Abstract
Fentanyl iontophoretic transdermal system (ITS) [IONSYS®, The Medicines Company, Parsippany, NJ, USA] is a needle-free, patient-controlled, postoperative opioid pain management treatment. It is indicated for the short-term management of acute postoperative pain in adults requiring opioid analgesia in the hospital. The safety and effectiveness of fentanyl ITS for acute postoperative pain management has been demonstrated in a range of surgery and patient types studied in seven phase 3 trials (three placebo-controlled trials and four active-comparator trials). The majority of the patients in the phase 3 trials had undergone either abdominal/pelvic, orthopedic, or thoracic surgery. Consistent with the prescribing information, physicians in clinical practice may treat patients with this system following any type of surgery including those that may not have been included in the phase 3 trials. The purpose of this case series is to illustrate how fentanyl ITS is being utilized for postoperative pain management in real-world clinical practice following a variety of surgeries and in current pain management protocols that may have evolved since the completion of the phase 3 program. There are seven cases from three clinical centers described within this case series, each using fentanyl ITS according to the prescribing information. The surgery types included are bariatric (N = 3), prostate (N = 2), colorectal (N = 1), and perirectal abscess drainage (N = 1). A systematic review of each patient chart was conducted via a standardized retrospective assessment by the clinicians who managed each patient. Additionally, each healthcare professional was interviewed regarding their overall experience and key learnings using fentanyl ITS. Overall, fentanyl ITS was effective and well tolerated in these case reports in current-day clinical practice settings. These case studies are informative about fentanyl ITS use shortly after product approval and set the stage for additional clinical research.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The management of acute postoperative pain remains a priority for patients and health care professionals; however, successful postoperative pain management continues to be challenging [1, 2]. Patient-controlled analgesia (PCA) was introduced in the late 1970s to improve postoperative pain management [3, 4]. It has been demonstrated that patients who receive PCA versus non-PCA analgesia have better pain control and are more satisfied with their method of pain control [5, 6].
New approaches, such as multimodal analgesia and enhanced recovery after surgery (ERAS) protocols, have been introduced to improve pain management and patient outcomes in the postoperative setting. At the same time, patients who are presenting for surgery have more co-morbidities (e.g., obesity) that make surgery and postoperative care more challenging. In addition, the US population is aging, and patients frequently have chronic concomitant illness. Opioids remain a cornerstone of postoperative pain management; however, their use must be balanced with the essential considerations about safe use and combination with other analgesic medications.
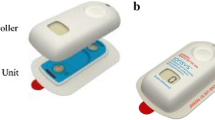
Fentanyl iontophoretic transdermal system (ITS) [IONSYS®, The Medicines Company, Parsippany, NJ, USA] was approved by both the US Food and Drug Administration (April 2015) and the European Medicines Committee (November 2015) for the short-term management of acute postoperative pain in adult patients requiring opioid analgesia in the hospital [7]. Fentanyl ITS can be used for a maximum of 72 h of treatment, while each individual system can be used for 24 h; for example, patients who require 72 h of treatment would require use of three systems consecutively. Fentanyl ITS delivers preprogrammed analgesic doses based on patient control. As with intravenous patient-controlled opioid analgesia, fentanyl ITS is to be used only in patients who are alert enough, have appropriate respiratory function (e.g., high oxygen saturation levels, no hypoventilation), and have adequate cognitive ability to understand the directions for use. Additionally, per the Prescribing Information, fentanyl ITS is to be used after patients have been titrated to an acceptable level of analgesia, typically using alternate opioid analgesics. While in clinical development, fentanyl ITS was studied in seven phase III placebo and active-controlled clinical trials [8–14]. In the active-comparator trials, fentanyl ITS was comparable to morphine IV PCA in terms of pain-control efficacy and safety and was superior for ease of care and nursing and patient satisfaction [8, 10–12].
The objective of this case series was to examine the “real-world” usage of the recently approved fentanyl ITS. It is important to note that, while this case series includes an evaluation of surgical types that would have fit the clinical study inclusion/exclusion criteria but may not have been specifically enrolled in the original clinical trials (i.e., bariatric surgery), fentanyl ITS is indicated across surgery types, and all cases were managed consistent with the Prescribing Information. This case series provides an opportunity to review experiences of early users of fentanyl ITS across a variety of surgical and patient types in a clinical practice setting.
Methods
A systematic retrospective review of each patient chart using a predefined set of variables was performed via a standardized case assessment form, with the data collected by interviewing the treating clinician(s). Additionally, each health care professional (HCP) being interviewed was asked an additional set of questions surrounding treatment decisions and clinical insights, and those findings are also summarized. All data were de-identified in the collection process. Sites were selected who had clinical experience with fentanyl ITS following approval and representing different surgery types. The sites selected the cases to be included in this case series.
Compliance with Ethics Guide
Data collected were all part of routine medical practice and documented in the patient medical records. Data were collected retrospectively by reviewing the charts, and were all de-identified. Therefore, Institutional Review Board approval was not obtained.
Results
This report includes seven cases from three sites. The focus of this case series is postoperative pain management using fentanyl ITS in the hospital setting, with use consistent with the Prescribing Information. The surgery types included are bariatric (N = 3) [Forest Health Medical Center, Ypsilanti, Michigan], prostate (N = 2) [Universitätsklinik Gießen und Marburg GmbH – Standort Marburg, Germany], colorectal (N = 1) [Landmark Medical Center, Woonsocket, RI], and perirectal abscess drainage (N = 1) [Landmark Medical Center, Woonsocket, RI]. The clinical course for each case is described in the narratives below, which are complemented by Table 1, which lists the details of each case side-by-side.
Bariatric
Case 1
A 64-year-old female patient (BMI 37 kg/m2) underwent a laparoscopic sleeve gastrectomy and repair of a paraesophageal hernia. She previously had a failed adjustable gastric band and the band and port were removed during this surgery. Her medical history included knee osteoarthritis, hypertension, and hypercholesterolemia. She had a history of obstructive sleep apnea. She previously had undergone bilateral total knee replacement surgery. She had no preoperative history of opioid tolerance. Fentanyl ITS was applied to the chest once the patient was on the medical-surgical unit mid-morning and was removed the following day (also mid-morning) just prior to a fluoroscopy of the upper gastrointestinal tract. The patient self-administered 34 doses of fentanyl over 24 h. She did not require any supplemental opioid medication in conjunction with fentanyl ITS. No concomitant medication was given for opioid-related adverse events including no signs of clinical relevant respiratory depression. The patient was transitioned to PO hydrocodone/acetaminophen and discharged on that regimen. The patient was very satisfied with fentanyl ITS, describing it as easy-to-use and preferable compared to her previous experience with IV PCA.
Case 2
A 45-year-old female patient (BMI 58 kg/m2) underwent a laparoscopic sleeve gastrectomy. Her medical history included osteoarthritis and obstructive sleep apnea. Surgical history included three Cesarean sections. She had no preoperative history of opioid tolerance. Fentanyl ITS was applied to the chest once the patient arrived on the medical-surgical unit late-morning and was removed the following day (early morning) just prior to a fluoroscopy swallowing study of the upper gastrointestinal tract. The patient self-administered 12 doses of fentanyl over 20 h. This patient reported no pain other than when walking, and consistently reported being comfortable at rest. The patient did not require any breakthrough medication, although perhaps could have benefited from a dose in conjunction with a single pain score of 7 out of 10 on a numeric rating scale (NRS). The patient had nausea on postoperative day (POD) 1, and a single dose of ondansetron 4 mg IV was administered approximately 2 h before the system was removed. There were no signs of respiratory depression. The patient was very satisfied with the pain control while receiving fentanyl ITS, noting that the pain control improved over time and that the system was very easy to use. However, it is important to note that this patient did require some additional training at the beginning of treatment to ensure that the patient “double-clicked” the device to activate a dose, but once that was completed she had no issues with using the system. The patient was discharged on PO hydrocodone/acetaminophen. The treating clinicians interpreted that this patient’s success with fentanyl ITS showed that effectiveness of the system was not related to BMI, considering the particularly high BMI (58 kg/m2) in this patient.
Case 3
A 42-year-old female patient (BMI 45 kg/m2) underwent a laparoscopic sleeve gastrectomy. Her medical history included obstructive sleep apnea and GERD. She had no preoperative history of opioid tolerance. Fentanyl ITS was applied to the chest once the patient was on the medical-surgical unit early afternoon and removed the following day (mid-morning) just prior to a fluoroscopy of the upper gastrointestinal tract. The patient administered 67 doses of fentanyl over 20 h. The patient did not require any breakthrough medication. No concomitant medication was given for opioid-related adverse events and there were no signs of respiratory depression. The patient was discharged on PO hydrocodone/acetaminophen. The patient was very satisfied with the pain control while receiving fentanyl ITS, and she reported that the system was very easy to use.
Abdominal: Colorectal and Perirectal
Case 4
A 71-year-old female patient (BMI 30 kg/m2) with acute diverticulitis underwent an elective laparoscopic, robotic-assisted low anterior resection (partial colon resection). She had a 14-year history of episodes of acute diverticulitis with hospitalizations, each managed non-operatively. Her medical history included diabetes, cardiovascular disease, chronic obstructive pulmonary disease, gastroesophageal reflux disease (GERD), depression, and anxiety. She had no prior surgeries. In the post-anesthesia care unit (PACU), she received 75 mcg of IV fentanyl and 1 g of IV acetaminophen for pain management. She was then transferred to the medical-surgical unit. She received standard bolus doses of morphine sulfate PRN and 1 g of IV acetaminophen every 6 h for pain management. The usual standard of care (SOC) in the hospital was IV PCA; however, there had been a shortage of medication cartridges and the hospital had switched to bolus doses of opioids for pain management. Her pain was not well controlled with this regimen. Due to her medical history, she was also being monitored for cardiovascular events. On post-op day (POD) 1, she had an asymptomatic elevated troponin. On POD 2 she had a cardiac catheterization that showed an occluded right coronary artery. The patient was diagnosed with a silent myocardial infarction (MI). Shortly thereafter, to achieve control of her postsurgical pain, bolus doses of morphine sulfate and IV acetaminophen were discontinued and fentanyl ITS (which had just become available in this hospital) was initiated for pain management on POD 3. At the time that fentanyl ITS was initiated, her pain score was 8 out of 10. Shortly after initiation of fentanyl ITS, her pain scores decreased and ranged between 3 to 5 during fentanyl ITS treatment. Fentanyl ITS was administered for 3 days (POD 3 to 6; total <72 h) and then discontinued; the three devices were applied to the upper arm on alternate sides. At that point, she was transitioned to oral (PO) hydrocodone/acetaminophen. The patient received no additional opioids while on fentanyl ITS. Throughout her stay, her diet was uneventfully advanced from clear liquids to full liquids, and then solids. There was some slight skin redness where the system had been applied, but it resolved without intervention by the next day. On POD 7 she underwent a percutaneous coronary intervention for further work-up of her silent MI. She was discharged on POD 8. The patient’s general impression of fentanyl ITS was that she was very happy with the method of pain control.
Case 5
A 63-year-old male patient (BMI 28 kg/m2) with a painful perirectal abscess (right posterolateral, multiloculated) was seen in the emergency room and underwent emergent surgery that evening, which involved an incision and drainage of the abscess with 150 ml of pus drained. He had a history of diabetes and thyroid disease. In the PACU, he received 100 mcg of fentanyl. He was transferred to the medical-surgical unit where the patient was trained on fentanyl ITS use and the system was applied to the upper arm. No other pain medications were used. Although the skin was normal, the system initially did not have good contact and then was successfully readjusted and adherence was then fine. Once the system was readjusted, his pain level quickly improved. The patient used two fentanyl ITS systems. On POD 1, the patient administered 19 doses of fentanyl; the number of doses decreased on POD 2, and the fentanyl ITS was used for most of that day and then discontinued. There was some slight skin redness at both administration sites where the system had been applied, but resolved without intervention within 24 h. The patient was then transitioned to PO hydrocodone/acetaminophen. On POD 4, the patient was discharged. The patient reported that he was very satisfied with the pain control that the system provided once it was adhered to his skin.
Prostate
Case 6
A 61-year-old male patient (BMI 28 kg/m2) with prostate cancer underwent a laparoscopic, robotic-assisted prostatovesiculectomy. He had a history of hypertension and hypercholesterolemia. Metamizole 1.25 g IV was given as a prophylactic non-opioid analgesic for postoperative pain. No analgesic medication was administered in the recovery room; however, the patient did receive 75 mcg of clonidine. Fentanyl ITS was applied at the upper left arm (right-handed patient) and the patient self-administered 41 doses. The patient received two IV pushes of piritramide 75 mg for breakthrough pain (a synthetic opioid analgesic with approximately two-thirds of the analgesic potency of morphine). In addition, 4 g of metamizole was used as part of a multimodal analgesia plan. Once the patient discontinued fentanyl ITS, he was transitioned to PO morphine 10 mg PRN. The patient experienced nausea and vomiting approximately 2 h after initiating fentanyl ITS; the nausea was associated with the IV administration of piritramide and was successfully treated with metoclopramide and granisetron. There were no skin irritations or other systemic side effects reported. The patient was very satisfied with the ease-of-use of the fentanyl ITS system. The patient indicated that he would like to use the same pain management regimen in the future if he ever needed another surgery.
Case 7
A 67-year-old male patient (BMI 25 kg/m2) with benign prostatic hypertrophy underwent open suprapubic resection of the prostate gland. Prior to surgery, he had urinary retention due to the hypertrophy that resulted in acute kidney failure requiring hemodialysis. He had been in the ICU due to substantial renal fluid loss (>30 l per day). He also had a history of hypertension and hypercholesterolemia. He was not a candidate for etoricoxib or morphine due to its active metabolite, considering the acute kidney failure. Metamizole IV was given as a prophylactic non-opioid analgesic for postoperative pain. The patient was titrated to comfort with 10 mg piritramide (synthetic opioid); then fentanyl ITS was applied to the upper left arm (right-handed patient) for 24 h and the patient administered 47 doses. No breakthrough opioid medication was required during the fentanyl ITS dosing. The patient received concomitant metamizole 4 g over the first 24 h. Once the patient discontinued fentanyl ITS he was transitioned to PO oxycodone PRN and metamizole. There was no skin irritation or other systemic side effects reported. The patient found the system very easy to use and was very satisfied with it for analgesia.
Clinical Practice Insights and Helpful Hints
Ease-of-Use
Overall, the clinicians interviewed felt that fentanyl ITS was easy to use for their patients. The patients in this case series consistently reported to their clinician that the product was easy to use. Nurses expressed relief at not having to program PCA settings or manage an additional IV line (or any IV line). One site also indicated that the use of fentanyl ITS with its fixed dosing is an asset as it eliminated unnecessary programming and variability in postoperative pain management. In addition, the physical therapists (physiotherapists) managing the prostate surgery patients noted that mobilizing patients was much easier without an IV line. Fentanyl ITS also reduced issues with IV access and workload associated with IV PCA devices.
Training
The sites emphasized the importance of staff training on how to manage the system. For example, at the site where the bariatric surgeries are conducted, all clinical team members (not limited to the physicians and nurses) who see these patients are trained on fentanyl ITS which allows them to reinforce the training when they see the patient. Also, the center now places a sign above each patient’s bed that says “IONSYS in place” as a reminder for the clinical staff to reinforce the key training points with the patients.
Patient Selection
The fentanyl ITS development program enrolled a broad range of surgery types. The labeled indication includes patients with moderate-to-severe postoperative pain without specification as to surgery type. However, when the phase 3 clinical studies were conducted, bariatric surgery was relatively uncommon. In this case series, three patients had bariatric surgical procedures with BMI ranging from 37 to 58 kg/m2. Fentanyl ITS was safe and effective for all three, supporting previous data [15] that higher BMI patients with postoperative pain can be appropriately treated with the system. Moreover, consistent with the characteristics of this patient population, all three bariatric patients had a history of sleep apnea and none developed signs of clinically relevant respiratory depression while fentanyl ITS was administered. Clinicians must routinely balance the benefit-risk of opioid postoperative pain management with the potential for respiratory depression.
Several of the patients in these case series had significant medical histories and co-morbidities. Two of the patients had a complicated hospital course prior to fentanyl ITS treatment: renal failure and silent MI. These experiences build upon the phase 3 study results and further show that fentanyl ITS can be used for pain management in more complex postoperative patients, within the parameters of the Prescribing Information.
Six of the seven cases were elective or semi-elective surgeries; the perirectal abscess case was an emergent case performed at night. This case was also managed successfully with fentanyl ITS and showed that training the staff in advance can enable appropriate use of the device in surgeries that are not pre-planned.
Titration to Comfort before Initiating Fentanyl ITS
It was repeatedly noted that patients responded better to fentanyl ITS if they were titrated to comfort prior to initiation, which is consistent with the Prescribing Information for fentanyl ITS. These clinicians initiated fentanyl ITS on the medical-surgical unit because they found that patients in the PACU may be too sedated to understand or operate the system, although the system can be applied in the PACU or on the patient floor.
Breakthrough Pain
In this series, most patients did not require additional analgesic medication during fentanyl ITS administration; however, the sites reported that it is worthwhile to have PRN orders of opioids and/or NSAIDs to assist with breakthrough pain in case it is required. However, it is important to remember that the concomitant use of fentanyl ITS with other central nervous system depressants can increase the risk of respiratory depression and therefore the clinicians must monitor patients even more closely if using a concomitant opioid.
Medication after Fentanyl ITS
Once patients were able to progress beyond parenteral opioids, they were easily transitioned from fentanyl ITS to the clinician’s standard pre-discharge oral analgesia regimen. Most were switched to the combination of hydrocodone and acetaminophen. This allowed fentanyl ITS to be readily incorporated into each center’s standard-of-care pain management regimen.
Application Site
When applying fentanyl ITS, it is important to press and hold the system firmly in place, with the sticky side down, onto the patient’s skin for at least 15 s. However, occasionally, the system may loosen from the skin and if this occurs, it should be secured to the patient’s skin by pressing the edges with fingers or securing with a non-allergenic tape to make sure that all edges have complete contact with the skin. It is important to remember not to place tape over the button, light, or digital display.
Application Site Skin Reactions
Consistent with the fentanyl ITS Prescribing Information, some patients experienced redness at the application site. In this case series, none of the site reactions were severe; none required intervention or discontinuation of the product. It is important to train and alert the clinical staff that this can be an adverse event associated with fentanyl ITS use; however, it is typically self-limited.
Discussion
Adequate analgesia in the postoperative setting is an integral part of patient recovery following surgery. Opioids continue to be a valuable component of multimodal treatment. As with all opioid medications, clinicians using fentanyl ITS should carefully select, monitor, and manage their patients for opioid-related adverse effects.
This report describes seven cases in clinical practice using fentanyl ITS for the management of post-operative pain in the acute care postoperative hospital setting. All patients described in this cases series were prescribed fentanyl ITS consistent with the Prescribing Information. In all cases, fentanyl ITS was effective, easy to use, and well tolerated; skin reactions were reported in two of the patients and were self-limited. This series details the differences in patient experiences based upon surgery type and shows the versatility of the fentanyl ITS system. For example, the laparoscopic bariatric surgery patients required 24 h or less of fentanyl ITS before conversion to PO opioids. Whereas, the patients who underwent colorectal surgery used the system for up to 3 days.
The main limitations of this case series are that the data was collected retrospectively in a small number of patients. However, the questionnaire was standardized and attempted to provide consistent information. The data collected in this case series were consistent with the phase 3 trials as well as the Prescribing Information and should provide treating clinicians with added confidence about the overall efficacy and safety of fentanyl ITS.
Conclusions
The clinical findings from the case studies are consistent with the data from the randomized clinical trials for fentanyl ITS in the management of postoperative pain. This case series adds to the clinical experience with fentanyl ITS and gives specific details of its use in recent patients who have undergone bariatric, colorectal, perirectal, or prostate surgeries. The efficacy and safety profile observed in these case studies was consistent across surgery types and patient characteristics. Perhaps most importantly, these reports support that the system can be practically integrated into current pain management regimens, including multimodal therapy and the subsequent switch to oral analgesics. Fentanyl ITS is a new patient-controlled option for postoperative pain management across a range of complex surgery and patient types, that may enable postoperative recovery.
References
Tan M, Law LS, Gan TJ. Optimizing pain management to facilitate enhanced recovery after surgery pathways. Can J Anaesth. 2015;62(2):203–18.
Tawfic QA, Faris AS. Acute pain service: past, present and future. Pain Manag. 2015;5(1):47–58.
Forrest WH Jr, Smethurst PW, Kienitz ME. Self-administration of intravenous analgesics. Anesthesiology. 1970;33(3):363–5.
Sechzer PH. Studies in pain with the analgesic-demand system. Anesth Analg. 1971;50(1):1–10.
McNicol ED, Ferguson MC, Hudcova J. Patient controlled opioid analgesia versus non-patient controlled opioid analgesia for postoperative pain. Cochrane Database Syst Rev. 2015;6:CD003348.
Bainbridge D, Martin JE, Cheng DC. Patient-controlled versus nurse-controlled analgesia after cardiac surgery—a meta-analysis. Can J Anaesth. 2006;53(5):492–9.
IONSYS (fentanyl iontophoretic transdermal system), CII Prescribing Information. http://www.ionsys.com/pdfs/ionsys-prescribing-information.pdf. Accessed 16 July 2016.
Viscusi ER, Reynolds L, Chung F, Atkinson LE, Khanna S. Patient-controlled transdermal fentanyl hydrochloride vs intravenous morphine pump for postoperative pain: a randomized controlled trial. JAMA. 2004;291(11):1333–41.
Viscusi ER, Reynolds L, Tait S, Melson T, Atkinson LE. An iontophoretic fentanyl patient-activated analgesic delivery system for postoperative pain: a double-blind, placebo-controlled trial. Anesth Analg. 2006;102(1):188–94.
Minkowitz HS, Rathmell JP, Vallow S, Gargiulo K, Damaraju CV, Hewitt DJ. Efficacy and safety of the fentanyl iontophoretic transdermal system (ITS) and intravenous patient-controlled analgesia (IV PCA) with morphine for pain management following abdominal or pelvic surgery. Pain Med. 2007;8(8):657–68.
Grond S, Hall J, Spacek A, Hoppenbrouwers M, Richarz U, Bonnet F. Iontophoretic transdermal system using fentanyl compared with patient-controlled intravenous analgesia using morphine for postoperative pain management. Br J Anaesth. 2007;98(6):806–15.
Hartrick CT, Bourne MH, Gargiulo K, Damaraju CV, Vallow S, Hewitt DJ. Fentanyl iontophoretic transdermal system for acute-pain management after orthopedic surgery: a comparative study with morphine intravenous patient-controlled analgesia. Reg Anesth Pain Med. 2006;31(6):546–54.
The Medicines Company: Data on file. 2016.
Chelly JE, Grass J, Houseman TW, Minkowitz H, Pue A. The safety and efficacy of a fentanyl patient-controlled transdermal system for acute postoperative analgesia: a multicenter, placebo-controlled trial. Anesth Analg. 2004;98(2):427–33.
Viscusi ER, Siccardi M, Damaraju CV, Hewitt DJ, Kershaw P. The safety and efficacy of fentanyl iontophoretic transdermal system compared with morphine intravenous patient-controlled analgesia for postoperative pain management: an analysis of pooled data from three randomized, active-controlled clinical studies. Anesth Analg. 2007;105(5):1428–36.
Acknowledgements
The data collection, writing of this manuscript, and publication charges were supported financially by The Medicines Company. Medical writing assistance was provided by Starr Lee Grundy of SD Scientific, Inc. This support was funded by The Medicines Company.
All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Disclosures
Steven Poplawski, Matthew Johnson, and Tilo Koch have nothing to disclose. Philip Philips serves on The Medicines Company Speaker’s Bureau. Leopold HJ Eberhart is a consultant for The Medicines Company. He has received lecture fees from Baxter, Grünenthal, Fresenius, and TEVA-ratiopharm. Loretta M. Itri is an employee of The Medicines Company.
Compliance with Ethics Guide
Data collected were all part of routine medical practice and documented in the patient medical records. Data were collected retrospectively by reviewing the charts, and were all de-identified. Therefore, Institutional Review Board approval was not obtained.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article go to www.medengine.com/Redeem/9417F060778E692D.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Poplawski, S., Johnson, M., Philips, P. et al. Use of Fentanyl Iontophoretic Transdermal System (ITS) (IONSYS®) in the Management of Patients with Acute Postoperative Pain: A Case Series. Pain Ther 5, 237–248 (2016). https://doi.org/10.1007/s40122-016-0061-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40122-016-0061-2