number of injections [38, 39]. More efforts from the healthcare authorities, physicians and patients should be conducted to avoid missing scheduled injections. Moreover, new therapies allowing greater durability of their beneficial effect might also provide a potential solution to this current need.
Finally, the present study showed that VA increased from the first monitoring visit after the lockdown until the last monitoring visit available (0.8 ETDRS letters) in those patients with delayed treatment who received timely re-treatment. This suggests a partial recovery of lost VA in the short-term, in line with prior observations [26].
The study had several limitations which need to be discussed. Some of these limitations are inherent to retrospective observational studies, such as missing data. Another limitation is the lack of structural outcomes, which could have provided valuable information regarding the impact of delaying treatment. We also recognize that although patients had similar demographic and clinical characteristics, they differ in some of these, and patients were not matched. Moreover, the study period was limited, and long-term consequences of treatment delay on VA could not be assessed and should be considered in future studies. Finally, these results are representative of the outcomes achieved in nAMD patients treated mainly in tertiary referral centres at a national level, which may not necessarily reflect the delivered care of most centres in Spain.
Conclusion
In conclusion, our results add to the body of evidence demonstrating that delayed anti-VEGF treatment due to barriers to care, such as the COVID-19 pandemic, has a detrimental effect on VA in nAMD. A higher proportion of scheduled injections were missed in the DTG due to patient’s decision, mainly for fear of COVID-19 infection. Understanding these reasons is key to developing effective measures aimed at improving treatment adherence. These results highlight the efforts that should be made by ophthalmologists, providers and nAMD patients not to delay in-person intravitreal injections.
References
European Centre for Disease Prevention and Control (ECDC). COVID-19. 2020. https://qap.ecdc.europa.eu/public/extensions/COVID-19/COVID-19.html#global-overview-tab. Accessed 29 Apr 2022.
Spanish Government. Real Decreto 463/2020, de 14 de marzo, por el que se declara el estado de alarma para la gestión de la situación de crisis sanitaria ocasionada por el COVID-19. 2020. https://boe.es/boe/dias/2020/03/11/pdfs/BOE-A-2020-3434.pdf#BOEn. Accessed 27 May 2020.
Teo KYC, Saxena N, Gan A, et al. Detrimental effect of delayed re-treatment of active disease on outcomes in neovascular age-related macular degeneration: the RAMPS study. Ophthalmology Retina. 2020;4:871–80.
Cohen SY, Mimoun G, Oubraham H, et al. Changes in visual acuity in patients with wet age-related macular degeneration treated with intravitreal ranibizumab in daily clinical practice: the LUMIERE study. Retina. 2013;33(3):474–81.
Eldem B, Lai TYY, Ngah NF, et al. An analysis of ranibizumab treatment and visual outcomes in real-world settings: the UNCOVER study. Graefes Arch Clin Exp Ophthalmol. 2018;256(5):963–73.
Holz FG, Tadayoni R, Beatty S, et al. Key drivers of visual acuity gains in neovascular age-related macular degeneration in real life: findings from the AURA study. Br J Ophthalmol. 2016;100(12):1623–8.
Gegúndez-Fernández JA, Llovet-Osuna F, Fernández-Vigo JI, et al. Recommendations for ophthalmologic practice during the easing of COVID-19 control measures. Acta Ophthalmol. 2021;99(7):e973–83.
Gegúndez-Fernández JA, Zarranz-Ventura J, Garay-Aramburu G, et al. Recommendations for eye care during the alarm state by the coronavirus disease pandemic COVID-19. Arch Soc Esp Oftalmol (Engl Ed). 2020;95(6):300–10.
Sociedades Científicas Españolas. Recomendaciones para la atención a pacientes oftalmológicos en relación con emergencia COVID-19. 2020. https://serv.es/oftalmologia-covid19/. Accessed 29 Apr 2020.
Rodríguez-Fernández CA, Varela-Agra M, Pérez-Roldán L, Álvarez-Reguera A, Martínez-Reglero C, Campo-Gesto A. Impact of COVID-19 on Eye care in spain during the first phase of the pandemic. J Clin Med. 2021;10(18):4087.
Olea JL, Tuñón J. Patients with neovascular age-related macular degeneration in Spain display a high cardiovascular risk. Eur J Ophthalmol. 2012;22(3):404–11.
World Health Organization. Statement—Older people are at highest risk from COVID-19, but all must act to prevent community spread. 2020. http://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/statements/statement-older-people-are-at-highest-risk-from-covid-19,-but-all-must-act-to-prevent-community-spread. Accessed 8 June 2020.
Naravane AV, Mundae R, Zhou Y, et al. Short term visual and structural outcomes of anti-vascular endothelial growth factor (anti-VEGF) treatment delay during the first COVID-19 wave: a pilot study. PLoS ONE. 2021;16(2): e0247161.
Borrelli E, Grosso D, Vella G, et al. Short-term outcomes of patients with neovascular exudative AMD: the effect of COVID-19 pandemic. Graefes Arch Clin Exp Ophthalmol. 2020;258(12):2621–8.
Borrelli E, Grosso D, Vella G, et al. Impact of COVID-19 on outpatient visits and intravitreal treatments in a referral retina unit: let’s be ready for a plausible “rebound effect.” Graefes Arch Clin Exp Ophthalmol. 2020;258(12):2655–60.
Borrelli E, Sacconi R, Querques L, et al. Taking the right measures to control COVID-19 in ophthalmology: the experience of a tertiary eye care referral center in Italy. Eye (Lond). 2020;34(7):1175–6.
Sekeroglu MA, Kilinc Hekimsoy H, Horozoglu Ceran T, Doguizi S. Treatment of neovascular age related macular degeneration during COVID-19 pandemic: the short term consequences of unintended lapses. Eur J Ophthalmol. 2022;32(2):1064–72.
Arruabarrena C, Toro MD, Onen M, et al. Impact on Visual acuity in neovascular age related macular degeneration (nAMD) in Europe due to COVID-19 Pandemic Lockdown. J Clin Med. 2021;10(15):3281.
Valverde-Megías A, Rego-Lorca D, Fernández-Vigo JI, Murciano-Cespedosa A, Megías-Fresno A, García-Feijoo J. Effect of COVID-19 lockdown in Spain on structural and functional outcomes of neovascular AMD patients. J Clin Med. 2021;10(16):3551.
Yeter DY, Dursun D, Bozali E, Ozec AV, Erdogan H. Effects of the COVID-19 pandemic on neovascular age-related macular degeneration and response to delayed Anti-VEGF treatment. J Fr Ophtalmol. 2021;44(3):299–306.
Sevik MO, Aykut A, Özkan G, Dericioğlu V, Şahin Ö. The effect of COVID-19 pandemic restrictions on neovascular AMD patients treated with treat-and-extend protocol. Int Ophthalmol. 2021;41(9):2951–61.
Stone LG, Grinton ME, Talks JS. Delayed follow-up of medical retina patients due to COVID-19: impact on disease activity and visual acuity. Graefes Arch Clin Exp Ophthalmol. 2021;259(7):1773–80.
Zarranz-Ventura J, Nguyen V, Creuzot-Garcher C, et al. International impact of the COVID-19 pandemic lockdown on intravitreal therapy outcomes: Fight Retinal Blindness registry. Retina. 2022;42:616–27.
Ramakrishnan MS, Yu Y, VanderBeek BL. Association of visit adherence and visual acuity in patients with neovascular age-related macular degeneration: secondary analysis of the comparison of age-related macular degeneration treatment trial. JAMA Ophthalmol. 2020;138(3):237–42.
Wasser LM, Weill Y, Brosh K, et al. The impact of COVID-19 on intravitreal injection compliance. SN Compr Clin Med. 2020;2:1–4.
Billioti de Gage S, Drouin J, Desplas D, et al. Intravitreal Anti-vascular endothelial growth factor use in france during the coronavirus disease 2019 pandemic. JAMA Ophthalmol. 2021;139(2):240–2.
Muether PS, Hermann MM, Koch K, Fauser S. Delay between medical indication to anti-VEGF treatment in age-related macular degeneration can result in a loss of visual acuity. Graefes Arch Clin Exp Ophthalmol. 2011;249(5):633–7.
Real JP, Granero GE, De Santis MO, et al. Rate of vision loss in neovascular age-related macular degeneration explored. Graefes Arch Clin Exp Ophthalmol. 2015;253(11):1859–65.
Massamba N, Dirani A, Knoeri J, Pasquier B, Ingram A, Soubrane G. Evaluating the impact of summer vacation on the visual acuity of AMD patients treated with ranibizumab. Eye (Lond). 2015;29(11):1453–7.
García-Layana A, García-Arumí J, Figueroa MS, et al. Management of wet age-related macular degeneration in spain: challenges for treat and extend implementation in routine clinical practice. J Ophthalmol. 2019;2019:9821509.
Teo KYC, Nguyen V, Barthelmes D, AExtended intervals for wet AMD patients with high retreatment needs: informing the risk during COVID-19, data from real-world evidence. Eye (Lond). 2021;35(10):2793–801.
Moussa M, Elshorbagy MS, Emarah A, et al. Intravitreal injections of anti-VEGF agents during COVID-19 pandemic: clinical audit from Tanta University Hospital. Int Ophthalmol. 2021;41(4):1437–43.
Pijls BG, Jolani S, Atherley A, et al. Demographic risk factors for COVID-19 infection, severity, ICU admission and death: a meta-analysis of 59 studies. BMJ Open. 2021;11(1):e044640.
Brant AR, Pershing S, Hess O, et al. The impact of COVID-19 on missed ophthalmology clinic visits. Clin Ophthalmol. 2021;15:4645–57.
Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med. 2006;355(14):1419–31.
Brown DM, Michels M, Kaiser PK, Heier JS, Sy JP, Ianchulev T. Ranibizumab versus verteporfin photodynamic therapy for neovascular age-related macular degeneration: two-year results of the ANCHOR study. Ophthalmology. 2009;116(1):57-65.e5.
García-Layana A, García-Arumí J, Figueroa MS, et al. Management of wet age-related macular degeneration in spain: challenges for treat and extend implementation in routine clinical practice. J Ophthalmol. 2019;2019:9821509.
Zarranz-Ventura J, Parrado-Carrillo A, Nguyen V, et al. Creation of a neovascular age-related macular degeneration national database using a web-based platform: Fight Retinal Blindness Spain. Report 1: visual outcomes. Clin Exp Ophthalmol. 2022;50(3):312–24.
Pina Marín B, Gajate Paniagua NM, Gómez-Baldó L, Gallego-Pinazo R. Burden of disease assessment in patients with neovascular age-related macular degeneration in Spain: Results of the AMD-MANAGE study. Eur J Ophthalmol. 2022;32(1):385–94.
Sociedad Española de Retina y Vítreo (SERV). Unidad de Terapia Intravítrea-Consenso Clínico. 2021. https://serv.es/pdf/UTIV_Doc_Completo.pdf. Accessed Mar 2021.
Monés J, Singh RP, Bandello F, Souied E, Liu X, Gale R. Undertreatment of neovascular age-related macular degeneration after 10 years of anti-vascular endothelial growth factor therapy in the real world: the need for a change of mindset. Ophthalmologica. 2020;243(1):1–8.
Acknowledgements
The authors would like to thank all the investigators and centers that have participated in the present study: Saturnino Gismero Moreno (Hospital Costa del Sol), Oscar Ruiz Moreno (Hospital Universitario Miguel Servet), Laura Broc Iturralde (Hospital Universitario Germans Trias i Pujol), Helena Brosa Morros (Hospital Universitario Vall d’Hebron), Laura Sararols Ramsay (Hospital General de Granollers), César Azrak Haskour (Hospital de Vinalopó), Maximino J. Abraldes (Hospital Clínico Universitario de Santiago), Beatriz Gonzalo Suárez (Clínica Oftalvist Madrid), Mónica Asencio Durán (Hospital Universitario La Paz), Carolina Arruabarrena Sánchez (Hospital Universitario Principe de Asturias), Jose Mª Ruiz Moreno (Hospital Universitario Puerta de Hierro), Marta S Figueroa (Hospital Universitario Ramón y Cajal), Arantzazu Larrauri Arana (Hospital Universitario de Áraba).
Funding
This work was supported by Novartis. The journal’s Rapid Service Fee was funded by Novartis.
Medical Writing, Editorial, and Other Assistance
Medical writing assistance was provided by Laura Prieto del Val from Dynamic Science S.L.U. (Evidenze Clinical Research) during the preparation of the study protocol and this manuscript, funded by Novartis, according to Good Publication Practice guidelines. We also thank Cristina Martínez García and Sonia de Orte-Seemann from Syneos Health for their invaluable help in the overall management of this study, as well as Ana Vazquez and Teresa Guix from Novartis Spain for their collaboration and assistance in the project management and manuscript review.
Author Contributions
Javier Zarranz-Ventura was responsible for conceptualization, methodology, validation, formal analysis, investigation, resources, writing—original draft, writing—review and editing, visualization, supervision, project administration and funding acquisition. José Juan Escobar-Barranco was responsible for conceptualization, methodology, validation, investigation, resources, writing—original draft, writing—review and editing, visualization and supervision. Saturnino Gismero Moreno, Oscar Ruiz Moreno, Laura Broc Iturralde, Helena Brosa Morros, Laura Sararols Ramsay, César Azrak Haskour, Maximino J Abraldes, Beatriz Gonzalo Suárez, Mónica Asencio Durán, Carolina Arruabarrena Sánchez, Jose Mª Ruiz Moreno, Marta S. Figueroa and Arantzazu Larrauri Arana were responsible for investigation and writing—review and editing. Laia Gómez-Baldó was responsible for conceptualisation, methodology, resources, writing—original draft, writing—review and editing, visualization, supervision, project administration and funding acquisition. Roberto Gallego-Pinazo was responsible for conceptualisation, methodology, validation, investigation, resources, writing—original draft, writing—review and editing, visualization and supervision.
Disclosures
Javier Zarranz-Ventura is a consultant for Novartis Pharmaceuticals, Bayer, Alcon, Alimera Sciences, Bausch and Lomb, Brill Pharma, DORC, Preceyes, Roche, Topcon and Zeiss. José Juan Escobar-Barranco declares no conflicts. Laia Gómez-Baldó is an employee of Novartis Pharmaceuticals. Roberto Gallego-Pinazo: is a consultant for Boheringer Ingelheim, Novartis Pharmaceuticals, Roche and Zeiss.
Compliance with Ethics Guidelines
The study was approved by the Independent Ethics Committee of Clinical Research of Hospital Clinic de Barcelona on 30 November 2020 and adhered to the Declaration of Helsinki and its later amendments. All patients had signed the informed consent before their inclusion in the FRB registry.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
The names of the participating Study Investigators and their institutions are listed in the Acknowledgements section.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Zarranz-Ventura, J., Escobar-Barranco, J.J., Gómez-Baldó, L. et al. Reasons for Delayed Anti-VEGF Treatment During COVID-19 Lockdown and Clinical Impact in Neovascular Age-Related Macular Degeneration. Ophthalmol Ther 12, 2537–2555 (2023). https://doi.org/10.1007/s40123-023-00757-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-023-00757-2