Abstract
Introduction
Multidisciplinary patient-reported outcomes are a critical part of assessing patients to better understand their well-being during treatment. The use of multidisciplinary patient-reported outcomes is recommended in many areas of medicine. The Patient-Reported Outcomes Measurement Information System-29 (PROMIS-29) has been utilized as a common measurement language across universally relevant domains, including pain, mood, sleep, social participation, and function.
Methods
Retrospective analysis of prospectively collected data was performed. Subjects were identified and consecutively enrolled upon entry into chronic pain centers across 24 sites in the United States. The PROMIS-29 v2.1 and the numerical rating scale (NRS) were recorded. The pain impact score and the Health Utilities Index Mark 3 (HUI-3) were calculated. Statistical differences were assessed between genders and among age groups comprising subjects less than 40, 41–60, 61–80, and over 80 years of age.
Results
A total of 19,546 patients were assessed over the enrollment period from 2018 to 2020. The PROMIS-29 v2.1 was evaluated across the seven domains, along with the numerical rating sale (NRS). The mean scores of the population for PROMIS SF v1.0 Pain Interference 4a, PROMIS SF v1.0 Sleep Disturbance 4a, PROMIS SF v1.0 Fatigue 4a, PROMIS SF v1.0 Anxiety 4a, PROMIS SF V1.0 Depression 4a, PROMIS SF v2.0 Ability to Participate in Social Roles and Activities 4a, and PROMIS SF v1.0 Physical Function 4a, measuring pain interference, sleep disturbance, fatigue, anxiety, depression, social participation, and physical function, were 64.61, 57.19, 58.50, 53.94, 54.45, 40.06, and 36.23, respectively. Pain intensity was 6.38 on an 11-point NRS scale. The pain impact score (PIS) and health utilities index mark 3 (HUI-3) scores, calculated across the designated age groups, were 33.19 and 0.67, respectively. Statistical differences were observed for the domains of sleep disturbance and physical function for age groups less than 40 and greater than 80 years of age.
Conclusion
This data set is the first published normative data set describing the PROMIS-29 assessment in the chronic pain population. The patient population is more homogeneous than expected, and females were found to have higher levels of dysfunction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
The assessment of outcomes in the field of pain management is rapidly evolving. |
Chronic pain is a global health concern, with extensive economic and societal impacts, afflicting nearly 120 million Americans, with estimates of over 40% of adults experiencing pain for over 3–6 months. |
Linear and one-dimensional pain assessments are outdated and inaccurate, whereas multidimensional validated patient-reported outcome assessments allow for a more complete view of the patient’s pain experience. |
To our knowledge, there has never been an investigation into the normative values for the chronic pain population so that we can better understand and benchmark our treatment algorithms. The purpose of this study is to survey and report on PROMIS-29, the pain impact score, and estimated HUI-3 scores of patients entering into chronic pain treatment in the United States. |
What was learned from the study? |
This data set is the first published normative data set describing the PROMIS-29 assessment in the chronic pain population. |
The patient population is more homogeneous than expected, and females were found to have higher levels of dysfunction. |
Digital Features
This article is published with digital features, including a summary slide, to facilitate understanding of the article. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.13643255.
Introduction
The assessment of outcomes in the field of pain management is rapidly evolving. Once dependent on linear numerical rating scales (NRS) and visual analog scales (VAS), emphasis within the specialty has shifted toward multidimensional patient-reported outcomes (PRO) [1]. This strategy of assessing pain from a multidimensional perspective in the context of the biopsychosocial model is recommended by every recent major consensus panel, including the US Department of Health and Human Services (HHS) [2], the National Institutes of Health (NIH) Research Task Force (RTF) for Chronic Low Back Pain [3], the Institute of Medicine (IOM) [4], the Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials (IMMPACT) [5], and more.
A common measurement language is critical to galvanizing innovation, research, and care improvement. The Patient-Reported Outcomes Measurement Information System (PROMIS) was created by the NIH in 2004, and several studies were initiated, focusing on heart failure, chronic obstructive pulmonary disease, rheumatoid arthritis, cancer, chronic low back pain, and major depression [6]. The goal of PROMIS was to update longer-form legacy instruments with validated outcomes measures using fewer questions with greater accuracy. Many familiar legacy measures were used to create the PROMIS item banks, and crosswalk tables are available to allow for conversion to legacy measures on the NIH-funded PROsetta Stone® website (www.prosettastone.org, NIH 1RC4CA157236, PI: David Cella, PhD). The PROMIS-29 battery assesses mental health, physical health, and social health through seven 4-question instruments, including assessments of fatigue, pain intensity, pain interference, physical function, sleep disturbance, anxiety, depression, and ability to participate in social roles and responsibilities [7]. The PROMIS-29 and its subdomains have been validated and/or cross-walked to a host of legacy measures in a variety of populations, including the Oswestry Disability Index [8], Roland-Morris Disability Questionnaire [9], Brief Pain Inventory-Pain Interference domain [10], the EuroQol Research Foundation five-dimension (EQ-5D) instrument [11], the Health Utilities Index Mark 3 (HUI-3) [12], and quality-adjusted life year [13] calculations.
The pain impact score (PIS) was defined and validated in 2014 by the NIH Research Task Force for Low Back Pain [3, 14]. The PIS is a calculated measure derived from the PROMIS-29 Pain Interference (PI), Physical Function (PF), and Pain Intensity (NRS) subdomain raw scores. The score for PF is inverted so that higher scores indicate more severe pain impact and physical dysfunction. The PIS is scored as follows:
PIS = NRS + PROMIS-PI + PROMIS-PF.
Score range is 8–50 points: NRS (0–10), PROMIS-PI (4–20), PROMIS-PF (4–20).
Estimated minimal clinically important difference (MCID) = 3 points [14].
The use of the PROMIS-29 and PIS as a multidimensional assessment and outcomes tool was previously compared to subjective percent pain relief (PPR) after a trial of spinal cord stimulation in 127 subjects [15]. PIS was found to correlate more strongly with PPR than did the change in NRS pain score or any PROMIS-29 subdomain alone [15].
The HUI-3 is a widely used measure in health-related quality of life (HRQOL) studies that can be used to estimate quality-adjusted life years (QALYs) [16]. The HUI-3 assesses eight health status dimensions on a 5- or 6-point scale (normal to highly impaired): vision, hearing, speech, ambulation, dexterity, emotion, and cognition [16, 17]. Hays et al. [12] described regression models on data from the original PROMIS cohort to accurately estimate HUI-3 scores across a representative US national sample of 3000 subjects. This same methodology was applied in this study to calculate HUI-3 preference scores in this sample of patients presenting to a chronic pain clinic.
The PROMIS-29 has been used in thousands of studies, published in over 150 journals, and is validated with cross-talks to other instruments. To our knowledge, there has never been an investigation into the normative values for the chronic pain population so that we can better understand and benchmark our treatment algorithms. The purpose of this study was to survey and report on PROMIS-29, the pain impact score, and estimated HUI-3 scores of patients entering into chronic pain treatment in the United States.
Methods
Study Design and Ethics Statement
Subjects were enrolled in a consecutive fashion throughout all participating sites from March 2019 to May 2020, after obtaining a waiver of consent and a full waiver of HIPAA authorization through the Western Institutional Review Board (WIRB) under Common Rule 45 CFR 46.116. The institutional review board (IRB) was happy to accept the decision of WIRB without any further determinations. Inclusion criteria included new patients entering into pain practice, patients > 18 years of age, and completion of the PROMIS-29. The PROMIS-29 has evolved into its third generation, v2.1, and comprises the following assessments: PROMIS Short Form (SF) v2.0—Physical Function 4a, PROMIS SF v1.0—Anxiety 4a, PROMIS SF v1.0—Depression 4a, PROMIS SF v1.0—Sleep Disturbance 4a, PROMIS SF v1.0—Ability to Participate in Social Roles and Activities 4a, PROMIS SF v1.0—Pain Interference 4a, and PROMIS Pain Intensity item (Global07).
PROMIS-29 data, along with patient demographic data including age and gender, was captured at all sites using a digital outcomes capture system (Real World Outcomes™, Celéri Health, Wilmington, DE, USA) within 3 weeks of the initial consultation for pain care. Statistical analysis was performed, calculating descriptive statistics of the median, mean, and mode of the reported T-scores of the PROMIS-29, its seven independent domains, and the calculated pain impact score.
Statistics
When comparing different age groups across the seven subdomains, along with the gender assessment, Welch’s t-test was employed to test the hypothesis that the two populations would have equal means.
Results
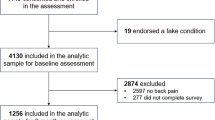
Between 2018 and 2020, 19,546 patients were enrolled across the 12 participating sites, obtaining baseline PROMIS-29 v2.1 upon entry into chronic pain practices across the United States. Patients were categorized by age group (less than 40 years, 41–60 years, 61–80 years, 81 years and older), along with an assessment of the effects of gender on the measured sample. Of the 19,546 patients assessed, 9165 were between the ages of 61 and 80, followed by the 40–60 group with 6840. In Fig. 1, we demonstrate PROMIS-29 score distributions in all subjects who presented to an outpatient chronic pain management clinic during the period 2018–2020, along with gender analysis (Fig. 1). Spanning all age groups, the average PROMIS-29 T-scores for the chronic pain population are as follows: pain interference, sleep disturbance, fatigue, anxiety, depression, social participation, and physical function are 64.61, 57.19, 58.50, 53.94, 54.45, 40.06, 36.23, respectively, with a pain impact score of 33.19 and pain intensity of 6.38 on an 11-point NRS scale (Fig. 1). The PROMIS-29 v2.1 battery consists of seven instruments sorted by subject age, assessing the mean, median, and mode for each age group. These groups were analyzed to detect differences amongst these cohorts in terms of their population profile related to mental, physical, and social health. A summary of this data is presented in Fig. 2. Figures 1 and 2 depict the proportion of all subjects achieving the calculated raw scores and T-scores in the respective domains. The color coding scheme represents the categorization of score ranges in the respective domains as severe, moderate, mild, and normal (Figs. 1, 2). Figure 3 shows the proportion of all subjects achieving the calculated impact scores and HUI-3 scores, for the total cohort as well as subcategories, based on age group (< 40, 41–60, 61–80, > 81) and gender. The mean score for each respective domain and subgroup is shown (Figs. 1, 2, 3). This was used to determine normative values for the chronic pain population and to demonstrate the heterogeneity of the population.
a Demonstrates PROMIS-29 score distributions in all subjects presenting to an outpatient chronic pain management clinic between 2018 and 2020. Spanning all age groups, the average PROMIS-29 T-scores for the chronic pain population are as follows: pain interference, sleep disturbance, fatigue, anxiety, depression, social participation, and physical function are 64.61, 57.19, 58.50, 53.94, 54.45, 40.06, 36.23, respectively, with a pain impact score of 33.19 and pain intensity of 6.38 on an 11-point NRS scale. b PROMIS-29 score distributions in female subjects presenting to an outpatient chronic pain management clinic between 2018 and 2020. The profile of these subjects (n = 11,310) indicates that on average, this population displays mild–moderate dysfunction across all PROMIS-29 domains, as noted above. c PROMIS-29 score distributions in male subjects presenting to an outpatient chronic pain management clinic between 2018 and 2020. The profile of these subjects (n = 8235) indicates that on average, this population displays mild–moderate dysfunction across all PROMIS-29 domains, as noted above
a The patient cohort aged 0–40 was evaluated by age to determine normative values for the chronic pain population to detect significant differences among those referred and seen in the chronic pain population. b The patient cohort aged 41–60 was evaluated by age to determine normative values for the chronic pain population to detect significant differences among those referred and seen in the chronic pain population. c The patient cohort aged 61–80 was evaluated by age to determine normative values for the chronic pain population to detect significant differences among those referred and seen in the chronic pain population. d The patient cohort aged 81 and older was evaluated by age to determine normative values for the chronic pain population to detect significant differences among those referred and seen in the chronic pain population
Mental Health
The depression assessment from the PROMIS SF v1.0 Depression scale demonstrates T-scores averages of 54.92, 54.75, 54.13, and 54.39 for those aged less than 40, 41–60, 61–80, and above 80 years, respectively. Interestingly, the scores are not statistically different among age groups or gender. The mean anxiety assessment scores from the PROMIS SF v1.0 Anxiety 4a are 55.45, 54.61, 53.29, and 52.72 for those aged less than 40, 41–60, 61–80, and above 80 years, respectively. Again, interestingly, among the age and gender assessed, there is no statistically significant difference, although females tended to have higher dysfunction and greater pain described.
Physical Health
The pain interference assessment from the PROMIS SF v1.0 Pain Interference 4a scale demonstrates T-scores of 64.73, 65.23, 64.26, and 63.71 for those aged less than 40, 41–60, 61–80, and above 80 years, respectively. Mean gender differences among the respondents were 64.95 versus 64.14. Fatigue scores assessed via the PROMIS SF v1.0 Fatigue 4a demonstrated in the chronic pain population average scores of 59.23, 59.26, 57.84, and 57.92 for those aged less than 40, 41–60, 61–80, and above 80 years, respectively. Gender differences were not statistically significant, as demonstrated with average scores of 59.49 and 56.99 for females and males, respectively. Physical function, measured from the PROMIS SF v1.0 Physical Function 4a, with high scores being supportive of care and low scores representing more dysfunction, are 37.96, 36.70, 35.85, and 34.00 for those aged less than 40, 41–60, 61–80, and above 80 years, respectively. Gender differences were not realized, with scores 35.61 for women and 36.96 for men. Mean sleep disturbance scores, as measured by the PROMIS-29 SF v1.0 Sleep Disturbance 4a, are 59.42, 59.11, 55.99, and 52.73 for those aged less than 40, 41–60, 61–80, and above 80 years, respectively. Gender differences for the aforementioned average scores are 57.64 and 56.63 for women and men, respectively.
Social Function
The mean scores for PROMIS-29 SF v2.0 4a Ability to Participate in Social Roles and Activities for the age groups less than 40, 41–60, 61–80, and above 80 years are 40.72, 39.55, 40.19, and 40.69, respectively, and for females and males are 39.90 and 40.24, respectively, with lower scores representing greater dysfunction.
Pain Intensity
The mean global pain impact scores, calculated as pain intensity + PROMIS Pain Interference + inverse of Physical Function, are 32.25, 33.41, 33.08, and 34.11, for those aged less than 40, 41–60, 61–80, and above 80 years. Pain intensity, based on a numerical rating scale of 0–10, is 6.50, 6.56, 6.24, and 6.24 for age groups of less than 40, 41–60, 61–80, and above 80 years, and gender grouping mean scores of 6.53 and 6.19 for females and males, respectively.
Gender Difference Analysis
Gender influence on PROMIS-29 assessment across the seven domains for patients entering into a chronic pain practice in the United States was evaluated as described above.
Discussion
This study represents the first time that this calculation has been performed, and has significant implications in measuring treatment success. This then serves as benchmark normative data on the chronic pain population entering into chronic pain and neurosurgery practices in the real world. The PROMIS-29 is generalized to the entire US population, and as such can serve as a common measurement language that is universally relevant across disease states. By creating a normative data set for the chronic pain population at multiple sites, this study serves to evaluate the typical PROMIS-29 presentation in this population so as to aid in the identification of outliers and to allow for more translational research. This has never been performed in a community-based, multicenter fashion for the chronic pain population.
For most PROMIS instruments, a T-score of 50 is the average for the US general population, with a standard deviation of 10. The T-score is reported with a standard error. A larger T-score represents more of the domain being measured, such that measures for negatively worded concepts represent worse than average. Larger scores of positively worded attributes represent a score better than average. As compared to the general population, all the PROMIS-29 T-scores measured in the population of chronic pain patients presenting to clinics seeking treatment are worse by a standard of deviation of 0.5–1.4 in general. It is interesting to note that although we assessed median and mean for all scores and instruments within the PROMIS-29, along with gender influence, the data is strikingly similar, spanning multiple different diagnoses, with little difference detected amongst age groups. We, however, did not specifically compare diagnosis or medication usage to the cohort studied, but follow-up studies are pending. Chronic pain patients demonstrate statistically higher reported dysfunction than the general population across the multiple domains assessed by the PROMIS-29 (Table 1).
When comparing different age groups across the seven subdomains, we found that there were significant differences between the < 40 and 80+ age groups in both sleep disturbance and physical function. For sleep disturbance, the mean T-score for < 40 was 59.59, and the mean T-score for 80+ was 52.93, demonstrating an average difference of 6.66 (0.67 standard deviation [SD]) between the two age groups. Likewise, for physical function, the mean T-score for < 40 was 37.99, and the mean T-score for 80+ was 34.06, meaning on average a difference of 3.93 (0.40 SD). This resulted in an MCID greater than 3. In addition, Welch’s t-test was performed to compare the age groups. In Table 2, we show a 95% confidence interval between the < 40 and 80+ age groups in sleep disturbance of between 5.95 and 7.94, with a p value of 5.58E–78. For physical function, the 95% confidence interval between the two age groups is 3.73 to 4.75, with a p value of 1.77E–54.
Overall, the PROMIS-29 showed great generalizability across a diverse cohort, with fairly homogeneous findings. Patients were examined in clinics across the United States, spanning previous treatments, medications, surgeries, and diagnoses. The HUI-3 assessment of the eight health status dimensions on a 5- or 6-point scale (normal to highly impaired), namely vision, hearing, speech, ambulation, dexterity, emotion, and cognition, was assessed based on age and gender and demonstrated no statistically significant differences, with mean scores of 0.67, based on assessment from Welch’s t-test.
There are several limitations to this study. As these assessments were collected prospectively for those new patients entering into pain practice, there may have been selection bias regarding those new patients that completed the assessment. Further, it must be appreciated that there are many diagnoses and pain qualifiers that differ for pain therapies, including duration of pain, location of pain, and previous treatments that are typically representative of those patients seeking care in these centers, although interestingly, the results are very homogeneous.
Conclusion
Patient outcome assessment is changing, focused on the complete survey of patient well-being using universally relevant measures rather than linear scores. This data set is the first published normative data set describing the PROMIS-29 chronic pain population referred to chronic pain centers in the United States in a multicenter, retrospective fashion spanning over 2 years, defining a fairly homogeneous chronic pain population as measured by the PROMIS-29 and its calculated representations, across the seven domains. Future study is needed to fully appreciate the implications of the norms of this population, which is descriptively more homogeneous than expected.
References
Pope JE, Fishman M. Redefining success: longitudinal patient reported outcome measures and the importance of psychometric testing for optimization in neuromodulation. Neuromodul Technol Neural Interface. 2018. https://doi.org/10.1111/ner.12907.
US Dept of Health and Human Services Pain Task Force. Pain Management Best Practices: Updates, Gaps, Inconsistencies, and Recommendations. 2018.
Deyo RA, Dworkin SF, Amtmann D, et al. Report of the NIH Task Force on research standards for chronic low back pain. J Pain. 2014;15(6):569–85.
Institute of Medicine. Relieving pain in America: a blueprint for transforming prevention, care, education, and research, vol. 26. Washington, DC: The National Academies Press; 2012.
Dworkin RH, Turk DC, Peirce-Sandner S, et al. Research design considerations for confirmatory chronic pain clinical trials: IMMPACT recommendations. PAIN®. 2010;149(2):177–93.
Cella D, Riley W, Stone A, et al. The Patient-Reported Outcomes Measurement Information System (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005–2008. J Clin Epidemiol. 2010;63(11):1179–94.
Hays RD, Spritzer KL, Schalet BD, Cella D. PROMIS®-29 v2.0 profile physical and mental health summary scores. Qual Life Res. 2018;27(7):1885–91.
Pennings JS, Devin CJ, Khan I, Bydon M, Asher AL, Archer KR. Prediction of Oswestry Disability Index (ODI) using PROMIS-29 in a national sample of lumbar spine surgery patients. Qual Life Res. 2019;28(10):2839–50.
Karp JF, Yu L, Friedly J, Amtmann D, Pilkonis PA. Negative affect and sleep disturbance may be associated with response to epidural steroid injections for spine-related pain. Arch Phys Med Rehabil. 2014;95(2):309–15.
Cook KF, Schalet BD, Kallen MA, Rutsohn JP, Cella D. Establishing a common metric for self-reported pain: linking BPI Pain Interference and SF-36 Bodily Pain Subscale scores to the PROMIS Pain Interference metric. Qual Life Res. 2015;24(10):2305–18.
Hartman JD, Craig BM. Comparing and transforming PROMIS utility values to the EQ-5D. Qual Life Res. 2018;27(3):725–33.
Hays RD, Revicki DA, Feeny D, Fayers P, Spritzer KL, Cella D. Using linear equating to map PROMIS® global health items and the PROMIS-29 V2.0 profile measure to the Health Utilities Index Mark 3. Pharmacoeconomics. 2016;34(10):1015–22.
Craig BM, Reeve BB, Brown PM, et al. US valuation of health outcomes measured using the PROMIS-29. Value Health. 2014;17(8):846–53.
Deyo RA, Ramsey K, Buckley DI, et al. Performance of a patient reported outcomes measurement information system (PROMIS) short form in older adults with chronic musculoskeletal pain. Pain Med. 2016;17(2):314–24. https://doi.org/10.1093/pm/pnv046.
Heros RD, Fishman MA, Pope JE, Falowski SM, Patterson DG. Pain impact scores are an effective predictive tool for neurostimulator trial success: initial results of the STIMPACT study. In: NANS Annual Meeting. Las Vegas; 2020.
Horsman J, Furlong W, Feeny D, Torrance G. The Health Utilities Index (HUI®): concepts, measurement properties and applications. Health Qual Life Outcomes. 2003;1:1–13.
Feeny D, Furlong W, Torrance GW, et al. Multiattribute and single-attribute utility functions for the Health Utilities Index Mark 3 system. Med Care. 2002;40(2):113–28.
Acknowledgements
The authors would like to thank the efforts of Chris Hanes for his significant work and statistical analysis on the project.
Funding
No funding or sponsorship was received for this study or publication of this article
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Dr. Pope is a consultant for Abbott, Medtronic, Flowonix, Ethos, PainTeq, Thermaquil, Vertos, Boston Scientific, Saluda, SpineThera, SPR Therapeutics, WISE, Stimgenics, and Aurora Spine. He has equity in Stimgenics, SPR Therapeutics, SpineThera, Thermaquil, Vertos, Neural Integrative Solutions, AGR, PainTeq, and Celéri Health, and his institution receives research grants from Painteq, Boston Scientific, Abbott, Medtronic, Saluda, Vertos, Ethos, Flowonix, and AIS. Dr. Fishman is a consultant to Abbott, Biotronik, Braeburn, Cornerloc, Medtronic, Thermaquil, and Nevro. His institution received research grants and payments from Abbott, Biotronik, Lumbrera, Medtronic, Stimgenics, Thermaquil, and Vertiflex. He has equity in Celéri Health and Thermaquil. Dr. Fishman’s spouse is an employee of Globus Medical. Dr. Chakravarthy is a consultant to Abbott, Bioness, Medtronic, Boston Scientific, MedinCell, Omnia Medical, Saluda Medical, SPR Therapeutics. He has stock options in Higgs Boson Health, Nalu Medical, and Oska Wellness. He is founder/co-founder of Douleur Therapeutics, Newrom Biomedical, Stimlock, Innovate Digital Ventures. His institution receives research grants from Boston Scientific, Abbott, Medtronic, Bioness. Dr. Hanes is a consultant for Abbott and Medtronic and has equity in Celéri Health. Dr. Gerling is a consultant for RCI Implants, Wolf Endoscopic, and Integrity Implants; and has intellectual property with Integrity Implants. Dr. Heros is a consultant for Abbott and Boston Scientific and performs research with Abbott and Nevro. Dr. Falowski is a consultant for Abbott, Medtronic, Boston Scientific, and Vertos, has equity in CornerLoc, PainTeq, Saluda, SpineThera, Thermaquil, SPR therapeutics, Stimgenics, AGR, Celéri Health, and Neural Integrative Solutions, and performs research with Abbott, Medtronic, Boston Scientific, Saluda, Biotronik, and Stimgenics. Dr. Deer is a consultant for Abbott, Axonics, Flowonix, Saluda Medical, Vertos, SpineThera, Nalu, Medtronic, Nevro, SI Bone, Stimgenics, SPR Therapeutics, Cornerloc Boston Scientific, PainTeq, and Vertiflex. Dr. Deer is a member of the advisory board for Abbott, Vertos, Flowonix, Nalu, SPR Therapeutics, and Vertiflex. Dr. Deer has equity options in Bioness, Vertiflex, Axonics, Vertos, SpineThera, Saluda Medical, Nalu, Cornerloc, PainTeq, and SPR Therapeutics. He is a research consultant for Abbott, Vertos, Mainstay Medical, Saluda, SPR Therapeutics, Boston Scientific, and Vertiflex. Dr Deer has a patent pending for the DRG paddle lead with Abbott. Dr. Shah, Dr. Orhurhu, and Dr. Urits have no disclosures.
Compliance with Ethics Guidelines
All participants were recruited after obtaining a waiver of consent and a full waiver of HIPAA authorization through the WIRB under Common Rule 45 CFR 46.116. The IRB was happy to accept the decision of WIRB without taking any further determinations.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Pope, J.E., Fishman, M., Chakravarthy, K. et al. A Retrospective, Multicenter, Quantitative Analysis of Patients’ Baseline Pain Quality (PROMIS-29) Entering into Pain and Spine Practices in the United States (ALIGN). Pain Ther 10, 539–550 (2021). https://doi.org/10.1007/s40122-021-00238-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40122-021-00238-z