Abstract
Introduction
Although the indications for antiviral therapy for patients with chronic hepatitis B have been gradually expanded in different guidelines, antiviral treatment efficacy remains unclear among HBeAg-seropositive patients with alanine aminotransferase (ALT) < 2 upper limits of normal (ULN). This study aimed to evaluate the efficacy of antiviral therapy for these patients.
Methods
In total, 102 treatment-naive patients who were HBeAg seropositive with ALT < 2 ULN and had received nucleotide analogs were included, and their clinical data were retrospectively analyzed.
Results
After 96-week treatment, 84.3% (n = 86), 26.5% (n = 27) and 20.6% (n = 21) patients achieved virological response, HBeAg seroclearance and HBeAg seroconversion, respectively. Logistic regression analysis revealed that baseline AST (odds ratio [OR] = 1.069, 95% confidence interval [CI] 1.014–1.127, p = 0.014), serum HBV DNA (OR = 0.540, 95% CI 0.309–0.946, p = 0.031) and quantitative HBsAg levels (OR = 0.147, 95% CI 0.036–0.597, p = 0.007) were independent factors for virological response. At baseline, HBsAg < 4.63 log10 IU/ml was identified as a strong predictor for the 96-week virological response, with a concordance rate of 0.902. Moreover, the levels of liver stiffness values (8.30 ± 3.86 vs. 6.17 ± 1.91, p < 0.001) at week 96 had significantly declined compared to baseline.
Conclusion
Nucleotide analog treatment effectively suppressed HBV DNA in patients with HBeAg-seropositive chronic hepatitis B with ALT < 2 × ULN and greatly improved liver fibrosis. The study also found that HBsAg < 4.63 log10 IU/ml was a strong predictor of the virological response.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Antiviral treatment efficacy remains unclear among patients who are HBeAg-seropositive with ALT < 2 ULN. |
This study analyzed the efficacy of antiviral therapy for these patients and found independent factors related to treatment. |
What was learned from the study? |
This article demonstrates that NA therapy is effective in suppressing HBV DNA and greatly improves liver fibrosis. |
It also shows that ALT levels do not affect the effectiveness of antiviral treatment and HBsAg < 4.63 log10 IU/ml was a strong predictor of the virological response. |
Introduction
Hepatitis B virus (HBV) infection is a global public health concern. Approximately 2 billion people worldwide have been infected with HBV, of which 240 million are chronically infected [1]. According to the 2017 WHO report, 325 million people worldwide are living with hepatitis B or C [2]. HBV infection contributes to 880,000 deaths every year [3]. The majority of previous studies were concerned with antiviral therapies in patients with alanine aminotransferase (ALT) more than two times the upper limit of normal (ULN) and have shown good anti-viral efficacy [4, 5]. In these patients, the higher the levels of ALT are, the better effect of antiviral therapy. Specifically, the seroconversion rates of HBeAg are higher in patients with ALT > 5 × ULN than in patients with ALT ≥ 2 and < 5 × ULN [6]. However, in patients with ALT < 2 × ULN, the initiation of antiviral therapy relies heavily on confirmation of moderate to severe necrotizing inflammation or fibrosis by using liver biopsy or FibroScan. Nevertheless, the invasiveness and expense of liver biopsy limit its clinical application. Similarly, non-invasive FibroScan often lacks accuracy, and this test is unavailable in some hospitals. There are also studies suggesting slow disease progression in patients with ALT < 2 × ULN [7]. As a result, the number of patients with ALT < 2 × ULN who need treatment is underestimated. Notably, studies have shown that patients with persistent normal levels of ALT may progress to cirrhosis and hepatocellular carcinoma (HCC), while effective treatment can reduce the incidence of disease progression [8].
Recent studies have shown that antiviral therapy may benefit patients with ALT < 2 × ULN; patients can achieve virological response and improvement in liver fibrosis [9,10,11]. However, the criteria for antiviral therapy for patients with ALT < 2 × ULN are inconsistent among different guidelines, including the Asian Pacific Association for the Study of the Liver (APASL), European Association for the Study of the Liver (EASL) and American Association for the Study of Liver Diseases (AASLD) [12]. When and whether indications of treatment for HBeAg-seropositive chronic hepatitis B (CHB) should be expanded remain controversial. Research has so far proven insufficient data to answer the question. This study aimed to retrospectively analyze the effects of antiviral therapy in real-world patients with HBV infection and persistent ALT < 2 × ULN.
Methods
Patient Population
Patients with chronic HBV infection attending hospital outpatient clinics (general specialists or liver specialists) without distinction were included from Nanfang Hospital of Southern Medical University in Guangzhou, China, and Shunde Hospital of Southern Medical University in Foshan, China. Retrospective data were collected from December 2008 to February 2022. The study was approved by the Institutional Ethics Committee of Nanfang Hospital (study identifier: NFEC-2020-290) and Shunde Hospital (study identifier: 20211129), and the study protocol was performed in accordance with the Helsinki Declaration of 1964 and its later amendments. Details from all patients' medical records were published anonymously.
Inclusion Criteria and Exclusion Criteria and Study Design
The inclusion criteria were as follows: (1) patients were HBsAg positive for > 6 months, HBeAg seropositive at the screening period and HBV DNA ≥ 2000 IU/ml; (2) within 6 months prior to treatment baseline, the levels of ALT were < 2 × ULN with an interval of > 14 days; (3) patients were treated with entecavir (ETV), tenofovir disoproxil (TDF) or tenofovir alafenamide (TAF) monotherapy; (4) baseline data within 3 months prior to treatment were available. The exclusion criteria were as follows: (1) previously treated with nucleoside or nucleotide analog (NA) therapy or interferon therapy; (2) pregnancy; (3) ultrasound or radiological examination revealing suspicious nodules or AFP > 50 ng/ml; (4) clinical symptoms of decompensated liver disease before treatment; (5) patients co-infected with hepatitis A virus (HAV), hepatitis C virus (HCV), hepatitis E virus (HEV) or human immunodeficiency virus (HIV) and patients with autoimmune diseases (including autoimmune liver disease). The specific data collection procedures were as follows: baseline, 12 weeks, 24 weeks, 36 weeks, 48 weeks, 60 weeks, 72 weeks, 84 weeks and 96 weeks. All planned visits have a ± 7-day visit window. The virological response (VR) was defined as the HBV DNA level below the detection limit (20 IU/ml). The HBeAg seroclearance was defined as a HBeAg level below the detection limit with or without HBeAg seroconversion (1 S/CO). The HBeAg seroconversion was defined as loss of HBeAg and appearance of HBeAb.
Biochemical Tests
Clinical and laboratory data were gathered from outpatient medical records. Data were collected using the following biochemical tests and guidelines: (1) serum HBV DNA (Roche COBAS TaqMan version 2.0—detection limit 20 IU/ml) [13], with the lower limit of detection defined as 20 IU/ml; (2) HBV markers: HBeAg was quantified by enzyme immunoassay (Alisei Quality System; RADIM, Rome, Italy), with the lower limit of detection being 1 S/CO. HBsAg quantification was assessed using Elecsys HBsAg II (Roche Diagnostics, Germany), and the lower limit of detection was 0.05 IU/ml.
Liver Stiffness Measurement
Liver stiffness measurement (LSM) was performed using the FibroScan system (Echosens, Paris, France) as a non-invasive method to estimate the severity of liver fibrosis. The range of results is expressed between 2.5 and 75 kPa. According to the LSM, F0–F4 were defined as: < 7.3 kPa for no/mild fibrosis (F0/F1), ≥ 7.3 and < 9.7 kPa for moderate fibrosis (F2), ≥ 9.7 and < 12.4 kPa for severe fibrosis (F3) and ≥ 12.4 kPa for cirrhosis (F4) [14].
Statistical Analysis
Quantitative variables were expressed as mean ± standard deviation (SD), and continuous variables were analyzed using the Student t-test. The VR time was calculated from the start of NA treatment to the first date when HBV DNA was not detected. Logistic regression analysis was used to explore the influencing factors of VR and HBeAg seroclearance. Odds ratio (OR) and 95% confidence intervals (95% CI) were calculated to assess relative risk and confidence intervals. p < 0.05 was considered statistically significant. Receiver-operating characteristic (ROC) curves were created to assess the diagnostic power of various serum biomarkers. Moreover, the above statistical analyses were performed in strict accordance with the statistical analysis methods using GraphPad Prism (version 8.0, GraphPad software) and the Statistical Package for Social Sciences (SPSS) (version 25.0).
Results
Baseline Characteristics of Patients
Of the initial 268 patients who were HBeAg-seropositive who met the inclusion criteria, 166 patients were excluded after screening for previous received therapy (n = 37), decompensated liver cirrhosis (n = 19), hepatocellular carcinoma (HCC) (n = 28), pregnancy (n = 26), co-infected with HAV, HCV, HEV, HIV and autoimmune diseases (n = 18) as well as incomplete data (n = 38). The flowchart is shown in Fig. 1. Finally, 102 patients who were HBeAg seropositive and had completed the 96-week treatment were included for analysis in this study.
The baseline characteristics of the study population are summarized in Table 1. Of all patients, 62.7% (n = 64) were men, and the age ranged from 18 to 62 years. There were 40, 49 and 13 patients using ETV, TDF and TAF, respectively. The mean baseline levels of ALT, HBV DNA and HBsAg were 52.69 ± 24.91 U/l, 6.53 ± 1.72 log10 IU/ml and 3.67 ± 0.87 log10 IU/ml, respectively. At baseline, 51.94% (n = 40), 18.2% (n = 14), 18.2% (n = 14) and 11.7% (n = 9) patients were in stage F0/F1, F2, F3 and F4 of liver stiffness assessment, respectively.
Clinical Outcomes of Patients During NA Treatment
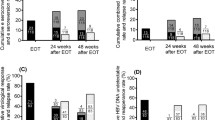
After 96-week treatment, 86 patients (84.3%) achieved virological response, 27 (26.5%) gained serum HBeAg seroclearance, and 21 (20.6%) achieved HBeAg seroconversion, respectively. No patient showed HBsAg loss. Furthermore, there was no significant difference between patients with normal or mildly elevated ALT during the treatment (normal ALT < 40U/l; 40 U/l ≤ mildly elevated ALT < 80 U/l, Fig. 2a–c).
Dynamic Changes of Viral Markers During NA Treatment
The serum markers of patients during treatment are presented in Table S1. The levels of ALT (baseline vs. 48 weeks vs. 96 weeks: 52.69 ± 24.91 vs. 26.81 ± 10.76 vs. 21.19 ± 10.12 U/l, p < 0.001), HBV DNA (baseline vs. 48 weeks vs. 96 weeks: 6.53 ± 1.72 vs. 1.79 ± 1.89 vs. 1.27 ± 1.11 log10 IU/ml, p < 0.001) and HBsAg (baseline vs. 48 weeks vs. 96 weeks: 3.67 ± 0.87 vs. 3.39 ± 0.81 vs. 3.26 ± 0.97 log10 IU/ml, p < 0.001) decreased significantly at week 48 and 96 compared to baseline, respectively. The level of HBV DNA decline in normal or mildly elevated ALT groups shows no difference (Figure S1). Likewise, FibroScan showed significant improvement in liver stiffness values at 48 weeks (6.33 ± 2.16 kPa) and 96 weeks (6.17 ± 1.91 kPa), respectively. Among 68 patients with pair values of liver stiffness assessment before and after treatment, the liver stiffness values decreased significantly after 48 weeks of treatment in patients with VR (Fig. 3a, p < 0.001). Meanwhile, 15 of 20 patients with liver stiffness values in stage F3 or F4 had improved to stage F0 to F2 (Fig. 3b).
Analysis of Factors Associated with Virological Response and HBeAg Seroclearance
As shown in Table 2, univariate analysis indicated that baseline AST levels (OR 1.072, 95% CI 1.019–1.127, p = 0.007), HBV DNA levels (OR 0.536, 95% CI 0.337–0.852, p = 0.008) and HBsAg levels (OR 0.070, 95% CI 0.016–0.299, p < 0.001) were closely associated with VR, whereas other clinical variables, including age, treatment strategy and liver stiffness value, did not show significant difference. Subsequent multivariate analysis showed that baseline AST levels (OR 1.069, 95% CI 1.014–1.127, p = 0.014), HBV DNA (OR 0.540, 95% CI 0.309–0.946, p = 0.031) and HBsAg (OR 0.147, 95% CI 0.036–0.597, p = 0.007) were significantly independently correlated with VR. However, no baseline factor was found to be significantly associated with 96-week HBeAg seroclearace (Table S2).
Analysis of the Predictors of Virological Response
Baseline levels of AST, HBV DNA and HBsAg for predicting VR were assessed by receiver-operating characteristic (ROC) analysis. As illustrated in Fig. 4a–c, the area under the ROC curve (AUROC) of AST, HBV DNA and HBsAg for predicting 96-week VR was 0.774 (95% CI 0.651–0.897), 0.755 (95% CI 0.618–0.894) and 0.902 (95% CI 0.788–1.00), respectively (all p < 0.05). Obviously, HBsAg was the strongest predictor for VR. Using maximized Youden’s index, an optimal cut-off value of 4.63 log10 IU/ml was defined for HBsAg to predict VR, with the sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of 96.5%, 81.3%, 96.5% and 81.3%, respectively (Table S3). Furthermore, the optimal cut-off values of AST and HBV DNA were 32 U/l and 7.92 log10 IU/ml, respectively.
By using the cut-off values as mentioned, the VR rates were significantly higher in patients with AST ≥ 32 U/l (95.0% vs. 69.0%, p = 0.01), HBV DNA < 7.92 log10 IU/ml (93.3% vs. 59.3%, p < 0.001) and HBsAg < 4.63 log10 IU/ml (97.3% vs. 18.6%, p < 0.001) compared to those with AST < 32 U/l, HBV DNA ≥ 7.92 log10 IU/ml and HBsAg ≥ 4.63 log10 IU/ml, respectively (Fig. 4d–f).
Discussion
This is a real-world study on patients with HBeAg-seropositive CHB with ALT < 2 ULN. This article demonstrates that NA therapy is effective in suppressing HBV DNA and greatly improves liver fibrosis. It also shows that ALT levels do not affect the effectiveness of antiviral treatment.
In clinical practice, VR is a very important indicator for evaluating efficacy. This study showed a VR rate of 84.3% after 96 weeks of treatment, which was similar to patients with HBeAg-seropositive CHB with ALT > 2 × ULN, approximately 67–89% [15,16,17]. The results showed that NA treatment could effectively suppress HBV viral replication in patients with CHB despite ALT levels < 2 × ULN. Importantly, the existence of serum HBV DNA levels is a strong predictor of risk for HCC [18, 19], and the decline of DNA levels could reduce the incidence of HCC risk [20]. It can be shown that decreasing viral loads can benefit patients by reducing the occurrence of adverse events.
On the other hand, this study also identified baseline HBsAg levels rather than ALT levels as a strong predictor of VR at 96 weeks through ROCs, which are significant for guiding clinical treatment. Similar arguments have been made in previous articles [21, 22], which may correlate with decreased HBsAg levels with decreased levels of cccDNA [22] (covalently closed cyclic DNA, the nuclear form of hepatitis B virus). In our study, the VR rates of the two groups with 4.63 log10 IU/ml as the boundary are significantly different (P < 0.05). Therefore, for patients who are HBeAg seropositive, treatment with HBsAg < 4.63 log10 IU/ml is worthwhile, regardless of the ALT level.
It is well known that the immune tolerance (IT) phase is characterized by high HBV DNA levels and low DNA levels. Therefore, effective treatment may break immune tolerance to some extent [23]. Previous major guidelines used ALT and HBV DNA to define the IT phase, but the standards were ambiguous. This study provides a more accurate cut-off value for defining the IT phase. Moreover, this may be a major innovation of this study.
In previous studies, patients who were HBeAg seropositive achieved a 67% VR rate and a 21% HBeAg seroconversion rate after 1 year of treatment [24], and 23% of the patients had HBeAg seroconversion after 5 years of treatment [25]. Although people, races and regions differ, our study has similar outcomes: 26.5% (n = 27) patients achieved HBeAg seroclearance and 20.6% (n = 21) patients HBeAg seroconversion. Patients with early seroconversion have a lower incidence of adverse clinical outcomes (cirrhosis, HCC or liver-related death) [7, 26, 27]. Therefore, it may be necessary for patients who are HBeAg seropositive with low ALT levels to take antiviral treatment.
We acknowledge that ALT is associated with host innate immunity, and elevated ALT may elicit potent host immunity and shows enhanced therapeutic efficacy [28, 29]. However, in this study, the normal and mildly elevated ALT groups achieved similar therapeutic outcomes. The results were not statistically different, so patients with normal or mildly elevated ALT were recommended to take antiviral therapy. Besides, our research also shows that after 96 weeks of treatment, patients obtained histological improvement, which was statistically significant.
The reason for not recommending treatment in previous studies is that VR rate was not ideal before 48 weeks because of the short follow-up time and insufficient data [4, 30, 31]. The results of our study were similar; however, good therapeutic results can be achieved after 96 weeks. Another reason is that ALT levels have previously been used as a surrogate marker to assess hepatocellular injury. Recent studies have shown that patients with normal and mildly elevated ALT have varying degrees of inflammation or fibrosis in the liver [32, 33]. Furthermore, a South Korean study with a median follow-up of 6.3 years shows that untreated patients with normal ALT (females, < 19 IU/ml; males, < 30 IU/ml) were at higher risk of clinical outcomes compared to treated patients with ALT ≥ 80 IU/ml. They were all patients with HBeAg-seropositive CHB with high HBV DNA levels (≥ 20,000 IU/ml) [34]. Therefore, it is recommended that patients with HBeAg-seropositive CHB with ALT < 2 × ULN be treated with antiviral therapy to suppress viral replication, reduce liver inflammation and ultimately slow down the progression of the disease.
This study had several limitations. First, the follow-up period was not long enough, only 96 weeks, and the long-term clinical outcomes of the patients (such as cirrhosis and HCC) could not be observed. However, this study used VR and HBeAg seroclearance rates as indicators of disease progression. Second, such patients are less likely to have received antiviral therapy in the past because of the current guidelines. Therefore, the data volume in this retrospective study is not immense. Finally, considering the risk and cost of repeated liver biopsy, our research used a non-invasive liver stiffness scan and not liver biopsy for the assessment of liver fibrosis in patients.
Conclusions
NA treatment was not only effective for HBV DNA suppression in patients with HbeAg-seropositive CHB with ALT < 2 × ULN but also significantly improved liver fibrosis. Meanwhile, there was no significant difference in the VR rate, HBeAg seroclearance and seroconversion between the normal ALT group and mildly elevated ALT group. This article provides real-world clinical data for the current China Guidelines and evidence for expanded treatment indications.
References
European Association for the Study of the Liver. EASL 2017 clinical practice guidelines on the management of hepatitis B virus infection. J Hepatol 2017;67(2):370–98.
World Health Organization. Global hepatitis report 2017. World Health Organization (2017). https://apps.who.int/iris/handle/10665/255016.
Howell J, Pedrana A, Schroeder S, Scott N, Aufegger L, Atun R, et al. A global investment framework for the elimination of hepatitis B. J Hepatol. 2021;74(3):535–49.
Wu I, Lai C, Han S, Han K, Gordon S, Chao Y, et al. Efficacy of entecavir in chronic hepatitis B patients with mildly elevated alanine aminotransferase and biopsy-proven histological damage. Hepatology (Baltimore, MD). 2010;51(4):1185–9.
Wong V, Hui A, Wong G, Chan R, Chim A, Lo A, et al. Four-year outcomes after cessation of tenofovir in immune-tolerant chronic hepatitis B patients. J Clin Gastroenterol. 2018;52(4):347–52.
Lv G, Ma W, Ying L, Jin X, Zheng L, Yang Y. Efficacy of telbivudine in HBeAg-positive chronic hepatitis B patients with high baseline ALT levels. World J Gastroenterol. 2010;16(32):4095–9.
Chu C, Hung S, Lin J, Tai D, Liaw Y. Natural history of hepatitis B e antigen to antibody seroconversion in patients with normal serum aminotransferase levels. Am J Med. 2004;116(12):829–34.
Sarin S, Kumar M. Should chronic HBV infected patients with normal ALT treated: debate. Hepatol Int. 2008;2(2):179–84.
Liu N, Yang N, Ma W, Yang S, Hu C, Li J, et al. Efficacy of antiviral treatment in liver biopsy-proven immune-tolerant chronic hepatitis B patients: a retrospective cohort study. Front Med. 2021;8: 655530.
Zhao Q, Liu K, Zhu X, Yan L, Ding Y, Xu Y, et al. Anti-viral effect in chronic hepatitis B patients with normal or mildly elevated alanine aminotransferase. Antiviral Res. 2020;184: 104953.
Ji J, Gao J, Wang C, Ouyang L, Liu Z, Liu Z. Characteristics and outcome of exertional heatstroke patients complicated by acute hepatic injury: a cohort study. J Clin Transl Hepatol. 2021;9(5):655–60.
Jeng W, Lok A. Should treatment indications for chronic hepatitis B be expanded? Clin Gastroenterol Hepatol. 2021;19(10):2006–14.
Jia W, Song L, Fang Y, Wu X, Liu D, Xu C, et al. Antibody to hepatitis B core antigen levels in the natural history of chronic hepatitis B: a prospective observational study. Medicine. 2014;93(29): e322.
Sandrin L, Fourquet B, Hasquenoph J, Yon S, Fournier C, Mal F, et al. Transient elastography: a new noninvasive method for assessment of hepatic fibrosis. Ultrasound Med Biol. 2003;29(12):1705–13.
Gish R, Lok A, Chang T, de Man R, Gadano A, Sollano J, et al. Entecavir therapy for up to 96 weeks in patients with HBeAg-positive chronic hepatitis B. Gastroenterology. 2007;133(5):1437–44.
Liu Y, Corsa A, Buti M, Cathcart A, Flaherty J, Miller M, et al. No detectable resistance to tenofovir disoproxil fumarate in HBeAg+ and HBeAg− patients with chronic hepatitis B after 8 years of treatment. J Viral Hepatitis. 2017;24(1):68–74.
Chan H, Fung S, Seto W, Chuang W, Chen C, Kim H, et al. Tenofovir alafenamide versus tenofovir disoproxil fumarate for the treatment of HBeAg-positive chronic hepatitis B virus infection: a randomised, double-blind, phase 3, non-inferiority trial. Lancet Gastroenterol Hepatol. 2016;1(3):185–95.
Zhang Q, Peng H, Liu X, Wang H, Du J, Luo X, et al. Chronic hepatitis B infection with low level viremia correlates with the progression of the liver disease. J Clin Transl Hepatol. 2021;9(6):850–9.
Chan H, Tse C, Mo F, Koh J, Wong V, Wong G, et al. High viral load and hepatitis B virus subgenotype ce are associated with increased risk of hepatocellular carcinoma. J Clin Oncol. 2008;26(2):177–82.
Paik N, Sinn D, Lee J, Oh I, Kim J, Kang W, et al. Non-invasive tests for liver disease severity and the hepatocellular carcinoma risk in chronic hepatitis B patients with low-level viremia. Liver Int. 2018;38(1):68–75.
Shin J, Jung S, Park B, Kim C, Eum J, Kim B, et al. Prediction of response to entecavir therapy in patients with HBeAg-positive chronic hepatitis B based on on-treatment HBsAg, HBeAg and HBV DNA levels. J Viral Hepatitis. 2012;19(10):724–31.
Chan H, Wong V, Tse A, Tse C, Chim A, Chan H, et al. Serum hepatitis B surface antigen quantitation can reflect hepatitis B virus in the liver and predict treatment response. Clin Gastroenterol Hepatol. 2007;5(12):1462–8.
Kim J, Ghosh A, Ayithan N, Romani S, Khanam A, Park J, et al. Circulating serum HBsAg level is a biomarker for HBV-specific T and B cell responses in chronic hepatitis B patients. Sci Rep. 2020;10(1):1835.
Cai S, Yu T, Jiang Y, Zhang Y, Lv F, Peng J. Comparison of entecavir monotherapy and de novo lamivudine and adefovir combination therapy in HBeAg-positive chronic hepatitis B with high viral load: 48-week result. Clin Exp Med. 2016;16(3):429–36.
Chang T, Lai C, Kew Yoon S, Lee S, Coelho H, Carrilho F, et al. Entecavir treatment for up to 5 years in patients with hepatitis B e antigen-positive chronic hepatitis B. Hepatology (Baltimore, MD). 2010;51(2):422–30.
Chen Y, Chu C, Liaw Y. Age-specific prognosis following spontaneous hepatitis B e antigen seroconversion in chronic hepatitis B. Hepatology (Baltimore, MD). 2010;51(2):435–44.
Chen J, Yang H, Iloeje U, You S, Lu S, Wang L, et al. Carriers of inactive hepatitis B virus are still at risk for hepatocellular carcinoma and liver-related death. Gastroenterology. 2010;138(5):1747–54.
Chang M, Liaw Y. Hepatitis B flare in hepatitis B e antigen-negative patients: a complicated cascade of innate and adaptive immune responses. Int J Mol Sci. 2022;23(3):1552.
Su X, Chen H, Zhu Z, Xie W, Peng J, Ma X, et al. Clinical diagnostic value of quantitative hepatitis B virus core antibody test in chronic viral hepatitis B. Bioinorg Chem Appl. 2021;2021:3720571.
Kwon J, Song M, Jang J, Bae S, Choi J, Yoon S, et al. Efficacy and safety of tenofovir disoproxil fumarate in treatment-naïve patients with chronic hepatitis B in Korea. Dig Dis Sci. 2019;64(7):2039–48.
Suzuki F, Suzuki Y, Hosaka T, Sezaki H, Akuta N, Fujiyama S, et al. Efficacy of long-term tenofovir-based rescue therapy in patients with chronic hepatitis B refractory to nucleoside/nucleotide analogs. J Gastroenterol. 2017;52(5):641–51.
Chen J, Xu C, Xi M, Hu W, Tang Z, Zang G. Predictors of liver histological changes and a sustained virological response to peginterferon among chronic hepatitis B e antigen-positive patients with normal or minimally elevated alanine aminotransferase levels. J Viral Hepatitis. 2017;24(7):573–9.
Wang H, Xue L, Yan R, Zhou Y, Wang M, Cheng M, et al. Comparison of histologic characteristics of Chinese chronic hepatitis B patients with persistently normal or mildly elevated ALT. PLoS ONE. 2013;8(11): e80585.
Kim GA, Lim YS, Han S, Choi J, Shim JH, Kim KM, et al. High risk of hepatocellular carcinoma and death in patients with immune-tolerant-phase chronic hepatitis B. Gut. 2018;67(5):945–52.
Acknowledgements
We appreciate the contributions of all patients and support of Nanfang Hospital and Shunde Hospital affiliated with Southern Medical University.
Funding
This research, and the journal’s Rapid Service Fee, was supported by the grants from the National Natural Science Foundation of China (81971949) and Clinical Research Program of Nanfang Hospital, Southern Medical University (no. 2018CR026).
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, had full access to the final version of the manuscript and agreed to its submission.
Author Contributions
Meixin Hu, Guichan Liao, and Jie Peng were involved in the study design. Meixin Hu and Sufang Wei collected the data. Meixin Hu, Guichan Liao, Muye Xia and Hongjie Chen analyzed and interpreted the data. Meixin Hu, Zhe Qian and Guichan Liao wrote the manuscript.
Disclosures
All named authors confirm that they have no financial and personal relationships with other persons or organizations that could improperly influence this research.
Compliance with Ethics Guidelines
The study was approved by the Institutional Ethics Committee of Nanfang Hospital (study identifier: NFEC-2020-290) and Shunde Hospital (study identifier: 20211129), and the study protocol was performed in accordance with the Helsinki Declaration of 1964 and its later amendments. All patients have details from their medical records published anonymously.
Data Availability
All data and analysis results are included in this article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Hu, M., Liao, G., Wei, S. et al. Effective Analysis of Antiviral Treatment in Patients with HBeAg-Seropositive Chronic Hepatitis B with ALT < 2 Upper Limits of Normal: A Multi-center Retrospective Cohort Study. Infect Dis Ther 12, 637–647 (2023). https://doi.org/10.1007/s40121-022-00757-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40121-022-00757-y