Abstract
Introduction
Laser photocoagulation has been the standard treatment for diabetic macular edema (DME) and proliferative diabetic retinopathy (PDR) for several decades. The discovery of vascular endothelial growth factor (VEGF) and the subsequent determination of its critical role in the development DME and PDR has led to the development of VEGF inhibitory drugs. Ranibizumab was the first anti-VEGF drug approved for the treatment of both DME and diabetic retinopathy in eyes with DME.
Methods
Medline searches with the keywords "ranibizumab," "diabetic macular edema," and "proliferative diabetic retinopathy" were performed to identify pertinent pre-clinical studies and clinical trials. Top-line data, with emphasis on pivotal trials, was identified and incorporated into this manuscript. Findings from small uncontrolled trials were generally not used unless they filled important gaps in our understanding of anti-VEGF therapy.
Results
Ranibizumab is a recombinant humanized antibody fragment that binds all isoforms of VEGF-A with high affinity. Three parallel lines of clinical research have produced level I evidence supporting the superiority of ranibizumab over laser photocoagulation for the treatment of DME. Regular injections also lead to improvement in diabetic retinopathy severity scores in a large minority of eyes. Ranibizumab is effective for PDR and produces less visual field loss than laser photocoagulation. It has an excellent safety profile, with low incidence of ocular and systemic adverse events.
Conclusions
Ranibizumab has become a frequently used first-line therapy for the treatment of DME. Emerging data suggest that it may become an important treatment for DR and PDR.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetes mellitus (DM) is responsible for more vision loss among working-aged individuals in industrialized nations than any other condition [1, 2]. Diabetes affects nearly every part of the visual system, but most vision loss results from complications of diabetic retinopathy (DR). Improved access to care and newly developed drugs are reducing the prevalence of DR within diabetic populations, but because the incidence of both type 1 and type 2 DM is increasing [3, 4], the total number of patients with DR is rapidly rising throughout the world. The resulting demand for endocrinology and ophthalmology services is straining healthcare delivery systems [5] and threatens to increase the number of patients with significant vision loss and blindness.
The lack of effective treatment for DR throughout much of the twentieth century meant that many patients with DR suffered complete loss of sight. Laser photocoagulation was introduced in the 1960s and rapidly became the standard of care for the treatment of diabetic macular edema (DME) [6] and proliferative diabetic retinopathy (PDR) [7]. Laser therapy frequently delayed or prevented vision loss, but significant improvement in visual acuity was uncommon [8]. Only with the development and introduction of intraocular pharmacotherapy have patients been able to reasonably hope for recovery of pre-existing visual acuity.
Ranibizumab (Lucentis®, Genentech, South San Francisco, CA, USA/Roche, Basel, Switzerland), a recombinant humanized antibody fragment that binds vascular endothelial growth factor (VEGF), was the first drug approved for the treatment of both DME and DR. This paper will focus on the clinical use of ranibizumab to treat vision-threatening complications of DR. This article is based on previously conducted studies and does not involve any new studies of human or animal subjects performed by any of the authors.
Development of Ranibizumab
Michaelson [9] first proposed the notion that a soluble vasoproliferative factor produced neovascularization in eyes with advanced DR, and Folkman [10] subsequently linked tumor growth to angiogenesis. The vasopermeability factor (VPF) was discovered in 1983 [11], and the vascular endothelial growth factor, determined by protein sequencing analysis to be identical to VPF, was discovered independently by Ferrara and Connolly [12, 13]. Researchers demonstrated that vascular endothelial growth factor concentrations were elevated in the vitreous of eyes with several chorioretinal vascular conditions, including DME and PDR [14]. Injecting VEGF into rat eyes was found to produce retinal hemorrhages and vascular changes similar to those seen in patients with DR [15], and was able to be prevented with the co-administration of a VEGF trap [16]. Injecting VEGF into monkey eyes produced neovascularization of the iris [17].
Several different bioengineering strategies have been used to develop VEGF-binding drugs. Scientists at Genentech produced bevacizumab, a full-length, recombinant, humanized, murine anti-VEGF antibody for the intravenous treatment of advanced solid malignancies. For several reasons, including concerns over a limited ability to penetrate the inner layers of the retina, possible production of an inflammatory response within the eye, and prolonged serum half-life, bevacizumab was not deemed appropriate for chorioretinal vascular conditions, so ranibizumab was developed exclusively for intraocular use [18].
Ranibizumab has a molecular weight of 48 kDa, approximately one-third that of a full-length antibody (Table 1). Six amino acids in the Fab originally used to construct bevacizumab were substituted to produce the affinity-enhanced ranibizumab molecule. Despite having only one VEGF binding site, ranibizumab is 5 to 20 times as potent as bevacizumab (two binding sites) when evaluated in similar bioassays [18]. Ranibizumab possesses a high binding affinity for VEGF165 (46–172 pM) [19, 20].
Research has shown that after the intravitreal injection of ranibizumab into monkey eyes, drug penetration of the outer retina occurs quickly, with resulting tissue concentrations approximately one-third of those seen in the vitreous [21]. Various studies suggest that the intravitreal half-life of ranibizumab ranges from 2.8 to 2.88 days [22, 23] in rabbits and 2.6 to 3.2 days in monkeys [21, 24]. Estimates of ranibizumab’s intraocular half-life in human eyes vary from 7.19 days (based on anterior segment samples) [25] to 9 days (population pharmacokinetic study) [26]. Ranibizumab is believed to pass from the eye into the systemic circulation without undergoing metabolic changes. Because of its short serum half-life of 2 h, ranibizumab does not diminish serum VEGF concentrations [27] before being removed from the body by ultrafiltration through the kidneys.
Figure 1 presents optical coherence tomography images illustrating improvements in a patient receiving monthly ranibizumab injections.
Optical coherence tomography scans through the macula of a 68-year-old woman with type 2 diabetes mellitus for 15 years. Her visual acuity at the baseline examination (top row) measured 20/40 −2, and the central subfield thickness measured 414 µm. After 6 monthly injections of 0.3 mg ranibizumab (middle row), her visual acuity had improved to 20/40 + 2 and the central subfield thickness had improved to 350 µm. Monthly injections were continued, and by 14 months (bottom row), the macular edema had largely resolved except for a small residual cystoid space. At this time, her visual acuity measured 20/30 and central subfield thickness measured 260 µm
Clinical Trials for Diabetic Macular Edema
Pilot Trials
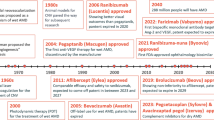
Ranibizumab entered clinical trials for neovascular age-related macular degeneration (nAMD) in 2000, an investigative sequence that culminated with the publication of the ANCHOR [28] and MARINA [29] trials in 2006 and approval by the US Food and Drug Administration (FDA) in the same year. At approximately the same time, two small pilot studies explored the safety and efficacy of ranibizumab in patients with DME. In the READ-1 study [30], ten patients with chronic DME received intravitreal injections of 0.5 mg ranibizumab at baseline and 1, 2, 4, and 6 months. By the 7-month endpoint, mean best-corrected visual acuity (BCVA) had improved by +12.3 letters. Mean macular volume improved from 9.22 to 7.47 mm2 (77% reduction in excess volume) and mean foveal thickness improved from 503 µm to 257 µm (85% reduction in excess thickness). The other study enrolled ten patients in an open-label, dose-escalation protocol [31]. Patients received low-dose (0.3 mg) or high-dose (0.5 mg) ranibizumab at baseline, and 1 and 2 months. The mean improvements in BCVA and macular thickness at 3 months for the 0.3-mg and 0.5-mg arms were +12 and +7.8 letters and −45.3 and −197.8 µm, respectively. Improvements in BCVA decreased slightly between 3 and 6 months.
These studies demonstrated that DME responds well to ranibizumab, but they failed to demonstrate superiority of either the 0.3-mg or 0.5-mg dose and suggested that 2-month injection intervals may be sufficient. Subsequent development of ranibizumab for DME proceeded along three research lines: READ-2/RISE and RIDE (Genentech-sponsored in the USA and South America); RESOLVE/RESTORE (Novartis-sponsored ex-US); and Diabetic Retinopathy Clinical Research Network (DRCR.net) Protocol I (National Eye Institute-sponsored). Table 2 lists the important DR clinical trials with ranibizumab and details their key findings.
READ/RISE and RIDE
Following successful completion of the READ-1 trial, the phase II READ-2 trial was performed at 14 sites [32]. This prospective controlled trial randomized 126 patients to receive 0.5 mg ranibizumab at baseline and 1, 3, and 5 months (group 1), laser at baseline and at month 3 if needed (group 2), and ranibizumab combined with laser at baseline and month 3 if needed (group 3). At the 6-month primary endpoint, the improvement in BCVA for group 1 was significantly greater than for group 2 (+7.24 vs. −0.43 letters; P = 0.0001) but not significantly different from that of group 3 (+3.8 letters). Fifteen-letter improvement in BCVA was achieved by 22%, 0%, and 8%, and excess macular thickness was reduced in 50%, 33%, and 45%, respectively.
Between months 6 and 24, all patients became eligible to receive ranibizumab injections every 2 months. At 24 months, mean improvements in BCVA were +7.7, +5.1, and +6.8 letters, and the proportions of patients improving by 15 letters were 24%, 18%, and 26%. Mean central subfield thickness (CST) measurements were 340, 286, and 258 µm. By month 24, mean visual acuity improvement experienced by the cohort initially randomized to laser approached that of the ranibizumab group, but the excess macula thickness in the ranibizumab monotherapy group indicated that these patients were probably under-treated [33].
At 24 months, the protocol was amended a second time to allow all patients to receive monthly pro re nata (PRN) ranibizumab in year 3. From the 24-month to the 36-month visits, patients had mean BCVA gains of +3.1, −1.6, and +2.0 letters. An average of 5.4, 2.3, and 3.2 ranibizumab injections were administered during the third year [34].
A higher dose of ranibizumab (2.0 mg) had been tested during the exploratory dosing nAMD trials, but it was quickly abandoned because of its high viscosity and tendency to cause inflammation. Several years later, however, the 2.0-mg dose was re-evaluated in the HARBOR trial [35] for nAMD and the READ-3 trial [36] for DME. Patients with DME received either the 2.0-mg or the 0.5-mg ranibizumab dose monthly for 6 months, followed by monthly PRN injections through month 12. Patients receiving 0.5 mg experienced a non-statistically significant greater improvement in BCVA (+9.34 letters vs. +7.04 letters), and more patients receiving the 2.0-mg dose died from myocardial infarction (4% vs. 1%). Because neither the READ-3 nor HARBOR trial showed that high-dose ranibizumab offered a clinical advantage, further development of the 2.0-mg dose was halted. The READ trials suggested that monthly ranibizumab was needed to produce optimal visual acuity results, but that higher doses provided no added benefit and might actually increase the risk of serious systemic adverse events.
As a result, the multi-center, double-masked parallel 3-year phase III RISE and RIDE registration trials compared the efficacy of lower-dose (0.3 mg and 0.5 mg) monthly ranibizumab against sham for patients with center-involving DME [37]. Seven hundred fifty-nine patients were randomized to one of three treatment arms: monthly 0.3 mg ranibizumab, monthly 0.5 mg ranibizumab, or sham. Patients were eligible for rescue laser photocoagulation at 3 months if the central retinal thickness (CRT) was >250 µm and if the change in CRT following the previous injection was <50 µm. The primary endpoint was the proportion of subjects improving by at least +15 letters at 24 months, and secondary endpoints included improvement in BCVA, improvement in macular thickness, and safety measures.
At the 24-month primary endpoint, the proportion of patients who improved by at least +15 letters was significantly greater in patients receiving 0.3 mg and 0.5 mg ranibizumab than in patients who received sham injections in both RISE (44.8%, 39.2%, and 18.1%) and RIDE (33.6%, 45.7%, and 12.3%). Mean improvement in BCVA was also greater in the groups receiving ranibizumab in both RISE (+12.5, +11.9, and +2.6 letters) and RIDE (+12.0, +10.9, and +2.3 letters), as were corresponding mean improvements in central foveal thickness (CFT) in RISE (−250.6 µm, −253.1 µm, and −133.4 µm) and RIDE (−259.8 µm, −270.7 µm, and −125.8 µm). Fewer patients receiving ranibizumab required rescue laser (means of 0.3 to 0.8) compared to those randomized to sham injections (means of 1.8 and 1.6). In the groups treated with 0.3 mg ranibizumab, 37.2% experienced a two-step or greater improvement in diabetic retinopathy severity score (DRSS), and 13.2% experienced a three-step or greater improvement.
The median DRSS remained stable at moderately severe NPDR in the sham groups, whereas scores improved from moderately severe to mild NPDR in the ranibizumab groups. Fewer patients receiving ranibizumab experienced a two-step worsening in the DRSS (1.7% to 2.1% vs. 9.6% in sham), and fewer ranibizumab patients developed vitreous hemorrhage. Since changes in BCVA were the same for patients receiving the 0.3-mg and 0.5-mg doses of ranibizumab, and the incidence of several categories of adverse events including central nervous system and cerebrovascular hemorrhage appeared to increase in a dose-dependent fashion (although the numbers were low), Genentech recommended the 0.3-mg dose for approval. The US FDA ultimately approved the use of 0.3 mg ranibizumab for DME on August 10, 2012. The label was subsequently expanded (2015) to include the treatment of diabetic retinopathy (NPDR and PDR) in patients with DME.
During year 3, patients continued receiving monthly ranibizumab injections, and those randomized to sham were eligible to receive monthly 0.5 mg ranibizumab [38]. From the 24-month to the 36-month visits, patients in the ranibizumab arms had stable BCVA, whereas those in the sham arms improved to +4.3 (RISE) and +4.7 (RIDE) letters above baseline.
After the 36-month visit, 500 patients were followed in the extension study for a mean of 14.1 months [39]. All patients were eligible to receive monthly PRN 0.5 mg ranibizumab if DME was identified by the investigator or BCVA worsened by at least five letters compared to month 36. Patients received a mean of 4.5 injections (annualized: 3.8), but 24.2% of patients did not require any injections. BCVA remained stable in all groups and mean CFT increased slightly. Few patients developed PDR, and those originally randomized to ranibizumab had a lower overall rate of progression to PDR than those originally randomized to sham.
RESOLVE/RESTORE
The phase II RESOLVE and phase III RESTORE trials that were performed in Europe, Asia, and Australia served as the basis for regulatory approval of ranibizumab in these regions for treatment of DME. In the 12-month RESOLVE trial, 151 patients were randomized to receive monthly 0.3 mg ranibizumab, 0.5 mg ranibizumab, or sham injections [40]. After 1 month, the drug dose could be doubled if the CRT was greater than 300 µm, or greater than 225 µm if the CRT decreased by <50 µm following the previous injection. At 3 months, patients received ranibizumab injections or sham injections on a monthly PRN basis, and all were eligible for rescue laser photocoagulation. At the 12-month primary temporal endpoint, mean BCVA improved by +10.3 letters in the pooled ranibizumab groups but declined by −1.4 letters in the sham group. BCVA gains of +2 lines and +3 lines were achieved by 60.8% and 32.4% of ranibizumab-treated eyes, but only 18.4% and 10.2% of sham-treated eyes, respectively. Mean improvements in CRT were −194.2 µm for the pooled ranibizumab groups and −48.4 µm for the sham group. Eighty-six percent of all eyes received a ranibizumab dose of 0.5 to 1.0 mg at some point during the trial. The mean number of administered injections was 10.2, and only 4.9% of ranibizumab treated-eyes (compared to 34.7% of sham eyes) required rescue laser.
Since nearly all patients in the RESOLVE trial eventually received a ranibizumab dose of at least 0.5 mg, only the larger (0.5 mg) dose was included in the design of the RESTORE trial. RESTORE was the multi-center (73 sites) phase III ranibizumab registration trial for the Eastern Hemisphere [41]. Three hundred forty-five patients were randomized to receive ranibizumab + sham laser, ranibizumab + laser, or sham injections + laser. Ranibizumab injections were administered monthly for three months, then monthly PRN; laser was performed at baseline and then every 3 months PRN. The trial’s primary objective was to demonstrate superior mean BCVA improvement at 12 months in patients treated with ranibizumab monotherapy or ranibizumab combined with laser compared to laser monotherapy. Secondary objectives included the proportions of patients achieving BCVA of at least 73 letters (20/40), the time course of the change in mean BCVA and CRT, patient-reported outcomes relative to laser photocoagulation, and safety measures.
At the 12-month primary endpoint, patients in the ranibizumab monotherapy, ranibizumab + laser, and sham/laser groups demonstrated improvements in mean BCVA (+6.1, +5.9, and +0.8 letters), BCVA score >73 letters (53.0%, 44.9%, and 23.6%) and CRT (−118.7 µm, −128.3 µm, and −61.3 µm). Health-related quality-of-life scores (measured by the 25-item National Eye Institute Visual Function Questionnaire [NEI VFQ-25]) improved more in the ranibizumab monotherapy and ranibizumab + laser groups than in the sham/laser group (P < 0.05 for each). Subgroup analyses showed that patients with baseline BCVA of ≥73 Early Treatment Diabetic Retinopathy Study (ETDRS) letters or CRT <400 µm achieved final BCVA that was as good with laser as with ranibizumab. Patients received a mean of seven ranibizumab/sham injections.
After the 12-month primary endpoint, 240 patients were enrolled in the 24-month extension trial. All patients were eligible to receive 0.5 mg ranibizumab according to BCVA and disease progression criteria, and at the investigators’ discretion. Additional laser photocoagulation was allowed according to ETDRS guidelines. At the pre-planned 24-month interim analysis, patients who originally received ranibizumab monotherapy and ranibizumab + laser maintained improvements in mean BCVA (+7.9 letters, +6.7 letters), CRT (−140.6 µm, −133.0 µm), and NEI VFQ-25 composite scores (5.6, 5.8) [42]. Between the 12- and 24-month examinations, patients originally treated with sham/laser experienced significant improvement in these measures (+5.4 letters, −126.6 µm, 4.3) after receiving ranibizumab. Similar numbers of injections were performed in each treatment arm (3.9, 3.5, and 4.1).
Most of the patients (208; 86.7%) were rolled into and completed the 24-month extension study. While patients received as-needed ranibizumab injections during the period between the 12- and 36-month examinations, BCVA and CRT were maintained [43]. At 36 months, improvements in BCVA were +8.0 letters (ranibizumab monotherapy), +6.7 letters (ranibizumab + previous laser), and +6.0 letters (original sham/laser arm). The mean numbers of injections over the final 2 years varied from 6.0 to 6.8 for each of the three treatment arms.
DRCR.net Protocol I
The DRCR.net Protocol I trial provided the first level I evidence that supported the use of ranibizumab as primary treatment for DME. Protocol I was a 5-year multi-center study that randomized 854 eyes with center-involving DME to receive 0.5 mg ranibizumab with prompt macular laser photocoagulation, 0.5 mg ranibizumab with deferred laser (for at least 6 months), intravitreal triamcinolone with prompt laser, or sham injections with prompt laser [44]. Patients received ranibizumab injections during the first year according to the 4:2:7 rule—four monthly injections, followed by two additional injections if fluid persisted, followed by seven visits during which the drug could be administered at the investigator’s discretion if insufficient improvement was noted. Laser photocoagulation of the macula and intravitreal triamcinolone (4 mg) injections could be repeated every 16 weeks and 13 weeks, respectively, as needed. Patients randomized to the ranibizumab + deferred laser group were not required to receive laser treatment at 6 months and beyond if the macula was dry.
At the 12-month examination, mean improvements in BCVA in the ranibizumab + prompt laser, ranibizumab + deferred laser, triamcinolone + laser, and sham + laser groups were +9, +9, +4, and +3 letters, respectively. Most of the gains in BCVA occurred by the 8-week visit. During the first 3 months of the trial, patients who were treated with triamcinolone + laser experienced improvement in BCVA that were similar to those receiving ranibizumab. However, BCVA in the triamcinolone group worsened over the subsequent 9 months because of the development of corticosteroid-induced cataracts. On the other hand, patients in the triamcinolone/laser group who were pseudophakic at baseline experienced 1-year improvement in BCVA comparable to that in the ranibizumab arms. The following subgroup analyses found no significant differences in 1-year visual acuity outcomes: prior treatment for DME, baseline visual acuity, baseline CST, and baseline severity of DR. Improvements in CST were comparable in the groups receiving ranibizumab and triamcinolone, all of which exceeded those in the group receiving sham/laser. Eyes treated with ranibizumab were less likely to experience increases in DRSS.
During the second year of the trial, the interval between examinations could be extended to 8 weeks if treatment had been deferred for two consecutive visits, and to 16 weeks if treatment was not performed at the 8-week visit. Patients in the triamcinolone/laser and laser/sham groups were eligible to receive ranibizumab as early as week 74 for persistent edema without improved BCVA. The 2-year BCVA outcomes were similar to those at 1 year, in that 44–49% of ranibizumab-treated eyes improved by at least +10 letters and 28–29% improved by at least +15 letters [45]. Compared to the sham/laser group, the mean changes in BCVA in patients receiving ranibizumab + prompt laser, ranibizumab + deferred laser, and triamcinolone + prompt laser were +3.7, +5.8, and −1.5 letters. Within the first 2 years of the trial, 43 eyes in the sham/laser group were switched to ranibizumab because of treatment “failure,” whereas only one of the patients randomized to ranibizumab required switching.
At the 3-year visit, the median numbers of injections given to patients in the ranibizumab + prompt laser and ranibizumab + deferred laser groups were 12 and 15, respectively [46], and the median numbers of laser treatments were 3 and 0, respectively. Only 46% of patients in the ranibizumab + deferred laser group had been treated with laser. Patients randomized to ranibizumab + deferred laser had BCVA improvements that were +2.9 letters greater than those randomized to ranibizumab + prompt laser (P = 0.02). The proportions of eyes with CST ≥250 µm were 36% in each of the ranibizumab arms.
At the 5-year visit, the mean BCVA improvements from baseline were +7.2 letters in the ranibizumab + prompt laser group and +9.8 letters in the ranibizumab + deferred laser group (P = 0.09) [47]. Visual acuity loss of ≥10 letters was experienced by 9% and 8% of patients, improvement of ≥10 letters by 46% and 58%, and improvement of ≥15 letters by 27% and 38%, respectively. Only 44% of patients who were randomized to ranibizumab + deferred laser received laser photocoagulation through 5 years. The median numbers of ranibizumab injections administered during the trial were 13 and 17, 54% and 45% of patients did not receive ranibizumab during year 4, and 62% and 52% did not receive ranibizumab during year 5.
RETAIN
The previously discussed trials demonstrated the efficacy of both monthly and PRN therapy for DME, but most physicians treat chorioretinal vascular conditions according to a treat-and-extend (T&E) regimen. The RETAIN trial randomized 372 patients to receive T&E 0.5 mg ranibizumab + laser (G1), T&E 0.5 mg ranibizumab (G2), or monthly PRN 0.5 mg ranibizumab (G3) [48]. Patients received monthly injections until BCVA stabilized, after which patients in G1 and G2 could be extended at 1-month intervals up to a maximum of 3 months. At the 24-month endpoint, median BCVA changes in the G1, G2, and G3 groups were +8.3, +6.5, and +8.1 letters, respectively. Seventy percent of patients receiving T&E were extended to a treatment interval of at least 2 months, and they required 40% fewer clinic visits than those receiving PRN injections.
Comparison Studies
Few prospective randomized DME trials have directly compared ranibizumab with bevacizumab and aflibercept. A 48-week randomized prospective trial compared bevacizumab with ranibizumab in 45 patients with DME [49]. BCVA improved significantly in both groups at all visits (P < 0.05), but those receiving ranibizumab experienced greater increases at weeks 8 (P = 0.0318) and 32 (P = 0.0415). Patients receiving bevacizumab required more injections than those receiving ranibizumab (means 9.84 vs. 7.67).
The prospective randomized multi-center DRCR.net Protocol T trial was the only trial to directly compare all three anti-VEGF drugs [50]. Six hundred sixty patients at 89 sites received 1.25 mg bevacizumab, 0.3 mg ranibizumab, or 2 mg aflibercept. Patients were treated according to a complicated algorithm that featured monthly injections until BCVA and CST were stable, and laser photocoagulation at 24 weeks for persistent edema.
The median numbers of injections through 52 weeks were nine (aflibercept), ten (bevacizumab), and ten (ranibizumab) (P = 0.045 for overall comparison), and laser photocoagulation was performed in 37%, 56%, and 46% of eyes, respectively (P < 0.001). Mean changes in BCVA were +13.3 letters (aflibercept), +9.7 letters (bevacizumab), and +11.2 letters (ranibizumab). A pre-planned subgroup analysis found that for eyes with baseline BCVA of 20/32 to 20/40, mean BCVA changes were +8.0 (aflibercept), +7.5 (bevacizumab), and +8.3 letters (ranibizumab). For eyes with baseline visual acuity of ≤20/50, mean changes in BCVA were +18.9 (aflibercept), +11.8 (bevacizumab), and +14.2 letters (ranibizumab), and the average changes in CST were −169 µm, −101 µm, and −147 µm.
The differences in BCVA improvements among the drugs had narrowed by 2 years and were no longer statistically significant for aflibercept vs. ranibizumab [51]. Mean improvements in BCVA for patients receiving aflibercept, bevacizumab, and ranibizumab were +12.8, +10.0, and +12.3 letters, and were +18.1, +13.3, and +16.1 letters in patients with baseline acuity of 20/50 or worse (aflibercept vs. ranibizumab, P = 0.18; ranibizumab vs. bevacizumab, P = 0.18). The median numbers of injections were 15, 16, and 15 (global: P = 0.08), and laser photocoagulation was performed in 41%, 64%, and 52% of patients.
Up to 40% of patients receiving ranibizumab injections experience a suboptimal response to therapy, with persistent macular edema [52]. Some authors recommend that these patients receive laser photocoagulation, intraocular corticosteroids, or a different anti-VEGF drug, but since randomized controlled trials have not adequately addressed this, physicians have insufficient guidance regarding the best strategy. A post hoc analysis of the DRCR.net Protocol I data evaluated the effects of “switching” therapy in patients treated with ranibizumab [53]. Switching rules were applied to patients at the 3- and 6-month visits. Over the subsequent 3 months, these patients experienced improvement in BCVA of three to five letters and CST of −40 to −70 µm. The authors point out the need for comparison groups, since improvement with switching may be confused with delayed improvement from ranibizumab therapy.
Proliferative Diabetic Retinopathy
In addition to restoring the integrity of the blood–retinal barrier and enabling resorption of macular edema, ranibizumab possesses potent anti-angiogenic activity. Ranibizumab has been used as monotherapy and in combination with panretinal photocoagulation to treat PDR. Retinal neovascularization rapidly involutes after ranibizumab injections, but these effects are usually transient, and pan-retinal photocoagulation (PRP) may still be necessary to permanently close new vessels.
Ranibizumab combined with PRP was compared to PRP alone for the treatment of high-risk PDR in 40 patients [54]. Split-session PRP was performed in all eyes at baseline and week 2, and 0.5 mg ranibizumab was given to the “PRP Plus” group at the completion of the first laser session and again at weeks 16 and 32 if needed. Patients were evaluated with ETDRS BCVA measurements, fluorescein angiography (FA), and OCT. Significant reductions in FA leakage were seen at all visits though 48 weeks in both groups, but the reduction was significantly greater in the PRP Plus group. BCVA worsened in the laser monotherapy group at 16, 32, and 48 weeks, but was unchanged in the PRP Plus group. The CST increased at all visits in the laser group but decreased at week 16 and stabilized through week 48 in the PRP Plus group. The authors concluded that the addition of ranibizumab to PRP reduces PDR-related FA leakage, and improves visual acuity and central macular thickness.
The DRCR.net Protocol S trial randomized 305 patients at 55 sites to receive panretinal photocoagulation or intravitreal 0.5 mg ranibizumab for PDR [55]. Complete photocoagulation was performed at baseline, and ranibizumab was injected every 4 weeks through 12 weeks, at weeks 16 and 20 if neovascularization persisted, and then monthly at the discretion of the investigator. All eyes with DME were eligible to receive ranibizumab. The primary outcome was change in BCVA and secondary outcomes included visual acuity area under the curve, peripheral visual field loss (as measured on Humphrey automated visual field testing), the need for vitrectomy, development of DME, and persistent or new neovascularization. Improvements in BCVA for the ranibizumab and PRP groups were +2.8 and +0.2 letters respectively (95% CI, −0.5 to +5.0). The ranibizumab group experienced less peripheral visual field sensitivity loss (−23 vs. −422 dB; 95% CI, 213–531 dB; P < 0.001), required fewer vitrectomy procedures (4% vs. 15%; 95% CI 4–15%; P < 0.001) and were less likely to develop DME (9% vs. 28%). For eyes without DME at baseline, a median of 7 ranibizumab injections were administered through year 1 and ten injections through year two. Forty-five percent of PRP eyes required additional laser and 35% of eyes in the PRP group received ranibizumab at baseline for DME and an additional 18% received ranibizumab because they developed DME during the 2 year trial.
Safety
Approximately 1% of ranibizumab has an excellent safety profile with low incidence of both ocular and systemic adverse events. Approximately 2% of patients in the RESOLVE and Protocol I trials developed endophthalmitis [40, 46] but incidence has subsequently fallen as physicians have become more proficient at delivering injections.
Patients with diabetes mellitus are at high risk of cardiovascular embolic events so adverse event rates must be viewed in the correct context. In the RESOLVE trial, the incidence rates of hypertension and arterial thromboembolic events were similar in the ranibizumab and sham groups [40]. By 3 years, the rates of Antiplatelet Trialists' Collaboration (APTC)-related events in the sham/laser, 0.3-mg ranibizumab, and 0.5-mg ranibizumab groups were 7.2%, 10.8%, and 10.4%, and the overall rates of death were 2.8%, 4.4%, and 6.4% [38]. In the Protocol T trial, APTC events were seen in 5% of patients receiving aflibercept, 8% receiving bevacizumab, and 12% receiving ranibizumab (global P = 0.047) [50]. These results must be contrasted with those from READ-2, Protocol I, and RESTORE, in which the incidence of APTC events in ranibizumab groups did not differ significantly from that in laser-treated patients. In aggregate, the incidence of serious systemic adverse events in patients treated with ranibizumab varies among trials, but does not appear to differ markedly from that associated with other anti-VEGF drugs.
Discussion
Several published trials have produced level I evidence showing that ranibizumab is superior to laser photocoagulation for the treatment of DME. Ranibizumab has thus emerged as an excellent first-line therapy for DME, either as monotherapy or in combination with laser photocoagulation of the macula. Initial intensive therapy (monthly injections) appear to produce the best short-term and long-term results. Following initial resolution of the macular edema, physicians have the discretion to continue monthly therapy, treat recurrent edema as needed, or pursue a treat-and-extend strategy. Fortunately, excellent long-term results have been reported with each strategy.
Following initial ranibizumab therapy, severity of the retinopathy frequently improves and the need for frequent injections lessens. Nonetheless, patient compliance remains critical to the delivery of effective therapy, and patients benefit when physicians carefully outline both the short-term and long-term strategies when initiating ranibizumab therapy.
Despite the best efforts of physicians and their staff, treatment of patients may be insufficient for several reasons. Many patients in both industrialized and developing nations have insufficient resources to cover the long-term costs incurred for the treatment of DME. This may be due to excessive travel costs to the physician’s office, insufficient health care insurance to pay for professional services, or inadequate access to effective drugs. As a result, many patients are unable to see physicians in a timely manner, are able to be diagnosed but cannot afford treatment, or are able to receive only a limited number of intravitreal injections that are insufficient for permanently resolving DME. Because the number of patients with DME in most nations will continue to grow, these inadequacies in health care delivery are not likely to be easily remedied.
In addition to center-involving DME, ranibizumab has also been approved for the treatment of diabetic retinopathy in eyes with non-center-involving DME. Treating these eyes may prevent or delay vision loss, but most physicians wait for the development of center-involving DME or PDR before initiating ranibizumab therapy.
Ranibizumab may be a reasonable alternative to PRP for the treatment of PDR. Ranibizumab-treated patients experience less DME and require fewer vitrectomy procedures, but compliance is critical for the prevention of severe vision loss. The efficacy of this strategy has not been determined beyond 2 years.
Conclusion
Ranibizumab is an effective and safe first-line therapy for the treatment of DME. It may provide advantages over PRP for the treatment of PDR, but patient selection is important.
References
Congdon N, O’Colmain B, Klaver CC, et al. Eye Diseases Prevalence Research Group. Causes and prevalence of visual impairment among adults in the United States. Arch Ophthalmol. 2004;122(4):477–85.
Roy MS, Janal MN. High caloric and sodium intakes as risk factors for progression of retinopathy in type 1 diabetes mellitus. Arch Ophthalmol. 2010;128:33–9.
Alwakeel JS, Sulimani R, Al-Asaad H, et al. Diabetes complications in 1952 type 2 diabetes mellitus patients managed in a single institution in Saudi Arabia. Ann Saudi Med. 2008;28:260–6.
Borchers AT, Uibo R, Gershwin ME. The geoepidemiology of type 1 diabetes. Autoimmun Rev. 2010;9:A355–65.
Stuckler D, Basu S, McKee M. Drivers of inequality in Millennium Development Goal progress: a statistical analysis. PLoS Med. 2010;7:e1000241.
Early Treatment Diabetic Retinopathy Study Research Group. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study Research Group report number 1. Arch Ophthalmol. 1985;103(12):1796–806.
The Diabetic Retinopathy Study Research Group. Photocoagulation treatment of proliferative diabetic retinopathy: the second report of diabetic retinopathy study findings. Trans Am Acad Ophthalmol Otolaryngol. 1978;85:82.
Early Treatment Diabetic Retinopathy Study Research Group. Early photocoagulation for diabetic retinopathy: ETDRS report number 9. Ophthalmology. 1991;98(Suppl):766–85.
Michaelson IC. The mode of development of the vascular system of the retina with some observations on its significance for certain retinal disorders. Trans Ophthalmol Soc UK. 1948;68:1625–710.
Folkman J. Tumor angiogenesis: therapeutic implications. N Engl J Med. 1971;285:1182–6.
Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey VS, Dvorak HF. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science. 1983;12:983–5.
Ferrara N, Henzel WJ. Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells. Biochem Biophys Res Commun. 1989;161:851–8.
Connolly DT, Heuvelman DM, Nelson R, et al. Tumor vascular permeability factor stimulates endothelial cell growth and angiogenesis. J Clin Invest. 1989;84:1470–8.
Aiello LP, Avery RL, Arrigg PG, et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med. 1994;331:1480–7.
Roberts WG, Palade GE. Increased microvascular permeability and endothelial fenestration induced by vascular endothelial growth factor. J Cell Sci. 1995;108:2369–79.
Qaum T, Xu Q, Joussen AM, et al. VEGF-initiated blood-retinal barrier breakdown in early diabetes. Invest Ophthalmol Vis Sci. 2001;42:2408–13.
Tolentino MJ, McLeod DS, Taomoto M, Otsuji T, Adamis AP, Lutty GA. Pathologic features of vascular endothelial growth factor-induced retinopathy in the nonhuman primate. Am J Ophthalmol. 2002;133:373–85.
Ferrara N, Damico L, Shams N, Lowman H, Kim R. Development of ranibizumab, an anti-vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina. 2006;26:859–70.
Lowe J, Araujo J, Yang J, et al. Ranibizumab inhibits multiple forms of active vascular endothelial growth factor in vitro and in vivo. Exp Eye Res. 2007;85(4):425–30.
Papadopoulos N, Martin J, Ruan Q, et al. Binding and neutralization of vascular endothelial growth factor (VEGF) and related ligands by VEGF Trap, ranibizumab and bevacizumab. Angiogenesis. 2012;15(2):171–85.
Gaudreault J, Fei D, Rusit J, Suboc P, Shiu V. Preclinical pharmacokinetics of ranibizumab (rhuFabV2) after a single intravitreal administration. Invest Ophthalmol Vis Sci. 2005;46:726–33.
Bakri SJ, Snyder MR, Reid JM, Pulido JS, Ezzat MK, Singh RJ. Pharmacokinetics of intravitreal ranibizumab (Lucentis). Ophthalmology. 2007;114:2179–82.
Christofordis JB, Carlton MM, Knopp MV, Hinkle GH. Pet/CT imaging of I-124-radiolabeled bevacizumab and ranibizumab after intravitreal injection in a rabbit model. Invest Ophthalmol Vis Sci. 2011;52:5899–903.
Mordenti J, Cuthbertson RA, Ferrara N, et al. Comparisons of the intraocular tissue distribution, pharmacokinetics, and safety of 125I-labeled full-length and Fab antibodies in rhesus monkeys following intravitreal administration. Toxicol Pathol. 1999;27:536–44.
Krohne TU, Liu Z, Holz FG, Meyer CH. Intraocular pharmacokinetics of ranibizumab following a single intravitreal injection in humans. Am J Ophthalmol. 2012;154(4):682–6.
Xu L, Lu T, Tuomi L, et al. Pharmacokinetics of ranibizumab in patients with neovascular age-related macular degeneration: a population approach. Invest Ophthalmol Vis Sci. 2013;54(3):1616–24.
Avery RL, Castellarin AA, Steinle NC, et al. Systemic pharmacokinetics following intravitreal injections of ranibizumab, bevacizumab or aflibercept in patients with neovascular AMD. Br J Ophthalmol. 2014;98(12):1636–41.
Brown DM, Kaiser PK, Michels M, et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med. 2006;334:1432–44.
Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for age-related macular degeneration. N Engl J Med. 2006;355:1419–31.
Nguyen QD, Tatlipinar S, Shah SM, et al. Vascular endothelial growth factor is critical stimulus for diabetic macular edema. Am J Ophthalmol. 2006;142:961–9.
Chun DW, Heier JS, Topping TM, Duker JS, Bankert JM. A pilot study of multiple intravitreal injections of ranibizumab in patients with center-involving clinically significant diabetic macular edema. Ophthalmology. 2006;113:1706–12.
Nguyen QD, Shah SM, Heier JS, et al. Primary end point (six months) results of the ranibizumab for edema of the macula in diabetes (READ-2) study. Ophthalmology. 2009;116:2175–81.
Nguyen QD, Shah SM, Khwaja AA, et al. Two-year outcomes of the ranibizumab for edema of the macula in diabetes (READ-2) study. Ophthalmology. 2010;117:2146–51.
Do DV, Nguyen QD, Khwaja AA, et al. Ranibizumab for edema of the macula in diabetes study: 3-year outcomes and the need for prolonged frequent treatment. JAMA Ophthalmol. 2013;131(2):139–45.
Busbee BG, Ho AC, Brown DM, et al. HARBOR Study Group. Twelve-month efficacy and safety of 0.5 mg or 2 mg ranibizumab in patients with subfoveal neovascular age-related macular degeneration. Ophthalmology. 2013;120(5):1046–56.
Do DV, Sepah YJ, Boyer D, et al. Month-6 primary outcomes of the READ-3 study (Ranibizumab for Edema of the mAcula in Diabetes-Protocol 3 with high dose). Eye (Lond). 2015;29(12):1538–44.
Nguyen QD, Brown DM, Marcus DM, et al. Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119:789–801.
Brown DM, Nguyen QD, Marcus DM, et al. Long-term outcomes of ranibizumab therapy for diabetic macular edema: the 36-month results from two phase III trials. Ophthalmology. 2013;120:2013–22.
Boyer DS, Nguyen QD, Brown DM, et al. Outcomes with as-needed ranibizumab after initial monthly therapy: long-term outcomes of the phase III RIDE and RISE trials. Ophthalmology. 2015;122:2504–13.
Massin P, Bandello F, Garweg JG, et al. Safety and efficacy of ranibizumab in diabetic macular edema (RESOLVE Study): a 12-month, randomized, controlled, double-masked, multicenter phase II study. Diabetes Care. 2010;33:2399–405.
Mitchell P, Bandello F, Schmidt-Erfurth U, et al. On behalf of the RESTORE study group. The RESTORE Study. Ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology. 2011;118(4):615–25.
Lang GE, Berta A, Eldem BM, et al. On behalf of the RESTORE Extension Study Group. Two-year safety and efficacy of ranibizumab 0.5 mg in diabetic macular edema. Interim analysis of the RESTORE extension study. Ophthalmology. 2013;120(10):2004–12.
Schmidt-Erfurth U, Lang GE, Holz FG, et al. RESTORE extension study group. Three-year outcomes of individualized ranibizumab treatment in patients with diabetic macular edema: the RESTORE extension study. Ophthalmology. 2014;121:1045–53.
Diabetic Retinopathy Clinical Research Network. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117:1064–77.
Diabetic Retinopathy Clinical Research Network Writing Committee, Elman MJ, Bressler NM, Qin H, et al. Expanded 2-year follow-up of ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2011;118:609–14.
Diabetic Retinopathy Clinical Research Network Writing Committee, Elman MJ, Qin H, Aiello LP, et al. Intravitreal ranibizumab for diabetic macular edema with prompt versus deferred laser treatment. Three-year randomized trial results. Ophthalmology. 2012;119(11):2312–8.
Elman MJ, Ayala A, Bressler NM, et al. For the Diabetic Retinopathy Clinical Research Network. Intravitreal ranibizumab for diabetic macular edema with prompt vs. deferred laser treatment: 5-year randomized trial results. Ophthalmology. 2015;122(2):375–81.
Prünte C, Fajnkuchen F, Mahmood S, et al. Ranibizumab 0.5 mg treat-and-extend regimen for diabetic oedema: the RETAIN study. Br J Ophthalmol. 2016;100(6):787–95.
Nepomuceno AB, Takaki E, Paes de Almeida FP, et al. A prospective randomized trial of intravitreal bevacizumab versus ranibizumab for the management of diabetic macular edema. Am J Ophthalmol. 2013;156:502–10.
Diabetic Retinopathy Clinical Research Network, Wells JA, Glassman AR, Ayala AR, et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. N Engl J Med. 2015;372(13):1193–203.
Wells JA, Glassman AR, Ayala AR, et al. Diabetic Retinopathy Clinical Research Network. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema: two-year results from a comparative effectiveness randomized clinical trial. Ophthalmology. 2016;123(6):1351–9.
Bressler SB, Ayala AR, Bressler NM, et al. Diabetic Retinopathy Clinical Research Network. Persistent macular thickening after ranibizumab treatment for diabetic macular edema with vision impairment. JAMA Ophthalmol. 2016;134(3):278–85.
Ferris FL 3rd, Maguire MG, Glassman AR, Ying GS, Martin DF. Evaluating effects of switching anti-vascular endothelial growth factor drugs for age-related macular degeneration and diabetic macular edema. JAMA Ophthalmol. 2016 [Epub ahead of print].
Filho JA, Messias A, Almeida FP, et al. Panretinal photocoagulation (PRP) versus PRP plus ranibizumab for high-risk proliferative diabetic retinopathy. Acta Ophthalmol. 2011;89:e567–72.
Writing Committee for the Diabetic Retinopathy Clinical Research Network, Gross JG, Glassman AR, Jampol LM, et al. Panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy. A randomized clinical trial. JAMA. 2015;314(20):2137–46.
Acknowledgements
No funding or sponsorship was received for this study or publication of this article. During the peer review process, Genentech was offered an opportunity to comment on the scientific accuracy of the article. Changes resulting from comments received were made by the author based on their scientific and editorial merit. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Disclosures
M. W. Stewart: Allergan: advisory board, institutional research support. Boehringer-Ingelheim: consultant. Momenta: consultant. Regeneron: advisory board, institutional research support.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not involve any new studies of human or animal subjects performed by any of the authors.
Data Availability
Data sharing is not applicable to this article, as no datasets were generated or analyzed during the current study.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article, go to http://www.medengine.com/Redeem/84F7F0603F049DFA.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Stewart, M.W. A Review of Ranibizumab for the Treatment of Diabetic Retinopathy. Ophthalmol Ther 6, 33–47 (2017). https://doi.org/10.1007/s40123-017-0083-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-017-0083-9