Abstract
Introduction
This study describes real-world treatment patterns in Germany for brolucizumab, an anti-vascular endothelial growth factor (anti-VEGF) therapy for neovascular age-related macular degeneration (nAMD).
Methods
This single-arm retrospective cohort study used German patient-level prescription data. Patients aged ≥ 50 years, who received ≥ 1 brolucizumab prescription in one eye only (unilateral) and had a minimum of 12 months follow-up were included. Three cohorts were defined from the overall population: anti-VEGF treatment-naive patients (“treatment-naive”); anti-VEGF treatment-experienced patients (“treatment-experienced”); and of the treatment-experienced cohort, patients persistent on brolucizumab for 12 months (“treatment-experienced persistent”), i.e. who received ≥ 2 brolucizumab injections and did not discontinue or use other anti-VEGF agents in that period. Descriptive statistics were used to analyse patient characteristics and injection intervals.
Results
A total of 2089 patients with at least 12 months follow-up and one brolucizumab injection were analysed. Most were female (58.1%), aged 80+ years (54.7%). A total of 539 (25.8%) were treatment-naive, 1550 (74.2%) treatment-experienced and, of those, 787 (50.8%) were persistent. Overall, the median (interquartile range, IQR) number of brolucizumab injections during the 12 months follow-up was 5.0 (3.0–8.0). In the treatment-naive and treatment-experienced sub-cohorts it was 5.0 (3.0–8.0) and 5.0 (3.0–9.0) injections, respectively. In the treatment-experienced persistent cohort the median (IQR) number of injections was 8.0 (5.0–10.0). In this same cohort, the median (IQR) treatment interval between anti-VEGF injections before switch to brolucizumab was 5.1 (4.0–8.0) weeks, whilst the brolucizumab interval at 12 months after switch was 8.0 (6.0–11.9) weeks. Of treatment-experienced patients, 67% extended their treatment interval and those with pre-switch intervals less than 6 weeks (< q6w), in particular, had meaningful treatment interval extensions.
Conclusion
Patients who switched to brolucizumab had a median treatment interval extension of about 3 weeks at 12 months. These results show that treatment with brolucizumab has the potential to reduce treatment burden in patients with nAMD in routine clinical practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
A major challenge in neovascular age-related macular degeneration (nAMD) management is the high treatment burden on patients and caregivers. |
Pivotal trials have suggested that brolucizumab could reduce the treatment burden of nAMD. Large real-world studies on treatment patterns with brolucizumab to date are limited and are needed to inform its use in clinical practice. |
What was learned from the study? |
This retrospective cohort study using prescription data from Germany assessed treatment patterns with unilateral brolucizumab injections in real-world clinical practice for nAMD. |
Patients who switched from other anti-vascular endothelial growth factor (anti-VEGFs) to brolucizumab extended their injection interval by a median of 2.9 weeks at 12 months, from 5.1 weeks pre-switch to 8.0 weeks at 12 months post-switch to brolucizumab. |
Findings suggest that brolucizumab may allow for a reduced treatment burden in patients with nAMD in routine clinical practice. |
Introduction
Intravitreal anti-vascular endothelial growth factor (anti-VEGF) therapies have become the standard of care for patients with neovascular age-related macular degeneration (nAMD) owing to clinical trial evidence supporting their ability to improve visual acuity and quality of life [1]. There are several anti-VEGF treatment regimens for nAMD including fixed dosing, treat-and-extend and pro re nata [2,3,4]. Evidence from real-world clinical practice has shown that, whilst treatment with ranibizumab and aflibercept have transformed the visual outcomes of patients with nAMD, maintenance of visual acuity involves burdensome treatment schedules. A significant proportion of patients require treatment more frequently than every 8 weeks (< q8w) [2, 3, 5]. This high injection burden may result in missed or discontinued treatment, leading to poorer visual acuity outcomes in real-world clinical practice compared to clinical trials [2, 6]. Therefore, the current challenge is to maintain visual acuity while minimising treatment burden on patients and caregivers [7].
Brolucizumab (Beovu®) is an anti-VEGF therapy for nAMD approved by the US Food and Drug Administration (FDA) [8], the European Medicines Agency (EMA) [9], and other national regulatory authorities after its efficacy and safety were demonstrated in the pivotal phase 3 HAWK and HARRIER trials [10]. In the first year of HAWK and HARRIER follow-up, brolucizumab demonstrated non-inferior visual acuity gains from baseline with greater fluid reduction vs aflibercept. These results were achieved with over 50% of patients treated with brolucizumab maintained on a 12-weekly (q12w) treatment interval after the loading phase (every 4 weeks [monthly] for the first three doses). This suggests that brolucizumab may allow for a reduced treatment burden for patients with nAMD, although no direct comparison regarding treatment intervals among the two drugs within the trials could be made [11]. Also, the generalisability of the HAWK and HARRIER pivotal trials to real-world clinical practice is limited. First, injection intervals are not as strictly protocol driven in real-world practice and may vary. Second, since patients who had received prior treatment for nAMD were excluded from the HAWK and HARRIER clinical trial population, there is also limited information on brolucizumab switching patterns and on the outcomes of patients who switch to brolucizumab. There have been a number of case studies of brolucizumab in pre-treated populations of patients with nAMD that showed meaningful interval extensions post-switch to brolucizumab [12,13,14,15,16,17]; however, the sample sizes are relatively small and come from single centres.
Therefore, robust and larger real-world evidence studies on treatment scheduling are needed to understand the extent to which brolucizumab can increase treatment intervals between injections in clinical practice. The objective of the study was to investigate real-world treatment patterns with brolucizumab in Germany, where it has been available for use since March 2020.
Methods
REALIZE is a single-arm retrospective cohort study which aims to describe treatment patterns with brolucizumab, including treatment intervals between anti-VEGF injections before and after a switch to brolucizumab.
Database Description
This study was conducted using the IQVIA Longitudinal Prescription Database (LRx) recording health data from patients in Germany [18]. The LRx database contains fully anonymised patient-level longitudinal data on patient characteristics and complies with relevant regulations for protecting patient privacy. Data were analysed in aggregated form without individual data becoming available. Individual consent forms were not obtained in line with German national and European Union legislation. Patient prescriptions are included as covered by the Statutory Health Insurance (SHI). The SHI covers 90% of the German population, and LRx covers approximately 80% of all prescriptions reimbursed by SHI funds in 2021 [19]. The prescription date was used as a proxy for anti-VEGF injection date.
Study Period and Cohort Description
The study period was defined from the date of the first available anti-VEGF injection in the data set to 30 November 2021. The index date for each patient was the date of the first brolucizumab injection, which could be anytime between 1 March 2020 (since brolucizumab became available in Germany for use outside of clinical trials in March 2020) and 30 November 2021. The date of the patient’s first nAMD diagnosis was assumed to be the date of the first anti-VEGF prescription in the database for that patient, from January 2015 onwards.
Patients aged 50 years or older with a diagnosis of neovascular AMD at index date with at least one brolucizumab prescription regardless of prior anti-VEGF treatment status and a minimum follow-up of 12 months from index date were included in the study. Patients without a record of prescription of any drug in the 6 months before the index date (pre-index period) were excluded from the study (Fig. 1). Because no diagnosis information was available, patients were assumed to have a nAMD diagnosis given that during the study period brolucizumab only had marketing authorisation for nAMD. As a result of the lack of eye-level data, patients were excluded from the study if there was evidence of bilateral treatment with brolucizumab; patients were assumed to have received bilateral treatment if they had more than one brolucizumab unit in a single prescription or at least one interval between two brolucizumab injections shorter than 20 days.
Definition of study population cohorts. *3 months and 12 months exclusion before index date were also used to create different cohorts for sensitivity analysis. Moreover, persistent patients are the patients who do not switch to any other anti-VEGF, do not discontinue anti-VEGF treatment and have at least two brolucizumab prescriptions over the follow-up. **Persistence on brolucizumab for a time period is defined as having at least two brolucizumab prescriptions, no switch to other anti-VEGF and no discontinuation during that period
We created three patient sub-cohorts: those with no previous anti-VEGF treatment before starting brolucizumab were referred to as the “treatment-naive cohort”; those with a record of previous anti-VEGF treatment before starting brolucizumab were referred to as the “treatment-experienced cohort”; and lastly, a subset of the treatment-experienced cohort, who were persistent on brolucizumab for at least 12 months post-index date were referred to as the “treatment-experienced persistent cohort” (Fig. 1). Finally, two additional sub-cohorts with at least 18 months follow-up (“18-month follow-up cohort”, and “treatment-experienced 18-month persistent cohort”) were defined. During the study period, patients may have discontinued treatment or switched to another anti-VEGF, except for those in the treatment-experienced persistent cohorts. Persistence on brolucizumab was defined as having at least two prescriptions of brolucizumab within 12 months of follow-up, without record of a different anti-VEGF prescription and without discontinuing brolucizumab treatment during those 12 months (discontinuation was defined as having no subsequent anti-VEGF injection within 6 months since the prior anti-VEGF injection).
Outcomes
Patient characteristics (age, gender) are reported overall and by sub-cohort. For the treatment-experienced cohort, we also report the type and number of previous anti-VEGF agents, and time since first anti-VEGF.
The primary outcome of the study was the number of brolucizumab injections in the 12 months following the first brolucizumab injection, overall and by sub-cohort. The secondary outcome was the time difference between the last anti-VEGF treatment interval before switch to brolucizumab and the last brolucizumab treatment interval at 12 months. The last anti-VEGF interval before switch was defined as treatment interval between the last two anti-VEGF injections before switch to brolucizumab. The last brolucizumab interval at 12 months was defined as the time between the last two brolucizumab injections before month 12. This secondary outcome was only analysed in the treatment-experienced persistent cohort. Moreover, for this cohort, patients were grouped and analysed according to the duration of their last treatment interval before switch to brolucizumab.
Statistical Methods
Descriptive statistics were used to analyse the outcomes of this study. Continuous variables were summarised by reporting the number of observations, means and standard deviations (SD), median and interquartile range (IQR), and minimum and maximum values and the proportion of missing data. Categorical variables were summarised by reporting counts and proportions. No imputation method was used for missing data and missingness was considered as a separate category. Treatment interval, defined as the time between two successive anti-VEGF injections, was calculated as the time between prescriptions in the database in weeks. For the treatment-experienced cohort, the length of the last interval before switch to brolucizumab was categorised as less than 4 weeks, 4–6 weeks, 6–8 weeks, 8–10 weeks, 10–12 weeks and 12 weeks or more. Treatment switches are described using Sankey diagrams that connect each treatment switch event over time.
Results
Patient Characterisation
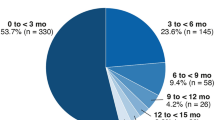
A total of 2089 patients, with at least one brolucizumab injection and a minimum of 12 months follow-up, were included in the study. Most patients were female (58.1%), aged 80 years and older (54.7%). A total of 539 (25.8%) were treatment-naive, 1550 (74.2%) treatment-experienced and, of those, 787 (50.8%) were treatment-experienced persistent (Table 1). Most of the treatment-experienced cohort previously received one 783 (50.5%) or two 743 (47.9%) anti-VEGF agents, and less than 2% received three. Patients in the overall treatment-experienced cohort received aflibercept (56.1%), ranibizumab (42.6%) and bevacizumab (1.3%) before their first brolucizumab prescription (Table 2). Finally, the characteristics of patients in the 18-month follow-up cohort were similar to those of the aforementioned 12-month cohorts (Tables S1, S2 in the supplementary material).
Description of Treatment Patterns
Overall, the median (IQR) number of brolucizumab injections during the 12-month follow-up period was 5.0 (3.0–8.0). The treatment-naive and treatment-experienced sub-cohorts had a median (IQR) of 5.0 (3.0–8.0) and 5.0 (3.0–9.0) brolucizumab injections, respectively. For the treatment-experienced persistent cohort, patients had a median of 8.0 (5.0–10.0) injections (Table 3, Fig. S1 in the supplementary material). For the treatment-experienced persistent cohort the median (IQR) treatment interval between the last anti-VEGF injections before switch to brolucizumab was 5.1 (4.0–8.0) weeks (Table 4). After switch to brolucizumab, the median (IQR) interval between injections was 8.0 (6.0–11.9) weeks at 12 months. Sixty-seven percent (527/787) of patients in the treatment-experienced persistent cohort achieved longer treatment intervals at 12 months with brolucizumab when compared with the last treatment interval prior to switch to brolucizumab.
When results were stratified by pre-switch interval length, notable brolucizumab interval extensions were achieved in those who had a treatment interval prior to switch shorter than 6 weeks (Fig. 2; Table 4).
Duration of treatment intervals at 12 months for the treatment-experienced persistent cohort stratified by the duration of the last treatment interval between anti-VEGF injections before switch to brolucizumab. Every boxplot represents distributions of the data reported in Table 4. (i.e. last treatment interval at 12 months grouped by last treatment interval before switch to brolucizumab). On the horizontal axis, N stands for the size of the sub-cohort (using 787 as denominator) based on the length of last treatment interval before switch to brolucizumab (where a total of 762 patients from the treatment-experienced persistent cohort had at least two anti-VEGF prescriptions before index date and were considered for the stratification). Each box covers the interquartile interval, where 50% of the data is found. The horizontal line in the box represents the median. The whiskers are the two lines outside the box that go from the minimum to the lower quartile (the start of the box) and then from the upper quartile (the end of the box) to the maximum
The results on interval extensions were replicated in the treatment-experienced 18-month persistent cohort (Table S3 in the supplementary material).
Patterns of Discontinuation and Switch
Overall, 16.0% (330/2089) of patients discontinued brolucizumab treatment within the first 12 months. The 12-month discontinuation rates were 17.6% (95/539) for the treatment-naive cohort and 15.2% (235/1550) for the treatment-experienced cohort.
The proportion of patients who switched at least once to a different anti-VEGF during the 12 months after the first brolucizumab injection was 40.4% (843/2089) overall, and 19.5% (105/539) and 47.6% (738/1550) for treatment-naive and treatment-experienced cohorts, respectively. The switching path is represented in Sankey plot diagrams (Fig. S2 in the supplementary material).
Discussion
The REALIZE study informs on patient characteristics and treatment patterns with brolucizumab in real-world clinical practice in Germany. The main analyses were conducted for the overall cohort with 12 months follow-up. Sub-cohorts of patients based on prior anti-VEGF treatment status and persistence were further analysed. In REALIZE, brolucizumab patients were mostly female (58.1%) and 80 years old and above (54.7%). These patient characteristics were comparable to those of other real-world studies with brolucizumab [20]. In the treatment-experienced cohort, approximately half of the patients (49.5%) had at least two different anti-VEGFs before switch to brolucizumab and had been on treatment on average for 3 years. Additionally, most patients (72.0%) of the treatment-experienced persistent cohort had treatment intervals below 8 weeks (< q8w) prior to switch. This is in line with smaller case series and observational studies across the world where most patients switching to brolucizumab are those experiencing a high treatment burden prior to switch to brolucizumab, indicating a patient population with significant unmet needs. For example, in the SHIFT study, Bulirsch et al. focused on patients with recalcitrant nAMD who were switched to brolucizumab therapy, reporting that all patients assessed had been previously treated but had recalcitrant fluid despite a high treatment frequency with ranibizumab, aflibercept and bevacizumab prior to switch to brolucizumab [20]. Another example is the REBA study, a retrospective, multicentric study, which included patients who switched to brolucizumab because of either fluid recurrence, fluid recalcitrance, or inability to extend beyond four or six weekly treatment intervals (q4w–q6w) prior to brolucizumab [13].
Our REALIZE study provides further clarity on brolucizumab use and benefit amongst treatment-experienced patients. The median interval extension of patients who switched to brolucizumab was about 3 weeks and approximately two-thirds (67.0%) of treatment-experienced patients extended their treatment interval. Similarly, several US single practice real-world studies have reported comparable interval extensions. Hamou et al. reported an average interval extension in previously treated eyes in the maintenance phase of 3 weeks [16]. Zubricky et al. concluded that previously treated patients switching to brolucizumab were able to extend their intervals by approximately 4 weeks [12]. Abdin et al. showed that patients with refractory nAMD who switched to brolucizumab reduced their annual injection number per eye by three injections in a small sample (n = 21 eyes) which further indicates interval extensions with brolucizumab [21]. Therefore, clinical trials and results from other real-world evidence studies indicate that results seen in REALIZE may be attributable to a better fluid response to brolucizumab compared to other anti-VEGF drugs [14, 15]. This may be explained by the fact that the presence of fluid is a key criterion, in clinical guidelines, when determining treatment interval for a patient [22, 23]. Therefore, persistent fluid is a common barrier to extending treatment interval. Interestingly, patients with a pre-switch anti-VEGF treatment interval of less than 6 weeks had particularly meaningful interval extensions in REALIZE, which shows that patients who respond poorly to other anti-VEGFs can benefit from switching to brolucizumab [12]. Reassuringly, results on interval extensions were replicated in the treatment-experienced 18-month persistent cohort which indicates that alleviation of treatment burden is durable. Our results therefore add further evidence that brolucizumab can extend treatment intervals, thus reducing the number of injections which lowers treatment burden for patients with nAMD [2,3,4,5,6] and demonstrates that a switch to brolucizumab may be an effective treatment option. Recalcitrant fluid, particularly in outer compartments such as the subretinal and sub-RPE (retinal pigment epithelium) space with fluid alterations can be better addressed with brolucizumab [11, 13, 14, 24,25,26] which makes it a useful addition to the armamentarium of choices in treatment of nAMD. Strengths of the study lie in the strong representativity and large coverage of the database for the German patient population, the large sample size and length of follow-up time. Our results can as such be considered a good representation of the way brolucizumab is currently used in clinical practice in Germany.
Our REALIZE study results should be nonetheless interpreted in the context of the limitations of the real-world data and analysis used. Firstly, all patients were assumed to be diagnosed with nAMD because the marketing authorisation of brolucizumab was for nAMD only during the study period. While we cannot rule out the possible inclusion of patients using brolucizumab off-label in a compassionate care setting for other retinal conditions, it is fair to assume that these patients without nAMD would make up a very small proportion of our sample. Secondly, since time intervals between anti-VEGF injections were inferred from prescription dates, we cannot be certain when or whether the prescribed injections were administered. In clinical practice in Germany, however, anti-VEGF injection is usually combined with the issuance of a prescription, so that the prescription of brolucizumab is always associated with the dispensing of the drug that occurs with it, which makes the prescription date a good proxy for injection date. Another limitation consists of the lack of information at the eye level, which led to the exclusion of patients who received brolucizumab less than 20 days apart and because we assumed they had received bilateral anti-VEGF injections. Despite this exclusion, it is possible that some bilateral patients were still included in the study, which would have led to underestimation of the interval length between brolucizumab injections at the eye level. On the other hand, it is also possible that unilateral patients were excluded through the same exclusion criterion, which would have led to overestimation of the time between brolucizumab injections at the eye level. While these limitations related to prescription information exist (i.e. lack of anti-VEGF injection date or bilaterality of the patients), our study design and analytical assumptions aimed at limiting their impact on the interpretation of results.
Safety of brolucizumab treatment could not be investigated in the present work because safety data are not documented in the prescription database. There has been an extensive amount of research in this area which has generated patient management learnings and guidance, to support healthcare professionals in mitigating and managing the risk of inflammatory adverse events and vision loss [27,28,29,30,31,32,33,34,35]. These learnings include patients with a history of previous intraocular inflammation (IOI) being at higher risk, the importance of monitoring for the signs of IOI prior to anti-VEGF injection, educating patients to report any symptoms of IOI as soon as they arise and that prompt intensive treatment of IOI could help prevent or recover vision loss [20, 27,28,29,30,31,32,33,34,35,36,37]. Finally, since brolucizumab is a relatively new therapy launched in March 2020, treatment patterns observed in routine care may not have stabilised yet, and in particular they may have been influenced by the confirmation of the safety signal of retinal vasculitis and/or retinal vascular occlusion, which may have prompted treatment switches or discontinuation, and the negative perception of a new drug with a higher potency, which may have created a reservation to initiate brolucizumab. Further analyses are recommended when newer data on real-world brolucizumab treatment patterns with even longer follow-up periods are available.
Conclusion
This is the largest European study to date of treatment patterns in brolucizumab patients. Treatment-experienced patients who switched to brolucizumab had a median treatment interval extension of about 3 weeks with particularly meaningful treatment interval extensions seen in patients with pre-switch intervals of less than 6 weeks. These results provide real-world evidence suggesting that brolucizumab has the potential to reduce treatment burden on patients with nAMD in routine clinical practice and can enhance patient care when supplemented with patient management learnings.
References
Assi L, Chamseddine F, Ibrahim P, et al. A global assessment of eye health and quality of life: a systematic review of systematic reviews. JAMA Ophthalmol. 2021;139(5):526–41.
Holz FG, Tadayoni R, Beatty S, et al. Multi-country real-life experience of anti-vascular endothelial growth factor therapy for wet age-related macular degeneration. Br J Ophthalmol. 2015;99(2):220–6.
Essex RW, Nguyen V, Walton R, et al. Treatment patterns and visual outcomes during the maintenance phase of treat-and-extend therapy for age-related macular degeneration. Ophthalmology. 2016;123(11):2393–400.
Holz FG, Schmitz-Valckenberg S, Fleckenstein M. Recent developments in the treatment of age-related macular degeneration. J Clin Invest. 2014;124(4):1430–8.
Khanani AM, Skelly A, Bezlyak V, Griner R, Torres LR, Sagkriotis A. SIERRA-AMD: a retrospective, real-world evidence study of patients with neovascular age-related macular degeneration in the United States. Ophthalmol Retina. 2020;4(2):122–33.
Kim LN, Mehta H, Barthelmes D, Nguyen V, Gillies MC. Meta analysis of real-world outcomes of intravitreal ranibizumab for the treatment of neovascular age-related macular degeneration. Retina Phila Pa. 2016;36(8):1418–31.
Spooner KL, Mhlanga CT, Hong TH, Broadhead GK, Chang AA. The burden of neovascular age-related macular degeneration: a patient’s perspective. Clin Ophthalmol Auckl NZ. 2018;4(12):2483–91.
Novartis. Beovu: Highlights of prescribing information. 2019. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/761125s000lbl.pdf. Accessed 3 June 2022.
European Medicines Agency (EMA). Beovu (brolucizumab) An overview of Beovu and why it is authorised in the EU. 2020. https://www.ema.europa.eu/en/documents/overview/beovu-epar-medicine-overview_en.pdf. Accessed 6 Mar 2022.
Dugel PU, Koh A, Ogura Y, et al. HAWK and HARRIER: phase 3, multicenter, randomized, double-masked trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2020;127(1):72–84.
Dugel PU, Singh RP, Koh A, et al. HAWK and HARRIER: ninety-six-week outcomes from the phase 3 trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2021;128(1):89–99.
Zubricky R, McCoy J, Sinha SB, et al. Injection intervals in real-world neovascular age-related macular degeneration (nAMD) switch brolucizumab (BROL) patients with at least 12 months of follow-up. Invest Ophthalmol Vis Sci. 2022;63(7):1313-F0147.
Bilgic A, Kodjikian L, March de Ribot F, et al. Real-world experience with brolucizumab in wet age-related macular degeneration: the REBA study. J Clin Med. 2021;10(13):2758.
Haensli C, Pfister IB, Garweg JG. Switching to brolucizumab in neovascular age-related macular degeneration incompletely responsive to ranibizumab or aflibercept: real-life 6 month outcomes. J Clin Med. 2021;10(12):2666.
Walter SD, Saba NJ. Real-world efficacy and safety of brolucizumab. Invest Ophthalmol Vis Sci. 2021;62(8):456.
Hamou SJ, Raimondo CD, Weber P, Woods BC. A retrospective study on the use of brolucizumab for the treatment of NVAMD: a 1-year private practice experience. Invest Ophthalmol Vis Sci. 2021;62(8):286.
Kishimoto M, Miki A, Chubachi A, et al. One-year results of switching to brolucizumab in exudative age-related macular degeneration. Invest Ophthalmol Vis Sci. 2022;63(7):323-F0154.
Richter H, Dombrowski S, Hamer H, Hadji P, Kostev K. Use of a German longitudinal prescription database (LRx) in pharmacoepidemiology. GMS Ger Med Sci. 2015;25:13. https://doi.org/10.3205/000218.
Jacob L, Yakkali B, Parekh M, Kostev K. No effects of the COVID-19 pandemic on the prescription of insulin in Germany. Prim Care Diabetes. 2022;16(4):588–90.
Bulirsch LM, Saßmannshausen M, Nadal J, Liegl R, Thiele S, Holz FG. Short-term real-world outcomes following intravitreal brolucizumab for neovascular AMD: SHIFT study. Br J Ophthalmol. 2021. https://doi.org/10.1136/bjophthalmol-2020-318672.
Abdin AD, Aljundi W, El Jawhari K, Suffo S, Weinstein I, Seitz B. First year real life experience with intravitreal brolucizumab for treatment of refractory neovascular age-related macular degeneration. Front Pharmacol. 2022. https://doi.org/10.3389/fphar.2022.860784.
Schmidt-Erfurth U, Chong V, Loewenstein A, et al. Guidelines for the management of neovascular age-related macular degeneration by the European Society of Retina Specialists (EURETINA). Br J Ophthalmol. 2014;98(9):1144–67.
Kodjikian L, Parravano M, Clemens A, et al. Fluid as a critical biomarker in neovascular age-related macular degeneration management: literature review and consensus recommendations. Eye. 2021;35(8):2119–35.
Ito A, Maruyama-Inoue M, Kitajima Y, Ikeda S, Inoue T, Kadonosono K. One-year outcomes of intravitreal brolucizumab injections in patients with polypoidal choroidal vasculopathy. Sci Rep. 2022;12(1):7987.
Schmidt-Erfurth U, Mulyukov Z, Gerendas BS, et al. Therapeutic response in the HAWK and HARRIER trials using deep learning in retinal fluid volume and compartment analysis. Eye. 2022;6:1–10.
Khanani A, Srinivas RS, Sarraf D, et al. Comparative assessment of the maximum thickness of pigment epithelial detachments and sub-RPE fluid in patients treated with brolucizumab versus aflibercept in the HAWK & HARRIER studies. 2021. https://doi.org/10.1097/IAE.0000000000002195.
Baumal CR, Bodaghi B, Singer M, et al. Expert opinion on management of intraocular inflammation, retinal vasculitis, and vascular occlusion after brolucizumab treatment. Ophthalmol Retina. 2021;5(6):519–27.
Holz FG, Heinz C, Wolf A, Hoerauf H, Pleyer U. Intraocular inflammation with brolucizumab use: patient management-diagnosis-therapy. Ophthalmol Z Dtsch Ophthalmol Ges. 2021;118(3):248–56.
Kilmartin D. Proposed best practice for Irish ophthalmologists: monitoring of patients following intravitreal brolucizumab therapy. SSRN. 2021. https://papers.ssrn.com/abstract=3937320. Accessed 13 May 2022.
Monés J, Srivastava SK, Jaffe GJ, et al. Risk of inflammation, retinal vasculitis, and retinal occlusion-related events with brolucizumab: post hoc review of HAWK and HARRIER. Ophthalmology. 2021;128(7):1050–9.
Pearce I, Amoaku W, Bailey C, et al. The changing landscape for the management of patients with neovascular AMD: brolucizumab in clinical practice. Eye. 2022;21:1–10.
Khanani AM, Brown DM, Jaffe GJ, et al. MERLIN: phase 3a, multicenter, randomized, double-masked trial of brolucizumab in participants with nAMD and persistent retinal fluid. Ophthalmology. 2022;129(9):974–85.
Khanani AM, Zarbin MA, Barakat MR, et al. Safety outcomes of brolucizumab in neovascular age-related macular degeneration. JAMA Ophthalmol. 2022;140(1):20–8.
Fonollosa A, Gallego-Pinazo R, Sararols L, Adán A, López-Gálvez M, Figueroa MS. Guidance on brolucizumab management recommendations. Arch Soc Esp Oftalmol Engl Ed. 2022. https://doi.org/10.1016/j.oftale.2022.05.006.
Aziz AA, Khanani AM, London N, et al. Real world efficacy and safety of brolucizumab in neovascular AMD: the REBEL study. Invest Ophthalmol Vis Sci. 2021;62(8):451.
Matsumoto H, Hoshino J, Mukai R, Nakamura K, Akiyama H. One-year results of treat-and-extend regimen with intravitreal brolucizumab for treatment-naïve neovascular age-related macular degeneration with type 1 macular neovascularization. Sci Rep. 2022;12(1):8195.
Kataoka K, Horiguchi E, Kawano K, et al. Three cases of brolucizumab-associated retinal vasculitis treated with systemic and local steroid therapy. Jpn J Ophthalmol. 2021;65(2):199–207.
Acknowledgements
Funding
The study and medical writing/editorial support was funded by Novartis Pharma AG. The journal’s Rapid Service fees were also funded by Novartis Pharma AG.
Editorial, Medical Writing, and Other Assistance
Authors would like to thank Cátia Bandeiras from Novartis for her project management, interpretation of results and writing support in this project. The authors would like to thank IQVIA (Anna Cascarano, Jo Wilfer and Sofia Zappacosta) for their joint support with analysis, interpretation of results and manuscript development.
Author Contributions
All authors contributed towards study conception and design, analysis, interpretation of data, drafting of paper and revising it critically for intellectual content. All authors were involved in the final approval for submission and agree to be accountable for all aspects of the work.
Disclosures
Raffael Liegl is a senior attending at University of Bonn, Dept. of Ophthalmology. Rita Freitas, Franklin Igwe and Helene Karcher are employees and shareholders of Novartis. Shala Chetty-Mhlanga is an employee of IQVIA, who received funding from Novartis Pharma AG for the conduct of this study.
Prior Presentation
This study was presented as an Audio Narrated Free Paper presentation at the 22nd EURETINA Congress; September 1–4, 2022; Hamburg, Germany and virtual.
Compliance with Ethics Guidelines
The German LRx database, used for analysis in this study, contains fully anonymised electronic prescription information and complies with relevant regulations for protecting patient privacy. Data were analysed in aggregated form without individual data becoming available. Individual consent forms were not obtained in line with German national and European Union legislation.
Data Availability
The data sets generated during and/or analysed during the current study are available from IQVIA. Restrictions might apply to the availability of these data, which were used under license for this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
The tREAtment patterns with broLucIZumab in GErmany (REALIZE) study.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Liegl, R.G., Karcher, H., Chetty-Mhlanga, S. et al. The Treatment Patterns with Brolucizumab in Germany (REALIZE) Study: A Retrospective Cohort Study Based on Longitudinal Prescription Data. Ophthalmol Ther 12, 195–208 (2023). https://doi.org/10.1007/s40123-022-00596-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-022-00596-7