Abstract
Pain-insomnia-depression syndrome (PIDS) is a complex triad of chronic pain, insomnia, and depression that has profound effects on an individual's quality of life and mental health. The pathobiological context of PIDS involves complex neurobiological and physiological mechanisms, including alterations in neurotransmitter systems and impaired pain processing pathways. The first-line therapeutic approaches for the treatment of chronic pain, depression, and insomnia are a combination of pharmacological and non-pharmacological therapies. In cases where patients do not respond adequately to these treatments, additional interventions such as deep brain stimulation (DBS) may be required. Despite advances in understanding and treatment, there are still gaps in knowledge that need to be addressed. To improve our understanding, future research should focus on conducting longitudinal studies to uncover temporal associations, identify biomarkers and genetic markers associated with PIDS, examine the influence of psychosocial factors on treatment responses, and develop innovative interventions that address the complex nature of PIDS. The aim of this study is to provide a comprehensive overview of these components and to discuss their underlying pathobiological relationships.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Pain-insomnia-depression syndrome (PIDS) is a group of symptoms characterized by the coexistence of chronic pain, insomnia, and depression in individuals with chronic nonmalignant pain (CNMP). |
Pain, insomnia, and depression have an interactive relationship, with each condition contributing to the development or worsening of the other. |
The initial treatment strategies for chronic pain, depression, and insomnia include the use of noninvasive treatment methods. |
Invasive treatments such as deep brain stimulation (DBS) may only be considered for selected patients who have demonstrated resistance to noninvasive approaches. |
Introduction
Pain-insomnia-depression syndrome (PIDS) could be considered a group of symptoms observed in individuals with chronic nonmalignant pain (CNMP) [1]. In the term “PIDS,” “chronic pain” is defined as a feeling of discomfort that persists or recurs beyond the usual recovery period, usually exceeding a time threshold of 3 months [2]. Pain, in all its sensory, cognitive, motivational, and emotional aspects, refers to an unpleasant sensation caused by actual or potential damage to body tissues [3]. It includes not only the perception of the characteristics, location, causes, duration, and intensity of the stimulus, but also the responses and reactions evoked by the stimulus, including involuntary reflexes and autonomic processes [4, 5]. Pain prevalence standardized by age and gender varies significantly across countries, with an estimated overall weighted average of 27.5%. This prevalence ranges from as low as 9.9% to as high as 50.3% in different countries [6]. There is evidence that chronic pain has reached epidemic proportions in the United States and Europe [7]. Insomnia is different from sleep deprivation and is defined by a feeling of dissatisfaction with the amount or quality of sleep. It is characterized by significant stress or dysfunction, as well as daytime symptoms such as fatigue, excessive sleepiness, impaired cognitive performance, and mood disturbances [8]. Approximately 6% of individuals meet strict diagnostic criteria for insomnia, while up to 48% experience insomnia symptoms [9].
Depression is characterized by a pervasive feeling of sadness, anhedonia, difficulty concentrating or making decisions, feelings of worthlessness or excessive guilt, recurrent thoughts of death or self-harm, changes in sleep patterns or appetite, restlessness or slow movements, and a persistent feeling of tiredness or lack of energy [10, 11]. It is one of the most common manifestations of psychiatric pathologies and is closely associated with significant disability, impaired health-related quality of life, and increased mortality [12]. The prevalence of depression varies depending on the population group. Recent estimates suggest that approximately 10.4% of people are affected within 12 months and 20.6% during their lifetime [13].
Given the importance of interrelated symptoms for developing treatment plans, as Williams emphasizes, co-occurring symptoms should be considered when selecting appropriate treatment strategies [14]. With the well-documented coexistence of pain, insomnia, and depression [9, 15,16,17,18,19], and the complex relationships between pain, insomnia, and depression [20,21,22], there is a need to explore the triangular relationships, pathobiological correlations, current treatment modalities, and future prospects associated with these phenomena. The primary objective of this study is not to generate novel knowledge, but rather to present a contextualized compilation of previously relevant materials within the scope of the study topic. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Neurobiological Correlations
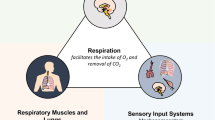
The complex relationships between chronic pain, insomnia, and depression are currently being investigated, and ongoing efforts are being made to elucidate their precise relationships. However, existing evidence suggests the presence of common brain structures, neural pathways, and neurobiological processes underlying these disorders [23] (Fig. 1). In the context of chronic pain [24], insomnia [25], and depression [24, 25], the hippocampus, which is an important component of the limbic system, has been observed to exhibit abnormalities. These abnormalities have been documented in academic research and suggest possible underlying mechanisms linking these disorders [24, 26].
In all three diseases, atrophy of the hippocampus [27] and increased activation of the limbic area are observed, with abnormal patterns in similar brain regions. Patients with depression and insomnia experience decreased activity in the prefrontal cortex (PFC), which leads to increased limbic activity through disinhibition. This increased activation may be a common mechanism contributing to the symptoms of all three disorders, with depression increasing emotional responses and chronic pain leading to increased emotional symptoms, insomnia, and sleep disturbances [28]. Furthermore, reduced hippocampal volume, also affected by chronic pain and depression, has been linked to reduced neurogenesis and neuroplasticity in both human patients and rodent models. This could potentially indicate a neuropathological mechanism underlying comorbid chronic pain and depression [29].
Furthermore, inflammation is recognized as an important regulator of hippocampal neurogenesis. Pain [30], insomnia [31], and depression are thought to be related to inflammatory responses in the hippocampus and may influence hippocampal neurogenesis [30]. Pain, insomnia, and depression are all associated with increased sensitization of the central nervous system, manifested by increased responsiveness to pain signals, sleep disturbances, and altered emotional processing [32]. In addition, chronic pain, insomnia, and depression are closely related to changes in neurotransmitter levels in the central nervous system, including serotonin (5-HT) [33], dopamine (DA), and norepinephrine (NE), which are critical for pain signaling, sleep regulation, and mood control [34,35,36,37,38]. Imbalances in these neurotransmitters can lead to the development of chronic pain, insomnia, and depression [34, 37, 38].
Chronic pain and depression are influenced by various molecular mechanisms, including monoamine neurotransmitters, inflammatory factors, and glutamate receptor subtypes. The dopaminergic system in the midbrain influences neuroplastic changes, with persistent pain negatively affecting DA activity. Decreased monoamine neurotransmitters in the central nervous system increase susceptibility to depression. Inflammatory factors cross the blood–brain barrier and alter neurotransmitter metabolism and neuroendocrine function. Glutamate, a primary excitatory neurotransmitter, is also linked to their development. Therapeutic medications can be developed to target specific areas common to both diseases, allowing for more precise treatment strategies. Epigenetic modifications alter chromatin structure and influence the accessibility of transcription factors to DNA promoter regions. MicroRNAs target histone deacetylase (HDAC) factors in both neuronal and non-neuronal cells and play a crucial role in gene regulation. The role of adult neurogenesis in chronic pain-related depression is not fully understood, but ongoing research suggests its possible importance [39].
Proinflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), nerve growth factor (NGF), interleukin-1 (IL-6), leukemia inhibitory factor (LIF), bradykinin, and prostanoids are neurobiological factors that are associated with chronic pain, insomnia, and depression. These cytokines act on nociceptive terminals in inflamed tissues, contribute to axonal damage, and modulate spontaneous nociceptor activity [28]. Nadeau et al. found that the preoperative level of mechanical pain hypersensitivity was almost indistinguishable in IL-1/TNF receptor knockout mice and wild-type littermates before partial sciatic nerve ligation [40]. However, both groups showed a significant reduction in the paw withdrawal threshold when exposed to a mechanical touch stimulus. The absence of interleukin-1 receptor (IL-1R1) and TNFR1 is necessary for pain relief, highlighting the critical role of proinflammatory cytokines in mediating neuropathic pain effects. Depression is associated with increased levels of proinflammatory cytokines that activate the hypothalamic–pituitary–adrenal axis (HPA or HTPA axis) and affect serotonergic, noradrenergic, and dopaminergic neurotransmission. The proinflammatory cytokines IL-1 and TNF-α are essential for the regulation of sleep patterns, particularly non-rapid eye movement sleep (NREMS), even in the absence of immune stress. A study by Moldofsky et al. showed a significant increase in IL-1 activity during sleep, accompanied by changes in immune function and the initiation of slow-wave sleep (SWS). IL-1β and TNF-α are substances that regulate sleep, and their introduction can mimic symptoms associated with poor sleep, such as depression, sleepiness, and fatigue. The exact mechanism by which these cytokines influence sleep regulation is not yet fully understood. However, they directly affect neurons in specific brain regions, such as the hypothalamic preoptic nucleus and basal forebrain [28].
Pain, insomnia, and depression may be related to brain-derived neurotrophic factor (BDNF), which increases the excitability of spinal neurons and mimics the effects of peripheral nerve injury. In vitro studies show increased BDNF levels in animal models of acute and chronic spinal cord pain. However, gene expression of BDNF and its receptor (neurokinin-1) was decreased in the hippocampus but increased in the spinal cord, suggesting an increase in BDNF in the spinal cord and a possible decrease in the hippocampus. Research shows that BDNF is crucial in the neurobiology of depression, with stressors such as forced swimming and chronic immobilization leading to declines in BDNF messenger RNA (mRNA) levels and reductions in BDNF stores in the hippocampus. However, this downregulation can be reversed by antidepressants that increase BDNF synthesis, neurogenesis, and signaling. BDNF also plays an important role in sleep regulation, with release increasing during both the NREMS and REMS phases in rabbits. Sleep deprivation can cause disruptions in BDNF levels, resulting in increases in BDNF protein and mRNA levels in the hippocampus but not the cerebellum or brainstem [28].
The orexin system is also involved in various physiological functions, including pain regulation [41], insomnia [42], and depression [43]. There are two types of orexin, namely orexin-A and orexin-B [44]. Both regulate their different influences through the binding and activation of two G protein-coupled receptors (GPCRs), orexin receptor type 1 (OX1R) and orexin receptor type 2 (OX2R) [45]. Orexin-producing cell bodies are located in particular in the lateral and perifornical regions of the hypothalamus. These cells send out fibers that extend throughout the central nervous system, forming a wide network. Orexin receptors are distributed throughout the brain and spinal cord. They are abundantly expressed in various regions, including the cerebral cortex, basal ganglia, ventral tegmental area (VTA), nucleus accumbens (NC), hippocampus, hypothalamic and thalamic nuclei, dorsal and median raphe, locus coeruleus (LC), des preoptic area, and the periaqueductal gray (PAG) and reticular formation. The different functions of the orexin receptors in many areas are due to the coexpression of OX1R and OX2R receptors, while in some areas only one of the two orexin receptors is predominantly expressed [41]. Numerous studies have provided evidence that injection of exogenous orexin at specific sites in the spinal cord and supraspinal sites associated with the descending pain regulatory circuits can result in significant reductions in nociceptive pain in various animal models of formalin-induced inflammatory pain responses [46]. Both OX1R and OX2R play a crucial role in the initiation and maintenance of sleep [47].
Preclinical models have shown reciprocal interactions between OX1R and OX2R [48]. It is worth noting that OX2R is particularly involved in the inhibition of NREM, while both receptors [48] contribute equally to the suppression of REM [49]. In addition, there is evidence that partial, prolonged, or total sleep deprivation is associated with increased orexin levels in the cerebrospinal fluid (CSF) [50]. Additionally, numerous animal studies have shown that both total sleep deprivation and selective REM deprivation result in increased orexin tone. This is supported by increased firing and projections of orexinergic neurons [51,52,53]. This evidence demonstrates the potential of exploiting antagonism at orexin receptors to regulate the orexin system and restore sleep in individuals suffering from insomnia [54,55,56]. Several experimental studies suggest a close connection between orexin and depression [57,58,59]. Consequently, continuous inhibition of orexin signaling could potentially lead to a state of lack of motivation or depression. It is worth noting that the function of the orexin system is impaired in both rats and individuals showing depression-like symptoms [60]. In the clinic, patients suffering from major depressive disorder and suicidal ideation show a decrease in orexin levels in their cerebrospinal fluid [57]. In contrast, it is worth noting that orexin has a direct stimulatory effect on GABAergic ventral pallidum (VP) neurons, thereby inhibiting depressive behavior in rats [61].
Tridirectional Associations
PIDS are comorbidities with complex relationships [9, 15,16,17,18,19]. These symptoms influence each other [62], increase the risk that other symptoms will appear or worsen [9], and produce synergistic effects on health outcomes according to the unpleasant symptom theory [62]. Although a comprehensive understanding of the precise mechanisms underlying these relationships has not yet been achieved, studies have proposed three different approaches to elucidate these relationships. While studies have suggested that pain intensity may be influenced by insomnia associated with depression [63] (Fig. 2A), other studies suggest that chronic pain disrupts sleep and may be influenced by mood disorders such as depression [64]. Undoubtedly, mood disorders have been suggested as a potential mediator in the interaction between sleep and pain [65] (Fig. 2B). In addition, there is evidence suggesting that chronic pain affects sleep quality [66], which in turn may contribute partially [67] or fully to the development or exacerbation of depression [13, 23, 66, 68, 69], either partially [67] or completely [70], whichever frequency is higher (Fig. 2C). Chronic pain leads to poorer sleep quality and ultimately more severe depression symptoms [23]. This mechanism has attracted increased attention. Studies have provided evidence of the connection between pain intensity, sleep quality, and depression. In particular, the association between pain disorders and depression appears to be influenced by the individual's sleep quality. In particular, improved sleep quality has been found to attenuate the negative effects of pain disorders on depressive symptoms, while poorer sleep quality increases these effects [71]. Likewise, sleep quality was shown to reduce the influence of pain intensity on negative mood (similar to depression) in individuals with rheumatoid arthritis and fibromyalgia [72]. Likewise, experiencing insomnia is associated with being twice as likely to suffer from depression and anxiety compared to people who report no symptoms of insomnia [73]. This finding is strongly supported by a community-based prospective study indicating that the occurrence of sleep disturbances in individuals with chronic musculoskeletal pain is associated with a threefold increased risk of developing depression [74]. These results confirm previous research highlighting pain as an indicator of depression, as well as several studies demonstrating an association between poor sleep quality and the occurrence of depressive symptoms [75, 76]. Experimental research has also uncovered several mechanisms through which sleep may influence depression, such as an increased negative reaction to stressful stimuli, an improvement in depressive symptoms, reduced frustration tolerance, and reduced emotional intelligence [77,78,79,80]. As a result, taking into account the cumulative results of theoretical reviews, epidemiological studies, cross-sectional analyses, and prospective studies, a comprehensive understanding is emerging that confirms the complex interplay between pain, insomnia, and depression [9, 15,16,17,18,19].
Potential relationship between chronic pain-insomnia-depression (CPID). A Intensity of chronic pain may be influenced by insomnia accompanied by depression; B chronic pain impacts sleep quality by influencing mood factors like depression; C chronic pain has a detrimental impact on sleep quality, which in turn may contribute to the development or exacerbation of depression
Contemporary Treatment Approaches
Treating pain, insomnia, and depression typically requires a comprehensive treatment approach. A multidisciplinary approach is typically used in the treatment of chronic pain [65], including both pharmacological [81] and noninvasive non-pharmacological therapies (NINPT). Noninvasive, non-pharmacological therapies include various modalities such as physical interventions, psychological interventions, complementary and alternative therapies [82], education, and occupational therapy. In addition, complementary and alternative medicine (CAM) techniques, including practices such as meditation, acupuncture, or music therapy, may be considered, although the level of evidence for their effectiveness is often limited [83]. Treatment of insomnia usually involves a combination of pharmacological and non-pharmacological interventions. The choice of treatment depends on factors such as the specific symptoms and severity of insomnia, the presence of coexisting disorders, the expected duration of treatment, the patient's preference for cognitive therapies, and the individual's susceptibility to medication side effects. Non-pharmacological approaches primarily focus on cognitive behavioral therapy for insomnia (CBTI), which is preferred due to its low side effects. However, there are limitations to the accessibility and availability of qualified CBTI facilitators. Pharmacologic treatment requires careful selection of popular drug categories such as benzodiazepines, non-benzodiazepines, antidepressants, and antihistamines for short-term use. These medications carry a higher risk of side effects, addiction, and abuse [84]. The currently accepted treatment approaches for depression include electroconvulsive therapy, cognitive behavioral therapy, interpersonal psychotherapy, and pharmacological interventions that modulate neurotransmission dependent on biogenic amines. The monoamine hypothesis, which proposes imbalances in the monoamine brain system as a factor contributing to depression, has been a major research focus in this area [85]. Despite all of these, pain management specialists should regularly assess patients with nonmalignant chronic pain for pain, insomnia, and depression [1].
Prospective Directions
Although chronic pain affects a significant number of people and is estimated to affect over 100 million people in the United States alone, study-level treatment failure rates are already significant. They are likely to be even more pronounced in the clinical setting. Evidence of treatment failure is usually derived from controlled trials, such as those evaluating gabapentin in neuropathic pain, where treatment can provide up to 50% relief compared to placebo [86]. The American Academy of Sleep Medicine and the European guidelines for the diagnosis and treatment of insomnia do not provide a clear definition of individuals who continue to experience insomnia despite appropriate therapy. In clinical trials, various terms such as treatment-resistant insomnia, refractory insomnia, residual insomnia, or persistent insomnia are used interchangeably to describe these patients [87]. Treatment-resistant depression (TRD) is characterized by a lack of response to two appropriate antidepressant treatment trials. TRD is often associated with prolonged and costly inpatient treatment. Despite various proposed definitions and criteria for identifying true TRD, no consensus has been reached. Consequently, the management of TRD presents challenges in terms of formulating therapeutic strategies and discovering effective treatments [88]. Given the significant burden of pain experienced by the population and the limited treatment options available, there is growing interest in exploring more radical approaches, including deep brain stimulation (DBS), or transcranial brain stimulation, as well as ketamine infusions, which are offered to patients with pain described as treatment-resistant [86].
Examining the associations between PIDS and individuals with chronic pain is a key area of research. Future research directions in PIDS will focus on improving the understanding of this complicated syndrome and providing a basis for groundbreaking interventions that reduce the burden of chronic pain, insomnia, and depression on those affected. Currently, brain stimulation (BS) methods such as DBS have attracted widespread attention and cover a wide range of applications, ranging from basic scientific investigations to the practical implementation of therapeutic interventions. DBS is a neurosurgical technique that involves inserting electrodes into specific regions of the brain and delivering continuous or intermittent electrical pulses from an implanted battery source [89].
From a clinical perspective, it offers several advantages over alternative surgical methods of neuromodulation. These advantages include the nonlesional nature of DBS, the ability to adjust stimulation parameters to optimize efficacy and minimize side effects, and the ability to directly interact with the circuit pathology responsible for apparent symptoms. From a scientific perspective, DBS serves as a valuable tool for studying the underlying physiological mechanisms of brain dysfunction. This allows for the identification and correction of pathological neuronal patterns, promotes technological advancement, and improves safety and clinical outcomes [90]. In addition, DBS has proven to be a highly targeted intervention, focusing on specific anatomical areas, often in the millimeter range. In this way, it has provided valuable insights into circuit theories of brain dysfunction. This research has shown that localized dysfunction and subsequent interventions can have significant effects on the broader networks in the brain [91]. The dual nature of DBS, acting as both a probe and modulator of brain circuits, has led to further investigation into its therapeutic potential for a variety of disorders. These include disorders that affect motor skills, emotional regulation, memory, and cognitive function [89]. DBS has been extensively studied as a potential therapeutic approach for the treatment of chronic pain [92]. While the specific mechanism of DBS for pain management is still unknown, several plausible theories have been proposed based on recent studies. DBS is believed to be able to modulate electrical activity and neurochemical transmission in the brain. Abnormal pain signals can be interrupted by electrical pulses via the electrodes, restoring normal brain activity. In addition, DBS can alter the abnormal oscillatory activity in the brain that is associated with chronic pain. DBS can reduce pain perception by altering neuronal oscillation patterns. Additionally, studies have shown that DBS can cause synaptic plasticity, the ability of synapses to strengthen or weaken over time [93,94,95].
DBS has been shown to be effective in the treatment of various pain syndromes, including post-stroke pain, phantom limb pain, brachial plexus injuries, failed back surgery syndrome, and pain associated with Parkinson’s disease (PD). The results suggest that applying DBS to different brain nuclei associated with chronic pain produces different results. In particular, the ventral posterolateral nucleus (VPL), anterior cingulate cortex (ACC), periventricular gray (PVG), and PAG were found to be the most effective targets and showed significant or moderate treatment effects. These identified targets are involved in both the sensory-discriminatory and affective aspects of pain perception. DBS can modulate the activity of neural circuits involved in pain processing and perception. The exact mechanism underlying the pain-relieving effects of DBS is not fully understood, but several factors are thought to be involved. These factors include changing the balance between inhibitory and excitatory neurotransmitters within pain pathways, reducing the activity of neurons that transmit nociceptive signals, improving the function of endogenous opioid systems responsible for pain relief, and changing emotional and cognitive aspects of pain such as anxiety and depression and catastrophic. The effectiveness of DBS in relieving chronic pain may vary depending on the brain target stimulated. Among the most common targets is the sensory thalamus (particularly the ventral-posterior-lateral and ventral-posterior-medial nuclei), which primarily contributes to the sensory-discriminatory aspects of pain, such as location, intensity, and quality. The PAG and periventricular gray matter are also targeted because of their role in descending pain modulation and the release of endogenous opioids. In addition, the ACC, which is involved in the emotional and motivational aspects of pain such as discomfort, suffering, and coping, is another commonly targeted area [96].
DBS therapy is effective in treating sleep–wake disorders in PD, including sleep fragmentation, rapid eye movement (REM) behavior disorder, excessive daytime sleepiness, periodic limb movements, and restless legs syndrome [97]. The exact mechanisms behind this improvement are not fully understood, but several possible causes include alteration of neural circuits involved in sleep regulation, control of abnormal oscillatory activity in the brain, and impairment of the production and activity of neurotransmitters such as dopamine, serotonin, and GABA. DBS can also help control sleep–wake cycles and promote better sleep by altering the balance of these neurotransmitters. Additionally, DBS can reduce hyperarousal and increase activity in certain brain regions, such as the PFC and limbic system, which are often associated with insomnia, resulting in less hyperarousal and better sleep initiation and maintenance. Overall, DBS therapy has the potential to significantly improve sleep quality and sleep management in PD [98].
DBS is effective in treating severe, treatment-resistant depression [99]. The exact mechanism of action is still unclear, but it has been suggested that it may modulate dysfunctional neural circuits associated with depression. This can be achieved by electrically stimulating specific brain regions, altering the activity and connectivity of these circuits, and improving depressive symptoms. DBS also affects the release and activity of neurotransmitters involved in mood regulation, such as 5-HT, DA, and norepinephrine. This stimulation can increase the release of these neurotransmitters or modulate their receptors, resulting in an antidepressant effect. DBS has also been found to induce neuroplastic changes in the brain, promoting the growth of new neurons and strengthening existing connections. It targets specific brain regions involved in mood regulation, such as the subcallosal cingulate region, NC, ventral capsule/striatum, medial forebrain bundle, inferior thalamic peduncle, and lateral habenula, to restore neuronal activity and relieve depressive symptoms. Additionally, DBS may have anti-inflammatory effects on the brain, which could contribute to its antidepressant effects [100,101,102].
Conclusion
PIDS exhibits intricate triangular relationships, with pain, insomnia, and depression influencing and exacerbating each other. The pathobiological correlations underlying PIDS involve complex mechanisms including neurophysiology, endocrine regulation, and psychological factors. Current treatment modalities for PIDS encompass a multidisciplinary approach, including both pharmacological and NINPT, which is considered first-line treatment. Invasive treatments such as DBS may only be considered for selected patients in cases where unresponsive patients have shown resistance to noninvasive methods. Overall, advancing our knowledge of the triangular relationships, pathobiological correlations, and current treatment modalities for PIDS is crucial for providing effective and comprehensive care to individuals affected by this syndrome. Continued research and collaboration among various disciplines are key to paving the way for future advancements in the field and ultimately improving the lives of those suffering from PIDS.
Data Availability
Upon request, the data will be made accessible to interested parties.
References
Collen M. The case for Pain Insomnia Depression Syndrome (PIDS): a symptom cluster in chronic nonmalignant pain. J Pain Palliat Care Pharmacother. 2008;22(3):221–5.
Pain IAftSo. Definitions of chronic pain syndromes: IASP. https://www.iasp-pain.org/advocacy/definitions-of-chronic-pain-syndromes/.
Chen J, Kandle PF, Murray I, Fitzgerald LA, Sehdev JS. Physiology, pain. 2019.
Lund JP. Orofacial pain: from basic science to clinical management: the transfer of knowledge in pain research to education. Quintessence Publishing; 2001.
Greenberg MS. Ulcerative, vesicular, and bullous lesions. Burket's Oral Medicine Diagnosis & Treatment. 2003.
Zimmer Z, Fraser K, Grol-Prokopczyk H, Zajacova A. A global study of pain prevalence across 52 countries: examining the role of country-level contextual factors. Pain. 2022;163(9):1740–50.
Courtney CA, Fernández-de-Las-Peñas C, Bond S. Mechanisms of chronic pain - key considerations for appropriate physical therapy management. J Man Manip Ther. 2017;25(3):118–27.
Levenson JC, Kay DB, Buysse DJ. The pathophysiology of insomnia. Chest. 2015;147(4):1179–92.
Finan PH, Smith MT. The comorbidity of insomnia, chronic pain, and depression: dopamine as a putative mechanism. Sleep Med Rev. 2013;17(3):173–83.
Gaynes BN, Lux L, Gartlehner G, Asher G, Forman-Hoffman V, Green J, et al. Defining treatment-resistant depression. Depress Anxiety. 2020;37(2):134–45.
Busch CA, Mohammed TF, Nadile EM, Witt ML, Vargas C, Tran M, et al. Costs and benefits of undergraduates revealing depression to online science instructors. CBE—Life Sci Educ. 2023;22(1):ar9.
Burke NN, Finn DP, Roche M. Neuroinflammatory Mechanisms Linking Pain and Depression. Mod Trends Pharmacopsychiatry. 2015;30:36–50.
Lorenzo EC, Kuchel GA, Kuo C-L, Moffitt TE, Diniz BS. Major depression and the biological hallmarks of aging. Ageing Res Rev. 2023;83: 101805.
Williams LA, editor. Clinical management of symptom clusters. Seminars in oncology nursing. Elsevier; 2007.
Wilson KG, Eriksson MY, Joyce L, Mikail SF, Emery PC. Major depression and insomnia in chronic pain. Clin J Pain. 2002;18(2):77–83.
O’Brien EM, Waxenberg LB, Atchison JW, Gremillion HA, Staud RM, McCrae CS, et al. Negative mood mediates the effect of poor sleep on pain among chronic pain patients. Clin J Pain. 2010;26(4):310–9.
Miró E, Martínez MP, Sánchez AI, Prados G, Medina A. When is pain related to emotional distress and daily functioning in fibromyalgia syndrome? The mediating roles of self-efficacy and sleep quality. Br J Health Psychol. 2011;16(4):799–814.
Chung K-F, Tso K-C. Relationship between insomnia and pain in major depressive disorder: a sleep diary and actigraphy study. Sleep Med. 2010;11(8):752–8.
Vgontzas A, Cui L, Merikangas KR. Are sleep difficulties associated with migraine attributable to anxiety and depression? Headache J Head Face Pain. 2008;48(10):1451–9.
Dunietz GL, Swanson LM, Jansen EC, Chervin RD, O'Brien LM, Lisabeth LD, et al. Key insomnia symptoms and incident pain in older adults: direct and mediated pathways through depression and anxiety. Sleep. 2018;41(9).
Liu M, McCurry SM, Belza B, Buchanan DT, Dobra A, Von Korff M, et al. Effects of pain, insomnia, and depression on psychoactive medication supply in older adults with osteoarthritis. Med Care. 2018;56(12):1024–31.
Liu M, McCurry SM, Belza B, Dobra A, Buchanan DT, Vitiello MV, et al. Effects of osteoarthritis pain and concurrent insomnia and depression on health care use in a primary care population of older adults. Arthritis Care Res (Hoboken). 2019;71(6):748–57.
Alhalal EA, Alhalal IA, Alaida AM, Alhweity SM, Alshojaa AY, Alfaori AT. Effects of chronic pain on sleep quality and depression: a cross-sectional study. Saudi Med J. 2021;42(3):315–23.
Anand KS, Dhikav V. Hippocampus in health and disease: an overview. Ann Indian Acad Neurol. 2012;15(4):239.
Xu M, Wang Q, Li B, Qian S, Wang S, Wang Y, et al. Cerebellum and hippocampus abnormalities in patients with insomnia comorbid depression: a study on cerebral blood perfusion and functional connectivity. Front Neurosci. 2023;17:1202514.
Yang S, Chang MC. Chronic pain: structural and functional changes in brain structures and associated negative affective states. Int J Mol Sci. 2019;20(13):3130.
Vaculik MF, Noorani A, Hung PS-P, Hodaie M. Selective hippocampal subfield volume reductions in classic trigeminal neuralgia. NeuroImage Clin. 2019;23:101911.
Boakye PA, Olechowski C, Rashiq S, Verrier MJ, Kerr B, Witmans M, et al. A critical review of neurobiological factors involved in the interactions between chronic pain, depression, and sleep disruption. Clin J Pain. 2016;32(4):327–36.
Sheng J, Liu S, Wang Y, Cui R, Zhang X. The link between depression and chronic pain: neural mechanisms in the brain. Neural Plastic. 2017.
Mokhtari T, Tu Y, Hu L. Involvement of the hippocampus in chronic pain and depression. Brain Sci Adv. 2019;5(4):288–98.
Zielinski MR, Gibbons AJ. Neuroinflammation, sleep, and circadian rhythms. Front Cell Infect Microbiol. 2022;12: 853096.
Campos ACP, Antunes GF, Matsumoto M, Pagano RL, Martinez RCR. Neuroinflammation, pain and depression: an overview of the main findings. Front Psychol. 2020;11:1825.
Karimi R, Mallah N, Scherer R, Rodríguez-Cano R, Takkouche B. Sleep quality as a mediator of the relation between depression and chronic pain: a systematic review and meta-analysis. Br J Anaesth. 2023;130(6):747–62.
Hao S, Shi W, Liu W, Chen Q-Y, Zhuo M. Multiple modulatory roles of serotonin in chronic pain and injury-related anxiety. Front Synaptic Neurosci. 2023;15:1122381.
ShV V. Insomnia, serotonin and depression. Georgian Med News. 2007;150:22–4.
Kesner AJ, Lovinger DM. Wake up and smell the dopamine: new mechanisms mediating dopamine activity fluctuations related to sleep and psychostimulant sensitivity. Neuropsychopharmacology. 2021;46(4):683–4.
Mitchell HA, Weinshenker D. Good night and good luck: norepinephrine in sleep pharmacology. Biochem Pharmacol. 2010;79(6):801–9.
Moret C, Briley M. The importance of norepinephrine in depression. Neuropsychiatr Dis Treat. 2011;7(sup1):9–13.
Meda RT, Nuguru SP, Rachakonda S, Sripathi S, Khan MI, Patel N. Chronic pain-induced depression: a review of prevalence and management. Cureus. 2022;14(8): e28416.
Nadeau S, Filali M, Zhang J, Kerr BJ, Rivest S, Soulet D, et al. Functional recovery after peripheral nerve injury is dependent on the pro-inflammatory cytokines IL-1β and TNF: implications for neuropathic pain. J Neurosci. 2011;31(35):12533–42.
Razavi BM, Hosseinzadeh H. A review of the role of orexin system in pain modulation. Biomed Pharmacother. 2017;90:187–93.
Fragale JE, James MH, Avila JA, Spaeth Andrea M, Aurora RN, Langleben D, et al. The insomnia-addiction positive feedback loop: role of the orexin system. In: Steiner MA, Yanagisawa M, Clozel M, editors., et al., The orexin system basic science and role in sleep pathology. 45. S. Karger AG; 2021.
Fagan H, Jones E, Baldwin DS. Orexin receptor antagonists in the treatment of depression: a leading article summarising pre-clinical and clinical studies. CNS Drugs. 2023;37(1):1–12.
Sun Y, Tisdale Ryan K, Kilduff Thomas S. Hypocretin/orexin receptor pharmacology and sleep phases. In: Steiner MA, Yanagisawa M, Clozel M, editors. The orexin system basic science and role in sleep pathology. 45. S. Karger AG; 2021.
Wang C, Wang Q, Ji B, Pan Y, Xu C, Cheng B, et al. The orexin/receptor system: molecular mechanism and therapeutic potential for neurological diseases. Front Mol Neurosci. 2018;11:220.
Kang X, Tang H, Liu Y, Yuan Y, Wang M. Research progress on the mechanism of orexin in pain regulation in different brain regions. Open Life Sci. 2021;16(1):46–52.
Lecea Ld, Huerta R. Hypocretin (orexin) regulation of sleep-to-wake transitions. Front Pharmacol. 2014;5:16.
Dugovic C, Shelton JE, Aluisio LE, Fraser IC, Jiang X, Sutton SW, et al. Blockade of orexin-1 receptors attenuates orexin-2 receptor antagonism-induced sleep promotion in the rat. J Pharmacol Exp Ther. 2009;330(1):142–51.
Mieda M, Hasegawa E, Kisanuki YY, Sinton CM, Yanagisawa M, Sakurai T. Differential roles of orexin receptor-1 and-2 in the regulation of non-REM and REM sleep. J Neurosci. 2011;31(17):6518–26.
Allen R, Mignot E, Ripley B, Nishino S, Earley C. Increased CSF hypocretin-1 (orexin-A) in restless legs syndrome. Neurology. 2002;59(4):639–41.
Briggs C, Bowes SC, Semba K, Hirasawa M. Sleep deprivation-induced pre-and postsynaptic modulation of orexin neurons. Neuropharmacology. 2019;154:50–60.
Alexandre C, Andermann ML, Scammell TE. Control of arousal by the orexin neurons. Curr Opin Neurobiol. 2013;23(5):752–9.
Mehta R, Khanday MA, Mallick BN. REM sleep loss associated changes in orexin-A levels in discrete brain areas in rats. Neurosci Lett. 2015;590:62–7.
Xue T, Wu X, Chen S, Yang Y, Yan Z, Song Z, et al. The efficacy and safety of dual orexin receptor antagonists in primary insomnia: a systematic review and network meta-analysis. Sleep Med Rev. 2022;61: 101573.
Janto K, Prichard JR, Pusalavidyasagar S. An update on dual orexin receptor antagonists and their potential role in insomnia therapeutics. J Clin Sleep Med. 2018;14(8):1399–408.
Winrow C, Renger J. Discovery and development of orexin receptor antagonists as therapeutics for insomnia. Br J Pharmacol. 2014;171(2):283–93.
Brundin L, Björkqvist M, Petersén Å, Träskman-Bendz L. Reduced orexin levels in the cerebrospinal fluid of suicidal patients with major depressive disorder. Eur Neuropsychopharmacol. 2007;17(9):573–9.
Ito N, Yabe T, Gamo Y, Nagai T, Oikawa T, Yamada H, et al. Icv administration of orexin-A induces an antidepressive-like effect through hippocampal cell proliferation. Neuroscience. 2008;157(4):720–32.
Salomon RM, Ripley B, Kennedy JS, Johnson B, Schmidt D, Zeitzer JM, et al. Diurnal variation of cerebrospinal fluid hypocretin-1 (Orexin-A) levels in control and depressed subjects. Biol Psychiat. 2003;54(2):96–104.
James MH, Campbell EJ, Walker FR, Smith DW, Richardson HN, Hodgson DM, et al. Exercise reverses the effects of early life stress on orexin cell reactivity in male but not female rats. Front Behav Neurosci. 2014;8:244.
Ji M-J, Zhang X-Y, Chen Z, Wang J-J, Zhu J-N. Orexin prevents depressive-like behavior by promoting stress resilience. Mol Psychiatry. 2019;24(2):282–93.
Liu M, Hou T, Nkimbeng M, Li Y, Taylor JL, Sun X, et al. Associations between symptoms of pain, insomnia and depression, and frailty in older adults: a cross-sectional analysis of a cohort study. Int J Nurs Stud. 2021;117: 103873.
Harrison L, Wilson S, Heron J, Stannard C, Munafò MR. Exploring the associations shared by mood, pain-related attention and pain outcomes related to sleep disturbance in a chronic pain sample. Psychol Health. 2016;31(5):565–77.
Valrie CR, Gil KM, Redding-Lallinger R, Daeschner C. Brief report: daily mood as a mediator or moderator of the pain–sleep relationship in children with sickle cell disease. J Pediatr Psychol. 2008;33(3):317–22.
Herrero Babiloni A, Beetz G, Bruneau A, Martel MO, Cistulli PA, Nixdorf DR, et al. Multitargeting the sleep-pain interaction with pharmacological approaches: a narrative review with suggestions on new avenues of investigation. Sleep Med Rev. 2021;59: 101459.
Turk DC, Fillingim RB, Ohrbach R, Patel KV. Assessment of psychosocial and functional impact of chronic pain. J Pain. 2016;17(9 Suppl):T21-49.
Juan W, Rui L, Wei-Wen Z. Chronic neck pain and depression: the mediating role of sleep quality and exercise. Psychol Health Med. 2020;25(8):1029–35.
Stein DJ, Kupfer DJ, Schatzberg AF. The American Psychiatric Publishing textbook of mood disorders. Berlin: American Psychiatric Publishing, Inc.; 2006.
Gil KM, Carson JW, Porter LS, Ready J, Valrie C, Redding-Lallinger R, et al. Daily stress and mood and their association with pain, health-care use, and school activity in adolescents with sickle cell disease. J Pediatr Psychol. 2003;28(5):363–73.
Amtmann D, Askew RL, Kim J, Chung H, Ehde DM, Bombardier CH, et al. Pain affects depression through anxiety, fatigue, and sleep in multiple sclerosis. Rehabil Psychol. 2015;60(1):81.
Zambelli Z, Halstead EJ, Fidalgo AR, Dimitriou D. Good sleep quality improves the relationship between pain and depression among individuals with chronic pain. Front Psychol. 2021;12:1555.
Hamilton NA, Catley D, Karlson C. Sleep and the affective response to stress and pain. Health Psychol. 2007;26(3):288.
Marcuzzi A, Skarpsno ES, Nilsen TIL, Mork PJ. The interplay between multisite pain and insomnia on the risk of anxiety and depression: the HUNT study. BMC Psychiatry. 2022;22(1):124.
Campbell P, Tang N, McBeth J, Lewis M, Main CJ, Croft PR, et al. The role of sleep problems in the development of depression in those with persistent pain: a prospective cohort study. Sleep. 2013;36(11):1693–8.
Buysse DJ, Angst J, Gamma A, Ajdacic V, Eich D, Rössler W. Prevalence, course, and comorbidity of insomnia and depression in young adults. Sleep. 2008;31(4):473–80.
Jaussent I, Bouyer J, Ancelin M-L, Akbaraly T, Peres K, Ritchie K, et al. Insomnia and daytime sleepiness are risk factors for depressive symptoms in the elderly. Sleep. 2011;34(8):1103–10.
Daniela T, Alessandro C, Giuseppe C, Fabio M, Cristina M, Michele F. Lack of sleep affects the evaluation of emotional stimuli. Brain Res Bull. 2010;82(1–2):104–8.
Killgore WD. Self-reported sleep correlates with prefrontal-amygdala functional connectivity and emotional functioning. Sleep. 2013;36(11):1597–608.
Finan PH, Quartana PJ, Smith MT. The effects of sleep continuity disruption on positive mood and sleep architecture in healthy adults. Sleep. 2015;38(11):1735–42.
Killgore WD, Balkin TJ, Yarnell AM, Capaldi VF II. Sleep deprivation impairs recognition of specific emotions. Neurobiol Sleep Circadian Rhythms. 2017;3:10–6.
Park HJ, Moon DE. Pharmacologic management of chronic pain. The Korean journal of pain. 2010;23(2):99–108.
Shi Y, Wu W. Multimodal non-invasive non-pharmacological therapies for chronic pain: mechanisms and progress. BMC Med. 2023;21(1):372.
Cheung JMY, Bartlett DJ, Armour CL, Laba T-L, Saini B. To drug or not to drug: a qualitative study of patients’ decision-making processes for managing insomnia. Behav Sleep Med. 2018;16(1):1–26.
Paul S, Vidusha K, Thilagar S, Lakshmanan DK, Ravichandran G, Arunachalam A. Advancement in the contemporary clinical diagnosis and treatment strategies of insomnia disorder. Sleep Med. 2022;91:124–40.
Wong M-L, Licinio J. Research and treatment approaches to depression. Nat Rev Neurosci. 2001;2(5):343–51.
Borsook D, Youssef AM, Simons L, Elman I, Eccleston C. When pain gets stuck: the evolution of pain chronification and treatment resistance. Pain. 2018;159(12):2421–36.
Akinnusi ME, El-Solh AA. Treatment-resistant insomnia: a common undefined condition. Am J Med. 2021;134(12):1447–8.
Voineskos D, Daskalakis ZJ, Blumberger DM. Management of treatment-resistant depression: challenges and strategies. Neuropsychiatr Dis Treat. 2020;16:221–34.
Lozano AM, Lipsman N. Probing and regulating dysfunctional circuits using deep brain stimulation. Neuron. 2013;77(3):406–24.
Kühn AA, Kempf F, Brücke C, Gaynor Doyle L, Martinez-Torres I, Pogosyan A, et al. High-frequency stimulation of the subthalamic nucleus suppresses oscillatory beta activity in patients with Parkinson’s disease in parallel with improvement in motor performance. J Neurosci. 2008;28(24):6165–73.
Lipsman N, Woodside DB, Giacobbe P, Hamani C, Carter JC, Norwood SJ, et al. Subcallosal cingulate deep brain stimulation for treatment-refractory anorexia nervosa: a phase 1 pilot trial. Lancet. 2013;381(9875):1361–70.
Frizon LA, Yamamoto EA, Nagel SJ, Simonson MT, Hogue O, Machado AG. Deep brain stimulation for pain in the modern era: a systematic review. Neurosurgery. 2020;86(2):191–202.
Herrington TM, Cheng JJ, Eskandar EN. Mechanisms of deep brain stimulation. J Neurophysiol. 2016;115(1):19–38.
Staudt MD, Yaghi NK, Mazur-Hart DJ, Shirvalkar P. Advancements in deep brain stimulation for chronic pain control. Front Pain Res. 2023;4.
Pereira EAC, Green AL, Aziz TZ. Chapter 23 - Deep brain stimulation for pain. In: Lozano AM, Hallett M, editors. Handbook of clinical neurology. 116. Berlin; 2013. p. 277–94.
Shaheen N, Shaheen A, Elgendy A, Bezchlibnyk YB, Zesiewicz T, Dalm B, et al. Deep brain stimulation for chronic pain: a systematic review and meta-analysis. Front Hum Neurosci. 2023;17:1297894.
Sharma VD, Sengupta S, Chitnis S, Amara AW. Deep brain stimulation and sleep-wake disturbances in Parkinson disease: a review. Front Neurol. 2018;9:697.
Bjerknes S, Skogseid IM, Hauge TJ, Dietrichs E, Toft M. Subthalamic deep brain stimulation improves sleep and excessive sweating in Parkinson’s disease. npj Parkinson’s Dis. 2020;6(1):29.
Drobisz D, Damborská A. Deep brain stimulation targets for treating depression. Behav Brain Res. 2019;359:266–73.
Torres-Sanchez S, Perez-Caballero L, Berrocoso E. Cellular and molecular mechanisms triggered by Deep Brain Stimulation in depression: a preclinical and clinical approach. Prog Neuropsychopharmacol Biol Psychiatry. 2017;73:1–10.
Schlaepfer TE, Bewernick BH, Kayser S, Hurlemann R, Coenen VA. Deep brain stimulation of the human reward system for major depression—rationale, outcomes and outlook. Neuropsychopharmacology. 2014;39(6):1303–14.
Wu Y, Mo J, Sui L, Zhang J, Hu W, Zhang C, et al. Deep brain stimulation in treatment-resistant depression: a systematic review and meta-analysis on efficacy and safety. Front Neurosci. 2021;15: 655412.
Acknowledgements
The authors would like to thank the Medical Research and Technology Center of Khatam Al-Nabieen University for their collaboration and provision of the necessary facilities to conduct this study.
Funding
No funding or sponsorship was received for this study or publication of this article.
Author information
Authors and Affiliations
Contributions
Murtaza Haidary: Supervision, Conceptualization, Methodology, Writing—Original Draft Preparation. Shamim Arif and Shekiba Madadi: Data Analysis, Writing—Review & Editing. Dawood Hossaini and Elham Akbari: Data Interpretation, Writing—Review & Editing. Hossain Rezayee: Writing—Review & Editing. All authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors disclose no conflicts of interest.
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Haidary, M., Arif, S., Hossaini, D. et al. Pain-Insomnia-Depression Syndrome: Triangular Relationships, Pathobiological Correlations, Current Treatment Modalities, and Future Direction. Pain Ther (2024). https://doi.org/10.1007/s40122-024-00614-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40122-024-00614-5