Abstract
Introduction
Chronic neck pain (CNP) is a global public health problem, with high prevalence and absenteeism rates. Central sensitization (CS) as a basis for chronic pain may play an essential role in its development and progression. It is often comorbid with low conditioned pain modulation (CPM) effects, cognitions, and psychological problems.
Objectives
The purposes of this study were to (1) explore the relationship between pain-related cognitions and psychological factors, CPM effects, and the central sensitization inventory (CSI) scores; and (2) determine whether cognitions and psychological factors can predict CSI scores and CPM effects in individuals with CNP.
Methods
Fifty-four individuals with CNP were recruited for this cross-sectional study. The following outcome measures were evaluated: The CSI (screening tool) was compared with the cold pressor test (CPT), which was the psychophysical test used to assess the CPM; neck pain intensity using the visual analogue scale (VAS), as well as pain-related cognitions (including kinesiophobia and pain catastrophization) and psychological states (including anxiety and depression) using self-report questionnaires.
Results
CSI score was not associated with the CPM effect (r = 0.257, p > 0.05), and no cognitions or psychological factors were associated with CPM (p > 0.05), but CSI score was moderately positively correlated with kinesiophobia (r = 0.554, p < 0.01), lowly positively correlated with pain catastrophization (r = 0.332, p = 0.017) and anxiety (r = 0.492, p < 0.01), but not depression (r = 0.207, p = 0.132). Multiple linear regression analysis showed that kinesiophobia (B = 1.308, p < 0.01) and anxiety (B = 1.806, p = 0.02) were significant positive predictors of CSI score.
Conclusions
The findings confirm some of our hypotheses. Accordingly, the findings inferred that the CSI does not seem to respond to CPM effect in patients with CNP effectively. In addition, CSI score was associated with cognitions and psychological factors, of which kinesiophobia and anxiety were effective predictors. In clinical practice, pain-related cognitions and psychological factors should be fully considered to manage neck pain efficiently.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Neck pain imposes a significant medical and economic burden on individuals and society, and central sensitization plays an important role in the development of neck pain chronicity and is often comorbid with poorer cognitive and psychological factors. |
We aimed to explore, in individuals with chronic neck pain, the correlation between the central sensitization inventory (CSI; screening tool), the conditioned pain modulation (CPM; psychophysical test), and pain-related cognitions and psychological factors. |
The results of the study disproved our hypothesis that CPM is not associated with any cognitions or psychological factors in individuals with chronic neck pain (CNP). In the future, we need to find other tools or methods to assess the pain-suppression system. |
Pain-related cognitions and psychological factors were associated with CSI, including kinesiophobia, anxiety and pain catastrophizing, but not depression. In addition, the first two were also valid predictors of CSI score. |
Given the multifactorial nature of CNP, barriers that may affect pain and rehabilitation outcomes should be well understood, along with consideration of potential treatment targets other than pain to aid in neck pain management. |
Introduction
Neck pain, one of the five most common chronic pain disorders [15], imposes a significant economic burden on individuals and society. According to a survey [59], the global age-standardized rate for point prevalence of neck pain per 100,000 population was 3551.1, and for incidence of neck pain per 100,000 population was 806.6, and about 50–85% of them may develop chronic neck pain (CNP) [15]. The International Association for the Study of Pain (IASP) defines central sensitization (CS) as “increased responsiveness of nociceptive neurons in the central nervous system to their normal or sub-threshold afferent inputs” [20], which is characterized by widespread nociceptive sensitization, temporal accumulation of pain, and inefficiency of endogenous pain inhibitory systems [72], as well as impacting on psychological well-being with increased risk of anxiety and/or depression [9, 23]. It has been shown that CS can be used to explain the long-term recurrent nature of CNP and the failure of treatment effects to be sustained [74].
The Central Sensitization Inventory (CSI) is a self-report screening tool [49]. It has been widely used in clinical practice because of its low cost and effectiveness. It has also demonstrated the validity and reliability of the CSI in identifying symptoms associated with CS in healthy populations, and patients with chronic pain [11, 49]. However, it is debatable whether the CSI truly reflects the CS [35]. Evidence suggests that this is associated with increased vulnerability to other central nervous system disorders, such as anxiety, depression, and stress [2], and that the relationship is bidirectional [3]. Considering that CSI focuses on hypersensitivity in a broader sense [1], including pain-related cognitions and psychological factors, it becomes challenging to quantify CS and its associated symptoms.
Quantitative sensory testing (QST) is considered to be the best measure of CS neurophysiology [5], and conditioned pain modulation (CPM), one of the components assessed by QST, is used to measure the net effect of endogenous pain regulation on both chemotaxis and inhibition. The clinical significance of CPM as a surrogate indicator of the brain's ability to activate endogenous analgesia may critically impact chronic pain via the descending pathway [44]. Impairment of the descending pathway is believed to promote the occurrence and development of CS [52, 69]. In patients with chronic pain, the low CPM has been associated with multiple factors, including more sites of pain [65], a higher degree of disability [47], and pain-related cognitions and psychological factors such as anxiety, depression, and catastrophizing [36, 40]. This might suggest a similar link in CNP individuals. The prefrontal lobe not only plays an important role in the regulation of mood and behavior, but also influences pain processing and downstream pathways [24]. Functional brain imaging shows overlapping of the corresponding brain regions [10, 43], which has also been well documented in studies of knee pain [39], chronic osteoarthritis of the knee [53], and chronic low back pain. A relationship between CPM and psychological factors had been investigated, but evidence was equivocal. A large cohort study showed [55] that cognitions and psychological factors were found to be unrelated to CPM in individuals with acute, subacute, and chronic musculoskeletal pain, in addition to similar findings in pain-free and chronic pain patients [48].
We have previously investigated individuals with CS pain [33, 39, 66]. That information, on pain-related cognitions (kinesiophobia, pain catastrophizing) and emotional distress (symptoms of depression and anxiety) has also been collected via clinically commonly used self-reported questionnaires. Still, similar exploratory studies have not yet been conducted in chronic neck pain populations. Given the clinical research and practice of the CSI and CPM in CS, and considering the crucial of cognitions and psychological factors in CS, the primary purposes of this study were (1) to further explore the correlations between pain-related cognitions and psychological factors, psychophysical measures of CS, and self-report screening tool in patients with CNP; and (2) to determine whether pain-related cognitions and psychological factors predicted CSI scores as well as CPM effects in patients with CNP. We hypothesized that in patients with CNP, pain-related cognitions and psychological factors are associated with CSI and also influence CSI score, and can also influence their CPM. This study’s results may help us to better understand the importance of paying adequate attention to patients' cognitions and psychological factors when treating them.
Methods
Ethical Approval
This clinical cross-sectional study was approved by the Ethics Committee of the Institute of Sports Science of the State General Administration of Sport of China (code: 20230327) following the Declaration of Helsinki. All eligible subjects were informed about the study and signed an informed consent form before the start of the study.
Participants
Patients with CNP were recruited in July of 2023 at the Institute of Sports Science of the National Sports Administration. The criteria for CNP were as follows. Participants included were: (1) Aged between 18 and 60; (2) The average resting neck pain intensity was between 3 and 7 points (ten-point visual analog scale), and the pain duration is more than three months [25]; (3) No history of cervical surgery and fracture. Participants were excluded if they displayed: (1) Signs of nerve root compression (e.g., paresthesia, numbness, or weakness in the upper extremities); (2) Inflammatory arthritis affecting the cervical spine (e.g., rheumatoid arthritis, ankylosing spondylitis); (3) Neurological diseases affecting the cervical spine (such as Parkinson’s disease, stroke, and multiple sclerosis); (4) During pregnancy or lactation.
Procedure
Participants underwent all assessments, including clinical history taking, physical examination, and psychophysical testing on the same day. All of the above assessments were completed under the guidance of professionally trained researchers.
Demographic Information Collection
Socio-demographic information was obtained by completing a questionnaire containing demographic and CNP characteristics. Information on age, gender, BMI, and pain duration was collected.
Variables
Pain Intensity
The Visual Analogue Scale (VAS) is the patient-reported outcome measure (PROM) most frequently used to measure pain intensity in chronic pain, which has good reliability, validity, and response sensitivity [21, 28]. The VAS used here is a bidirectional 10-cm straight line with two labels, that is, “no pain” and “the worst pain”, located at the either end of the line. Individuals were requested to make a mark on the line based on the average pain intensity in the last month, and the distance between the pain-free and the marked point was the VAS pain intensity score; 0–3 cm indicates mild, 3–7 cm indicates moderate, and > 7 cm indicates severe pain intensity [30].
Central Sensitization
The Central Sensitization Inventory (CSI) is a two-part self-report questionnaire that consists of an assessment of common health-related symptoms in patients with CS (Part A) and a question asking whether the patient has been diagnosed with a specific disease (Part B). Only part A was used for assessment in this study. This section consists of 25 items that subjects were asked to rate on a five-point Likert scale from 0 to 4, where ‘0’ is ‘never’ and ‘4’ is ‘always.’ A total score of 100 was assigned, where the severity of CS-related symptoms was categorized as subclinical (0–29), mild (30–39), moderate (40–49), severe (50–59), and very severe (60–100) [50, 63], with higher scores representing greater self-reported severity of symptoms. In identifying patients with central sensitivity syndrome (CSS), the 40/100 threshold showed an acceptable psychometric effect. In addition, the CSI has good retest reliability and internal consistency, and the Chinese version of the CSI also shows good psychometric characteristics [22, 37].
Psychophysical Measurement of Descending Pain Inhibition Pathways—CPM
The efficacy of the descending pain suppression system was assessed using the cold pressor test (CPT), where cold pain is the conditioned stimulus and the pressure pain threshold (PPT) is the test stimulus. After following the standardized procedure, the subjects submerge their non-dominant side hand in water at room temperature (22 ± 1 °C) for 1 min prior to the rest. Conditional stimulation was then applied, with the subject’s dominant side hand immersed in temperature-controlled cold water (12 ± 1 °C) monitored by a thermometer (BATTERY TP101) for 1 min. The cold water should be over the wrist, the fingers should be spread, and any hand movements and position movements should be avoided. 30 s later, the PPT value was measured at the superior trapezius muscle on the dominant side (that is, the midpoint of the line between C7 and the lateral border of acromion), and the average value of three consecutive measurements was taken as the test stimulus for analysis. The CPM effect was evaluated by PPT difference before and during the CPT test. A negative value represents a low CPM effect, and a zero or positive value means a typical effect [34]. Room temperature, humidity, and brightness remain constant throughout the process.
Pain-Related Psychosocial Factors
Anxiety and depression symptoms were assessed using the depression subscale of the Hospital Anxiety and Depression Scale (HADS). The scale consists of two subscales, the anxiety subscale (HADS-A) and the depression subscale (HADS-D), each of which consists of seven items, each of which is rated on a four-point scale from 0 to 3, and a total score of ≥ 8 for each subscale is considered to be anxiety or depressive symptoms [7].
Pain-Related Cognitions
Pain Catastrophizing
Worries or catastrophizing thoughts related to pain were assessed using the Pain Catastrophizing Scale (PCS) [62]. The scale consists of 13 entries evaluated in three dimensions, namely rumination (entries 8, 9, 10, 11), magnification (entries 6, 7, 13), and helplessness (entries 1, 2, 3, 4, 5, 12) [62]. Each entry included a score of 0 (never)—4 (always) and a total score of 0–52, with > 25 considered to have a high level of pain catastrophizing [55]. The Chinese version of the PCS showed good reliability [61].
Kinesiophobia
The Tampa Scale for Kinesiophobia (TSK) is used to measure the fear of exercise/(re) injury in patients with chronic pain, and associated with fear avoidance measures. The Chinese version of the questionnaire has excellent internal consistency (Cronbach’s alpha of 0.74) and test–retest reliability (intraclass correlation coefficient of 0.86) [70]. The scale consists of 17 items, each item from 1 to 4 points, and the total score from 17 to 68 points. The greater the score indicates, the greater the fear of exercise; 37/68 is considered to be the critical value of high and low fear [19].
Statistical Analysis
The sample size was referenced to previous similar studies [41, 57], and 54 participants were included. Statistical analyses were performed in SPSS version 27.0 (IBM, New York, NY). Continuous variables were expressed as mean ± standard deviation, and categorical variables were expressed as frequencies and percentages. The Shapiro–Wilk test was used to detect the normality of all variables visually. Using box-plot to identify outliers. Due to the non-normality of the parameters, correlations of CSI scores, CPM effects, and self-report questionnaires were assessed using Spearman's correlation coefficients. According to Hinkle’s recommendations [27], correlation coefficients < 0.3 were interpreted as negligible correlations, 0.3 ≤ to < 0.5 as low correlations, 0.5 ≤ to < 0.7 as moderate correlations, 0.7 ≤ to < 0.9 as high correlations, and ≥ 0.9 as very high correlations. To explore the potential predictive value of cognition and psychological factors in CPM and CSI scores, multiple linear regression analyses were performed. Which CPM effect and CSI score were used as dependent variables, and kinesiophobia, pain catastrophizing, anxiety, and depression as independent variables, respectively. The Durbin–Waston test was used to test sample independence, and variance inflation factor (VIF) ≤ 5 was used for the multicollinearity test. The significance was set as p < 0.05.
Results
Fifty-four patients with CNP were included in the study, of whom 35 (64.8%) were female, with a mean age of 39.5 (± 8.8) years and a mean BMI of 22.8 (± 2.9) kg/m2. The mean duration of pain at study inclusion was 4.7 (± 1.2) months, and the mean VAS was 4.5 (± 1.5) points (Table 1). The effect of CPM is shown in Table 2. According to the scale threshold, 42.6% of the subjects were considered to have symptoms related to CS or CSS, of which 74.1 and 1.9% were considered to have motor fear and pain catastrophizing beliefs, and 27.8 and 24.0% were considered to have anxiety and depression, respectively.
Correlation Between CSI Scores and CPM Effects
Spearman's correlation coefficient showed no statistically significant correlation between CSI scores and CPT test results (r = 0.257, p > 0.05).
Correlation of Pain-Related Cognitions and Psychological Factors with CPM Effects
When controlling for gender, age, BMI, and pain duration, there was no statistically significant correlation between any of the cognitive and psychological factors and the results of the CPM test (r = − 1.72–0.14, p > 0.05).
Correlation of Pain-Related Cognitions and Psychological Factors with CSI Scores
When controlling for gender, age, BMI, and pain duration, there was a moderate correlation between CSI scores and kinesiophobia (rs = 0.554, p < 0.01), a low correlation with pain catastrophizing and anxiety (r = 0.332, p = 0.017; r = 0.492, p < 0.01), and no statistically significant correlation with depression (r = 0.207, p > 0.05)(Table 3).
Pain-Related Cognitions and Psychological Factors as Potential Predictors of CSI
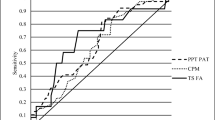
In the study, the results of the multiple regression linear analysis between cognitive and psychological factors and CSI scores were presented in Table 4 and Fig. 1. The analysis revealed that both kinesiophobia (B = 1.308, p < 0.01) and anxiety (B = 1.806, p = 0.02) had a positive prediction on CSI scores. However, pain catastrophizing (B = 0.129, β = 0.074, p > 0.05) and depression (B = − 0.902, β = − 0.190, p > 0.05) did not have a significant influence on CSI scores. After removing outliers, the results still showed significant predictive roles for kinesiophobia (B = 1.084, p = 0.002) and anxiety(B = 1.567, p = 0.008)in CSI scores, with neither pain catastrophizing nor depression (p > 0.05) had no predictive role.
Multiple linear regression analysis model. The Central Sensitization Inventory (CSI) scores was the dependent variable, and self-report questionnaires were the independent variables, including the Tampa Scale for Kinesiophobia (TSK), The Pain Catastrophizing Scale (PCS), The Hospital Anxiety and Depression Scale- Anxiety (HADS-A) and The Hospital Anxiety and Depression Scale- Depression (HADS-D)
Discussion
The results of this study showed that CSI scores were not statistically significantly correlated with the CPM effect, and neither cognitions nor psychological factors were statistically significantly associated with the CPM effect. In contrast, CSI scores were associated with kinesiophobia, pain catastrophizing, and anxiety, but not with depression. The present study also showed that kinesiophobia and anxiety significantly predicted CSI scores in patients with CNP when age, gender, BMI, and pain duration were considered. To our knowledge, this is the first study to explore the correlation between pain-related cognitive and psychological factors, CSI scores, and CPM effects in a CNP population.
The present study failed to reveal any correlation between CSI scores and CPM effects. Although CPT is not the gold standard for the diagnosis of CS, and it is not possible to avoid the risk of distraction induced by the conditioned stimulus in this parallel CPT paradigm [68], we still believe that CPT is the best testing paradigm and elicits the most substantial CPM effect [56]. Bezerra et al.[14] conducted a cross-sectional study of 267 patients with chronic musculoskeletal pain, comparing CSI scores with CPT and compared and did not find a correlation between the two. Coronado et al. [16] found no relationship between CSI scores and QST in patients with shoulder pain either, and these studies confirm the results of the present study. Even though the CPM paradigm is subject to multifactorial influences and the degree of response to CPM varies from one conditioned stimulus to another. Nevertheless, quantifying CPM effects makes it more likely to infer changes in the CNS processing of pain in patients with chronic pain. We did not find any cognitive or psychological factors associated with CPM effects. Nahman-Averbuch et al. [48] explored the relationship between anxiety, depression, pain catastrophizing, and CPM in a review, and their findings were consistent with this research. Furthermore, an exploratory study by Plinsinga et al. [55] showed that cognitive and psychological factors were not associated with CPM in 1142 patients with acute, subacute, and chronic pain, while secondary analyses also showed that there was no cumulative effect of any negative cognitive and psychological presence on CPM. It has been suggested that cognitive processes during testing and acute processing of pain-related cognitions may affect CPM, specifically that the brain rather than the spinal cord plays a role in the regulation of anticipation-induced pain [58]. In contrast, in patients with chronic widespread pain, it has also been demonstrated that catastrophizing may predominantly influence injurious processing at the supraspinal rather than spinal level [58], whereas CPM responds to downstream pain pathways at the spinal level.
In addition, the correlation of CPM effects with cognitions and psychological factors cannot be explained singularly because of the overlap and complexity of the areas involved in CPM activation, in the assessment of pain sensations, and in the areas that play a role in the emotional/affective components associated with pain, and in the higher sensory structures involved in sensory vigilance processing [46]. Considering the differences in study protocols and the different test and conditioned stimuli used during CPM testing, these differences may all affect the interpretation of the results.
As shown previously, cognitions and psychological factors play an essential role in CSI score, which may be an important reason for the lack of correlation between CSI score results and CPM effects. Notably, the results of the present study show that kinesiophobia is moderately correlated with CSI scores and is also a significant potential predictor of CSI scores. On the one hand, patients with chronic musculoskeletal pain (CMP) usually initially show kinesiophobia for pain avoidance purposes, which leads to the modification of exercise behavior and influences pain management and its related behaviors. On the other hand, pain and the processing of pain-related information in patients with CMP may be related to how movement fear is perceived. Asiri et al. [6] used kinesiophobia as a predictor of pain intensity, proprioception, and functioning in patients with CNP, and concluded that patients with higher level of kinesiophobia faced more pain and poorer proprioception and functional performance. Wertli et al. [71] explored the prognostic value of motor fear in low back pain, and a systematic review also assessed the role of motor fear on pain, disability, and quality of life in patients with CMP [38]. Therefore, it is reasonable to believe that higher TSK scores are likely to predict higher CSI scores in patients with CNP, more likely to develop CS and related symptoms, as well as affecting function, rehabilitation outcomes, and quality of life in patients with CNP.
A correlation between pain catastrophizing and CSI scores was also found in our findings, although this relationship was weak. Pain catastrophizing has been defined as an exaggerated negative state of mind that arises during painful experiences [17] and influences the perception and understanding of pain. The relationship between catastrophizing and pain intensity has been confirmed in studies [67], which have shown that the higher the level of catastrophizing, the more severe the pain is approximately. Therefore, we hypothesized that in patients with CNP, the persistence of pain leads to an excessive preoccupation with pain and an exaggeration of the threats associated with it. The industry and worsening of these features, in turn, results in lower pain thresholds and increased pain sensitivity, and therefore, patients are more likely to exhibit higher CSI scores.
Considering that clinical guidelines recommend exercise as an effective treatment for neck pain [8], and that higher levels of exercise fear and pain catastrophizing may affect treatment adherence, their assessment and intervention are essential. Studies have shown that virtual reality (VR)-based interactive exercise training [12, 64], global postural re-education (GPR) [42, 54], pain neuroscience education (PNE) [65], and cognitive behavioral therapy (CBT) [4, 45] are all effective in improving kinesiophobia and pain intensity in CNP, which not only help to rationalize patients’ beliefs and perceptions of kinesiophobia, but also support multidisciplinary interventions for neck pain.
Among the psychological factors associated with pain, this study explored anxiety and depression. We found that anxiety was moderately correlated with CSI and effectively predicted CSI scores. On the one hand, anxiety is the second most common complication associated with neck pain [73], and the longer the duration of pain, the greater the risk of anxiety in patients [29]. Pain relief is the main complaint of CNP patients, and if unresolved for an extended period, then they will face both physical and psychological stress. In a recent study, Clark et al. [13] explored the relationship between physical and emotional sensitivity and showed that trait anxiety may be associated with the development of CS. In addition, the CSI was developed with anxiety, a psychological factor related to CS, in mind, along with a specialized entry (item 3) to facilitate direct responses. It is reasonable to believe that anxiety plays a vital role in predicting CSI scores. Therefore, healthcare professionals must provide appropriate psychological guidance or interventions for CNP patients in clinical care to reduce the risk of anxiety.
We did not find a correlation between depression and CSI. A national survey of chronic spinal pain showed that persistent depression was more strongly associated with chronic spinal pain than major depression [60], yet more studies of chronic pain and depression have focused primarily on major or persistent depression, and the evidence from studies of time series of depression and chronic pain is ambiguous [18, 26]. Unlike these studies, included were assessed as having a depressive state, but to a lesser extent. Moreover, our study did not consider potential moderators or mediating effects between chronic neck pain and depression, such as exercise and sleep disorders [31, 32]. It thus could not accurately conclude whether CS in the chronic neck pain population is truly independent of depression.
Strengths and Limitations
This study is the first to explore the correlation between pain-related cognitions and psychological factors, CPM, and CSI in patients with CNP and to affirm the predictive value of kinesiophobia and anxiety for CSI score. Other potential factors affecting CSI scores deserve further study in the future.
The present study was design was cross-sectional, and while several cognitions and psychological factors were identified, it was not possible to determine whether there was a causal relationship between them and the development of CS in a CNP population. Nevertheless, cross-sectional studies could serve as an initial stage for future research while continuing to explore other potential factors. Secondly, when performing the CPT, we assessed the CPM effect while the dominant hand was still immersed in cold water and thus could not avoid the risk of distraction induced by the conditioned stimulus, which could have impacted the CPM effect. Furthermore, confounding variables such as age, gender, the intensity of pain that may have affected these findings were not considered by the authors. As a result, future studies should focus on these confounding variables and see how they affect the outcomes. We identified and processed the outliers in this study, and the statistics and analyses similarly supported the use of motor fear and anxiety as significant predictors of CSI scores. We believe that the occurrence of outliers was random and unavoidable and did not affect the overall trend and predictors of the model. Therefore, all sample sizes were ultimately retained in this study to ensure data integrity and authenticity. Finally, the results of this study should be interpreted with caution due to the small sample size of our research and the fact that we did not include a healthy population as a control, which may limit the generalizability of the findings to some extent. Further studies in the broader population are needed to confirm these findings.
Clinical Implications
The study’s results validate our hypothesis that CPM is not associated with any cognitions or psychological factors in individuals with CNP. In the future, we need to find other tools or methods to quantify the pain suppression system. In clinical practice, the use of CSI for the assessment of somatic symptoms based on the interaction of biopsychosocial factor should be combined with a history of previous disease and clinical examination to improve the validity and accuracy of identifying and assessing CS. Given the growing body of evidence supporting the important role of CS in pain chronicity, including CNP, it may be useful to consider CS as a potential target to reduce pain sensitivity and improve central inhibition [51]. In addition, extensive attention should be paid to patients' psychological, cognitive, and social factors to provide multidisciplinary interventions. Therefore, understanding the potential mechanisms and clinical significance of CS may help manage individuals with CNP and provide new ideas for their clinical diagnosis and treatment.
Conclusions
This cross-sectional study reports for the first time the limitations of using the CSI and CPM to assess CS and its associated symptoms in patients with chronic neck pain. The results indicated that cognitive and psychological factors were not associated with CPM effects but were related to CSI scores. Among these, kinesiophobia and anxiety positively predicted CSI scores but did not seem related to depression. All these results suggest that CS in patients with chronic neck pain is multifactorial, and its associated treatment should also consider potential targets other than pain factors, but larger studies are needed for further confirmation.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adams GR, Gandhi W, Harrison R, et al. Do “central sensitization” questionnaires reflect measures of nociceptive sensitization or psychological constructs? Protocol for a systematic review. PAIN Reports. 2021;6(4): e962.
Adams LM, Turk DC. Psychosocial factors and central sensitivity syndromes. Curr Rheumatol Rev. 2015;11(2):96–108.
Ahmad AH, Zakaria R. Pain in times of stress. Malays J Med Sci. 2015;22(Spec Issue):52–61.
Andias R, Sa-Couto P, Silva AG. Blended-Learning pain neuroscience education and exercise in high school students with chronic neck pain: a randomized controlled trial. Phys Ther. 2022;102(6): pzac048.
Arendt-Nielsen L, Morlion B, Perrot S, et al. Assessment and manifestation of central sensitisation across different chronic pain conditions. Eur J Pain. 2018;22(2):216–41.
Asiri F, Reddy RS, Tedla JS, et al. Kinesiophobia and its correlations with pain, proprioception, and functional performance among individuals with chronic neck pain. PLoS One. 2021;16(7): e0254262.
Bjelland I, Dahl AA, Haug TT, et al. The validity of the Hospital Anxiety and Depression Scale. An updated literature review. J Psychosom Res. 2002;52(2):69–77.
Blanpied PR, Gross AR, Elliott JM, et al. Neck pain: revision 2017. J Orthop Sports Phys Ther. 2017;47(7):A1–83.
Breivik H, Collett B, Ventafridda V, et al. Survey of chronic pain in Europe: prevalence, impact on daily life, and treatment. Eur J Pain (London, England). 2006;10(4):287–333.
Buhle J, Wager TD. Performance-dependent inhibition of pain by an executive working memory task. Pain. 2010;149(1):19–26.
Caumo W, Antunes LC, Elkfury JL, et al. The Central Sensitization Inventory validated and adapted for a Brazilian population: psychometric properties and its relationship with brain-derived neurotrophic factor. J Pain Res. 2017;10:2109–22.
Chen KB, Sesto ME, Ponto K, et al. Use of virtual reality feedback for patients with chronic neck pain and kinesiophobia. IEEE Trans Neural Syst Rehabil Eng. 2017;25(8):1240–8.
Clark JR, Nijs J, Yeowell G, et al. Trait sensitivity, anxiety, and personality are predictive of central sensitization symptoms in patients with chronic low back pain. Pain Pract. 2019;19(8):800–10.
Cliton Bezerra M, Valentim Bittencourt J, Reis FJJ, et al. Central Sensitization Inventory is a useless instrument for detection of the impairment of the conditioned pain modulation in patients with chronic musculoskeletal pain. Joint Bone Spine. 2021;88(3): 105127.
Cohen SP, Hooten WM. Advances in the diagnosis and management of neck pain. BMJ. 2017. https://doi.org/10.1136/bmj.j3221.
Coronado RA, George SZ. The Central Sensitization Inventory and Pain Sensitivity Questionnaire: An exploration of construct validity and associations with widespread pain sensitivity among individuals with shoulder pain. Musculoskelet Sci Pract. 2018;36:61–7.
Craner JR, Gilliam WP, Sperry JA. Rumination, magnification, and helplessness: how do different aspects of pain catastrophizing relate to pain severity and functioning? Clin J Pain. 2016;32(12):1028–35.
Currie SR, Wang J. More data on major depression as an antecedent risk factor for first onset of chronic back pain. Psychol Med. 2005;35(9):1275–82.
Demirbüken İ, Özgül B, Kuru Çolak T, et al. Kinesiophobia in relation to physical activity in chronic neck pain. J Back Musculoskelet Rehabil. 2016;29(1):41–7.
Den Boer C, Dries L, Terluin B, et al. Central sensitization in chronic pain and medically unexplained symptom research: a systematic review of definitions, operationalizations and measurement instruments. J Psychosom Res. 2019;117:32–40.
Euasobhon P, Atisook R, Bumrungchatudom K, et al. Reliability and responsivity of pain intensity scales in individuals with chronic pain. Pain. 2022;163(12):e1184–91.
Feng B, Hu X, Lu WW, et al. Cultural validation of the Chinese central sensitization inventory in patients with chronic pain and its predictive ability of comorbid central sensitivity syndromes. J Pain Res. 2022;15:467–77.
Fine PG. Long-term consequences of chronic pain: mounting evidence for pain as a neurological disease and parallels with other chronic disease states. Pain Medicine (Malden, Mass). 2011;12(7):996–1004.
Goffaux P, Redmond WJ, Rainville P, et al. Descending analgesia–when the spine echoes what the brain expects. Pain. 2007;130(1–2):137–43.
Hague M, Shenker N. How to investigate: chronic pain. Best Pract Res Clin Rheumatol. 2014;28(6):860–74.
Hilderink PH, Burger H, Deeg DJ, et al. The temporal relation between pain and depression: results from the Longitudinal Aging Study Amsterdam. Psychosom Med. 2012;74(9):945–51.
Hinkle DE, Wiersma W, Jurs SG. Applied statistics for the behavioral sciences, Vol. 663. Boston: Houghton Mifflin College Division; 2003
Hjermstad MJ, Fayers PM, Haugen DF, et al. Studies comparing Numerical Rating Scales, Verbal Rating Scales, and Visual Analogue Scales for assessment of pain intensity in adults: a systematic literature review. J Pain Symptom Manage. 2011;41(6):1073–93.
Hu Y, Yang Z, Li Y, et al. Anxiety symptoms and associated factors among chronic low back pain patients in China: a cross-sectional study. Front Public Health. 2022;10: 878865.
Jensen MP, Chen C, Brugger AM. Interpretation of visual analog scale ratings and change scores: a reanalysis of two clinical trials of postoperative pain. J Pain. 2003;4(7):407–14.
Juan W, Rui L, Wei-Wen Z. Chronic neck pain and depression: the mediating role of sleep quality and exercise. Psychol Health Med. 2020;25(8):1029–35.
Karimi R, Mallah N, Scherer R, et al. Sleep quality as a mediator of the relation between depression and chronic pain: a systematic review and meta-analysis. Br J Anaesth. 2023;130(6):747–62.
Klyne DM, Moseley GL, Sterling M, et al. Individual variation in pain sensitivity and conditioned pain modulation in acute low back pain: effect of stimulus type, sleep, and psychological and lifestyle factors. J Pain. 2018;19(8):942.e1-942.e18.
Kregel J, Schumacher C, Dolphens M, et al. Convergent validity of the Dutch central sensitization inventory: associations with psychophysical pain measures, quality of life, disability, and pain cognitions in patients with chronic spinal pain. Pain Pract. 2018;18(6):777–87.
Latremoliere A, Woolf CJ. Central sensitization: a generator of pain hypersensitivity by central neural plasticity. J Pain. 2009;10(9):895–926.
Lewis GN, Rice DA, McNair PJ. Conditioned pain modulation in populations with chronic pain: a systematic review and meta-analysis. J Pain. 2012;13(10):936–44.
Liang D, Yu X, Guo X, et al. Adaptation and validation of the Chinese version of the Central Sensitisation Inventory in patients with chronic pain. General Psychiatry. 2022;35(6): e100919.
Luque-Suarez A, Martinez-Calderon J, Falla D. Role of kinesiophobia on pain, disability and quality of life in people suffering from chronic musculoskeletal pain: a systematic review. Br J Sports Med. 2019;53(9):554–9.
Maclachlan LR, Collins NJ, Hodges PW, et al. Psychological and pain profiles in persons with patellofemoral pain as the primary symptom. Eur J Pain (London, England). 2020;24(6):1182–96.
Manchikanti L, Fellows B, Pampati V, et al. Comparison of psychological status of chronic pain patients and the general population. Pain Physician. 2002;5(1):40–8.
Matesanz-García L, Cuenca-Martínez F, Simón AI, et al. Signs indicative of central sensitization are present but not associated with the central sensitization inventory in patients with focal nerve injury. J Clin Med. 2022;11(4):1075.
Mendes-Fernandes T, Puente-González AS, Márquez-Vera MA, et al. Effects of global postural reeducation versus specific therapeutic neck exercises on pain, disability, postural control, and neuromuscular efficiency in women with chronic nonspecific neck pain: study protocol for a randomized, parallel, clinical trial. Int J Environ Res Public Health. 2021;18(20):10704.
Miller EK, Cohen JD. An integrative theory of prefrontal cortex function. Annu Rev Neurosci. 2001;24:167–202.
Miranda J, Lamana SMS, Dias EV, et al. Effect of pain chronification and chronic pain on an endogenous pain modulation circuit in rats. Neuroscience. 2015;286:37–44.
Monticone M, Ambrosini E, Cedraschi C, et al. Cognitive-behavioral treatment for subacute and chronic neck pain: a Cochrane review. Spine. 2015;40(19):1495–504.
Moont R, Crispel Y, Lev R, et al. Temporal changes in cortical activation during conditioned pain modulation (CPM), a LORETA study. Pain. 2011;152(7):1469–77.
Morris MC, Walker LS, Bruehl S, et al. Impaired conditioned pain modulation in youth with functional abdominal pain. Pain. 2016;157(10):2375–81.
Nahman-Averbuch H, Nir RR, Sprecher E, et al. Psychological factors and conditioned pain modulation: a meta-analysis. Clin J Pain. 2016;32(6):541–54.
Neblett R, Cohen H, Choi Y, et al. The Central Sensitization Inventory (CSI): establishing clinically significant values for identifying central sensitivity syndromes in an outpatient chronic pain sample. J Pain. 2013;14(5):438–45.
Neblett R, Hartzell MM, Mayer TG, et al. Establishing clinically relevant severity levels for the central sensitization inventory. Pain Pract. 2017;17(2):166–75.
Nijs J, Meeus M, Van Oosterwijck J, et al. Treatment of central sensitization in patients with ‘unexplained’ chronic pain: what options do we have? Expert Opin Pharmacother. 2011;12(7):1087–98.
Ossipov MH, Morimura K, Porreca F. Descending pain modulation and chronification of pain. Curr Opin Support Palliat Care. 2014;8(2):143–51.
Parks EL, Geha PY, Baliki MN, et al. Brain activity for chronic knee osteoarthritis: dissociating evoked pain from spontaneous pain. Eur J Pain (London, England). 2011;15(8):843.e1-14.
Pillastrini P, De Lima E, Sá Resende F, Banchelli F, et al. Effectiveness of global postural re-education in patients with chronic nonspecific neck pain: randomized controlled trial. Phys Ther. 2016;96(9):1408–16.
Plinsinga ML, Vuvan V, Maclachlan L, et al. Pain-related cognitions and emotional distress are not associated with conditioned pain modulation: an explorative analysis of 1142 participants with acute, subacute, and chronic pain. Pain. 2023;164(7):1593–9.
Ramaswamy S, Wodehouse T. Conditioned pain modulation—a comprehensive review. Neurophysiol Clin. 2021;51(3):197–208.
Rampazo ÉP, da Silva VR, de Andrade ALM, et al. Sensory, motor, and psychosocial characteristics of individuals with chronic neck pain: a case-control study. Phys Ther. 2021;101(7): pzab104.
Rice DA, Parker RS, Lewis GN, et al. Pain catastrophizing is not associated with spinal nociceptive processing in people with chronic widespread pain. Clin J Pain. 2017;33(9):804–10.
Safiri S, Kolahi AA, Hoy D, et al. Global, regional, and national burden of neck pain in the general population, 1990–2017: systematic analysis of the Global Burden of Disease Study 2017. BMJ (Clinical research ed). 2020;368: m791.
Schmaling KB, Nounou ZA. Incident chronic spinal pain and depressive disorders: data from the National Comorbidity Survey. J Pain. 2019;20(4):481–8.
Shen B, Wu B, Abdullah TB, et al. Translation and validation of simplified Chinese version of the Pain Catastrophizing Scale in chronic pain patients: education may matter. Mol Pain. 2018;14:174480691875528.
Sullivan MJL, Bishop SR, Pivik J. The Pain Catastrophizing Scale: development and validation. Psychol Assess. 1995;7(4):524–32.
Tanaka K, Murata S, Nishigami T, et al. The central sensitization inventory predict pain-related disability for musculoskeletal disorders in the primary care setting. Eur J Pain (London, England). 2019;23(9):1640–8.
Tejera D, Beltran-Alacreu H, Cano-de-la-Cuerda R, et al. Effects of virtual reality versus exercise on pain, functional, somatosensory and psychosocial outcomes in patients with non-specific chronic neck pain: a randomized clinical trial. Int J Environ Res Public Health. 2020;17(16):5950.
Vaegter HB, Graven-Nielsen T. Pain modulatory phenotypes differentiate subgroups with different clinical and experimental pain sensitivity. Pain. 2016;157(7):1480–8.
Vaegter HB, Palsson TS, Graven-Nielsen T. Facilitated pronociceptive pain mechanisms in radiating back pain compared with localized back pain. J Pain. 2017;18(8):973–83.
Van Bogaert W, Liew B X W, Fernández-de-Las-Peñas C, et al. Exploring interactions between sex, pain characteristics, disability, and quality of life in people with chronic spinal pain: a structural equation model. J Pain. 2023; S1526-5900(23)00584-9.
Villemure C, Bushnell CM. Cognitive modulation of pain: how do attention and emotion influence pain processing? Pain. 2002;95(3):195–9.
Wang R, King T, De Felice M, et al. Descending facilitation maintains long-term spontaneous neuropathic pain. J Pain. 2013;14(8):845–53.
Wei X, Xu X, Zhao Y, et al. The Chinese version of the Tampa Scale for Kinesiophobia was cross-culturally adapted and validated in patients with low back pain. J Clin Epidemiol. 2015;68(10):1205–12.
Wertli MM, Rasmussen-Barr E, Weiser S, et al. The role of fear avoidance beliefs as a prognostic factor for outcome in patients with nonspecific low back pain: a systematic review. Spine J. 2014;14(5):816–36 (e4).
Woolf CJ. Central sensitization: implications for the diagnosis and treatment of pain. Pain. 2011;152(3 Suppl):S2–15.
Xu Y, Wang Y, Chen J, et al. The comorbidity of mental and physical disorders with self-reported chronic back or neck pain: results from the China Mental Health Survey. J Affect Disord. 2020;260:334–41.
Zabala Mata J, Lascurain-Aguirrebeña I, Dominguez López E, et al. Enhanced pronociceptive and disrupted antinociceptive mechanisms in nonspecific chronic neck pain. Phys Ther. 2021;101(3): e223.
Acknowledgements
We thank the participants of the study.
Funding
This research (including the journal’s Rapid Service Fee) was supported by the Fundamental Research Funds for the China Institute of Sport Science (Grant basic 23-43).
Author information
Authors and Affiliations
Contributions
Yuwei He and Jialin Wang served as principal authors, had full access to all the data in the study, and took responsibility for the accuracy of the data analysis and its integrity. Peng Zhao contributed to the conception and design. Ruirui Wang and Meng Li contributed to data acquisition and interpretation. Yuwei He and Jialin Wang prepared the initial manuscript draft, revised the article, and gave final approval.
Corresponding author
Ethics declarations
Conflict of Interest
Yuwei He, Jialin Wang, Peng Zhao, Meng Li and Ruirui Wang have no competing interests disclose.
Ethical Approval
This clinical cross-sectional study was approved by the Ethics Committee of the Institute of Sports Science of the State General Administration of Sport of China (code: 20230327) following the Declaration of Helsinki. All eligible subjects were informed about the study and signed an informed consent form before the start of the study.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
He, Y., Wang, J., Zhao, P. et al. Correlations of The Central Sensitization Inventory, conditioned pain modulation, cognitions and psychological factors in individuals with chronic neck pain: A cross-sectional study. Pain Ther 13, 843–856 (2024). https://doi.org/10.1007/s40122-024-00601-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40122-024-00601-w