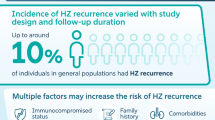
Abstract
Introduction
The objective of this work was to summarize the incidence of herpes zoster (HZ) complications in different populations.
Methods
Systematic literature review of PubMed, Embase, and Virtual Health Library records between January 1, 2002 and October 20, 2022 using search strings for HZ, complications, and frequency measurements.
Results
The review included 124 studies, most conducted in the general population (n = 93) and on individuals with comorbidities (n = 41) ≥ 18 years of age. Most studies were conducted in Europe (n = 44), Asia (n = 40), and North America (n = 36). Postherpetic neuralgia (PHN) was the most studied neurological complication. Variable relative PHN incidence was found in the general population (2.6–46.7%) or based on diagnosis: immunocompromised (3.9–33.8%), depression (0–50%), and human immunodeficiency virus (HIV) (6.1–40.2%). High incidence rates were observed in hematological malignancies (HM) and solid organ malignancies (132.5 and 93.7 per 1000 person-years, respectively). Ocular complications were frequently reported with herpes zoster ophthalmicus (HZO). The relative incidence (incidence rate) of HZO in the general population was reported as 1.4–15.9% (0.31–0.35 per 1000 person-years). High relative incidence was observed in HIV (up to 10.1%) and HM (3.2–11.3%). Disseminated HZ was the most frequently reported cutaneous complication. The relative incidence of disseminated HZ was 0.3–8.2% in the general population, 0–0.5% in the immunocompetent, and 0–20.6% in patients with comorbidities. High relative incidence was reported in HM and solid organ transplant (up to 19.3% and 14.8%, respectively).
Discussion
Most reported complications were neurological (n = 110), ocular (n = 48), and cutaneous (n = 38). Few studies stratified complications by age or gender (or both). Incidence appeared higher in select immunocompromised populations. Higher incidence was associated with older age in several studies; the general association with gender was unclear.
Conclusions
Variable incidence of HZ complications was reported by population subgroup. Further research is required to quantitatively analyze incidence by age, gender, and location.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
The burden of herpes zoster complications is poorly studied, with limited epidemiological information, except for postherpetic neuralgia. This impairs the quantitative analysis of complications incidence to be used in health economic and outcomes studies. |
A systematic literature review was conducted to summarize the incidence of herpes zoster complications in different populations between 2002 and 2022. |
What was learned from the study? |
Among 124 retrieved studies, most frequently reported complications were of neurological (n = 110), ocular (n = 48), and cutaneous (n = 38) origin. |
Postherpetic neuralgia, ocular complications associated with herpes zoster ophthalmicus, and disseminated zoster were the most reported complications. |
High incidence of herpes zoster complications was frequently reported in populations with immunocompromising conditions; however, intra-study comparisons between the general or immunocompetent population and immunocompromised subgroups were mostly not statistically tested. |
Inter-study variability of outcomes was high as a result of varying study design, setting, and sample sizes. |
Introduction
Herpes zoster (HZ), commonly referred to as shingles, is caused by reactivation of latent varicella-zoster virus (VZV). HZ frequently presents as a painful debilitating rash, including skin inflammation and blisters, and sometimes causes scarring and permanent pigment changes. Treatment of HZ with oral antiviral therapy is recommended within 72 h of rash onset, and usually given for 7 days in the absence of HZ complications. However, substantial pain and discomfort may be prolonged for weeks, months, or even years in severe cases, diminishing patient quality of life (QoL) [1].
The incidence of HZ ranges between 5.2 and 10.9 cases per 1000 person-years [2]. Anyone previously infected with VZV is at risk of developing HZ. The frequency and severity of HZ episodes increase with age as a result of age-related decline in immunity [3, 4]. Among adults who reach 85 years of age, approximately half will likely have suffered at least one episode of HZ [5]. Besides age, people with immunosuppression from any cause, including hematologic malignancies, human immunodeficiency virus (HIV), and immunosuppressive medications, have a higher risk of HZ [6]. Other commonly identified risk factors include female sex, white race, and presence of comorbid conditions [7,8,9].
HZ can give rise to various complications, some of which are serious or even life-threatening. The most common HZ complication is postherpetic neuralgia (PHN), which is frequently defined as chronic pain persisting or appearing 3 months after initial rash detection or HZ diagnosis. It is associated with an average worst pain score ≥ 3 on the Likert scale, as measured by the Zoster Brief Pain Inventory (ZBPI) [10]. HZ and PHN have been shown to adversely affect healthy aging and QoL [11]. Other common HZ complications include ocular, neurological, and cutaneous conditions [12]. Some of these consequently cause severe health impact, such as Ramsay Hunt syndrome, which can lead to facial paralysis, or delayed contralateral hemiparesis [13]. Various other chronic infections are important in clinical practice [14,15,16,17,18,19,20].
In general, the incidence and burden of HZ complications other than PHN are poorly studied and reliable epidemiological information is limited. Consequently, the incidence and burden of these conditions in many regions are unknown. This limits reliable modelling of complications in health economic evaluations examining the public health impact of HZ on diverse subject groups.
The objective of this systematic literature review (SLR) was to identify and summarize the incidence of HZ complications in different populations.
Methods
Objectives
The main objective of the present SLR was to collect and consolidate information on the incidence of HZ complications in adults ≥ 18 years of age by complication type (and subtype) and for a wide range of populations generally classified as (a) general, (b) healthy (immunocompetent), and (c) with selected comorbidities/underlying conditions (including immunocompromising and/or immunosuppressive ones). The secondary objective was to detect differences in the incidence of complications by (a) age and (b) sex, if supported by available data.
Specific to the main objective, a list of primary HZ complications was developed (see Supplementary material, Appendix SI, Table SI-1); additional HZ complications were extracted as reported in the included body of literature.
Design
SLR design followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and the Cochrane Collaboration guidelines [21, 22]. All articles retrieved from the electronic databases were screened against predefined eligibility criteria (detailed in the Supplementary material, Appendix SI, Table SI-2) using a modified PICO (population, intervention, comparator, outcomes) framework. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. Due to this reason an ethics committee approval was not required.
Electronic Databases
The search was conducted on records published between January1, 2002 and October 20, 022 in Medline (accessed via PubMed), Embase, and Virtual Health Library, combining keywords (free-text terms) and Medical Subject Headings (MeSH terms) specific to each database. The start date was chosen in alignment with a recent SLR of HZ incidence worldwide [2] resulting into an extensive search process of manageable size. The exact search strings are documented in the Supplementary material, Appendix SII, Tables SII-1 to SII-3.
Study Selection, Data Extraction, and Quality Control
Letters to the editor, editorials or comments, case reports, and SLRs or meta-analyses were excluded from this review. Additional exclusion criteria encompassed small sample size (fewer than 100 HZ cases in the case of relative incidence, a population sample size with underlying disease lower than 100 in the case of cumulative incidence or incidence rate), animal studies, purely pediatric studies (< 18 years of age), studies conducted in non-representative populations (e.g., military personnel, pregnant women, etc.), phase I/II/III clinical trials, modelling studies, studies on the occurrence of HZ after coronavirus disease 2019 (COVID-19) vaccination or on the relation of HZ with COVID-19 infection, and studies reporting on the risk of another disease after HZ (such as stroke, dementia, coronary artery syndrome, or other malignancy). Pediatric zoster [23,24,25,26,27,28], with an incidence reported as declining from 74 to 38 per 100,000 person-years between 2003 and 2014 [29] consistent with increased participation in pediatric varicella vaccination programs, was explicitly not in scope of this review.
A three-step study selection procedure was implemented as detailed in the Supplementary material, Appendix SIII, along with further data extraction and quality control methodology.
Data extraction was guided by the common definitions outlined in the Supplementary material, Appendix SIV. Critical study quality aspects were documented in a data-extraction sheet, including representativeness of the population, definition of HZ cases and its complications, loss to follow-up, misclassification, calculation of incidence, etc. The details are reported in the Supplementary material, Appendix SV.
Results
Out of 3855 unique search hits, 124 articles were included in this review [30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127,128,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150,151,152,153]. Figure 1 gives a schematic overview of the screening and selection procedure (PRISMA flowchart).
Study Characteristics
An overview of the baseline characteristics of the included studies can be found in the Supplementary material, Appendix SV. Most studies were conducted in Europe (n = 44), followed by Asia (n = 40) and North America (n = 36).
Most studies (76%) reported the age of the population under investigation, and most studies included adults ≥ 18 years of age. In 13 studies [44, 50, 59, 104, 106, 108, 114, 117, 126, 129, 142, 144, 145], the study population was ≥ 50 years of age, while one study [97] included only individuals ≥ 60 years of age, and three studies [42, 56, 128] included only individuals ≥ 65 years of age. In one article [66], the study population was ≥ 80 years of age.
The definition of HZ was variable: over 50% of the studies used International Classification of Diseases (ICD) codes or their equivalent solely, while seven studies [37, 42, 48, 50, 92, 103, 106] used antiviral prescription or laboratory tests in combination with ICD codes. One study used a natural language processing software algorithm based on Read codes [67]. In other studies (29%), clinical diagnosis criteria were used to define HZ cases, with seven studies [31, 46, 61, 85, 87, 96, 139] defining HZ cases using a laboratory test or self-reporting in addition to clinical diagnosis [31, 46, 61, 85, 87, 96, 139]. Only one study each relied solely on laboratory testing [100] or self-diagnosis [84] for case definition.
Most studies (n = 93) reported on HZ complications in the general population and 13 studies [44, 66, 67, 72, 79, 90, 100, 108, 113, 115, 125, 128, 139] reported HZ complications in an immunocompetent population. Populations with various comorbidities—including immunosuppressive or immunocompromising conditions—were examined in 41 studies. Nine studies included a patient population with diabetes mellitus [36, 43, 58, 76, 89, 97, 109, 119, 126], and eight studies [33, 37, 48, 56, 89, 97, 105, 109] included a population with a form of malignancy. Studies were conducted among rheumatic disorder populations [33, 37, 48, 56, 89, 97, 105, 109], individuals having undergone stem cell transplantation [73, 77, 92], and patients with HIV [46, 57, 146]. Five studies collected data on solid organ transplantation (SOT) patients [19, 57, 63, 103, 110]. Three studies [31, 34, 45] focused on patients with chronic obstructive pulmonary disease (COPD), and one study each collected data on patients with inflammatory bowel disease (IBD) [65] and chronic/end-stage renal disease [78].
The incidence of complications was reported as relative incidence (proportion or percentage of HZ cases leading to complications) in most studies (76%). Five studies [125, 131, 136, 145, 147] reported cumulative incidence (number of complication cases per N subjects), and 13 studies [36, 40,41,42, 56, 59,60,61, 70, 82, 92, 98, 101] reported incidence rates (number of complication cases per X person-years). Twelve studies [47, 55, 66, 67, 75, 86, 106, 113, 115, 126, 127, 138] reported incidence of complications in multiple ways, mainly a combination of relative incidence with incidence rate or cumulative incidence.
Overview of Complication Types
Reported complications were categorized into three main types: neurological (including PHN), ocular, and cutaneous. Visceral complications and miscellaneous other were also reported. Table 1 provides a summary by main type, subtype(s), and number of articles included in this review.
Main Results
Cutaneous Complications
The incidence of cutaneous complications is summarized in Table 2. Most frequently reported cutaneous complications were disseminated HZ, bacterial superinfection, pigmentary changes, and scarring.
The relative incidence of disseminated HZ in the general population ranged from 0.3% to 8.2%. The upper bound (8.2%) was reported in the hospital setting with a small-to-medium sample of total HZ cases investigated (n = 355) [135]; other studies reported lower relative incidence of disseminated HZ at 1.5% [123], 2.1% [134], and 2.1% [141], respectively. The incidence rate of disseminated HZ in the general population was reported as 0.04 per 1000 person-years [113]. Relative incidence ranged from 0 to 0.5% in the immunocompetent population, and the incidence rate varied from 0.03 to 0.04 per 1000 person-years. In populations with comorbidities, high relative incidence was reported in patients with solid organ transplantation (SOT) (14.8%) [118] and hematological malignancy (HM) (19.3%) [48]. Two studies [49, 136] presented disseminated zoster incidence results by age with no apparent trend identified.
The relative incidence of bacterial superinfection in the general population was in the range 0.1–38.2%; higher relative incidence was reported in hospital studies (12.5–38.2%) [140, 148, 149]. In patients with HIV, the relative incidence of bacterial superinfection was 3.8–6.3% [31, 34]. One study [53] stratified incidence by age and demonstrated highest incidence in the oldest age group. No data were found by gender.
The relative incidence of other cutaneous complications in the general population ranged from 0.7% to 10.5%. A high percentage of patients with rheumatoid arthritis experienced cutaneous complications (12.3%) [73]. The incidence rate appeared higher in HM (122.8 per 1000 person-years) and SOT (73.6 per 1000 person-years) compared to the general population (6.2 to 7.8 per 1000 person-years). In three studies [50, 62, 82], higher incidence of cutaneous complications was found in older patients and male patients.
Postherpetic Neuralgia (90-Day Definition)
The incidence of PHN is summarized in Table 3. The relative incidence of PHN (90-day definition) in the general population was 2.6–46.7%, with the lowest value of 2.6% reported in a retrospective analysis of a general practitioners’ database [119] possibly influenced by underreporting of less than severe pain [119]. The relative incidence of PHN in the immunocompetent population was 2.4–25.4%, with the lowest incidence of 2.4% reported in a nationwide health insurance research database study [79] using specific ICD9 classification of PHN possibly influenced by undercoding [79]. In a retrospective review of patients with HZ on tumor necrosis factor alpha (TNFα) inhibitors, less than 1% of cases developed PHN [38]. High PHN incidence rates were found in immunocompromised patients (up to 33.8% [115]) and patients with HIV (up to 40.2% [109]). A relative incidence rate of 50% was reported in patients with depression [97], albeit derived from a very small sample of HZ cases observed. PHN incidence rates were high in HM (132.5 per 1000 person-years) [56] and SOM (93.7 per 1000 person-years) [56].
A total of 22 studies stratified incidence by gender; 14/22 reported comparable incidence by gender, either statistically tested [75, 98, 106, 109, 114, 119, 143] or not [38, 43, 48, 61, 85, 128, 152]. Six studies found higher incidence in female patients, four statistically tested [62, 111, 126, 131], two not [101, 130]. One study [97] reported a statistically significant higher incidence in male patients.
PHN incidence increases with age, as evidenced in 28/34 studies, confirmed by a statistically significant result (n = 15) [36, 50, 62, 75, 97,98,99, 106, 111, 119, 125, 129, 130, 138, 143] or no statistical tests performed (n = 13) [48, 52, 61, 64, 79, 85, 101, 116, 127, 128, 131, 151, 153].
Neurological Complications Other than Postherpetic Neuralgia
The incidence of neurological complications other than PHN is summarized in Table 4. The relative incidence of encephalitis or meningitis in the general population ranged from 0.02% to 2.7%, and in immunocompetent individuals from 0.1% to 0.4%. The relative incidence of encephalitis ranged from 0.6% to 1.2% in SCT patients. The relative incidence of combined encephalitis and meningitis/meningoencephalitis ranged from 0 to 3.7% in the general population. The relative incidence of meningoencephalitis in patients with HIV was reported as 2.80% [31]. Patients with HM and SOM had high incidence rates at 13.0 and 6.5 per 1000 person-years, respectively [56]. Two studies [136, 138] reported incidence stratified by age; incidence appeared higher in the older population. Another two studies [113, 152] stratified complications by gender; no clear pattern could be observed.
The incidence of any other neurological complication was reported as 0.6–3.24% in the general and 0.5% [72] in the immunocompetent population, respectively. High relative incidence of neurological complications (10.2–26.3%) was reported in patients with RA. Patients with SOM appeared to have experienced those complications the least (0.4–3.2%).
Higher incidence was found in male patients in one study [82]. Higher incidence was reported in older patients in three studies [51, 63, 82].
Ocular Complications
The incidence of herpes zoster ophthalmicus (HZO) and that of ocular complications are reported in Table 5. The relative incidence of HZO varied widely from 1.4% to 15.9% in the general population. The lowest incidence of 1.4% [114] was reported in a prospective study of 513 patients with HZ.
The incidence rate of HZO in the general population ranged from 0.31 to 0.35 per 1000 person-years. Similar incidence was observed in the immunocompetent population (4.8% and 0.33 per 1000 person-years). High relative incidence was found in HM (3.2–11.3%); in HIV, one study reported 10.1% [34]; however, no ocular complications were recorded elsewhere [72].
Three studies reported incidence on gender stratification, with no differences between male and female patients found in two studies [60, 113], and higher incidence in female patients reported in the third [41]. Higher incidence was seen in older patients, as reported in four studies [41, 60, 138, 150].
The relative incidence of any ocular complication in the general population was 0–4.36%, and the incidence rate was 1.53–3.65 person-years. In the immunocompetent population, the relative incidence of any ocular complication was reported to be between 1.6% and 4.1% [72, 100]; however, another study [110] reported 12.3% based on a sample of 219 patients with HZ. In populations with comorbidities, high relative incidence of any ocular complication was found in HIV (5.7–6.3%) and SCT (0–5.7%). No clear patterns were found in age [49, 53, 55, 62, 82, 136] and gender [82, 152] comparisons.
Visceral Complications
The incidence of visceral (plus other systemic) complications is reported in Table 6. The relative incidence of any visceral complications in the general population was reported at 2.6% [62]. The relative incidence of hepatitis was 0.6%, 0.5%, and 0.8% in the general, immunocompetent, and immunocompromised populations, respectively. The corresponding incidence rates for pneumonia were 1.5%, 0.8%, and 2.5% [115].
Upper respiratory tract infections were reported in 5.6% of patients with HZ [121]; lower respiratory tract and gastrointestinal tract complication rates were 0.95% and 1.1%, respectively [121].
The cumulative incidence of hepatitis in the general population was 0.82/1000 [138].
The relative incidence of visceral zoster in SCT subjects was 1.2% [146].
Miscellaneous Other Complications
The incidence of miscellaneous other complications is reported in Table 7. Varicella-zoster virus (VZV) vasculopathy relative incidence was 0.7%, 0.5%, and 1.0% in the general, immunocompetent, and immunocompromised populations, respectively.
Any Complications
The incidence of any complications is reported in Table 8. The relative incidence of any HZ complication in the general population varied between 13.8% and 32.9%. One study [100] reported the relative incidence in immunocompetent patients at 16.5%. The highest relative incidence of HZ complications was reported in SOT (20–47%), ESRD (37–38.9%), and HIV (28.3–32.3%). Gender comparisons were performed in two studies [49, 128], indicating higher incidence in male patients. Four studies [49, 84, 126, 128] found higher incidence in older age groups.
Antiviral Medication
One study [144] examined the effect of antiviral drugs on HZ-related pain including PHN. The prevalence of PHN at 90 days was 27%, at least as frequent in patients treated by antivirals within 72 h of HZ rash onset as in patients treated later or not treated at all.
Discussion
Summary of Results
Complications arising from VZV reactivation infection were reviewed qualitatively in three major medical literature databases on records published in the English language between 2002 and 2022. A systematic exposition of HZ complications by main type and subtype, as originally scoped in the literature search from expert HZ reviews [13, 110], and validated empirically from the search outcomes, constitutes the main outcome of this work. The SLR included 124 studies, most conducted in the general population (n = 93) and on individuals with comorbidities (n = 41) ≥ 18 years of age, with most studies conducted in Europe (n = 44), Asia (n = 40), and North America (n = 36), and study populations aged ≥ 50 years frequently reported (n = 36).
The results indicate that neurological complications including PHN remain the most frequently reported ones (n = 110), with ocular (n = 48) and cutaneous (n = 38) complications following, and visceral (plus other systemic) HZ complications being less frequently studied and reported. High incidence of complications was observed in immunocompromised patients; mechanisms and possible causal relationships between partially functional immune response and dissemination of HZ infection to the central nervous system [154] or lungs [155] are proposed elsewhere [156].
PHN was the most frequently studied neurological complication. Variable relative PHN incidence was found in the general population (2.6–46.7%) or based on diagnosis: immunocompromised (3.9–33.8%), depression (0–50%), and HIV (6.1–40.2%). High incidence rates were observed in hematological (132.5 per 1000 person-years) and solid organ malignancies (93.7 per 1000 person-years) compared to the general population (0.15–2.1 per 1000 person-years) and the immunocompromised (0.42–0.48 per 1000 person-years).
Ocular complications were frequently reported with HZO. The relative incidence of HZO in the general population varied as 1.4–15.9% and the incidence rate ranged between 0.31 and 0.35 per 1000 person-years. High relative incidence was observed in HIV (up to 10.1%) and hematological malignancies (3.2–11.3%).
Disseminated zoster was the most frequently reported cutaneous complication. The relative incidence of disseminated zoster was 0.3–8.2% in the general population, 0–0.5% in the immunocompetent, and 0–20.6% in populations with comorbidities. High relative incidence was reported in patients with hematological malignancy (up to 19.3%) and patients with solid organ transplant (up to 14.8%).
A limited number of studies stratified complications by age or gender (or both). Higher incidence was associated with older age in several studies; the general association with gender was unclear.
One study [144] reported no significant effect of antiviral drugs on HZ-related pain including PHN. This observation is in line with a meta-analysis [157] of the effect of antiviral treatment on preventing PHN from six clinical trials, determining with high certainty that orally administered aciclovir does not reduce the incidence of PHN significantly, and stating insufficient evidence to determine the effect of other antiviral treatments. An apparent unmet need in managing PHN effectively is discussed elsewhere [158].
Comparisons to Other Reviews
Global systematic literature reviews of HZ complications are scarce; see for example [159], which reported the risk of PHN in the general population between 5% and 32.5% from 49 studies, and the risk of HZO between 10.1% and 14.9% from 4 studies. Regional reviews of the medical literature on the HZ burden of disease have recently included more confined expositions of HZ complications [160, 161], focusing mostly on HZO and PHN. Our review is generally inclusive of work examined therein published in the English language. In the south-east Asia review (72 papers but 22 being case studies) [161], HZ complications were reported in 10.2–21.2% of patients; the relative incidence of PHN varied widely between 6.3% and 50% from nine studies. In the China review [160], the percentage of patients developing PHN varied from 1.85% to 80.2% with a median of 22% based on 39 studies (18 in Chinese, 21 in English). Ophthalmic complications developed in 0.76–26.42% of patients with HZ, infections including pneumonia sepsis and encephalitis/meningoencephalitis in 1.11–4.7%, and dermatological complications including cellulitis and other skin infections in 2.17–12.86% of patients (eight studies in Chinese, six in English). A subset of HZ complications on immunocompromised patients was reviewed in the USA [162]; the risk of developing PHN was reported to be between 6% and 45% across the examined immunocompromising conditions (6–41% in HSCT, 7–45% in SOT, 6–40% in HM), and the risk of disseminated zoster in HSCT ranged from 0 to 32% with a median of 3%.
Limitations
Most studies (n = 83) used a retrospective design. Most retrospective database analyses (n = 53) utilized diagnostic codes to identify HZ and its complications, with the possibility of physician miscoding or undercoding. A few retrospective studies [42, 50, 92, 103] utilized diagnostic codes coupled with antiviral prescriptions, while some [31, 55, 57, 100, 118, 127, 141, 146] were based on clinical diagnoses not accompanied by coding. Two retrospective database analyses [84, 112] were based on self-reported outcomes. Several retrospective studies [61, 62, 64,65,66,67, 70, 72, 106, 107, 119, 121, 137, 147, 152] were performed on general practitioner (GP) or GP/outpatient/primary care databases, with the possibility of selection bias in the form of exclusion of older institutionalized subjects. Selection bias in the form of exclusion of unemployed/uninsured persons from a number of (n = 25) health insurance database analyses [30, 33, 35, 36, 41, 42, 44, 47, 52, 56, 58, 59, 73, 75, 77,78,79,80,81,82, 92, 101, 116, 117, 138] is also possible.
In addition, HZ case definition was frequently not available in the study report (n = 17), nor were specific inclusion (n = 15) or exclusion (n = 64) criteria. Several studies did not provide critical baseline characteristics such as participant age range (n = 31) and gender (n = 35). Most studies (n = 97) did not report on antiviral use for HZ episode treatment.
In populations with comorbidities, the specific conditions were not reported in several studies [44, 64, 95, 106, 111, 114, 116].
The majority of studies were conducted in Europe (n = 44), Asia (n = 40), and North America (n = 36). There were only two studies including data from South America [142, 143]. Multicountry studies [142, 143] did not stratify incidence of complications by country or geographical region.
All outcomes summarized in the present SLR should be placed in perspective under the various methodological and operational study limitations outlined above.
Conclusions
This systematic review synthesized the literature on incidence of HZ complications in adults ≥18 years of age from 124 studies. Complication types were grouped as ocular, neurological, cutaneous, visceral, and miscellaneous other. Neurological complications were the most studied complication type (n = 110), while visceral complications were minimally reported (n = 6). PHN remains the most studied HZ complication.
The incidence of HZ complications was higher in older age groups (although frequently not statistically tested), and it appeared higher in immunocompromised populations compared to the immunocompetent (cell-mediated immunity and VZV reactivation are discussed elsewhere [163]). The relationship between incidence levels in the general population and the immunocompromised was less clear, which may be attributed to varying proportions of immunocompromised individuals in the respective general population samples. Furthermore, HZ complications in immunocompromised populations were reported in only a few studies and the immunocompromising disease or condition was not always clearly defined. In studies examining the burden of HZ by underlying condition/diagnosis, and across many complication types, high incidence of complications was observed in patients with HM, SOT, and HIV. In studies that stratified incidence of complication by gender (n = 41), the association between gender and incidence was unclear.
In general, the incidence of HZ complications could not be sufficiently summarized by age, or gender, as a result of insufficient numbers of studies that provided complete data on these variables. In terms of geographical representation, the present data collection would not be globally representative as most studies were conducted in Europe, Asia, and North America, while there were no studies from Africa.
In short, further research would be required to quantitatively analyze HZ complications incidence by age, gender, and/or location, for effective utilization of aggregated values in health economic and outcomes studies. The collected body of evidence from this work can serve as the starting point towards that end.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available as the study constitutes an SLR of published literature. As such all data can be obtained from the published literature.
References
Katz JCE, Walther RR, Sweeney EW, Dworkin RH. Acute pain in herpes zoster and its impact on health-related quality of life. Clin Infect Dis. 2004;39(3):342–8. https://doi.org/10.1086/421942.
van Oorschot DVH, Bunge E, Diaz-Decaro J, Curran D, Yawn B. A systematic literature review of herpes zoster incidence worldwide. Hum Vaccin Immunother. 2021;17(6):1714–32. https://doi.org/10.1080/21645515.2020.1847582.
Schmader K. Herpes zoster. Ann Intern Med. 2018;169(3):Itc19–31. https://doi.org/10.7326/aitc201808070.
Yawn BP, Gilden D. The global epidemiology of herpes zoster. Neurology. 2013;81(10):928–30. https://doi.org/10.1212/WNL.0b013e3182a3516e.
Insinga RPIR, Pellissier JM, Saddier P, Nikas AA. The incidence of herpes zoster in a United States administrative database. J Gen Intern Med. 2005;20(8):748–53. https://doi.org/10.1111/j.1525-1497.2005.0150.x.
Mullooly JPRK, Chun C, Weinmann S, Houston H. Incidence of herpes zoster, 1997–2002. Epidemiol Infect. 2002;133(2):245–53. https://doi.org/10.1017/s095026880400281x.
Opstelten W, Van Essen GA, Schellevis F, Verheij TJM, Moons KGM. Gender as an independent risk factor for herpes zoster: a population-based prospective study. Ann Epidemiol. 2006;16:692–5. https://doi.org/10.1016/j.annepidem.2005.12.002.
Cadogan SL, Mindell JS, Breuer J, Hayward A, Warren-Gash C. Prevalence of and factors associated with herpes zoster in England: a cross-sectional analysis of the Health Survey for England. BMC Infect Dis. 2022. https://doi.org/10.1186/s12879-022-07479-z.
Joon Lee T, Hayes S, Cummings DM, et al. Herpes zoster knowledge, prevalence, and vaccination rate by race. J Am Board Fam Med. 2013;26(1):45–51. https://doi.org/10.3122/jabfm.2013.01.120154.
Coplan P, Schmader K, Nikas A, et al. Development of a measure of the burden of pain due to herpes zoster and postherpetic neuralgia for prevention trials: adaptation of the brief pain inventory. J Pain. 2004;5(6):344–56. https://doi.org/10.1016/j.jpain.2004.06.001.
Lang P-O, Aspinall R. Vaccination for quality of life: herpes–zoster vaccines. Aging Clin Exp Res. 2021;33(4):1113–22. https://doi.org/10.1007/s40520-019-01374-5.
Yawn BP SP, Wollan PC, St Sauver JL, Kurland MJ, Sy LS. A population-based study of the incidence and complication rates of herpes zoster before zoster vaccine introduction. Mayo Clin Proc. 2008;82(11):1341–9. https://doi.org/10.4065/82.11.1341. (Published correction appears in Mayo Clin Proc. 2008 Feb;83(2):255.)
Volpi A. Severe complications of herpes zoster. Herpes. 2007;14(Suppl 2):35–9. https://doi.org/10.1007/s11940-013-0246-5.
Hoesly FJ, Sluzevich JC. Chronic cutaneous varicella zoster virus infection complicating dermatomyositis. J Dermatol. 2014;41(4):334–6. https://doi.org/10.1111/1346-8138.12402.
Wauters O, Lebas E, Nikkels AF. Chronic mucocutaneous herpes simplex virus and varicella zoster virus infections. J Am Acad Dermatol. 2012;66(6):e217–27. https://doi.org/10.1016/j.jaad.2010.07.011.
Boussault P, Boralevi F, Labbe L, Sarlangue J, Taïeb A, Leaute-Labreze C. Chronic varicella-zoster skin infection complicating the congenital varicella syndrome. Pediatr Dermatol. 2007;24(4):429–32. https://doi.org/10.1111/j.1525-1470.2007.00471.x.
Matsuo K, Honda M, Shiraki K, Niimura M. Prolonged herpes zoster in a patient infected with the human immunodeficiency virus. J Dermatol. 2001;28(12):728–33.
Nikkels AF, Snoeck R, Rentier B, Pierard GE. Chronic verrucous varicella zoster virus skin lesions: clinical, histological, molecular and therapeutic aspects. Clin Exp Dermatol. 1999;24(5):346–53. https://doi.org/10.1046/j.1365-2230.1999.00498.x.
DeWane ME, Smith JS, DeSimone MS, Mostaghimi A. A case of refractory verrucous varicella zoster virus in a patient with persistent pancytopenia after CAR-T therapy. Br J Dermatol. 2022;187(3):e77. https://doi.org/10.1111/bjd.21609.
Tilley DH, Satter EK, Kakimoto CV, Lederman ER. Disseminated verrucous varicella zoster with exclusive follicular involvement. Arch Dermatol. 2012;148(3):405–7. https://doi.org/10.1001/archdermatol.2011.2801.
Page MJ, McKenzie KJ, Bossuyt PM, et al. The PRISMA 2020 statement: an updatedguideline for reporting systematic reviews. BMC Syst Rev. 2021;10:89. https://doi.org/10.1186/s13643-021-01626-4.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ. Cochrane Handbook for Systematic Reviews of Interventions version 6.3. 2022. https://training.cochrane.org/handbooks. Accessed 10 June 2024.
Smith CG, Glaser DA. Herpes zoster in childhood: case report and review of the literature. Pediatr Dermatol. 1996;13(3):226–9. https://doi.org/10.1111/j.1525-1470.1996.tb01208.x.
Nikkels AF, Nikkels-Tassoudji N, Piérard GE. Revisiting childhood herpes zoster. Pediatr Dermatol. 2004;21(1):18–23. https://doi.org/10.1111/j.0736-8046.2004.21104.x.
Katakam BK, Kiran G, Kumar U. A prospective study of herpes zoster in children. Indian J Dermatol. 2016;61(5):534–9. https://doi.org/10.4103/0019-5154.190121.
Leung AK, Robson WL, Leong AG. Herpes zoster in childhood. J Pediatr Health Care. 2006;20(5):300–3. https://doi.org/10.1016/j.pedhc.2006.01.004.
Brodell RT, Zurakowski JE. Childhood shingles. Herpes zoster can occur in healthy children too. Postgrad Med. 2004;115(4):63–5. https://doi.org/10.3810/pgm.2004.04.1493.
Quesada D, Morsky L, Aguìñiga-Navarrete P, Garrett MB. Pediatric herpes zoster. Clin Pract Cases Emerg Med. 2020;4(1):32–4. https://doi.org/10.5811/cpcem.2019.10.44301.
Weinmann S, Naleway AL, Koppolu P, et al. Incidence of herpes zoster among children: 2003–2014. Pediatrics. 2019. https://doi.org/10.1542/peds.2018-2917.
Berlinberg EJ, Kim E, Deiner MS, Patterson C, Porco TC, Acharya NR. Seasonality of herpes zoster and herpes zoster ophthalmicus. J Clin Virol. 2020;126:104306. https://doi.org/10.1016/j.jcv.2020.104306.
Blank LJ, Polydefkis MJ, Moore RD, Gebo KA. Herpes zoster among persons living with HIV in the current antiretroviral therapy era. J Acquir Immune Defic Syndr. 2012;61(2):203–7. https://doi.org/10.1097/QAI.0b013e318266cd3c.
Bruxvoort KJ, Liang AS, Harpaz R, et al. Patient report of herpes zoster pain: incremental benefits of zoster vaccine live. Vaccine. 2019;37(26):3478–84. https://doi.org/10.1016/j.vaccine.2019.04.095.
Chen SY, Suaya JA, Li Q, et al. Incidence of herpes zoster in patients with altered immune function. Infection. 2014;42(2):325–34. https://doi.org/10.1007/s15010-013-0550-8.
Gebo KA, Kalyani R, Moore RD, Polydefkis MJ. The incidence of, risk factors for, and sequelae of herpes zoster among HIV patients in the highly active antiretroviral therapy era. J Acquir Immune Defic Syndr. 2005;40(2):169–74. https://doi.org/10.1097/01.qai.0000178408.62675.b0.
Gudin J, Fudin J, Wang E, Haylon T, Patel K, Goss TF. Treatment patterns and medication use in patients with postherpetic neuralgia. J Manag Care Spec Pharm. 2019;25(12):1387–96. https://doi.org/10.18553/jmcp.2019.19093.
Guignard AP, Greenberg M, Lu C, Rosillon D, Vannappagari V. Risk of herpes zoster among diabetics: a matched cohort study in a US insurance claim database before introduction of vaccination, 1997–2006. Infection. 2014;42(4):729–35. https://doi.org/10.1007/s15010-014-0645-x.
Habel LA, Ray GT, Silverberg MJ, et al. The epidemiology of herpes zoster in patients with newly diagnosed cancer. Cancer Epidemiol Biomark Prev. 2013;22(1):82–90. https://doi.org/10.1158/1055-9965.Epi-12-0815.
Javed S, Kamili QU, Mendoza N, Tyring SK. Possible association of lower rate of postherpetic neuralgia in patients on anti-tumor necrosis factor-α. J Med Virol. 2011;83(11):2051–5. https://doi.org/10.1002/jmv.22182.
Katz J, McDermott MP, Cooper EM, Walther RR, Sweeney EW, Dworkin RH. Psychosocial risk factors for postherpetic neuralgia: a prospective study of patients with herpes zoster. J Pain. 2005;6(12):782–90. https://doi.org/10.1016/j.jpain.2005.07.006.
Klein NP, Bartlett J, Fireman B, et al. Long-term effectiveness of zoster vaccine live for postherpetic neuralgia prevention. Vaccine. 2019;37(36):5422–7. https://doi.org/10.1016/j.vaccine.2019.07.004.
Kong CL, Thompson RR, Porco TC, Kim E, Acharya NR. Incidence rate of herpes zoster ophthalmicus: a retrospective cohort study from 1994 through 2018. Ophthalmology. 2020;127(3):324–30. https://doi.org/10.1016/j.ophtha.2019.10.001.
Langan SM, Smeeth L, Margolis DJ, Thomas SL. Herpes zoster vaccine effectiveness against incident herpes zoster and post-herpetic neuralgia in an older US population: a cohort study. PLoS Med. 2013;10(4):e1001420. https://doi.org/10.1371/journal.pmed.1001420.
Marin M, Yawn BP, Hales CM, et al. Herpes zoster vaccine effectiveness and manifestations of herpes zoster and associated pain by vaccination status. Hum Vaccin Immunother. 2015;11(5):1157–64. https://doi.org/10.1080/21645515.2015.1016681.
Meyers JL, Madhwani S, Rausch D, Candrilli SD, Krishnarajah G, Yan S. Analysis of real-world health care costs among immunocompetent patients aged 50 years or older with herpes zoster in the United States. Hum Vaccin Immunother. 2017;13(8):1861–72. https://doi.org/10.1080/21645515.2017.1324373.
Moanna A, Rimland. Decreasing incidence of herpes zoster in the highly active antiretroviral therapy era. Clin Infect Dis. 2013;57(1):122–5. https://doi.org/10.1093/cid/cit165.
Sahoo F, Hill JA, Xie H, et al. Herpes zoster in autologous hematopoietic cell transplant recipients in the era of acyclovir or valacyclovir prophylaxis and novel treatment and maintenance therapies. Biol Blood Marrow Transplant. 2017;23(3):505–11. https://doi.org/10.1016/j.bbmt.2016.12.620.
Thompson RR, Kong CL, Porco TC, Kim E, Ebert CD, Acharya NR. Herpes zoster and postherpetic neuralgia: changing incidence rates from 1994 to 2018 in the United States. Clin Infect Dis. 2021;73(9):e3210–7. https://doi.org/10.1093/cid/ciaa1185.
Tran KD, Falcone MM, Choi DS, et al. Epidemiology of herpes zoster ophthalmicus: recurrence and chronicity. Ophthalmology. 2016;123(7):1469–75. https://doi.org/10.1016/j.ophtha.2016.03.005.
Tran TN, Ray GT, Horberg MA, et al. Complications of herpes zoster in cancer patients. Scand J Infect Dis. 2014;46(7):528–32. https://doi.org/10.3109/00365548.2014.901554.
Tseng HF, Bruxvoort K, Ackerson B, et al. The epidemiology of herpes zoster in immunocompetent, unvaccinated adults ≥50 years old: incidence, complications, hospitalization, mortality, and recurrence. J Infect Dis. 2020;222(5):798–806. https://doi.org/10.1093/infdis/jiz652.
Tseng HF, Lewin B, Hales CM, et al. Zoster vaccine and the risk of postherpetic neuralgia in patients who developed herpes zoster despite having received the zoster vaccine. J Infect Dis. 2015;212(8):1222–31. https://doi.org/10.1093/infdis/jiv244.
White RR, Lenhart G, Singhal PK, et al. Incremental 1-year medical resource utilization and costs for patients with herpes zoster from a set of US health plans. Pharmacoeconomics. 2009;27(9):781–92. https://doi.org/10.2165/11317560-000000000-00000.
Yawn BP, Saddier P, Wollan PC, St Sauver JL, Kurland MJ, Sy LS. A population-based study of the incidence and complication rates of herpes zoster before zoster vaccine introduction. Mayo Clin Proc. 2007;82(11):1341–9. https://doi.org/10.4065/82.11.1341.
Yawn BP, Wollan P, St SJ. Comparing shingles incidence and complication rates from medical record review and administrative database estimates: how close are they? Am J Epidemiol. 2011;174(9):1054–61. https://doi.org/10.1093/aje/kwr206.
Yawn BP, Wollan PC, St Sauver JL, Butterfield LC. Herpes zoster eye complications: rates and trends. Mayo Clin Proc. 2013;88(6):562–70. https://doi.org/10.1016/j.mayocp.2013.03.014.
Yenikomshian MA, Guignard AP, Haguinet F, et al. The epidemiology of herpes zoster and its complications in Medicare cancer patients. BMC Infect Dis. 2015;15:106. https://doi.org/10.1186/s12879-015-0810-6.
Erard V, Guthrie KA, Varley C, et al. One-year acyclovir prophylaxis for preventing varicella-zoster virus disease after hematopoietic cell transplantation: no evidence of rebound varicella-zoster virus disease after drug discontinuation. Blood. 2007;110(8):3071–7. https://doi.org/10.1182/blood-2007-03-077644.
Suaya JA, Chen SY, Li Q, Burstin SJ, Levin MJ. Incidence of herpes zoster and persistent post-zoster pain in adults with or without diabetes in the United States. Open Forum Infect Dis. 2014;1(2):ofu049. https://doi.org/10.1093/ofid/ofu049.
Meyers JL, Candrilli SD, Rausch DA, Yan S, Patterson BJ, Levin MJ. Cost of herpes zoster and herpes zoster-related complications among immunocompromised individuals. Vaccine. 2018;36:6810–8. https://doi.org/10.1016/j.vaccine.2018.08.080.
Borkar DS, Tham VM, Esterberg E, et al. Incidence of herpes zoster ophthalmicus: results from the Pacific Ocular Inflammation Study. Ophthalmology. 2013;120(3):451–6. https://doi.org/10.1016/j.ophtha.2012.09.007.
Amirthalingam G, Andrews N, Keel P, et al. Evaluation of the effect of the herpes zoster vaccination programme 3 years after its introduction in England: a population-based study. Lancet Public Health. 2018;3(2):e82–90. https://doi.org/10.1016/s2468-2667(17)30234-7.
Forbes HJ, Bhaskaran K, Grint D, et al. Incidence of acute complications of herpes zoster among immunocompetent adults in England: a matched cohort study using routine health data. Br J Dermatol. 2021;184(6):1077–84. https://doi.org/10.1111/bjd.19687.
Forbes HJ, Bhaskaran K, Thomas SL, et al. Quantification of risk factors for postherpetic neuralgia in herpes zoster patients: a cohort study. Neurology. 2016;87(1):94–102. https://doi.org/10.1212/wnl.0000000000002808.
Gauthier A, Breuer J, Carrington D, Martin M, Rémy V. Epidemiology and cost of herpes zoster and post-herpetic neuralgia in the United Kingdom. Epidemiol Infect. 2009;137(1):38–47. https://doi.org/10.1017/s0950268808000678.
Gupta G, Lautenbach E, Lewis JD. Incidence and risk factors for herpes zoster among patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2006;4(12):1483–90. https://doi.org/10.1016/j.cgh.2006.09.019.
Matthews I, Duong M, Parsons VL, et al. Burden of disease from shingles and post-herpetic neuralgia in the over 80 year olds in the UK. PLoS ONE. 2020;15(2):e0229224. https://doi.org/10.1371/journal.pone.0229224.
Matthews I, Lu X, Dawson H, et al. Assessing the effectiveness of zoster vaccine live: a retrospective cohort study using primary care data in the United Kingdom. Vaccine. 2018;36(46):7105–11. https://doi.org/10.1016/j.vaccine.2018.08.037.
Mikaeloff Y, Kezouh A, Suissa S. Nonsteroidal anti-inflammatory drug use and the risk of severe skin and soft tissue complications in patients with varicella or zoster disease. Br J Clin Pharmacol. 2008;65(2):203–9. https://doi.org/10.1111/j.1365-2125.2007.02997.x.
Scott FT, Leedham-Green ME, Barrett-Muir WY, et al. A study of shingles and the development of postherpetic neuralgia in East London. J Med Virol. 2003;70:S24–30. https://doi.org/10.1002/jmv.10316.
Walker JL, Andrews NJ, Amirthalingam G, Forbes H, Langan SM, Thomas SL. Effectiveness of herpes zoster vaccination in an older United Kingdom population. Vaccine. 2018;36(17):2371–7. https://doi.org/10.1016/j.vaccine.2018.02.021.
Coen PG, Scott F, Leedham-Green M, et al. Predicting and preventing post-herpetic neuralgia: are current risk factors useful in clinical practice? Eur J Pain. 2006;10:695–700. https://doi.org/10.1016/j.ejpain.2005.11.002.
Yanni EA, Ferreira G, Guennec M, et al. Burden of herpes zoster in 16 selected immunocompromised populations in England: a cohort study in the Clinical Practice Research Datalink 2000–2012. BMJ Open. 2018. https://doi.org/10.1136/bmjopen-2017-020528.
Chen JY, Wang LK, Feng PH, et al. Risk of shingles in adults with primary Sjogren’s syndrome and treatments: a nationwide population-based cohort study. PLoS ONE. 2015;10(8):e0134930. https://doi.org/10.1371/journal.pone.0134930.
Chen YT, Wang HH, Wang TJ, Li YC, Chen TJ. Early application of low-level laser may reduce the incidence of postherpetic neuralgia (PHN). J Am Acad Dermatol. 2016;75(3):572–7. https://doi.org/10.1016/j.jaad.2016.03.050.
Jih JS, Chen YJ, Lin MW, et al. Epidemiological features and costs of herpes zoster in Taiwan: a national study 2000 to 2006. Acta Derm Venereol. 2009;89(6):612–6. https://doi.org/10.2340/00015555-0729.
Liao TL, Chen YM, Liu HJ, Chen DY. Risk and severity of herpes zoster in patients with rheumatoid arthritis receiving different immunosuppressive medications: a case-control study in Asia. BMJ Open. 2017;7(1): e014032. https://doi.org/10.1136/bmjopen-2016-014032.
Liao TL, Lin CH, Chen HH, Chen YM, Lin CC, Chen DY. Significant associations of neurological complications of herpes zoster with stroke in rheumatoid arthritis patients. J Am Heart Assoc. 2017. https://doi.org/10.1161/jaha.117.006304.
Lin SY, Liu JH, Lin CL, et al. A comparison of herpes zoster incidence across the spectrum of chronic kidney disease, dialysis and transplantation. Am J Nephrol. 2012;36(1):27–33. https://doi.org/10.1159/000339004.
Lu WH, Lin CW, Wang CY, Chen LK, Hsiao FY. Epidemiology and long-term disease burden of herpes zoster and postherpetic neuralgia in Taiwan: a population-based, propensity score-matched cohort study. BMC Public Health. 2018;18(1):369. https://doi.org/10.1186/s12889-018-5247-6.
Sheu JJ, Keller JJ, Chen YH, Wu CS, Lin HC. No increased risk of sudden sensorineural hearing loss following recent herpes zoster: a nationwide population-based study. Acta Otolaryngol. 2012;132(2):167–72. https://doi.org/10.3109/00016489.2011.633227.
Wang TJ, Hu CC, Lin HC. Increased risk of anterior uveitis following herpes zoster: a nationwide population-based study. Arch Ophthalmol. 2012;130(4):451–5. https://doi.org/10.1001/archophthalmol.2011.357.
Wang WY, Liu SH, Lin MY, Lin CC, Wang IJ. Initial presentation sites as predictors of herpes zoster complications: a nationwide cohort study. PLoS ONE. 2016;11(10):e0164019. https://doi.org/10.1371/journal.pone.0164019.
Tsai TF, Yao CA, Yu HS, et al. Herpes zoster-associated severity and duration of pain, health-related quality of life, and healthcare utilization in Taiwan: a prospective observational study. Int J Dermatol. 2015;54(5):529–36. https://doi.org/10.1111/ijd.12484.
Li Y, An Z, Yin D, et al. Disease burden due to herpes zoster among population aged ≥50 years old in China: a community based retrospective survey. PLoS ONE. 2016;11(4):e0152660. https://doi.org/10.1371/journal.pone.0152660.
Lu L, Suo L, Li J, Pang X. A retrospective survey on herpes zoster disease burden and characteristics in Beijing, China. Hum Vaccin Immunother. 2018;14(11):2632–5. https://doi.org/10.1080/21645515.2018.1489193.
Sun X, Wei Z, Lin H, Jit M, Li Z, Fu C. Incidence and disease burden of herpes zoster in the population aged ≥50 years in China: data from an integrated health care network. J Infect. 2021;82(2):253–60. https://doi.org/10.1016/j.jinf.2020.12.013.
Suo L, Lu L, Li J, et al. A case control study on family history as a risk factor for herpes zoster and associated outcomes, Beijing, China. BMC Infect Dis. 2017;17(1):334. https://doi.org/10.1186/s12879-017-2416-7.
Xing X, Sun K, Yan M. Delayed initiation of supplemental pain management is associated with postherpetic neuralgia: a retrospective study. Pain Physician. 2020;23(1):65–72. https://doi.org/10.36076/PPJ.2020/23/65.
Zhang J, Ding Q, Li XL, Hao YW, Yang Y. Support vector machine versus multiple logistic regression for prediction of postherpetic neuralgia in outpatients with herpes zoster. Pain Physician. 2022;25(3):E481–8.
Wang X-X, Zhang Y, Fan B-F. Predicting postherpetic neuralgia in patients with herpes zoster by machine learning: a retrospective study. Pain Ther. 2020;9(2):627–35. https://doi.org/10.1007/s40122-020-00196-y.
Yang F, Yu S, Fan B, et al. The epidemiology of herpes zoster and postherpetic neuralgia in China: results from a cross-sectional study. Pain Ther. 2019;8:249–59. https://doi.org/10.1007/s40122-019-0127-z.
Sakai R, Kasai S, Hirano T, et al. No increased risk of herpes zoster in TNF inhibitor and non-TNF inhibitor users with rheumatoid arthritis: epidemiological study using the Japanese health insurance database. Int J Rheum Dis. 2018;21:1670–7. https://doi.org/10.1111/1756-185X.13300.
Asada H, Nagayama K, Okazaki A, et al. An inverse correlation of VZV skin-test reaction, but not antibody, with severity of herpes zoster skin symptoms and zoster-associated pain. J Dermatol Sci. 2013;69(3):243–9. https://doi.org/10.1016/j.jdermsci.2012.10.015.
Imafuku S, Nakayama J, Higa K, et al. One-year follow-up of zoster-associated pain in 764 immunocompetent patients with acute herpes zoster treated with famciclovir (FAMILIAR study). J Eur Acad Dermatol Venereol. 2014;28(12):1716–22. https://doi.org/10.1111/jdv.12379.
Kurokawa I, Kumano K, Murakawa K. Clinical correlates of prolonged pain in Japanese patients with acute herpes zoster. J Int Med Res. 2002;30(1):56–65. https://doi.org/10.1177/147323000203000109.
Nakamura Y, Miyagawa F, Okazaki A, et al. Clinical and immunologic features of recurrent herpes zoster (HZ). J Am Acad Dermatol. 2016;75(5):950–6.e1. https://doi.org/10.1016/j.jaad.2016.05.037.
Sato K, Adachi K, Nakamura H, et al. Burden of herpes zoster and postherpetic neuralgia in Japanese adults 60 years of age or older: results from an observational, prospective, physician practice-based cohort study. J Dermatol. 2017;44(4):414–22. https://doi.org/10.1111/1346-8138.13639.
Takao Y, Miyazaki Y, Okeda M, et al. Incidences of herpes zoster and postherpetic neuralgia in Japanese adults aged 50 years and older from a community-based prospective cohort study: the SHEZ study. J Epidemiol. 2015;25(10):617–25. https://doi.org/10.2188/jea.JE20140210.
Cho SI, Lee CH, Park GH, Park CW, Kim HO. Use of S-LANSS, a tool for screening neuropathic pain, for predicting postherpetic neuralgia in patients after acute herpes zoster events: a single-center, 12-month, prospective cohort study. J Pain. 2014;15(2):149–56. https://doi.org/10.1016/j.jpain.2013.10.006.
Choi WS, Kwon SS, Lee J, et al. Immunity and the burden of herpes zoster. J Med Virol. 2014;86(3):525–30. https://doi.org/10.1002/jmv.23830.
Hong MJ, Kim YD, Cheong YK, Park SJ, Choi SW, Hong HJ. Epidemiology of postherpetic neuralgia in Korea: an electronic population health insurance system based study. Medicine (Baltimore). 2016;95(14):e3304. https://doi.org/10.1097/md.0000000000003304.
Kim SY, Lee CH, Min C, Kim HJ, Choi HG. Herpes zoster does not increase the risk of sudden sensory neural hearing loss: a longitudinal follow-up study using a national sample cohort. Int J Audiol. 2020;59(3):173–8. https://doi.org/10.1080/14992027.2019.1666309.
Lee SM, Han J, Yang CM, et al. Chronic and recurrent herpes zoster ophthalmicus. Medicina (Kaunas). 2021;57(10):999. https://doi.org/10.3390/medicina57100999.
Song H, Lee J, Lee M, et al. Burden of illness, quality of life, and healthcare utilization among patients with herpes zoster in South Korea: a prospective clinical-epidemiological study. Int J Infect Dis. 2014;20:23–30. https://doi.org/10.1016/j.ijid.2013.11.018.
Woo YR, Jung YJ, Kim JS, et al. Cutaneous comorbidities in patients with multiple myeloma: a 10-year retrospective cohort study from a Korean population. Medicine (United States). 2018;97(43):e12825. https://doi.org/10.1097/MD.0000000000012825.
Alicino C, Trucchi C, Paganino C, et al. Incidence of herpes zoster and post-herpetic neuralgia in Italy: results from a 3-years population-based study. Hum Vaccin Immunother. 2017;13(2):399–404. https://doi.org/10.1080/21645515.2017.1264834.
Amicizia D, Domnich A, Arata L, et al. The role of age-sex interaction in the development of post-herpetic neuralgia. Hum Vaccin Immunother. 2017;13(2):376–8. https://doi.org/10.1080/21645515.2017.1264799.
Bricout H, Perinetti E, Marchettini P, et al. Burden of herpes zoster-associated chronic pain in Italian patients aged 50 years and over (2009–2010): a GP-based prospective cohort study. BMC Infect Dis. 2014;14:637. https://doi.org/10.1186/s12879-014-0637-6.
Parruti G, Tontodonati M, Rebuzzi C, et al. Predictors of pain intensity and persistence in a prospective Italian cohort of patients with herpes zoster: relevance of smoking, trauma and antiviral therapy. BMC Med. 2010;8:58. https://doi.org/10.1186/1741-7015-8-58.
Volpi A, Gatti A, Pica F, Bellino S, Marsella LT, Sabato AF. Clinical and psychosocial correlates of post-herpetic neuralgia. J Med Virol. 2008;80(9):1646–52. https://doi.org/10.1002/jmv.21254.
Gialloreti LE, Merito M, Pezzotti P, et al. Epidemiology and economic burden of herpes zoster and post-herpetic neuralgia in Italy: a retrospective, population-based study. BMC Infect Dis. 2010;10:230. https://doi.org/10.1186/1471-2334-10-230.
Caputo M, Horn J, Karch A, et al. Herpes zoster incidence in Germany—an indirect validation study for self-reported disease data from pretest studies of the population-based German National Cohort. BMC Infect Dis. 2019;19(1):99. https://doi.org/10.1186/s12879-019-3691-2.
Hillebrand K, Bricout H, Schulze-Rath R, Schink T, Garbe E. Incidence of herpes zoster and its complications in Germany, 2005–2009. J Infect. 2015;70(2):178–86. https://doi.org/10.1016/j.jinf.2014.08.018.
Schmidt-Ott R, Schutter U, Simon J, et al. Incidence and costs of herpes zoster and postherpetic neuralgia in German adults aged ≥ 50 years: a prospective study. J Infect. 2018;76(5):475–82. https://doi.org/10.1016/j.jinf.2018.02.001.
Schröder C, Enders D, Schink T, Riedel O. Incidence of herpes zoster amongst adults varies by severity of immunosuppression. J Infect. 2017;75(3):207–15. https://doi.org/10.1016/j.jinf.2017.06.010.
Ultsch B, Köster I, Reinhold T, et al. Epidemiology and cost of herpes zoster and postherpetic neuralgia in Germany. Eur J Health Econ. 2013;14(6):1015–26. https://doi.org/10.1007/s10198-012-0452-1.
Ultsch B, Siedler A, Rieck T, Reinhold T, Krause G, Wichmann O. Herpes zoster in Germany: quantifying the burden of disease. BMC Infect Dis. 2011;11:173. https://doi.org/10.1186/1471-2334-11-173.
Kho MML, Roest S, Bovée DM, et al. Herpes zoster in solid organ transplantation: incidence and risk factors. Front Immunol. 2021;12:645718. https://doi.org/10.3389/fimmu.2021.645718.
Opstelten W, Mauritz JW, de Wit NJ, van Wijck AJ, Stalman WA, van Essen GA. Herpes zoster and postherpetic neuralgia: incidence and risk indicators using a general practice research database. Fam Pract. 2002;19(5):471–5. https://doi.org/10.1093/fampra/19.5.471.
Opstelten W, Zuithoff NPA, van Essen GA, et al. Predicting postherpetic neuralgia in elderly primary care patients with herpes zoster: prospective prognostic study. Pain. 2007;132:S52–9. https://doi.org/10.1016/j.pain.2007.02.004.
Pierik JG, Gumbs PD, Fortanier SA, Van Steenwijk PC, Postma MJ. Epidemiological characteristics and societal burden of varicella zoster virus in the Netherlands. BMC Infect Dis. 2012;12:110. https://doi.org/10.1186/1471-2334-12-110.
van Wijck AJM, Aerssens YR. Pain, itch, quality of life, and costs after herpes zoster. Pain Pract. 2017;17(6):738–46. https://doi.org/10.1111/papr.12518.
Cebrián-Cuenca AM, Díez-Domingo J, Rodríguez MS, et al. Epidemiology of herpes zoster infection among patients treated in primary care centres in the Valencian community (Spain). BMC Fam Pract. 2010;11:33. https://doi.org/10.1186/1471-2296-11-33.
Díez-Domingo J, Curran D, Cambronero MDR, Garcia-Martinez JA, Matthews S. Economic burden and impact on quality of life of herpes zoster in spanish adults aged 50 years or older: a prospective cohort study. Adv Ther. 2021;38(6):3325–41. https://doi.org/10.1007/s12325-021-01717-7.
Muñoz-Quiles C, López-Lacort M, Díez-Domingo J, Orrico-Sánchez A. Herpes zoster risk and burden of disease in immunocompromised populations: a population-based study using health system integrated databases, 2009–2014. BMC Infect Dis. 2020;20(1):905. https://doi.org/10.1186/s12879-020-05648-6.
Muñoz-Quiles C, López-Lacort M, Orrico-Sánchez A, Díez-Domingo J. Impact of postherpetic neuralgia: a six year population-based analysis on people aged 50 years or older. J Infect. 2018;77(2):131–6. https://doi.org/10.1016/j.jinf.2018.04.004.
Salleras L, Salleras M, Salvador P, et al. Herpes zoster and postherpetic neuralgia in Catalonia (Spain). Hum Vaccin Immunother. 2015;11(1):178–84. https://doi.org/10.4161/hv.34421.
Buchan SA, Daneman N, Wang J, et al. Incidence of hospitalizations and emergency department visits for herpes zoster in immunocompromised and immunocompetent adults in Ontario, Canada, 2002–2016. Clin Infect Dis. 2020;71(1):22–9. https://doi.org/10.1093/cid/ciz769.
Drolet M, Brisson M, Schmader KE, et al. The impact of herpes zoster and postherpetic neuralgia on health-related quality of life: a prospective study. CMAJ. 2010;182(16):1731–6. https://doi.org/10.1503/cmaj.091711.
Drolet M, Brisson M, Schmader K, et al. Predictors of postherpetic neuralgia among patients with herpes zoster: a prospective study. J Pain. 2010;11(11):1211–21. https://doi.org/10.1016/j.jpain.2010.02.020.
Marra F, Chong M, Najafzadeh M. Increasing incidence associated with herpes zoster infection in British Columbia, Canada. BMC Infect Dis. 2016;16(1):589. https://doi.org/10.1186/s12879-016-1898-z.
Friesen KJ, Chateau D, Falk J, Alessi-Severini S, Bugden S. Cost of shingles: population based burden of disease analysis of herpes zoster and postherpetic neuralgia. BMC Infect Dis. 2017;17(1):69. https://doi.org/10.1186/s12879-017-2185-3.
El Hayderi L, Nikkels-Tassoudji N, Nikkels AF. Incidence of and risk factors for cutaneous scarring after herpes zoster. Am J Clin Dermatol. 2018;19(6):893–7. https://doi.org/10.1007/s40257-018-0385-2.
Schmidt SAJ, Kahlert J, Vestergaard M, Schønheyder HC, Sørensen HT. Hospital-based herpes zoster diagnoses in Denmark: rate, patient characteristics, and all-cause mortality. BMC Infect Dis. 2016. https://doi.org/10.1186/s12879-016-1369-6.
Bongers LD, Navarini A, Berger CT, Mueller SM. Complications and cost estimations in herpes zoster—a retrospective analysis at a Swiss tertiary dermatology clinic. Swiss Med Wkly. 2021;151(51): w30081. https://doi.org/10.4414/smw.2021.w30081.
Albrecht P, Patrzałek M, Goryński P. The burden of herpes zoster and its complications in Poland in according to the age. Przegl Epidemiol. 2015;69(4):693–7, 841–3.
Cebeci D, Karasel S. Retrospective analysis of 170 patients followed up with herpes zoster and postherpetic neuralgia in nothern Cyprus. Open Access Maced J Med Sci. 2021;9(B):583–7. https://doi.org/10.3889/oamjms.2021.6251.
Weitzman D, Shavit O, Stein M, Cohen R, Chodick G, Shalev V. A population based study of the epidemiology of herpes zoster and its complications. J Infect. 2013;67(5):463–9. https://doi.org/10.1016/j.jinf.2013.06.016.
Aunhachoke K, Bussaratid V, Chirachanakul P, et al. Measuring herpes zoster, zoster-associated pain, post-herpetic neuralgia-associated loss of quality of life, and healthcare utilization and costs in Thailand. Int J Dermatol. 2011;50(4):428–35. https://doi.org/10.1111/j.1365-4632.2010.04715.x.
Bajaj DR, Devrajani BR, Yousfani A, Shah SZA, Devrajani T, Bibi I. Frequency and pattern of herpes zoster at Liaquat University Hospital Hyderabad (six months hospital based descriptive study). J Liaquat Univ Med Health Sci. 2009;8(2):113–7.
Alakloby OM, AlJabre SH, Randhawa MA, Alzahrani AJ, AlWunais KM, Bukhari IA. Herpes zoster in eastern Saudi Arabia: clinical presentation and management. J Drugs Dermatol. 2008;7(5):457–62.
Rampakakis E, Pollock C, Vujacich C, et al. Economic burden of herpes zoster (“culebrilla”) in Latin America. Int J Infect Dis. 2017;58:22–6. https://doi.org/10.1016/j.ijid.2017.02.021.
Kawai K, Rampakakis E, Tsai TF, et al. Predictors of postherpetic neuralgia in patients with herpes zoster: a pooled analysis of prospective cohort studies from North and Latin America and Asia. Int J Infect Dis. 2015;34:126–31. https://doi.org/10.1016/j.ijid.2015.03.022.
Rabaud C, Rogeaux O, Launay O, et al. Early antiviral treatment fails to completely prevent herpes-related pain. Med Mal Infect. 2013;43(11–12):461–6. https://doi.org/10.1016/j.medmal.2013.07.012.
Stein AN, Britt H, Harrison C, Conway EL, Cunningham A, Macintyre CR. Herpes zoster burden of illness and health care resource utilisation in the Australian population aged 50 years and older. Vaccine. 2009;27(4):520–9. https://doi.org/10.1016/j.vaccine.2008.11.012.
Blennow O, Fjaertoft G, Winiarski J, Ljungman P, Mattsson J, Remberger M. Varicella-zoster reactivation after allogeneic stem cell transplantation without routine prophylaxis–the incidence remains high. Biol Blood Marrow Transplant. 2014;20(10):1646–9. https://doi.org/10.1016/j.bbmt.2014.06.002.
MacIntyre R, Stein A, Harrison C, Britt H, Mahimbo A, Cunningham A. Increasing trends of herpes zoster in Australia. PLoS ONE. 2015;10(4):e0125025. https://doi.org/10.1371/journal.pone.0125025.
Abdul Latheef E, Pavithran K. Herpes zoster: a clinical study in 205 patients. Indian J Dermatol. 2011;56(5):529–32. https://doi.org/10.4103/0019-5154.87148.
Malkud S, Dyavannanavar V, Purnachandra D, Murthy K. Clinical and morphological characteristics of herpes zoster—a study from tertiary care centre. J Pak Assoc Dermatol. 2016;26(3):219–22.
Duracinsky M, Paccalin M, Gavazzi G, et al. ARIZONA study: is the risk of post-herpetic neuralgia and its burden increased in the most elderly patients? BMC Infect Dis. 2014;14:529. https://doi.org/10.1186/1471-2334-14-529.
Pickering G, Gavazzi G, Gaillat J, Paccalin M, Bloch K, Bouhassira D. Is herpes zoster an additional complication in old age alongside comorbidity and multiple medications? Results of the post hoc analysis of the 12-month longitudinal prospective observational ARIZONA cohort study. BMJ Open. 2016;6(2):e009689. https://doi.org/10.1136/bmjopen-2015-009689.
Sundström K, Weibull CE, Söderberg-Löfdal K, Bergström T, Sparén P, Arnheim-Dahlström L. Incidence of herpes zoster and associated events including stroke—a population-based cohort study. BMC Infect Dis. 2015;15:488. https://doi.org/10.1186/s12879-015-1170-y.
Adhicari D, Agarwal D. A hospital-based clinical study of herpes zoster—a report of 113 cases. IOSR-JDMS. 2017;16(1):33–8. https://doi.org/10.9790/0853-1601093338.
Steiner I, Kennedy PG, Pachner AR. The neurotropic herpes viruses: herpes simplex and varicella-zoster. Lancet Neurol. 2007;6(11):1015–28. https://doi.org/10.1016/s1474-4422(07)70267-3.
Mirouse A, Vignon P, Piron P, et al. Severe varicella-zoster virus pneumonia: a multicenter cohort study. Crit Care. 2017;21(1):137. https://doi.org/10.1186/s13054-017-1731-0.
Gerada C, Campbell TM, Kennedy JJ, et al. Manipulation of the innate immune response by varicella zoster virus. Front Immunol. 2020;11:1. https://doi.org/10.3389/fimmu.2020.00001.
Chen N, Li Q, Yang J, Zhou M, Zhou D, He L. Antiviral treatment for preventing postherpetic neuralgia. Cochrane Database Syst Rev. 2014;2014(2):CD006866. https://doi.org/10.1002/14651858.CD006866.pub3.
Sacks GM. Unmet need in the treatment of postherpetic neuralgia. Am J Manag Care. 2013;19(1 Suppl):S207–13.
Kawai K, Gebremeskel BG, Acosta CJ. Systematic review of incidence and complications of herpes zoster: towards a global perspective. BMJ Open. 2014;4(6):e004833. https://doi.org/10.1136/bmjopen-2014-004833.
Yin D, Van Oorschot D, Jiang N, et al. A systematic literature review to assess the burden of herpes zoster disease in China. Expert Rev Anti Infect Ther. 2021;19(2):165–79. https://doi.org/10.1080/14787210.2020.1792290.
San Martin P, Aunhachoke K, Batac MCF, et al. Systematic literature review of herpes zoster disease burden in Southeast Asia. Infect Dis Ther. 2023;12(6):1553–78. https://doi.org/10.1007/s40121-023-00822-0.
McKay SL, Guo A, Pergam SA, Dooling K. Herpes zoster risk in immunocompromised adults in the united states: a systematic review. Clin Infect Dis. 2020;71(7):e125–34. https://doi.org/10.1093/cid/ciz1090.
Oxman MN. Herpes zoster pathogenesis and cell-mediated immunity and immunosenescence. J of Osteopat Med. 2009;109(s62):13–7. https://doi.org/10.7556/jaoa.2009.20009.
Medical Writing, Editorial and Other Assistance
The authors would like to thank Business & Decision Life Sciences Medical Communication Service Center for editorial assistance and manuscript coordination, on behalf of GSK.
Funding
GlaxoSmithKline Biologicals SA funded this study and took in charge all costs associated with the development and publication of this manuscript, including the journal’s Rapid Service Fee.
Author information
Authors and Affiliations
Contributions
Desmond Curran and Nikolaos Giannelos were involved in the design of the study. Carol Kagia, Chi Nguyen and Nikki Vroom collected or generated the data. Carol Kagia, Chi Nguyen, Desmond Curran, Hilde Vroling, Nikolaos Giannelos and Nikki Vroom analyzed and/or interpreted the data. All authors participated to the development of this manuscript and in its critical review with important intellectual contributions. All authors had full access to the data and gave approval of the final manuscript before submission. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of Interest
Nikolaos Giannelos and Desmond Curran are employed by and hold financial equities in GSK. The authors declare no other financial or non-financial relationships and activities. Carol Kagia, Chi Nguyen, Hilde Vroling and Nikki Vroom are employees of P95/Pallas. P95/Pallas received funding from GSK for the submitted work. P95/Pallas holds/held contracts with AstraZeneca, CEPI, GSK, Pfizer, Sanofi, Seqirus, Orchard, Biomarin, Daiichi, Bavarian Nordic and Bayer. Authors declare no other financial and non-financial relationships and activities.
Ethics/Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. Due to this reason an ethics committee approval was not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Giannelos, N., Curran, D., Nguyen, C. et al. The Incidence of Herpes Zoster Complications: A Systematic Literature Review. Infect Dis Ther 13, 1461–1486 (2024). https://doi.org/10.1007/s40121-024-01002-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40121-024-01002-4