Abstract
Invasive meningococcal disease (IMD) outbreaks associated with Hajj and Umrah pilgrimage events in the Kingdom of Saudi Arabia (KSA) are well recognized. Past outbreaks have been associated with substantial intercontinental spread of specific Neisseria meningitidis serogroups. The emergence of meningococcal serogroup W (MenW) was a global concern following the 2000/2001 Hajj outbreaks. Broader compulsory meningococcal serogroups A, C, W and Y (MenACWY) immunization strategies for pilgrims were introduced in response to these events and led to substantial declines in IMD cases associated with these mass gatherings. However, there remains potential for future outbreaks either within KSA during the Hajj or in local populations via pilgrim meningococcal transmission on their return. While the annual Hajj involves pilgrims from over 185 countries, two-thirds of these arrive from 13 countries, chiefly from across South-East Asia, the Middle East and North African (MENA) regions; for which we review the relevant epidemiology of IMD and meningococcal carriage. While disease surveillance is limited and data are often lacking, MenB is an important serogroup associated with IMD and carriage in a number of countries. Available literature suggests that most pilgrims receive polysaccharide MenACWY vaccines (which do not impact carriage and onward transmission) and incomplete compliance with visa/entry immunization regulations is reported. Existing preventative approaches for visiting pilgrims require continued oversight. More complete compliance and switching to the conjugated MenACWY vaccine can provide more robust and broader protection for pilgrims. Additional immunization options could also be considered.
Graphical Plain Language Summary

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Attendance at Hajj or Umrah pilgrimage events is a well-recognized risk in the relative importance of specific serogroups in local population carriage and Invasive meningococcal disease (IMD) epidemiology. |
Compulsory immunization against MenACWY serogroups for pilgrims has led to a substantial decline in IMD, although there remains the potential for future outbreaks during the Hajj. |
More complete compliance with immunization requirements and switching to the conjugated MenACWY vaccine can provide broader and more robust protection. |
Digital Features
As well as a Graphical Plain Language Summary, an audio narration of this feature from the authors is also available. To access the audio narration alongside the Graphical Plain Language Summary for this article, go to https://doi.org/10.6084/m9.figshare.19298804.
Introduction
Mass gatherings and those associated with religious events such as the Hajj and Umrah pilgrimages to Makkah, Kingdom of Saudi Arabia (KSA), are well recognized risk factors for Neisseria meningitidis (meningococcal) transmission and subsequent invasive meningococcal disease (IMD) outbreaks [1,2,3,4]. The annual Hajj pilgrimage lasts for 5 days (from day 8 to day 12 or 13 during the 12th month of the Islamic lunar calendar). Over 1.5 million external pilgrims (from more than 184 countries) enter the KSA to attend each year, with 1.86 million external pilgrims visiting in 2019 [2, 5, 6]. For Umrah, visitors attend in far greater numbers (7.2 million in 2019), although attendance is across a longer period with a peak during the month of Ramadan (almost 2 months before Hajj season) [7]. The often intense crowding that exists in the Hajj and Umrah setting, with large group congregation for prayers and with pilgrims sharing accommodations and dining facilities, and the broad mix of individuals from diverse geographical origins and the local KSA population all contribute to enhanced risk of meningococcal transmission [8, 9].
While 12 N. meningitidis serogroups have been identified [10], six serogroups are responsible for the great majority of IMD (serogroups A [MenA], B [MenB], C [MenC], W [MenW], X [MenX] and Y [MenY]) [11,12,13]. Substantial geographical variation exists in the relative importance of specific serogroups in local population carriage and IMD epidemiology. Consequently, the admixture of pilgrims from different countries attending the Hajj is associated with the circulation and exposure to a diverse range of serogroups across the broader pilgrim population. Due to the different epidemiological patterns within their country and disease unpredictability, some serogroups will be relatively unfamiliar to pilgrims from certain countries (in the sense of pilgrims’ local prevalent epidemiology), with the potential for carriage acquisition of unfamiliar meningococcal serogroups and subsequent onward transmission to their broader local population upon their return [5, 14]. Obviously, there is also the potential for IMD outbreaks, either at and during the Hajj or in the pilgrims’ homeland upon their return.
Previous IMD outbreaks associated with Hajj events have substantially impacted the global IMD epidemiology. Notably, the outbreaks in 2000 and again (although of lesser magnitude) in 2001, due predominantly to MenW, resulted in substantial pilgrim mortality [15]. They also served as the origin of subsequent intercontinental spread via pilgrims acting as a transmission vector on their return home. Indeed, this was the principal step in the emergence of MenW (previously of relatively limited concern) as an important global IMD serogroup [16,17,18], compounded by the emergence of a separate South American strain in 2003 [18].
This, in turn, has led to the implementation of comprehensive vaccination strategies (via quadrivalent MenACWY vaccine for pilgrims entering KSA and also for KSA residents) and other measures such as chemoprophylaxis to mitigate further outbreak risk [19]. As we outline below, while these have led to considerable success, some limitations remain in the current immunization approaches, and there remains an ongoing risk for future outbreaks arising at or from the Hajj [5, 20].
In the present manuscript, we provide a narrative overview of key lessons from past pilgrim IMD outbreaks and studies of meningococcal carriage, and then we focus on relevant epidemiologic aspects of IMD and carriage in those countries from which the majority of external pilgrims originate. This provides a perspective on the existing immunization strategies and how these could be strengthened to provide broader and more complete protection for pilgrims.
Data Sources
As a first step, we utilized data reported by the KSA Ministry of Health for the 2019 Hajj [6], where additional information supplied indicates that nearly two-thirds of visiting pilgrims arrive from 13 countries chiefly from across Southeast Asia and the Middle East and North Africa (MENA) region; Indonesia, India, Pakistan, Bangladeshi, Iran, Egypt, Turkey, Iraq, Algeria, Nigeria, Morocco, Sudan and Malaysia. We then searched PubMed to identify relevant literature on IMD and N. meningitidis carriage associated with Hajj or Umrah pilgrimage from these countries, along with articles reporting on these same characteristics within each of these countries. The search strategy included a range of free-text and Medical Subject Headings (MeSH) search terms, e.g., “Hajj”, “Umrah”, ‘‘meningococcal”, ‘‘N. meningitidis” and “carriage” and specific countries in various combinations. The identified articles were supplemented by snowball sampling of relevant citations from the identified papers (including a large number of comprehensive, systematic reviews). Our search covered the period through March 2020, and although comprehensive, our approach was not systematic per se; we did not have specific selection criteria or specific publication date limitations. Our aim was to identify the most relevant data for each specific country. Although for some, the available literature is limited and often rather outdated, whenever possible we focus on the more recent published data for each country.
In compliance with ethical guidelines, this article is based on previously conducted studies and reported data and does not contain any studies with human participants or animals performed by any of the authors.
IMD Hajj Outbreaks and Subsequent Immunization Policy
IMD outbreaks during the Hajj and responsive immunization strategies for at-risk pilgrims have been reviewed extensively [5, 14,15,16, 19,20,21]; a schematic of key events is shown in Fig. 1.
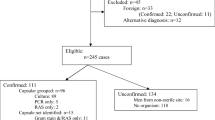
Awareness of the significance of Hajj as a focus for local disease and subsequent international spread was raised following MenA outbreaks in 1987 [22, 23]. This prompted the introduction of mandatory immunization against MenA for pilgrims arriving from the high-risk sub-Saharan meningitis belt countries in 1987, using a bivalent MenA-C vaccine, a measure extended in 1988 to include all visiting Hajj pilgrims [19]. In the same year, mass chemoprophylaxis with ciprofloxacin for pilgrims from sub-Saharan meningitis belt countries to reduce meningococcal nasopharyngeal carriage was also introduced (and continues to the present time). Following Umrah-related MenA outbreaks in 1992 (chiefly in unvaccinated pilgrims) [23], mandatory immunization was implemented for all pilgrims entering KSA [19]. Nevertheless, IMD outbreaks due to various serogroups continued to occur. In their extensive analysis of IMD surveillance within KSA across 1995–2011, Memish and colleagues reported that between 1995 and 1999, a total of 265 IMD cases occurred within KSA, of which 60 involved Hajj pilgrims and 46 involved Umrah pilgrims; most IMD was due to MenA (49% of serogrouped cases), although MenB (26%) and MenW (20%) were also noted [15].
The subsequent 2000 and 2001 outbreaks involved a total of 654 IMD cases, of which 249 (38.1%) occurred in external pilgrims, involving 223 Hajj pilgrims and 26 Umrah pilgrims, with the remainder chiefly occurring in KSA citizens/residents [15]. The IMD case fatality ratio (CFR) was substantially higher in external pilgrims than in KSA citizens/residents, and so the majority of IMD deaths in the 2000–2001 outbreaks were in visiting external pilgrims. This may be due in part to the delay in diagnosis and medical care in pilgrim patients on pilgrimage, and also the generally greater age of pilgrims than that of affected residents [15]. Although the great majority of IMD cases in 2000–2001 were due to MenW (78% of serogrouped cases), IMD due to MenA was also reported (accounting for 20% of serogrouped cases). Most MenA cases were observed in 2000, where in reality there was a MenA outbreak at the same time as the larger MenW outbreak. Six cases of MenB were also reported during these years [15]. These latter outbreaks led to greater reinforcement of the existing immunization requirements. Immediately following the 2000 outbreak, a shift towards a broader protection policy utilizing the quadrivalent MenACWY vaccine was partially implemented, but supply issues seriously hampered availability for pilgrims (which may have contributed to the later 2001 Hajj outbreak). Use of the MenACWY vaccine was compulsory from 2002 onwards for all pilgrims, and while initially only available as a polysaccharide vaccine, the use of a conjugated MenACWY vaccine has been widely available since 2010 (although, as we describe below, pilgrim uptake is low). In tandem with the external pilgrimage policy, expansion of immunization activities for all KSA citizens/residents with pilgrim interactions and introduction of infant quadrivalent MenACWY vaccination has occurred [19, 24].
At the present time, while both polysaccharide and conjugated ACWY vaccines are valid for external pilgrims, policy differences exist in the duration of validity of the vaccination. While pilgrims immunized with conjugated MenACWY vaccines must show evidence of vaccination within the past 5 years, those receiving the polysaccharide vaccine must show evidence of immunization within the past 3 years [4]. The requirement for chemoprophylaxis for all pilgrims arriving from the sub-Saharan meningitis belt countries remains in place. These protocols, along with continued expansion and widespread availability of conjugated MenACWY immunization for all at-risk KSA citizens/residents, have led to substantial success in curtailing further disease outbreaks amongst Hajj/Umrah pilgrims, with no outbreaks since 2001 [19]. This has been accompanied by a substantial decline in IMD cases within the KSA population over the past two decades [15, 24]. However, one cannot be complacent, for a number of reasons.
Firstly, although outbreaks have been curtailed, sporadic cases continue to occur, with 14 IMD cases reported in pilgrims attending Hajj between 2002 and 2011 [15]. Onward transmission from pilgrims is also observed, most recently with reports in 2019 of meningococcal infection due to a ciprofloxacin-resistant non-groupable N. meningitidis strain in two Umrah pilgrims returning to the United Kingdom (UK). A further close contact in the UK subsequently developed IMD due to this same strain [25, 26].
Secondly, the current strategies require strengthening. Conjugated MenACWY vaccines are preferable to polysaccharide vaccines (due to the induction of immunologic memory and the potential for reduction in nasopharyngeal carriage acquisition and onward transmission [27, 28]). Expert groups have called for the use of conjugated vaccines by pilgrims to be made a priority, especially for those pilgrims making regular visits, as repeated polysaccharide immunization may lead to weaker immune responses [29, 30]. However, this has not been formally actioned, and data indicate that most pilgrims receive the polysaccharide vaccine. In their 2014 study (published in 2017), Memish et al. reported that 98.2% of the 1055 arriving pilgrims (most from countries we also describe below) had received the polysaccharide vaccine, and the remainder had received a conjugate vaccine [31]. A more recent 2017 study (published in 2021) involving 2973 pilgrims arriving from a wide range of countries reported that between 60 and 70% of vaccinated pilgrims arriving from low- and middle-income countries (LIMCs) had received polysaccharide MenACWY vaccine [32]. Perhaps more worrying was the high proportion of unvaccinated pilgrims from LIMCs (20–25%) [32]. An earlier survey from 2009 reported that only 75% of pilgrims had received meningococcal vaccination before travel [33], while a more recent survey involving 1012 Umrah pilgrims visiting in 2019 reported that only 41.3% had actually received meningococcal vaccination [34]. While other data report 100% compliance in pilgrims from most countries surveyed in 2013 and 2014 [31, 35], these more recent data suggest a need to strengthen oversight of the existing regulations for pilgrims [32]. Domestic pilgrim vaccination (i.e., in KSA residents/citizens attending the Hajj) is also suboptimal. A recent study of immunization status in external and domestic pilgrims reported that 81.7% of domestic pilgrims had confirmed meningococcal vaccination status compared with 93.5% of overseas pilgrims [36]. Vaccine uptake by healthcare workers (HCWs) working/volunteering at the Hajj is inadequate, with one study reporting only 76.1% of HCWs surveyed between 2015 and 2017 indicating receipt of MenACWY [37].
Another issue is the role of chemoprophylaxis in pilgrims attending from sub-Saharan Africa, where concerns have been raised regarding the risk–benefit trade-off in continuing this policy, especially in light of increasing antimicrobial resistance [19, 31, 38]. Finally, there remains the potential for future Hajj IMD outbreaks and ongoing intercontinental spread due to serogroups not currently covered by current mandatory immunization (i.e., MenB and MenX) [14, 39]. Protein-based vaccines (4CMenB and MenB-FHbp) against a range of subcapsular antigens conferring protection against MenB are now available [12, 40, 41], although MenB vaccination is not included in current immunization strategies for individuals entering KSA for Hajj and Umrah pilgrimage [4, 20, 42].
Meningococcal Carriage
The intense social crowding and congregation that accompany the Hajj are recognized factors associated with higher rates of meningococcal transmission and carriage acquisition [30]. A recent review of carriage studies reported that meningococcal carriage is higher in the holy cities of Mecca and Medina compared to other KSA cities (e.g., Riyadh), and in general, carriage is greater in Hajj pilgrims compared with Umrah pilgrim populations [9]. This same review also noted that most studies in pilgrim populations report comparable carriage rates to those reported in other settings, although factors influencing carriage such as antibiotic use will have an impact [9]. Onward transmission to close contacts of returning pilgrims is well documented, with household contact carriage rates ranging between 8 and 25% [9]. While substantial temporal variation exists in carriage of specific serogroups, the broad picture is the predominance of MenW and MenB in carriage isolates [9].
Quantifying the impact of Hajj attendance on carriage is complex. A number of studies conducted in 2001, 2014 and 2017 specifically examined carriage rates in pilgrims before and after Hajj participation [31, 32, 43]. Carriage patterns from pilgrims in specific countries are described later, but these studies report overall carriage rates ranging from 3.4 to 8.0% in pilgrims prior to Hajj attendance, and between 1.2 and 10.4% in departing pilgrims. Two of these studies analysed carriage loss and acquisition during the Hajj using a paired-sample approach. In the earlier 2001 study, a broad pattern of higher carriage rates was seen in departing pilgrims, with some evidence of increased acquisition in pilgrims from countries with low carriage prior to the Hajj, such as among Malaysian and Indonesian pilgrims. MenW accounted for a far higher proportion of isolates identified in pilgrims after the Hajj prior to their departure home than that identified from pilgrims on arrival for the Hajj (44.2% and 5.3%, respectively) [43]. This is in keeping with the understanding that acquisition and onward transmission of MenW carriage was the principal driver of subsequent intercontinental spread. The later 2014 study found higher carriage among pilgrims arriving from countries with high endemic disease (although the 6.3% carriage rate was considered low) and generally lower carriage in departing pilgrims [31]. An important observation from this later study was that, while carriage was relatively low, there was a high prevalence of MenB isolates, accounting for 66.7% of all positive samples on arrival and for 100% of those from departing pilgrims [31]. A second important point is that antibiotic use during Hajj either as a compulsory prophylaxis or taken for other reasons, including illness, and as a general precautionary measure during the pilgrimage is high [44], and this can impact/reduce carriage rates [31, 45]. Recent data indicate that 55.8% of pilgrims report antibiotic use during the Hajj [32].
Importance in Selected Countries
In 2019, a total of 2,489,406 pilgrims participated in Hajj activity, which included 1,855,027 external pilgrims [6]. Of these we selected those 13 countries that account for the greatest numbers of pilgrims travelling to KSA: Indonesia, India, Pakistan, Bangladesh, Iran, Egypt, Turkey, Iraq, Algeria, Nigeria, Morocco, Sudan and Malaysia (Fig. 2). These account for 65.4% of all external pilgrims. For each country, we describe the relevant epidemiological aspects, including principal IMD causative serogroups (Table 1), and available data for meningococcal carriage in the local resident population (Table 2) and in pilgrims from these countries before or after attending the Hajj (Table 3). While our purpose was to report all relevant serogroups, we also wanted to evaluate patterns of MenB disease or carriage, and so relevant data for MenB are presented in bold across these tables. Where possible, we focus on the most recent data, although for some countries data are sparse and epidemiologic data are often outdated.
Pilgrims attending Hajj in 2019. Data as reported [6] supplemented by additional information provided by the Ministry of Health
It should be noted that routine meningococcal vaccine programmes vary widely in those countries. In most countries there is no formal national immunization policy, the exceptions being Egypt (infant polysaccharide MenA–C vaccine) and Sudan and Nigeria (infant conjugated MenA vaccine as part of the broader regional efforts, although coverage data are lacking). Consequently, it is chiefly only those pilgrims from these countries participating in Hajj or Umrah activities who receive any form of meningococcal vaccine, and as outlined earlier, the great majority receive polysaccharide MenACWY [31, 32].
Indonesia
Epidemiology
IMD is considered as occurring only sporadically; incidence data are not formally reported [46, 47]. Limited data indicate that IMD represents approximately 17% of such cases reported in children (< 5 years) in studies spanning 1995–2002, although absolute numbers are very low [48, 49], and broader population studies are lacking. In 2000, 14 IMD cases were reported to the World Health Organization (WHO), including one case due to MenB [47].
Carriage
While population carriage data are not reported, carriage data in Hajj pilgrims are available for 2001 and 2014 [31, 43]. For 2001, carriage was identified in 1.3% of Hajj pilgrims arriving from Indonesia prior to attendance, while unpaired sampling of pilgrims on departure returning to Indonesia indicated 7.7% carriage. MenW accounted for all identifiable strains (33.3% of positive samples), with the remainder being non-groupable [43]. In 2014, carriage was identified in 2/98 (2.0%) of the pilgrims sampled on arrival (both due to MenB). Paired post- versus pre-Hajj sampling found no evidence of carriage acquisition during the 2014 Hajj [31].
India
Epidemiology
Incidence data are limited, with recent and previous focused reviews unable to report formal incidence rates [50,51,52], although an incidence of 13.2 per 100,000 was reported by WHO in 2005 [47]. Surveillance limitations preclude accurate estimations of the burden of IMD in India [50]. Most data describe the relative proportions of IMD as the cause of bacterial meningitis, where the broad pattern is that pneumococcal and Haemophilus influenzae type b (Hib) meningitis are more common than meningococcal disease [50, 51]. IMD outbreaks are well documented (extensively reviewed by Sinclair et al. [51]), notably across 1985–1988, principally in northern India/New Delhi (> 6100 cases in 1985, with a CFR of 13%) but with smaller outbreaks across much of India, and then most recently in 2005–2008 in New Delhi (867 cases) and Meghalaya in the Northeast (2000 cases in 2008, with an estimated CFR of 10%) [51, 53]. The true magnitude of these outbreaks and resultant mortality is likely underestimated [51]. While both outbreaks were due to MenA [51, 53, 54], the principal serogroup identified across most other studies and disease reports [50], sporadic IMD cases and fatalities due to MenC and MenB are reported [47, 55]. Outbreaks have also been documented in military personnel sharing accommodation in 2006, with 17 cases occurring over 4 months (though most occurred within the first 3 weeks) [56].
Carriage
Population carriage data are limited. Carriage of 11.9% was found in military recruits aged 16–25 years sampled in 1994 [57], while 14.4% carriage was observed in randomly sampled soldiers during the field outbreak in 2006 (all MenA) [56]. In a 2014 study, 1.5% carriage was reported in college students living in dormitories in Kashmir, with all isolates being MenB [58].
Carriage data in Hajj pilgrims are available for 2001 and 2014 [31, 43]. For the 2001 Hajj, meningococcal carriage was identified in 13/61 of arriving Indian pilgrims (21.3%) prior to attendance; of these 13 isolates, MenB was the most frequent serogroup identified (30.8%). Unpaired sampling indicated carriage in 3/66 (4.5%) of pilgrims returning to India, all due to MenW [43]. In 2014, there was no carriage identified in any of the 100 pilgrims sampled on arrival, and none in any of the 73 pilgrims sampled on departure [31].
Pakistan
Epidemiology
Formal IMD incidence data are limited and outdated, with an incidence of 5.4 per 100,000 reported for 1995 [59]. Detailed serogroup information is lacking. Disease outbreaks are uncommon, the last being a MenA outbreak occurring in Karachi in 1988, with 122 cases chiefly occurring in children aged ≤ 10 years [60]. While some studies reporting on bacterial meningitis etiology in Pakistan indicate that pneumococcal and Hib meningitis are more frequent than IMD (especially in children) [61], others report IMD as an important cause; for example, N. meningitidis accounted for 31.5–36.7% of bacterial meningitis cases in studies conducted between 1995 and 2001 [62, 63] and 95% of cases in a later study (conducted between 2007 and 2012) [64]. Nevertheless, the absence of recent epidemiologic data precludes the ability to obtain reliable IMD incidence estimates or relative serogroup prevalence.
Carriage
Population carriage data are lacking. Carriage data in Hajj pilgrims are available for 2014, with carriage reported in 1/98 pilgrims sampled on arrival (1.0%), with MenB identified. No meningococcal carriage acquisition was observed during the Hajj [31].
Bangladesh
Epidemiology
Estimated incidence (based on limited data) is 1 per 100,000 [46]. IMD accounts for substantial proportions of meningitis in Bangladesh. One study reporting on meningitis hospital admissions between 2003 and 2005 reported that meningococcal disease represented 18% of all confirmed bacterial meningitis cases (with pneumococcal and Hib meningitis each accounting for 3%). The great majority of IMD cases were adults, with 65% ≥ 15 years of age [65]. MenA was the predominant serogroup identified, accounting for 121/139 (87.1%) of all IMD cases; MenB was reported in 4.3% [65]. A later broader study evaluating IMD causative serogroups between 1999 and 2006, reported that of 132 isolates analysed, 97.7% were due to MenA (and 2.3% MenB); 60% of all IMD isolates examined were in those aged ≥ 15 years of age [66]. Temporal patterns in case identification across both of these studies suggest that the far higher case numbers in 2002–2004 reflect MenB disease outbreaks in this period of cases [65, 66]. More recent data are lacking, although some authors indicate an increasing importance of MenB in Bangladesh [46].
Carriage
Population carriage data are lacking. Carriage data in Hajj pilgrims are available for 2001 and 2014 [31, 43]. For 2001, carriage was identified in 15/73 (20.5%) of pilgrims arriving from Bangladesh prior to Hajj (with MenB identified in two samples). Carriage was reported for 5/75 (6.7%) of departing pilgrims. MenB was identified in three of these samples (60%), while two samples were non-groupable [43]. In 2014, carriage was observed in 2/79 (2.5%) of arriving pilgrims, with MenB identified in both. Carriage was identified in 1/27 (3.7%, MenB) of returning pilgrims (again MenB), with no indication of carriage acquisition seen in the paired post- versus pre-Hajj sampling [31].
Iran
Epidemiology
IMD incidence rates are infrequently reported, although an incidence of 0.14 per 100,000 has been described (for 2005) [59]. A systematic review of studies published between 1994 and 2015 reported that N. meningitidis accounted for 13% of bacterial meningitis cases (with pneumococcal and Hib meningitis accounting for 30 and 15%, respectively) [67]. Serogroup data are limited. One study reported that of six IMD isolates serogrouped in military personnel in 2003–2006, MenC accounted for 83% and MenB for 17% [68]. A later study reporting on 16 isolates from IMD in children found the majority (51%) due to MenB, with MenC accounting for 24% [69].
Carriage
Population data indicate that carriage is relatively high. A study evaluating meningococcal carriage in 226 military recruits aged 19–28 years between 2015 and 2016 reported 8% carriage, chiefly MenC (50.0%); MenA accounted for 22.2% and MenY for 16.6% [70]. Another study evaluating 335 male dormitory students aged 18–34 years in 2017 and 2018 reported 6.9% carriage, chiefly MenC (43.5%), with the remainder non-groupable [71].
Although not included in the 2001 and 2014 studies described above [31, 43], carriage data in Iranian Hajj pilgrims are available from studies conducted before and after the Hajj in 2003 and 2012 [45, 72]. For 2003, of the 674 pilgrims sampled immediately prior to departure from Iran, 5.2% had meningococcal carriage [MenB (25.7% of strains); MenC (14.2%); MenW (2.9%); non-groupable (28.6%)]. The carriage rate of these same pilgrims on arrival in Iran post-Hajj was 4.6%; again, chiefly MenB (6.4% of strains), although far more isolates were non-groupable (67.7%) [45]. This same study also evaluated the impact of prophylactic ciprofloxacin on carriage rates in a study sub-cohort, where carriage rates fell from 8.1% (before leaving Iran) to zero (on their return) after receiving ciprofloxacin 24 h prior to departure [45]. In 2012, while there was no carriage identified in any of the 422 pilgrims sampled on arrival, 1.4% carriage was reported in departing pilgrims (although serogroup data were not reported) [72].
Egypt
Epidemiology
Formal IMD incidence data are limited and outdated, with an incidence of 0.1 per 100,000 reported for 2006 [59]. The introduction of infant MenA–C immunization in 1992 in response to previous outbreaks due to MenA has led to a relative decline in the prevalence of IMD and also a shift in causative serogroups [73, 74]. Neisseria meningitidis accounted for 18.9–38.2% of bacterial meningitis cases treated at a major hospital between 1997 and 2006, and while this constituted the major pathogen, the relative proportion was lower than that reported for 1988–1995 (35.2–72.5%) [75]. Serogroup data reported in studies characterizing IMD between 1998 and 2004 indicate that of 135 confirmed IMD cases, 51% of cases were due to MenB, 35% to MenA and 4% to MenW [76, 77].
Carriage
Population carriage data are scarce. Carriage data in Hajj pilgrims are available for 2014, where carriage was reported in 1/98 (1.0%) pilgrims on arrival (due to MenB). Paired pre- versus post-Hajj sampling found no evidence of carriage acquisition during Hajj attendance [31].
Turkey
Epidemiology
IMD is considered endemic, with an incidence rate ranging from 1 to 5.5 per 100,000 reported from 1985 to 2006 [78]. The robust surveillance system and frequent reporting of meningitis in Turkey provides clear evidence of shifts in bacterial meningitis etiology and in the relative importance of IMD causative serogroups [79,80,81,82,83]. Immunization activities directed against pneumococcal and Hib disease have led to a relative decline in bacterial meningitis due to these organisms, and while pneumococcal meningitis remains important, IMD has accounted for 70–90% of cases since 2013 [83]. Similarly, there has been a shift in IMD etiology, with a relative decline in disease due to MenW, and a far greater prevalence of MenB disease (increasing from 31.2% of serogrouped cases in 2005–2006 to 54.7% of serogrouped cases in 2017–2018) [83]. IMD due to MenB is also evident in refugee populations within Turkey, with three cases in children of Syrian/Turkoman origin reported since 2014 [84].
Carriage
Population carriage has been evaluated in a number of large-scale studies, each sampling > 1000 children or adults (Table 2) [85,86,87]. In 2001–2002, 6.2% carriage was found in schoolchildren; MenC (35.2%) was the most prevalent serogroup, followed by MenA (28.1%), MenB (22.5%) and MenW (11.2%) [85]. More recent studies indicate a decline in MenC carriage, and the increasing importance of MenB carriage. While a 2012–2013 study of adults found only 0.6% meningococcal carriage, all five of the isolates with available serogroup data were MenB [86]. A subsequent 2015 study reported 6.3% carriage in 1518 adolescents and young adults (aged 10–24 years). Carriage was greatest among subjects in the 21–24 age group (9.1%) and even higher (16.2%) in those aged 21–24 years who were a household contact of returning Hajj or Umrah pilgrims. MenW (66.6%) was the most prevalent serogroup, with 9.4% of isolates due to MenB [87].
Data for carriage in Turkish pilgrims are available for both the 2010 and 2018 Hajj [88, 89]. In 2010, carriage was identified in 13.3% of pilgrims prior to departure, the majority being MenW (82.5%) followed by MenB (14.3%). Following their return, 27.4% carriage was found: MenW (91.4%) and MenB (6.2%). MenW acquisition during the Hajj was observed in 13.2% of returning pilgrims who were negative before departure. This study also identified MenW carriage in 25.6% of household contacts of returning pilgrims [88]. For the 2018 Hajj, of 229 Hajj pilgrims aged 10–80 years (all of whom had received conjugated MenACWY vaccine at least 2 weeks prior to sampling), carriage was identified in 3.9% immediately prior to departure (all of which were MenB). Following their return, although overall carriage rates were not reported, paired sampling found MenB acquisition in 1/229 (0.4%) of returning pilgrims [89].
Iraq
Epidemiology
Data are limited, with reported incidence rates ranging from 5.9 to 30.4 per 100,000 between 1987 and 1991, while a far lower incidence was reported for later years: 0.18 per 100,000 reported for 2005 by WHO [59]. Serogroup data are also limited, although a small study evaluating pediatric meningitis cases identified N. meningitidis in 2/50 (4%) of cerebrospinal fluid samples; both isolates were MenW [90].
Carriage
Population data and data for pilgrim carriage are unavailable.
Algeria
Epidemiology
IMD has an estimated incidence rate of 8.0 per 100,000 reported for 2017 [91]. A study evaluating IMD cases between 1992 and 2001 reported that the majority of isolates were MenA (71.5%), with relatively similar proportions due to MenB (9.2%), MenC (9.2%) and MenW (8.5%) [92]. More recent data indicate a shift; a systematic review which reported on Algerian data from 2010, 2011 and 2015 found that MenB was the most frequently identified serogroup, accounting for 59.3% of IMD cases, with 11.9% due to MenW (and 10.8% and 6.4% due to MenA and MenC, respectively) [11]. Other recently reported data (for the 2003–2013 period) also indicate that MenB was the predominant strain isolated from IMD cases (46.7%), followed by MenW (14%) and MenC (8%) [93, 94].
Carriage
Population data and data for pilgrim carriage are unavailable.
Nigeria
Epidemiology
Although IMD incidence rates are not formally reported, the onset of recent outbreaks and epidemics in recent years (between 2014 and 2017) highlights the importance of IMD in Nigeria [95,96,97]. In regions most affected by recent outbreaks such as those in the Northwest, cumulative incidence rates exceeding 150 per 100,000 have been reported [96, 98]. The introduction of widespread MenA vaccination from 2013 onwards as part of the MenAfriVac campaign across the sub-Saharan meningitis belt has led to a decline in disease and disease outbreaks due to MenA, with a shift towards MenC as the major cause of IMD [95, 96]. While IMD due to MenW and MenA also occur, MenB disease is rarely reported [11, 96]. In the most recent epidemic, in late 2016–mid 2017 across northern Nigeria, the IMD burden comprised 14,518 suspected cases, with a CFR of 8%. Of 387 confirmed IMD cases, 92.5% were due to MenC (and 7.0% MenA) [97].
Carriage
Population data are limited. A relatively recent study evaluating meningococcal carriage in pupils attending Islamic boarding schools in Kano (northern Nigeria, conducted in 2016 prior to the more recent late 2016–2017 MenC outbreak in the region) reported carriage in 23/150 children (15.3%). MenB was the predominant carriage isolate identified, in 8.0% of children and accounting for 52.2% of all positive specimens; MenC accounted for 21.7% of carriage (with 8.7% being MenA) [99]. Data for carriage in Nigerian pilgrims are available for both the 2001 and 2014 Hajj. In 2001, carriage was identified in 6.8% of pilgrims on arrival, and in 6.3% of departing pilgrims. The majority of samples were non-groupable [43]. For the 2014 Hajj, carriage was identified in 16.5% of arriving pilgrims; 5/14 N. meningitidis isolates were MenB, with the remainder non-groupable. No data for departing pilgrims were reported [31].
Morocco
Epidemiology
IMD is considered endemic-sporadic with an incidence rate ranging from 2 to 3.6 per 100,000, with a CFR of 7–13% [78]. Serogroup data indicate that while historically MenA was the predominant strain until the mid-1990s [100], the vast majority of more recent cases are due to MenB [94, 100, 101]. The most recent serogroup study, evaluating IMD in Casablanca across 2010–2019, reported that of 245 confirmed IMD isolates (75% of which occurred in children ≤ 9 years of age), 93.1% were due to MenB, with MenW accounting for 3.7% [94].
Carriage
While population carriage data are unavailable, limited data indicate that carriage is a feature of returning Hajj pilgrims. A study initiated by WHO in response to the 2000 Hajj outbreak evaluated carriage in pilgrims returning to Morocco and their close family contacts (n = 1186; sampling 3 to 12 months afterwards). Meningococcal strains were isolated from 2.7% of specimens; while almost 50% were non-groupable, MenW accounted for 32.6% of positive carriage samples, with 12.6% due to MenB [102].
Sudan
Epidemiology
IMD is considered endemic, with epidemiology characterized by frequent cyclical outbreaks, for example in 1999 (> 33,000 cases), 2000 (> 4000 cases), 2003 (> 1000 cases), 2006 (≈ 5000 cases) and 2007 (≈ 7000 cases) [59]. In 2006, the estimated incidence rate was 13.3 per 100,000 [59]. While MenA was the principal serogroup identified across most of these outbreaks, MenW emerged as an important cause in 2000 (with a CFR of 8%). Serogroup data from 37 confirmed IMD cases from 2004 to 2006 indicated although MenA was the most frequent serogroup (78.4%), 15.5% of cases were due to MenW (and 8.1% to MenC) [103]. As described below, data suggest that the origin of MenW disease within Sudan stems directly from the Hajj outbreak in 2000 [102, 104]. More recent epidemiological data are lacking.
Carriage
Population data are unavailable. Data for carriage in Sudanese Hajj pilgrims are available for 2001, where carriage was identified in 11.1% of Hajj pilgrims arriving from Sudan prior to attendance, with unpaired sampling reporting lower carriage (2.7%) on departure [43]. Studies specifically evaluating potential carriage of Haj-associated MenW in pilgrims returning to Sudan in 2000 have reported carriage of identical clones in returning pilgrims [102, 104]. A WHO-initiated study evaluating carriage in 285 pilgrims and their close family contacts shortly after the 2000 Hajj reported N. meningitidis in 2.0% of specimens (n = 5), all of which were identical to the MenW clone associated with the 2000 Hajj outbreak [102]. Another analysis reported carriage in three returning pilgrims (and in two additional family members), with this same clone then subsequently identified in two entirely unconnected patients developing IMD in 2001 [104].
Malaysia
Epidemiology
Data for Malaysia are sparse and infrequently reported [46, 47, 105]. A single-centre study evaluating documented IMD presenting between 1987 and 2004 reported only 17 cases. Serogroup data were available for six cases: five MenW (two of which involved pilgrims returning from Hajj in 2000 and 2001, each of whom had received prior MenACWY vaccination) and one MenB [106]. Disease outbreaks have been reported (e.g., a bacterial meningitis outbreak among trainees in a government academy in 2009) but details or confirmation of IMD are lacking [47].
Carriage
Population carriage data are limited. A 2005 study evaluating meningococcal carriage in 3195 military recruits aged 17–24 years reported a 37% carriage rate. Of 210 isolates serogrouped, the great majority were serogroup X, Y or Z (81.4%); MenW accounted for 4.8% and MenA 3.3% [107]. A subsequent study from rural Borneo in 2016 sampling 140 children and adults reported carriage rates of 5%, although no serogroup data were reported [108].
Carriage data in Hajj pilgrims are available for 2001 and 2014 [31, 43]. For 2001, carriage was identified in 2.6% of Hajj pilgrims arriving from Malaysia prior to attendance, while unpaired sampling of Malaysian pilgrims on departure indicated substantially greater carriage (53.9%). The majority of isolates identified in departing pilgrims were MenW (63.4%), with the remainder being non-groupable [43]. In 2014, while there was no carriage identified in any of the 95 pilgrims sampled on arrival, carriage was reported in 1/68 pilgrims (1.6%) undergoing paired post- versus pre-Hajj sampling, with MenB the identified strain [31].
Observations Across Selected Studies
The data reported across these countries highlight substantial geographical and temporal heterogeneity patterns in IMD and meningococcal carriage. For some countries, data are sparse and limited, and often from some time ago, including those countries contributing substantial pilgrim numbers such as Indonesia, India and Pakistan. Efforts should be made to generate and publish more robust data on both the local IMD epidemiology and the relevant pilgrim characteristics, to allow a greater understanding of the current IMD landscape in such countries. Greater information is available for other countries such as Turkey and countries in North Africa. As a consequence, while much of the data we describe above are, to some extent, outdated, we believe that reporting across countries that contribute the great majority of Hajj pilgrims provides an additional perspective to the risk of future IMD outbreaks that have been the focus of previous and often comprehensive reviews [2, 3, 5, 9, 14, 19, 20, 29, 30].
Although such limitations mean we must be cautious about over-interpreting these data, some additional observations can be made. While a broad range of meningococcal serogroups are carried by and circulate among pilgrims attending Hajj and Umrah events, MenW and more recently MenB are important serogroups in some countries [9, 31]. Of the 13 selected countries, the available data indicate that MenB was the predominant IMD causative serogroup in Turkey [83], Iran [69] and across much of the MENA region; Morocco [94], Algeria [93, 94] and Egypt [77] (although with the caveat that recent data exist only for Turkey and Morocco). There is some, albeit limited, evidence for MenB carriage acquisition during the Hajj, although this is inconsistent, and patterns are undoubtedly influenced by factors such as antibiotic use [31, 45].
Additional Immunization Considerations
The existing immunization regulations for Hajj/Umrah pilgrims remain relatively unchanged since their development and introduction in 2001. These were implemented in response to the IMD disease patterns and vaccine options available at that time, and to some extent may not account for more recent IMD patterns [109] and the more recent development of MenB vaccines [110, 111]. Although effective in reducing IMD, there is limited evidence that MenB vaccines have any effect on N. meningitidis carriage [110,111,112,113], and so are unlikely to prevent any onward transmission of MenB. Novel pentavalent vaccines are in clinical development. These include a pentavalent vaccine targeting MenX (i.e., MenACWY and X), principally to address MenW emergence in sub-Saharan Africa [114, 115], and vaccines combining 4CMenB or MenB-FHbp vaccine components with MenACWY elements as MenABCWY formulations [116,117,118]. These pentavalent vaccines are in the latter stages of clinical development and could be available within the next 5 years. Although data are lacking for the specific benefits of use in pilgrim populations, existing MenB vaccines and novel pentavalent vaccines could offer additional immunization options for further consideration.
Conclusions
The introduction of mandatory MenACWY immunization for Hajj and Umrah pilgrims entering the KSA has realized substantial benefits in reducing IMD outbreaks and subsequent spread in returning travellers. While this represents a substantial achievement, the sustained success of immunization activities may require further actions. Continued emphasis on the value of conjugated ACWY vaccines is needed, along with greater monitoring of vaccine compliance. The use of conjugated MenACWY vaccines in place of polysaccharide vaccines in immunization protocols could reduce the requirement for chemoprophylaxis in pilgrims from sub-Saharan Africa, and so may reduce antibiotic use and antimicrobial resistance. The introduction of additional immunization options such as MenB vaccination could also be considered. The Graphical Plain Language Summary summarizes the context, outcomes and impact of this study for healthcare professionals.
References
Hoang VT, Gautret P. Infectious diseases and mass gatherings. Curr Infect Dis Rep. 2018;20(11):44.
Yezli S, Assiri AM, Alhakeem RF, Turkistani AM, Alotaibi B. Meningococcal disease during the Hajj and Umrah mass gatherings. Int J Infect Dis. 2016;47:60–4.
Yezli S, Assiri A, Nabulsi H, Awam A, Blumberg L, Endericks T, et al. From mass gatherings medicine to mass gatherings health: conclusions from the 3rd international conference on mass gatherings medicine, Riyadh, Kingdom of Saudi Arabia. Int J Infect Dis. 2018;66:128–30.
Muttalif AR, Presa JV, Haridy H, Gamil A, Serra LC, Cane A. Incidence and prevention of invasive meningococcal disease in global mass gathering events. Infect Dis Ther. 2019;8(4):569–79.
Yezli S. The threat of meningococcal disease during the Hajj and Umrah mass gatherings: a comprehensive review. Travel Med Infect Dis. 2018;24:51–8.
Kingdom of Saudia Arabia. Ministry of Health. Hajj statistics 2019–1440. https://www.stats.gov.sa/sites/default/files/haj_40_en.pdf. Accessed 31 May 2021
Rajab MH. A master of public health with a concentration in mass gatherings health. Cureus. 2019;11(10): e5944.
Peterson ME, Mile R, Li Y, Nair H, Kyaw MH. Meningococcal carriage in high-risk settings: a systematic review. Int J Infect Dis. 2018;73:109–17.
Yezli S, Wilder-Smith A, Bin Saeed AA. Carriage of Neisseria meningitidis in the Hajj and Umrah mass gatherings. Int J Infect Dis. 2016;47:65–70.
Harrison OB, Claus H, Jiang Y, Bennett JS, Bratcher HB, Jolley KA, et al. Description and nomenclature of Neisseria meningitidis capsule locus. Emerg Infect Dis. 2013;19(4):566–73.
Peterson ME, Li Y, Bita A, Moureau A, Nair H, Kyaw MH, et al. Meningococcal serogroups and surveillance: a systematic review and survey. J Glob Health. 2019;9(1): 010409.
Parikh SR, Campbell H, Bettinger JA, Harrison LH, Marshall HS, Martinon-Torres F, et al. The everchanging epidemiology of meningococcal disease worldwide and the potential for prevention through vaccination. J Infect. 2020;81(4):483–98.
Jafri RZ, Ali A, Messonnier NE, Tevi-Benissan C, Durrheim D, Eskola J, et al. Global epidemiology of invasive meningococcal disease. Popul Health Metr. 2013;11(1):17.
Yezli S, Gautret P, Assiri AM, Gessner BD, Alotaibi B. Prevention of meningococcal disease at mass gatherings: lessons from the Hajj and Umrah. Vaccine. 2018;36(31):4603–9.
Memish Z, Al Hakeem R, Al Neel O, Danis K, Jasir A, Eibach D. Laboratory-confirmed invasive meningococcal disease: effect of the Hajj vaccination policy, Saudi Arabia, 1995 to 2011. Euro Surveill. 2013;18(37):20581.
Lingappa JR, Al-Rabeah AM, Hajjeh R, Mustafa T, Fatani A, Al-Bassam T, et al. Serogroup W-135 meningococcal disease during the Hajj, 2000. Emerg Infect Dis. 2003;9(6):665–71.
Wilder-Smith A, Goh KT, Barkham T, Paton NI. Hajj-associated outbreak strain of Neisseria meningitidis serogroup W135: estimates of the attack rate in a defined population and the risk of invasive disease developing in carriers. Clin Infect Dis. 2003;36(6):679–83.
Booy R, Gentile A, Nissen M, Whelan J, Abitbol V. Recent changes in the epidemiology of Neisseria meningitidis serogroup W across the world, current vaccination policy choices and possible future strategies. Hum Vaccin Immunother. 2019;15(2):470–80.
Yezli S, Bin Saeed AA, Assiri AM, Alhakeem RF, Yunus MA, Turkistani AM, et al. Prevention of meningococcal disease during the Hajj and Umrah mass gatherings: past and current measures and future prospects. Int J Infect Dis. 2016;47:71–8.
Yezli S, Alotaibi B. Meningococcal disease during the Hajj and Umrah mass gatherings: A, C, W, Y may be covered but don’t forget the B and X factors! Travel Med Infect Dis. 2017;15:5–7.
Memish ZA. Meningococcal disease and travel. Clin Infect Dis. 2002;34(1):84–90.
Moore PS, Reeves MW, Schwartz B, Gellin BG, Broome CV. Intercontinental spread of an epidemic group A Neisseria meningitidis strain. Lancet. 1989;2(8657):260–3.
Al-Gahtani YM, El Bushra HE, Al-Qarawi SM, Al-Zubaidi AA, Fontaine RE. Epidemiological investigation of an outbreak of meningococcal meningitis in Makkah (Mecca), Saudi Arabia, 1992. Epidemiol Infect. 1995;115(3):399–409.
Badur S, Al Dabbagh MA, Shibl AM, Farahat FM, Ozturk S, Saha D, et al. The epidemiology of invasive meningococcal disease in the Kingdom of Saudi Arabia: a narrative review with updated analysis. Infect Dis Ther. 2021;14:1–15. https://doi.org/10.1007/s40121-021-00467-x.
Zumla A, Memish ZA. Risk of antibiotic resistant meningococcal infections in Hajj pilgrims. BMJ. 2019;366:l5260.
Willerton L, Lucidarme J, Campbell H, Caugant DA, Claus H, Jacobsson S, et al. Geographically widespread invasive meningococcal disease caused by a ciprofloxacin resistant non-groupable strain of the ST-175 clonal complex. J Infect. 2020;81(4):575–84.
Read RC, Baxter D, Chadwick DR, Faust SN, Finn A, Gordon SB, et al. Effect of a quadrivalent meningococcal ACWY glycoconjugate or a serogroup B meningococcal vaccine on meningococcal carriage: an observer-blind, phase 3 randomised clinical trial. Lancet. 2014;384(9960):2123–31.
Clark SA, Borrow R. Herd protection against meningococcal disease through vaccination. Microorganisms. 2020;8(11):1675.
Shibl A, Tufenkeji H, Khalil M, Memish Z. Consensus recommendation for meningococcal disease prevention in children and adolescents in the Middle East region. J Epidemiol Glob Health. 2012;2(1):23–30.
Shibl A, Tufenkeji H, Khalil M, Memish Z. Consensus recommendation for meningococcal disease prevention for Hajj and Umra pilgrimage/travel medicine. East Mediterr Health J. 2013;19(4):389–92.
Memish ZA, Al-Tawfiq JA, Almasri M, Azhar EI, Yasir M, Al-Saeed MS, et al. Neisseria meningitidis nasopharyngeal carriage during the Hajj: a cohort study evaluating the need for ciprofloxacin prophylaxis. Vaccine. 2017;35(18):2473–8.
Alasmari A, Houghton J, Greenwood B, Heymann D, Edwards P, Larson H, et al. Meningococcal carriage among Hajj pilgrims, risk factors for carriage and records of vaccination: a study of pilgrims to Mecca. Trop Med Int Health. 2021;26(4):453–61.
Memish ZA, Assiri AM, Hussain R, Alomar I, Stephens G. Detection of respiratory viruses among pilgrims in Saudi Arabia during the time of a declared influenza A(H1N1) pandemic. J Travel Med. 2012;19(1):15–21.
Tobaiqy M, Alhasan AH, Shams MM, Amer SA, MacLure K, Alcattan MF, et al. Assessment of Preventative Measures Practice among Umrah Pilgrims in Saudi Arabia, 1440H–2019. Int J Environ Res Public Health. 2020;18(1):257.
Memish ZA, Assiri A, Almasri M, Alhakeem RF, Turkestani A, Al Rabeeah AA, et al. Prevalence of MERS-CoV nasal carriage and compliance with the Saudi health recommendations among pilgrims attending the 2013 Hajj. J Infect Dis. 2014;210(7):1067–72.
Badahdah AM, Alghabban F, Falemban W, Albishri A, Rani Banik G, Alhawassi T, et al. Meningococcal vaccine for Hajj Pilgrims: compliance, predictors, and barriers. Trop Med Infect Dis. 2019;4(4):127.
Badahdah AM, Alfelali M, Alqahtani AS, Alsharif S, Barasheed O, Rashid H, et al. Mandatory meningococcal vaccine, and other recommended immunisations: uptake, barriers, and facilitators among health care workers and trainees at Hajj. World J Clin Cases. 2018;6(16):1128–35.
Al-Tawfiq JA, Memish ZA. The emergence, persistence, and dissemination of antimicrobial-resistant bacteria in environmental Hajj settings and implications for public health. Trop Med Infect Dis. 2021;6(1):33.
Memish ZA, Steffen R, White P, Dar O, Azhar EI, Sharma A, et al. Mass gatherings medicine: public health issues arising from mass gathering religious and sporting events. Lancet. 2019;393(10185):2073–84.
Borrow R, Taha MK, Giuliani MM, Pizza M, Banzhoff A, Bekkat-Berkani R. Methods to evaluate serogroup B meningococcal vaccines: from predictions to real-world evidence. J Infect. 2020;81(6):862–72.
Perez JL, Absalon J, Beeslaar J, Balmer P, Jansen KU, Jones TR, et al. From research to licensure and beyond: clinical development of MenB-FHbp, a broadly protective meningococcal B vaccine. Expert Rev Vaccines. 2018;17(6):461–77.
Al-Tawfiq JA, Memish ZA. The Hajj 2019 vaccine requirements and possible new challenges. J Epidemiol Glob Health. 2019;9(3):147–52.
Al-Azeri AAB, Altuhami H, Al-Rabeah A, Alhamdan N, Aljefri M. Meningococcal carriage among Hajjis in Makkah, 1421. Saudi Epidemiol Bull. 2002;9(1):3–4.
Hoang VT, Nguyen TT, Belhouchat K, Meftah M, Sow D, Benkouiten S, et al. Antibiotic use for respiratory infections among Hajj pilgrims: a cohort survey and review of the literature. Travel Med Infect Dis. 2019;30:39–45.
Alborzi A, Oskoee S, Pourabbas B, Alborzi S, Astaneh B, Gooya MM, et al. Meningococcal carrier rate before and after Hajj pilgrimage: effect of single dose ciprofloxacin on carriage. East Mediterr Health J. 2008;14(2):277–82.
Aye AMM, Bai X, Borrow R, Bory S, Carlos J, Caugant DA, et al. Meningococcal disease surveillance in the Asia-Pacific region (2020): the global meningococcal initiative. J Infect. 2020;81(5):698–711.
Vyse A, Wolter JM, Chen J, Ng T, Soriano-Gabarro M. Meningococcal disease in Asia: an under-recognized public health burden. Epidemiol Infect. 2011;139(7):967–85.
Gessner BD, Sutanto A, Linehan M, Djelantik IG, Fletcher T, Gerudug IK, et al. Incidences of vaccine-preventable Haemophilus influenzae type b pneumonia and meningitis in Indonesian children: hamlet-randomised vaccine-probe trial. Lancet. 2005;365(9453):43–52.
Pusponegoro HD, Oswari H, Astrawinata D, Fridawati V. Epidemiologic study of bacterial meningitis in Jakarta and Tangerang: preliminary report. Pediatr Infect Dis J. 1998;17(9 Suppl):S176–8.
Dutta AK, Swaminathan S, Abitbol V, Kolhapure S, Sathyanarayanan S. A comprehensive review of meningococcal disease burden in India. Infect Dis Ther. 2020;9(3):537–59.
Sinclair D, Preziosi MP, Jacob John T, Greenwood B. The epidemiology of meningococcal disease in India. Trop Med Int Health. 2010;15(12):1421–35.
Dubey AP, Hazarika RD, Abitbol V, Kolhapure S, Agrawal S. Mass gatherings: a review of the scope for meningococcal vaccination in the Indian context. Hum Vaccin Immunother. 2021;17(7):2216–24.
Nair D, Dawar R, Deb M, Capoor MR, Singal S, Upadhayay DJ, et al. Outbreak of meningococcal disease in and around New Delhi, India, 2005–2006: a report from a tertiary care hospital. Epidemiol Infect. 2009;137(4):570–6.
Negi S, Grover S, Rautela S, Rawat D, Gupta S, Khare S, et al. Direct detection and serogroup characterization of Neisseria meningitidis from outbreak of meningococcal meningitis in Delhi. Iran J Microbiol. 2010;2(2):73–9.
Aggarwal M, Manchanda V, Talukdar B. Meningitis due to Neisseria meningitidis serogroup B in India. Indian Pediatr. 2013;50(6):601–3.
Kushwaha AS, Aggarwal SK, Arora MM. Outbreak of meningococcal infection amongst soldiers deployed in operations. Med J Armed Forces India. 2010;66(1):4–8.
Jha D, Ghosh MK. Epidemiology of meningococcal carrier state amongst recruits of a military training centre. J Commun Dis. 1995;27(4):250–5.
Bali NKMH, Tantray VG, Ali S, Kakru DK, Koul PA. Meningococcal carriage among college freshmen in Kashmir, North India—a single centre study. J Clin Diagn Res. 2017;11:13–7.
Ceyhan M, Anis S, Htun-Myint L, Pawinski R, Soriano-Gabarro M, Vyse A. Meningococcal disease in the Middle East and North Africa: an important public health consideration that requires further attention. Int J Infect Dis. 2012;16(8):e574–82.
Dure-Samin A, Mubina A, Azra Y. An epidemic of meningococcal disease in Karachi (Pakistan): a study of children. Acta Paediatr Jpn. 1991;33(3):352–6.
Zaidi AK, Khan H, Lasi R, Mahesar W, Sindh Meningitis Study Group. Surveillance of pneumococcal meningitis among children in Sindh, southern Pakistan. Clin Infect Dis. 2009;1(48 Suppl 2):S129–35.
Shaikh S, Shaikh RB, Faiz MS. Seasonal paradox in acute meningitis at Nawabshah. J Coll Phys Surg Pak. 2003;13(4):207–9.
Rabbani MA, Khan AA, Ali SS, Ahmad B, Baig SM, Khan MA, et al. Spectrum of complications and mortality of bacterial meningitis: an experience from a developing country. J Pak Med Assoc. 2003;53(12):580–3.
Taj A, Jamil N. Detection of meningococcal meningitis in cerebrospinal fluid of patients with neurological disorders in government hospitals of Karachi. J Pak Med Assoc. 2016;66(11):1418–21.
Gurley ES, Hossain MJ, Montgomery SP, Petersen LR, Sejvar JJ, Mayer LW, et al. Etiologies of bacterial meningitis in Bangladesh: results from a hospital-based study. Am J Trop Med Hyg. 2009;81(3):475–83.
Hossain MA, Ahmed D, Ahmed T, Islam N, Breiman RF. Increasing isolations of Neisseria meningitides serogroup A from blood and cerebrospinal fluid in Dhaka, Bangladesh, 1999–2006. Am J Trop Med Hyg. 2009;80(4):615–8.
Houri H, Pormohammad A, Riahi SM, Nasiri MJ, Fallah F, Dabiri H, et al. Acute bacterial meningitis in Iran: systematic review and meta-analysis. PLoS ONE. 2017;12(2): e0169617.
Ataei RMTA, Hajia M, Ghorbani G, Hossaini S, Karami A. Serotyping of Neisseria meningitides in conscripts with meningitis admitted to five military hospital in Tehran between September 2004 and September 2006. J Army Med Sci. 2006;4(1):771–9.
Attarpour-Yazdi MM, Ghamarian A, Mousaviehzadeh M, Davoudi N. Identification of the serotypes of bacterial meningitis agents; implication for vaccine usage. Iran J Microbiol. 2014;6(4):211–8.
Ataee RA, Mehrabi-Tavana A, Hosseini SM, Kaviani F. Carriage rates of Neisseria meningitidis serogroups: determination among freshmen conscripts before vaccination. Iran J Microbiol. 2016;8(3):203–9.
Sadeghi M, Ahmadrajabi R, Dehesh T, Saffari F. Prevalence of meningococcal carriage among male university students living in dormitories in Kerman, southeast of Iran. Pathog Glob Health. 2018;112(6):329–33.
Metanat M, Sharifi-Mood B, Sanei-Moghaddam S, Rad NS. Pharyngeal carriage rate of Neisseria meningitidis before and after the Hajj pilgrimage, in Zahedan (southeastern Iran), 2012. Turk J Med Sci. 2015;45(6):1317–20.
Nakhla I, Frenck RW Jr, Teleb NA, El Oun S, Sultan Y, Mansour H, et al. The changing epidemiology of meningococcal meningitis after introduction of bivalent A/C polysaccharide vaccine into school-based vaccination programs in Egypt. Vaccine. 2005;23(25):3288–93.
Shaban L, Siam R. Prevalence and antimicrobial resistance pattern of bacterial meningitis in Egypt. Ann Clin Microbiol Antimicrob. 2009;8:26.
Mobarak EI. Trend, features and outcome of meningitis in the Communicable Diseases hospital, Alexandria, Egypt, 1997–2006. J Egypt Public Health Assoc. 2012;87(1–2):16–23.
Afifi S, Wasfy MO, Azab MA, Youssef FG, Pimentel G, Graham TW, et al. Laboratory-based surveillance of patients with bacterial meningitis in Egypt (1998–2004). Eur J Clin Microbiol Infect Dis. 2007;26(5):331–40.
Klena JD, Wasfy MO, Nada RA, Ahmed SF, Maksoud MA, Marfin A, et al. Characterization of Neisseria meningitidis isolates from Egypt using multilocus sequence typing. Trans R Soc Trop Med Hyg. 2012;106(5):309–14.
Borrow R, Caugant DA, Ceyhan M, Christensen H, Dinleyici EC, Findlow J, et al. Meningococcal disease in the Middle East and Africa: findings and updates from the Global Meningococcal Initiative. J Infect. 2017;75(1):1–11.
Toprak D, Soysal A, Torunoglu MA, Turgut M, Turkoglu S, Pimenta FC, et al. PCR-based national bacterial meningitis surveillance in Turkey: years 2006 to 2009. Pediatr Infect Dis J. 2014;33(10):1087–9.
Ceyhan M, Gurler N, Ozsurekci Y, Keser M, Aycan AE, Gurbuz V, et al. Meningitis caused by Neisseria Meningitidis, Hemophilus Influenzae Type B and Streptococcus Pneumoniae during 2005–2012 in Turkey. A multicenter prospective surveillance study. Hum Vaccin Immunother. 2014;10(9):2706–12.
Ceyhan M, Ozsurekci Y, Gurler N, Karadag Oncel E, Camcioglu Y, Salman N, et al. Bacterial agents causing meningitis during 2013–2014 in Turkey: a multi-center hospital-based prospective surveillance study. Hum Vaccin Immunother. 2016;12(11):2940–5.
Ceyhan M, Ozsurekci Y, Lucidarme J, Borrow R, Meningitis Surveillance Group. Characterization of invasive Neisseria meningitidis isolates recovered from children in Turkey during a period of increased serogroup B disease, 2013–2017. Vaccine. 2020;38(19):3545–52.
Ceyhan M, Ozsurekci Y, Tanir Basaranoglu S, Gurler N, Sali E, Keser Emiroglu M, et al. Multicenter hospital-based prospective surveillance study of bacterial agents causing meningitis and seroprevalence of different serogroups of Neisseria meningitidis, Haemophilus influenzae Type b, and Streptococcus pneumoniae during 2015 to 2018 in Turkey. mSphere. 2020;5(2):e00060-e120.
Dinleyici EC, Borrow R. Meningococcal infections among refugees and immigrants: silent threats of past, present and future. Hum Vaccin Immunother. 2020;16(11):2781–6.
Gazi H, Surucuoglu S, Ozbakkaloglu B, Akcali S, Ozkutuk N, Degerli K, et al. Oropharyngeal carriage and penicillin resistance of Neisseria meningitidis in primary school children in Manisa, Turkey. Ann Acad Med Singap. 2004;33(6):758–62.
Kepenekli Kadayifci E, Guneser Merdan D, Soysal A, Karaaslan A, Atici S, Durmaz R, et al. Prevalence of Neisseria meningitidis carriage: a small-scale survey in Istanbul, Turkey. J Infect Dev Ctries. 2016;10(4):413–7.
Tekin RT, Dinleyici EC, Ceyhan M, Karbuz A, Salman N, Sutcu M, et al. The prevalence, serogroup distribution and risk factors of meningococcal carriage in adolescents and young adults in Turkey. Hum Vaccin Immunother. 2017;13(5):1182–9.
Ceyhan M, Celik M, Demir ET, Gurbuz V, Aycan AE, Unal S. Acquisition of meningococcal serogroup W-135 carriage in Turkish Hajj pilgrims who had received the quadrivalent meningococcal polysaccharide vaccine. Clin Vaccine Immunol. 2013;20(1):66–8.
Tezer H, Gulhan B, Simge Gisi A, Nar Otgun S, Kanik-Yuksek S, Ozkaya-Parlakay A, et al. The impact of meningococcal conjugate vaccine (MenACWY-TT) on meningococcal carriage in Hajj Pilgrims returning to Turkey. Hum Vaccin Immunother. 2020;16(6):1268–71.
Razak AA, Al-Mathkhury HFJ, Jasim KA, Saber MQ, Al-Shammari AJN. Prevalence of Neisseria meningitidis in Iraqi children presented with meningitis. Int J Sci Technol. 2013;8(1):62–6.
Taha MK, Presa J, Serra L. A review of the epidemiology of invasive meningococcal disease and vaccination strategies in North Africa. Int J Infect Dis. 2021;104:189–97.
Tali-Maamar H, Rahal K. Étude de souches de Neisseria meningitidis isolées en Algérie entre 1992 et 2001 [Study of Neisseria meningitidis strains collected in Algeria between 1992 and 2001]. Méd Maladies Infect. 2003;33:640–3.
Tali-Maamar H, Laliam R, Guettou B, Rahal K. Molecular typing and antibiotic sensitivity of Neisseria meningitidis strains isolates in Algeria. 27th ECCMID; Vienna, Austria 2017.
Ait Mouss K, Razki A, Hong E, Zaki B, Maaloum F, Nzoyikorera N, et al. Epidemiological profile of Neisseria meningitidis in Casablanca, Morocco: 2010–2019. Access Microbiol. 2020;2(9): acmi000157.
Mustapha MM, Harrison LH. Vaccine prevention of meningococcal disease in Africa: major advances, remaining challenges. Hum Vaccin Immunother. 2018;14(5):1107–15.
Cooper LV, Ronveaux O, Fernandez K, Lingani C, Goumbi K, Ihekweazu C, et al. Spatiotemporal analysis of serogroup C meningococcal meningitis spread in Niger and Nigeria and implications for epidemic response. J Infect Dis. 2019;220(Suppl 4):S244–S52.
Nnadi C, Oladejo J, Yennan S, Ogunleye A, Agbai C, Bakare L, et al. Large outbreak of Neisseria meningitidis serogroup C—Nigeria, December 2016–June 2017. MMWR Morb Mortal Wkly Rep. 2017;66(49):1352–6.
Kwambana-Adams BA, Amaza RC, Okoi C, Rabiu M, Worwui A, Foster-Nyarko E, et al. Meningococcus serogroup C clonal complex ST-10217 outbreak in Zamfara State, Northern Nigeria. Sci Rep. 2018;8(1):14194.
Aminu AIYSM. Carriage rate of Neisseria meningitides among pupils of islamic boarding schools (Tsangaya Almajirai) in Kano, Nigeria. Bayero J Pure Appl Sci. 2017;10(1):239–42.
Zerouali K, Elmdaghri N, Boudouma M, Benbachir M. Serogroups, serotypes, serosubtypes and antimicrobial susceptibility of Neisseria meningitidis isolates in Casablanca, Morocco. Eur J Clin Microbiol Infect Dis. 2002;21(6):483–5.
Razki A, Hong E, Zerouali K, Belabbes H, Aitmouss K, Terrade A, et al. Molecular characterization of invasive isolates of Neisseria meningitidis in Casablanca, Morocco. J Clin Microbiol. 2018;56(7):e00445-e518.
Nicolas P, Ait M’barek N, Al-Awaidy S, Al Busaidy S, Sulaiman N, Issa M, et al. Pharyngeal carriage of serogroup W135 Neisseria meningitidis in Hajjees and their family contacts in Morocco, Oman and Sudan. APMIS. 2005;113(3):182–6.
Karsany MS, Elshayeb AA, Saeed ES, Elaagib R, Ibrahim SA, Elsamani E, et al. Patterns of meningococcal infection in Sudan with emergence of Neisseria meningitidis serogroup W135. East Mediterr Health J. 2013;19(10):843–6.
Issa M, Molling P, Unemo M, Backman A, Mosaad M, Sulaiman N, et al. Neisseria meningitidis serogroup W-135 isolated from healthy carriers and patients in Sudan after the Hajj in 2000. Scand J Infect Dis. 2003;35(4):230–3.
McNeil HC, Jefferies JM, Clarke SC. Vaccine preventable meningitis in Malaysia: epidemiology and management. Expert Rev Anti Infect Ther. 2015;13(6):705–14.
Raja NS, Parasakthi N, Puthucheary SD, Kamarulzaman A. Invasive meningococcal disease in the University of Malaya Medical Centre, Kuala Lumpur, Malaysia. J Postgrad Med. 2006;52(1):23–9.
Rohani MY, Ahmad Afkhar F, Amir MA, Muhd Amir K, Sahura H, Fairuz A, et al. Serogroups and antibiotic susceptibility patterns of Neisseria meningitidis isolated from army recruits in a training camp. Malays J Pathol. 2007;29(2):91–4.
Morris DE, McNeil H, Hocknell RE, Anderson R, Tuck AC, Tricarico S, et al. Carriage of upper respiratory tract pathogens in rural communities of Sarawak, Malaysian Borneo. Pneumonia (Nathan). 2021;13(1):6.
Sridhar S, Greenwood B, Head C, Plotkin SA, Safadi MA, Saha S, et al. Global incidence of serogroup B invasive meningococcal disease: a systematic review. Lancet Infect Dis. 2015;15(11):1334–46.
Martinon-Torres F, Banzhoff A, Azzari C, De Wals P, Marlow R, Marshall H, et al. Recent advances in meningococcal B disease prevention: real-world evidence from 4CMenB vaccination. J Infect. 2021;83(1):17–26.
Safadi MAP, Martinon-Torres F, Serra L, Burman C, Presa J. Translating meningococcal serogroup B vaccines for healthcare professionals. Expert Rev Vaccines. 2021;20(4):401–14.
Marshall HS, McMillan M, Koehler AP, Lawrence A, Sullivan TR, MacLennan JM, et al. Meningococcal B vaccine and meningococcal carriage in adolescents in Australia. N Engl J Med. 2020;382(4):318–27.
McMillan M, Marshall HS, Richmond P. 4CMenB vaccine and its role in preventing transmission and inducing herd immunity. Expert Rev Vaccines. 2021;29:1–12.
Chen WH, Neuzil KM, Boyce CR, Pasetti MF, Reymann MK, Martellet L, et al. Safety and immunogenicity of a pentavalent meningococcal conjugate vaccine containing serogroups A, C, Y, W, and X in healthy adults: a phase 1, single-centre, double-blind, randomised, controlled study. Lancet Infect Dis. 2018;18(10):1088–96.
Tapia MD, Sow SO, Naficy A, Diallo F, Haidara FC, Chaudhari A, et al. Meningococcal serogroup ACWYX conjugate vaccine in Malian Toddlers. N Engl J Med. 2021;384(22):2115–23.
Vesikari T, Brzostek J, Ahonen A, Paassilta M, Majda-Stanislawska E, Szenborn L, et al. Immunogenicity and safety of different schedules of the meningococcal ABCWY vaccine, with assessment of long-term antibody persistence and booster responses—results from two phase 2b randomized trials in adolescents. Hum Vaccin Immunother. 2021;17(11):4689–700.
Beran J, Drazan D, Enweonye I, Bhusal C, Toneatto D. Immunogenicity and safety of investigational MenABCWY vaccine and of 4CMenB and MenACWY vaccines administered concomitantly or alone: a phase 2 randomized study of adolescents and young adults. mSphere. 2021;6(6): e0055321.
Peterson J, Drazan D, Czajka H, Maguire J, Pregaldien J-L, Seppa I, et al. Pentavalent meningococcal (MenABCWY) vaccine is safe and well tolerated with immunogenicity noninferior to coadministered MenB-FHbp and MenACWY-CRM in a phase 2 study of healthy adolescents and young adults. Open Forum Infect Dis. 2020;1:S25–6.
Acknowledgements
Funding
GlaxoSmithKline Biologicals SA funded this literature review and took in charge all costs associated with the development and publication of this manuscript, including the journal’s Rapid Service Fee.
Medical writing, editorial, and other assistance
The authors would like to thank Business & Decision Life Sciences platform for editorial assistance and manuscript coordination and design support for the digital illustrations/animations, on behalf of GSK. Amandine Radziejwoski and Fanélie Bauer coordinated publication development and editorial support. The authors also thank Iain O’Neill (freelance on behalf of Business & Decision Life Sciences) for providing medical writing support.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors participated in the design or implementation or analysis, and interpretation of the study; and the development of this manuscript. All authors had full access to the data and gave final approval before submission.
Disclosures
Selim Badur, Mansour Khalaf and Serdar Öztürk are employed by the GSK group of companies and hold shares in the GSK group of companies; they each declare no other financial and non-financial relationships and activities. Fayssal Farahat, Atef Shibl, Ashraf Amir and Rajaa Al-Raddadi declare no financial and non-financial relationships and activities and no conflicts of interest.
Compliance with ethics guidelines
This article is based on previously conducted studies and does not contain any studies performed by any of the authors with human participants or animals.
Data availability
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Badur, S., Khalaf, M., Öztürk, S. et al. Meningococcal Disease and Immunization Activities in Hajj and Umrah Pilgrimage: a review. Infect Dis Ther 11, 1343–1369 (2022). https://doi.org/10.1007/s40121-022-00620-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40121-022-00620-0