Abstract
Introduction
To compare the results of an accelerated corneal cross-linking (CXL) protocol (9 mW/cm2, 10 min) with the standard CXL protocol (3 mW/cm2, 30 min) in patients with Down syndrome (DS) who have keratoconus (KC).
Methods
Twenty-seven 10- to 20-year-old patients with DS who had bilateral progressive KC were enrolled in a contralateral randomized trial and completed 2 years of follow-up examinations. Fellow eyes were randomly allocated to the accelerated CXL group or the standard CXL group. The main outcome measure was change in maximum keratometry (Kmax) centered on the steepest point (zonal Kmax − 3 mm) with a non-inferiority margin of 1.0 diopter (D). Vision and refraction tests, ophthalmic examinations, and corneal tomography were performed at baseline and at 6, 12, and 24 months after CXL. Failure was defined as an increase of ≥ 1.0 D in zonal Kmax − 3 mm within a 12-month period.
Results
The mean age (± standard deviation) of the patients was 15.71 ± 2.40 years. The within-group change in zonal Kmax − 3 mm was not significant after 2 years in either group, and within-group zonal Kmax − 3 mm remained stable. At 2 years after CXL, the mean change in the zonal Kmax − 3 mm was – 0.02 ± 0.81 D and – 0.31 ± 0.86 D in the accelerated CXL and standard CXL groups, respectively (P = 0.088). At 1 year of follow-up, three patients in the accelerated CXL group showed treatment failure (mean change in zonal Kmax − 3 mm + 2.12 ± 0.11 D); no patients in the standard CXL group showed treatment failure. At 2 years of follow-up, these three patients showed a decrease of – 0.43 ± 0.18 D in zonal Kmax − 3 mm from a baseline value of 55.11 ± 0.32 D. The 2-year trends of the inferior–superior asymmetry and vertical coma were statistically significantly different between the two groups, with the accelerated CXL protocol showing superiority in patients with higher baseline values.
Conclusion
In young patients with Down syndrome, the accelerated CXL protocol was able to halt disease progression and may be an alternative for the standard CXL protocol. In advanced KC, the efficacy of the accelerated approach was delayed and appeared later in the follow-up. In asymmetric cornea, the accelerated CXL resulted in centralization of the corneal cone.
Trial Registration
Iranian Registry of Clinical Trials, IRCT20100706004333N3
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
The incidence of keratoconus (KC) is sixfold higher in patients with Down syndrome (DS) than in the general population. |
Results from case reports on patients with DS who had KC and were treated with the standard corneal cross-linking (CXL) protocol are controversial, but to date the accelerated CXL protocol has not been evaluated in this patient population. |
What was learned from the study? |
In young patients with DS, the accelerated protocol could halt disease progression. |
In advanced KC, the efficacy of the accelerated approach was delayed and appeared later in the follow-up. |
In asymmetric cornea, the accelerated CXL protocol resulted in centralization of the corneal cone. |
Digital Features
This article is published with digital features to facilitate understanding of the article. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.12933461.
Introduction
The incidence of keratoconus (KC) is sixfold higher in patients with Down syndrome (DS) than in the general population [1]. In these patients, corneal cross-linking (CXL) is the preferred treatment for stopping disease progression and improving vision and quality of life [2]. Multiple meta-analyses have demonstrated the safety and efficacy of standard and accelerated CXL for the treatment of progressive KC [3,4,5,6], but studies in patients with DS are limited. The authors of one study on two cases suggested that standard CXL in these patients could prevent disease progression; however, one of the patients developed an infectious corneal ulcer [7]. Soeters et al. [8] reported acceptable 1-year results with standard CXL (performed under local anesthesia) in seven eyes. Sabti et al. [9] also reported desirable 3-year results using the standard CXL protocol in a 4-year-old girl. Faschinger et al. [10] described corneal melting after CXL in a patient with DS. Accelerated CXL protocols have the advantage of reducing surgical time, and shorter treatments are less demanding of resources and less stressful for patients. However, to our knowledge, no study has yet compared the efficacy of the standard protocol (3 mW/cm2, 30 min) with the accelerated protocol (9 mW/cm2, 10 min) in patients DS, which is the main objective of this study.
Methods
Study Design
This non-inferiority, contralateral, and randomized trial was conducted at Noor Eye Hospital in Tehran following the principles of Good Clinical Practice. The non-inferiority design was chosen to test the hypothesis that the accelerated CXL protocol is equally effective (or not unacceptably less effective) as the standard CXL procedure in patients with DS at 2 years after the procedure. The study protocol is shown in Electronic Supplementary material file.
Patients and Sampling
Cases were identified through a population-based study in which 250 patients with DS were recruited [11]. The diagnosis of DS, as indicated in their medical records, was confirmed by karyotype testing. Of those recruited, all patients with DS between the ages of 10 and 20 years who also had bilateral KC were considered for enrollment. Those patients aged ≤ 14 years underwent CXL immediately after enrollment, in accordance with national guidelines that take into account the rapid progression of this disease. Those patients > 14 years of age were monitored for progression every 6 months, up to 18 months. Inclusion criteria for this group was (1) the diagnosis of progressive KC defined as ≥ 1.0 diopter (D) increase in zonal maximum keratometry (Kmax) in a 3-mm zone around the steepest point (zonal Kmax − 3 mm) [11]; (2) ≥ 2.0% decrease in minimum corneal thickness (MCT), or an increase of ≥ 1.0 D in refractive astigmatism, within a 12-month period [12]. Exclusion criteria were having any concurrent intellectual or mental disorder, such as Klinefelter syndrome and autism, or any contraindication for surgery and anesthesia.
Randomization
A balanced block randomization with a block size of 4 was used in this study, such that the right eye of each patient was randomly allocated to one protocol, and the left eye was assigned to the other protocol. To maintain concealment of treatment allocation, randomization was performed by one person (SA) other than the surgeons. The surgeons were masked to treatment allocation until the time of surgery. The statistician (HZ) and ophthalmic healthcare providers who performed the study tests were also blinded to treatment allocation throughout the study.
Examinations
Since CXL was to be performed under general anesthesia, all patients were examined by a cardiologist and an anesthesiologist, and biochemistry and hematology laboratory tests were run. Vision and ophthalmic examinations were performed at baseline, and at 6, 12, and 24 months after the CXL procedure. In addition to slit lamp (Haag-Streit, Mason, OH, USA) examinations, the subjects were tested for uncorrected and corrected distance visual acuity (UDVA and CDVA, respectively) using the Snellen SC-2000 visual measurement system (Nidek Inc., Tokyo, Japan), refractive astigmatism by retinoscopy (ParaStop HEINE BETA 200; HEINE Optotechnik, Herrsching, Germany), and tomography using the Pentacam HR high-resolution rotating Scheimpflug camera system (Oculus Optikgeräte GmbH, Wetzlar, Germany). All tests were performed between 8 am and 12 noon. Imaging was repeated, if necessary, until an acceptable quality (minimum valid data: 93.0%) was acquired. If more than three attempts were needed, another appointment was scheduled for 2–3 days later to avoid participant fatigue and measurement error. The same optometrist performed the baseline and post-CXL tests with each device.
The indices extracted from the Pentacam system were maximum keratometry in the 3 mm zone around the point of maximum keratometry (zonal Kmax − 3 mm), MCT, inferior–superior asymmetry (I–S value), anterior and posterior elevation at the apex and thinnest point, anterior (ARC) and posterior (PRC) radius of curvature centered on the thinnest point, and anterior vertical coma.
Outcomes
The main outcome measure was an inter-group difference of ≤ 1.0 D in zonal Kmax − 3 mm. Secondary outcomes were 6-, 12-, and 24-month changes in UDVA, CDVA, refractive astigmatism, and tomographic indices. Based on the main outcome measure, failure was defined as a ≥ 1.0 D increase in zonal Kmax − 3 mm.
Interventions
All CXL procedures were performed under general anesthesia to standardize treatment conditions in a patient group with variable tolerance for CXL under local anesthesia. CXL was performed by two surgeons with similar skill and experience in performing the procedure (KA and MS). In both groups, after inducing general anesthesia, the central 9.0-mm epithelium was removed manually using a hockey knife. After removing the lid speculum, VibeX Rapid riboflavin 0.1% (Avedro Inc., Waltham, MA, USA) was instilled five times at 3-min intervals to soak the cornea, and those with a baseline pachymetry of < 400 µm were hydrated with sterile distilled water. Following anterior chamber saturation with riboflavin, the KXL System (Avedro Inc.) was used to deliver UV light onto the cornea at 3 mW/cm2 for 30 min in the standard CXL group and at 9 mW/cm2 for 10 min in the accelerated CXL group. Riboflavin instillation was repeated every 5 min during irradiation. After this step, the corneal surface was rinsed with balanced saline solution, a soft bandage contact lens (Ciba Vision, Duluth, GA) was placed on the eye, and one drop of levofloxacin (Sina Darou, Tehran, Iran) was instilled. Patients were monitored in the hospital until vital signs were stabilized. Postoperative treatment included levofloxacin eye drops four times daily, betamethasone 0.1% (Sina Darou) four times daily, and preservative-free artificial tears (Hypromellose) as needed. Parents were advised to take precautions against eye rubbing. Patients were examined on days 1 and 3 after the CXL procedure, and the bandage contact lens was removed if re-epithelialization was observed. If re-epithelialization was not observed by the third day, daily visits were continued until re-epithelialization was complete. After removal of the contact lens, levofloxacin was discontinued, and betamethasone 0.1% was continued for one more week. No complications were observed during or after the procedure in any of the patients. The CXL protocol details are shown in Electronic Supplementary material table.
Statistical Analysis
Given that the main effect of CXL is corneal flattening, zonal Kmax − 3 mm was considered to be the main outcome measure. Taking into account the contralateral design of the study, we calculated the sample size as n = 27 bilateral cases using:
where the non-inferiority margin or delta (δ) was calculated as 1.0 with a standard deviation (σ) of a 1.2 D reduction in zonal Kmax − 3 mm [11] for the null hypothesis, α = 0.0167 (0.05 divided by 3 for pre- and post-CXL measurements) and β = 0.05 (power = 95%).
Analyses were performed using R package version 3.5.2 (R Foundation for Statistical Computing, Vienna, Austria). A linear random mixed-effect model was used to compare the 2-year trends between the groups. The analyses focused on examining the correlation between fellow eyes and the follow-up times in an autoregressive correlation matrix, and baseline values of indices were entered into the model as covariates. Within-group changes were tested with repeated measures analysis of variance. The analysis approach was intention-to-treat. Refractive astigmatism was analyzed using the Alpins method [13]. The level of significance was set at 0.0167 for all main and secondary outcome indices.
Ethical Considerations
The goals and methods of the study were explained to the parents in the presence of the patients; signed informed consents were obtained from parents before enrollment into the study, and verbal assent was obtained from patients before every procedure. The study adhered to the tenets of the Declaration of Helsinki of 1964 and its later amendments. Approval for this study was obtained from the Ethics Committee of Tehran University of Medical Sciences (ID: IR.TUMS.MEDICINE.REC.1397.091), National Institute for Medical Research Development (IR.NIMAD.REC.1398.03), and the Iranian Registry of Clinical Trials, a member of the WHO Registry Network under registration number IRCT20100706004333N3.
Results
Twenty-seven patients with DS who had bilateral progressive KC were included in the study. A flow diagram of the enrollment procedure is shown in Electronic Supplementary Material Fig. 1. The mean age of the sample was 15.78 ± 2.46 (range 11–19) years, and 55.6% were male. Three patients were under 14 years of age, and the CXL procedure was performed immediately after the initial diagnosis in these three patients. All participants underwent all examinations during the 2-year follow-up, and no patient was lost to follow-up. Baseline vision tests were not successfully completed for six patients, and three patients had missing refraction data.
Main Outcome
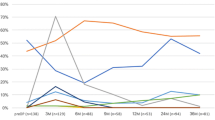
Baseline zonal Kmax − 3 mm was not statistically significantly different between the two groups (48.90 ± 3.12 D for the accelerated CXL group and 48.97 ± 3.04 D for the standard CXL group; P = 0.988). At 2 years after CXL, the change zonal Kmax − 3 mm was – 0.02 ± 0.81D in the accelerated group (P = 0.156) and − 0.31 ± 0.86D in the standard group (P = 0.446); the inter-group difference in 2-year change was not statistically significant (P = 0.088) (Fig. 1).
Secondary Outcomes
The studied indices measured at baseline and at 6, 12, and 24 months after the CXL procedure are summarized in Table 1. At baseline, only the I–S value (P < 0.001), anterior elevation at the thinnest point (P < 0.001), and anterior vertical coma (P < 0.001) were significantly different between the two groups, with higher values in the accelerated CXL group.
At 2 years after CXL, significant within-group changes were observed only in the accelerated group, with changes in the I–S value (− 0.66 ± 0.73 D; P < 0.001), PRC (− 0.13 ± 0.15 mm; P = 0.016), and anterior vertical coma (− 0.20 ± 0.17 µm; P < 0.001). The inter-group difference in 2-year changes in the I–S value (P = 0.008), PRC (P = 0.003), and anterior vertical coma (P < 0.001) were statistically significant. None of the other indices showed significant differences within or between the groups (all P > 0.0167).
Complications
At 1 year after CXL, mean change in zonal Kmax − 3 mm was + 2.12 ± 0.11 D in the subgroup of failed cases in the accelerated group (n = 3 patients, 11.1%). No treatment failure was observed in the standard group. At 2 years after CXL, no treatment failure was detected in either group, and the three patients for whom treatment failed showed a mean change of − 0.43 ± 0.18 D in zonal Kmax − 3 mm. Table 2 presents the baseline values of zonal Kmax − 3 mm and its changes in these two subgroups (failed and success cases) of each group.
No case of endothelial edema, corneal infection, scarring, or epithelial healing problems were observed in follow-up examinations.
Discussion
The cornea in patients with DS has a different structure than that in persons without DS, and even in the absence of pathologies, it can be thinner and steeper [14]. The reported prevalence of KC is also higher in these patients compared to normal subjects [1]. Despite the risk of certain complications, CXL can effectively prevent disease progression, depending on appropriate patient selection.
Few published studies have reported the results of CXL in patients with DS. In the study by Soeters et al. [8], the 1-year single-point Kmax reduction was 0.22 D in the seven eyes treated with standard CXL, and in the case study by Sabti et al. [9] the reduction was 0.8 D in one eye and 1.3 D in the fellow eye. In their review article, Perez-Straziota et al. [15] reported that single-point Kmax declined from 1.8 to 0.5 D 1 year after standard CXL in normal patients under 18 years of age. Single-point Kmax reductions of 1.2 D [15] and 0.7 D [16] have also been reported after accelerated CXL. It would appear that CXL protocols in individuals with DS have a smaller flattening effect as those in individuals without DS in the same age group. In our patient group, based on a non-inferiority margin of 1.0 D for zonal Kmax − 3 mm, the inferiority of accelerated CXL compared to the standard protocol was rejected, and the two protocols were not clinically different, especially in long-term follow-up.
Based on the 2-year results of our study, zonal Kmax − 3 mm was decreased by 0.02 D in the accelerated CXL group and by 0.31 D in the standard CXL group. In the accelerated CXL group, zonal Kmax − 3 mm had increased by 0.06 D at 1 year of follow-up and decreased by 0.04 D at 2 years of follow-up (average 0.02 D decrease). At 1 year after CXL, three patients were identified with treatment failure; these patients showed improvement in the second year, with all three showing flattening by the end of the second year. No intervention was done for these patients experiencing treatment failure after the first year, and they were only monitored. As Table 2 demonstrates, patients who failed treatment had a higher baseline zonal Kmax − 3 mm (55.1 D) and were older (17.3 years) than those for whom treatment was successful. These results show that the accelerated CXL protocol seems to have had a delayed effect in these patients. In the standard CXL group, the decrease was 0.10 D during the first year and 0.20 D in the second year. Our results indicate that longer follow-ups are needed to examine the possibly delayed effect of the accelerated CXL protocol.
Overall, in our study, the 1-year treatment failure rate (based on ≥ 1.0 D increase in zonal Kmax − 3 mm) was 11.1% in the accelerated CXL group and 0.0% in the standard group. The reported failure rate in similarly aged patients without DS is 15.4% [16] with the accelerated CXL protocol and 23.1% [16, 17] with the standard CXL protocol. From these results it would appear that the accelerated protocol has a lower flattening effect and lower failure rate in patients with DS. It should be noted that the single-point Kmax was used in the mentioned studies, and not the zonal Kmax − 3 mm.
The CDVA in both CXL groups decreased by about 1 Snellen line. In a meta-analysis of KC progression in subjects without DS, Ferdi et al. [18] showed that CVDA can decrease by 0.004 logMAR in 1 year, but this change was not statistically significant. In other words, the change is not due to disease progression and can be attributed to measurement repeatability. Given the subjectivity of the parameter and its dependence on the examiner and examinee, such observations can be expected. In patients with DS, measurement repeatability could be significantly reduced, but to our knowledge, no study has yet been performed that would help distinguish the contribution of repeatability from the success/failure of the procedure. Comparison of CDVA changes in our patients with DS with a sample of patients without DS [16] suggests that even with the standard CXL protocol, there is less improvement in patients with DS(0.06 vs. 0.14 logMAR). Given the level of concentration and cognitive ability of patients with DS and the subjectivity of the test, this difference is quite expected.
The I–S value, which is an index of the average power difference between the superior and inferior cornea, decreased in the accelerated CXL group (0.66 ± 0.73 D), with a significantly higher value at baseline and increased in the standard group (0.12 ± 1.64 D) after 2 years. In the study by Magli et al. [19], the Epi-Off group of patients without DS and aged < 18 years showed a decrease from 7.40 ± 0.7 to 6.84 ± 1.1 (0.54D decrease). To our knowledge, there is no published study on the repeatability of the measurements for this index. According to 2-year changes in the anterior vertical coma in our study, the decrease in the I–S value in the accelerated CXL group can be attributed to the centralization of the corneal cone.
The strength of this study is its randomized design in patients with DS who had bilateral KC; to our knowledge, this study is the first of this kind. However, having a larger sample size for the central and peripheral KC subgroups would have allowed for more accurate comparisons between the two protocols. Another limitation is the relatively small sample size and the low power of comparisons; also Bonferroni corrections, which are recommended for multiple comparisons, were not done to avoid further power reduction. The goal was to maintain a minimum power of 95% for the main outcome measure. Nonetheless, it should be noted that adjusting the P values and reducing power would generate the same results, thereby confirming the comparability of the two CXL approaches.
Conclusion
Overall, based on the observed changes in the studied indices, in DS patients under 20-years old, both standard and accelerated CXL protocols can halt disease progression. In more advanced cases of KC, the efficacy of the accelerated approach seems to be postponed until the second year. Also, in cases with higher I–S value and vertical coma, the accelerated protocol resulted to the centralization of the corneal cone.
References
Woodward MA, Blachley TS, Stein JD. The association between sociodemographic factors, common systemic diseases, and keratoconus: an analysis of a nationwide heath care claims database. Ophthalmology. 2016;123:457–65.
Hashemi H, Seyedian MA, Miraftab M, Fotouhi A, Asgari S. Corneal collagen cross-linking with riboflavin and ultraviolet A irradiation for keratoconus: long-term results. Ophthalmology. 2013;120:1515–20.
Wen D, Li Q, Song B, Wang Q, et al. Comparison of standard versus accelerated corneal collagen cross-linking for keratoconus: a meta-analysis. Invest Ophthalmol Vis Sci. 2018;59:3920–31.
Liu Y, Liu Y, Zhang Y-N, et al. Systematic review and meta-analysis comparing modified cross-linking and standard cross-linking for progressive keratoconus. Int J Ophthalmol. 2017;10:1419–29.
Shajari M, Kolb CM, Agha B, et al. Comparison of standard and accelerated corneal cross-linking for the treatment of keratoconus: a meta-analysis. Acta Ophthalmol. 2019;97:e22–35.
Miraftab M, Hashemi H, Abdollahi M, Nikfar S, Asgari S. The efficacy of standard versus accelerated epi-off corneal cross-linking protocols: a systematic review and sub-group analysis. Int Ophthalmol. 2019;39:2675–83.
Koppen C, Leysen I, Tassignon MJ. Riboflavin/UVA cross-linking for keratoconus in Down syndrome. J Refract Surg. 2010;26:623–4.
Soeters N, Bennen E, Wisse RPL. Performing corneal crosslinking under local anesthesia in patients with Down syndrome. Int Ophthalmol. 2018;38:917–22.
Sabti S, Tappeiner C, Frueh BE. Corneal cross-linking in a 4-year-old child with keratoconus and down syndrome. Cornea. 2015;34:1157–60.
Faschinger C, Kleinert R, Wedrich A. Corneal melting in both eyes after simultaneous corneal cross-linking in a patient with keratoconus and Down syndrome. Ophthalmologe. 2010;107(951–2):4–5.
Hashemi H, Miraftab M, Amanzadeh K, et al. Keratoconus detection by novel indices in patients with Down syndrome: a cohort population-based study. Jpn J Ophthalmol. 2020;64:285–91.
Burns DM, Johnston FM, Frazer DG, Patterson C, Jackson AJ. Keratoconus: an analysis of corneal asymmetry. Br J Ophthalmol. 2004;88:1252–5.
Alpins N. Astigmatism analysis by the Alpins method. J Cataract Refract Surg. 2001;27:31–49.
Asgari S, Mehravaran S, Fotouhi A, Makateb A, Hashemi H. Total corneal refractive power and shape in Down syndrome. Eur J Ophthalmol. 2019. https://doi.org/10.1177/1120672119883594.
Perez-Straziota C, Gaster RN, Rabinowitz YS. corneal cross-linking for pediatric keratcoconus review. Cornea. 2018;37:802–9.
Baenninger PB, Bachmann LM, Wienecke L, Thiel MA, Kaufmann C. Pediatric corneal cross-linking: comparison of visual and topographic outcomes between conventional and accelerated treatment. Am J Ophthalmol. 2017;183:11–6.
Henriquez MA, Villegas S, Rincon M, Maldonado C, Izquierdo L Jr. Long-term efficacy and safety after corneal collagen crosslinking in pediatric patients: three-year follow-up. Eur J Ophthalmol. 2018;28:415–8.
Ferdi AC, Nguyen V, Gore DM, Allan BD, Rozema JJ, Watson SL. Keratoconus natural progression: a systematic review and meta-analysis of 11529 eyes. Ophthalmology. 2019;126:935–45.
Magli A, Forte R, Tortori A, Capasso L, Marsico G, Piozzi E. Epithelium-off corneal collagen cross-linking versus transepithelial cross-linking for pediatric keratoconus. Cornea. 2013;32:597–601.
Acknowledgements
We thank the patients for participating in this trial.
Funding
The study was partially funded by National Institute for Medical Research Development (NIMAD)/# 973289. No funding or sponsorship was received for the publication of this article.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
HH and SA conceptualized and designed the study; HH, KA, and MAS acquired the data; SA and HZ performed the statistical analysis; SA, CR, SM, RA, RV, and PV interpreted the results; HH, CR, and SA drafted the manuscript; all authors critically revised and gave final approval to the manuscript.
Disclosures
Riccardo Vinciguerra, Paolo Vinciguerra, Renato Ambrosio Jr, and Cynthia J Roberts are consultants for Oculus Optikgeräte GmbH, Germany. Renato Ambrosio Jr is also a member of the journal’s Editorial Board. Hassan Hashemi, Kazem Amanzadeh, Mohammad Seyedian, Hojjat Zeraati, Shiva Mehravaran, and Soheila Asgari have nothing to declare.
Compliance with Ethics Guidelines
The goals and methods of the study were explained to the parents in the presence of the patients; signed informed consents were obtained from parents before enrollment into the study, and verbal assent was obtained from patients. The study adhered to the tenets of the Declaration of Helsinki of 1964 and its later amendments. Approval for this study was obtained from the Ethics Committee of Tehran University of Medical Sciences (ID:IR.TUMS.MEDICINE.REC.1397.091), National Institute for Medical Research Development (IR.NIMAD.REC.1398.03), and the Iranian Registry of Clinical Trials, a member of the WHO Registry Network under registration number IRCT20100706004333N3.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Digital Features
To view digital features for this article go to https://doi.org/10.6084/m9.figshare.12933461.
Electronic supplementary material
Below is the link to the electronic supplementary material.
40123_2020_303_MOESM1_ESM.tiff
Supplemental figure. Flow chart drawing the passage of patients in the randomized trial comparing accelerated 9 mW/cm2 and standard corneal cross-linking (TIFF 183 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Hashemi, H., Amanzadeh, K., Seyedian, M. et al. Accelerated and Standard Corneal Cross-Linking Protocols in Patients with Down Syndrome: A Non-inferiority Contralateral Randomized Trial. Ophthalmol Ther 9, 1011–1021 (2020). https://doi.org/10.1007/s40123-020-00303-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-020-00303-4