Abstract
Introduction
The REGAL (RSV Evidence—a Geographical Archive of the Literature) series provide a comprehensive review of the published evidence in the field of respiratory syncytial virus (RSV) in Western countries over the last 20 years. This fourth publication covers the risk and burden of RSV infection in infants with congenital heart disease (CHD).
Methods
A systematic review was undertaken for articles published between January 1, 1995 and December 31, 2015 across PubMed, Embase, The Cochrane Library, and Clinicaltrials.gov. Studies reporting data for hospital visits/admissions for RSV infection among children with CHD as well as studies reporting RSV-associated morbidity, mortality, and healthcare costs were included. The focus was on children not receiving RSV prophylaxis. Study quality and strength of evidence (SOE) were graded using recognized criteria.
Results
A total of 1325 studies were identified of which 38 were included. CHD, in particular hemodynamically significant CHD, is an independent predictor for RSV hospitalization (RSVH) (high SOE). RSVH rates were generally high in young children (<4 years) with CHD (various classifications), varying between 14 and 357/1000 (high SOE). Children (<6 years) with RSV infection spent 4.4–14 days in hospital, with up to 53% requiring intensive care (high SOE). Infants (<2 years) with CHD had a more severe course of RSVH than those without CHD (high SOE). Case fatality rates of up to 3% were associated with RSV infection in children with CHD (high SOE). RSV infection in the perioperative period of corrective surgery and nosocomial RSV infection in intensive care units also represent important causes of morbidity (moderate SOE).
Conclusion
CHD poses a significant risk for RSVH and subsequent morbidity and mortality. RSV infection often complicates corrective heart surgery. To reduce the burden and improve outcomes, further research and specific studies are needed to determine the longer-term effects of severe RSV infection in young children with CHD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Congenital heart disease (CHD) is the most common type of birth defect, accounting for one-third of all major congenital anomalies, and represents a major public health issue [1, 2]. The reported total CHD prevalence has increased substantially over the last century, most likely due to improved diagnostic methods and screening modalities, although prevalence rates vary widely among studies worldwide [2]. It is estimated that CHD affects 9 in 1000 children, although significant geographical differences in prevalence have been reported [2]. A systematic review of the literature found that Europe had the second highest reported total CHD birth prevalence [8.2 per 1000 live births, 95% confidence interval (CI) 8.1–8.3]; this was significantly higher than in North America (6.9 per 1000 live births, 95% CI 6.7–7.1; P < 0.001) [2].
Children with CHD are at risk for increased morbidity from viral lower respiratory tract infection (LRTI) because of anatomical cardiovascular lesions, which can cause pulmonary hypertension leading to increased ventilation–perfusion mismatch and, ultimately, hypoxia [3]. Globally, RSV is the most common cause of childhood acute LRTIs, with at least 3.4 million episodes necessitating admission to hospital each year [4]. CHD limits an infant’s ability to increase cardiac output, and concurrently oxygen delivery can be severely limited [5]. If an infant develops respiratory syncytial virus (RSV) LRTI, oxygen uptake can be further impaired and the work of breathing in these infants with compromised cardiac reserve is increased [5]. Infants and young children with CHD are especially at risk for severe disease and hospitalization and, in some instances, may require admission to the intensive care unit (ICU), supplemental oxygen therapy and prolonged mechanical ventilation [5, 6]. Furthermore, RSV LRTI can cause mortality in the immediate period surrounding either palliative or corrective cardiac surgery employing cardiopulmonary bypass [7]. Hemodynamically significant CHD (HS-CHD) in association with RSV infection can lead to prolonged hospitalization [8] and an increased risk of death [9]. RSV infection may also delay corrective cardiac surgery [10], potentially increasing CHD-associated morbidity.
Since treatment is largely supportive and there is no effective vaccine for RSV, prevention is critically important in this vulnerable population. Current guidelines focus on infants at high risk for severe RSV infection and recommend RSV immunoprophylaxis in children with HS-CHD aged <12 months at the start of the RSV season [11,12,13]. However, since a number of children with residual HS-CHD postoperatively or awaiting cardiac transplant still remain at risk for severe RSV LRTI, some position statements continue to support RSV prophylaxis in the second year of life [14,15,16,17].
Identification of risk factors for RSV hospitalization (RSVH) and implementation of targeted prophylaxis are important to reduce the burden of severe RSV disease on inpatient and outpatient services and to improve patient outcomes. A vast amount of literature on RSV has accumulated over the past 20 years, and to review, evaluate and interpret this evidence a panel of experts in RSV from the United States, Canada and Europe formed REGAL (RSV Evidence—a Geographical Archive of the Literature) [18]. REGAL provides a comprehensive understanding on a range of topics on RSV LRTI within Western societies. This, the fourth paper in the REGAL series [18,19,20], identifies and evaluates the risks and associated morbidity and mortality of severe RSV LRTI in infants and young children with CHD.
Methods
Study Objective
REGAL encompassed seven specific research questions on RSV related to the following topics: overall epidemiology [18], prematurity [19], chronic lung disease (CLD)/bronchopulmonary dysplasia (BPD) [20], CHD, long-term respiratory morbidity, other high-risk groups (e.g., Down syndrome), and prevention, management and future perspectives. For the purposes of REGAL, Western countries were defined as the United States, Canada, and Europe (including Turkey and the Russian Federation). The overall methodology for REGAL was described in full in the first publication [18]. In summary, to address each question a systematic review of the medical literature electronically indexed in PubMed, EMBASE, and the Cochrane Library was performed. In addition, clinicaltrials.gov was searched for any relevant studies that are currently being conducted.
Literature Search
The specific research question for this review was: “What is the predisposition and associated morbidity, long-term sequelae and mortality of infants and young children with CHD following severe RSV infection?” The target population for this systematic literature review was infants and young children with CHD/HS-CHD who had ‘proven’ or ‘probable’ RSV. CHD as part of syndrome (e.g., Down syndrome) was excluded from this review, as clearly some of the genetic risk factors have an independent impact on RSV acquisition. A separate review on Down syndrome and other special populations will be published in due course. The focus of the current review was children who had not received RSV immunoprophylaxis; however, to avoid the loss of significant studies, all publications were reviewed regardless of whether prophylaxis was given. Where possible, data for children not receiving prophylaxis were reported, or, if this was not possible, the level of prophylaxis was reported alongside the results.
We performed a literature search in MEDLINE (PubMed), EMBASE and the Cochrane Library from January 1, 1995 to December 31, 2015. The following general terms and limits were used: “RSV” OR “respiratory syncytial virus” AND “congestive heart disease” OR “CHD” AND “hospitalization” OR “predisposition” OR “risk factor” AND “limits: human, infant aged up to 1 year; child (<18 years)’’. ‘‘Bronchiolitis’’ and ‘‘pneumonia’’ were captured as part of the Medical Subject Headings (MeSH) terms. It is recognized that, while some relevant articles might have been missed by the searches, we are confident that the combined Boolean operators ‘‘AND’’ and ‘‘OR’’ of the key text words and index terms precisely captured the vast majority of relevant citations which were pertinent for this evidence-based review. Additional studies identified through review of bibliographies and as abstracts presented at relevant meetings were also included. To ensure that the review was as contemporary as possible, relevant studies published during the drafting of this paper were also included.
Definition of CHD
The working definitions of CHD have evolved over time and may not be consistent between studies. HS-CHD in relation to RSV has been defined as [21,22,23]:
-
Uncorrected or palliated cyanotic or acyanotic CHD with pulmonary hypertension
-
Systolic pulmonary arterial pressure ≥40 mmHg or mean pulmonary arterial pressure ≥25 mmHg, and/or
-
Need for medication to manage congestive heart failure.
This definition was not consistently used by all authors and manuscripts, but we suggest that this is a useful definition, and perhaps should be adopted in future manuscripts describing the epidemiology and management of CHD.
For completeness, all reports describing infants with CHD, regardless of definition, were considered for inclusion in this review in order to elucidate the additional risk of RSV infection in children with significant cardiac issues at birth. To facilitate interpretation of the data, the definitions of CHD used in individual studies, where stated, were documented.
Outcomes of Interest
Key outcomes for this review included: hospitalization rates due to severe RSV infection; hospital length of stay (LOS); ICU admission and LOS; oxygen requirement; need for and duration of mechanical ventilation and/or non-invasive ventilation; and case fatality rates.
Evaluation of Data
All included studies were graded according to the Oxford Centre for Evidence-Based Medicine Levels of Evidence [24, 25] (Supplementary Material 1—REGAL Protocol). Each study was also subject to a risk of bias assessment using the RTI Item Bank (score of 1 = very high risk of bias; score of 12 = very low risk of bias) for observational studies [26]. No quantitative data synthesis was conducted due to heterogeneity between studies in terms of design, patient populations, RSV testing, recording and availability of outcomes, and differences in clinical practice between countries and over time.
Statement of Ethics Compliance
The analysis in this article is based on previously published studies and does not involve any new studies of human subjects performed by any of the authors.
Results
Articles Selected
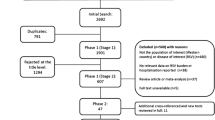
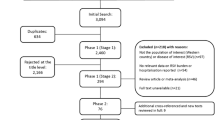
From a total of 1325 publications, 38 studies were included in the final review: 31 identified from the database searches and a further 7 from reference lists/other sources (Fig. 1). Details of all 36 studies, including evidence grades and risk of bias assessments, can be found in the online supplement.
Incidence of RSVH in Infants and Children with CHD
CHD, irrespective of hemodynamic significance, poses a significant risk for severe RSV infection requiring hospitalization [5, 8, 27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47]. RSVH rates ranged from 14 to 357 per 1000 (Table 1), although comparison of these studies is difficult as a result of differences in the study populations, some of which were not specifically CHD patients, methods used and inclusion criteria.
Several studies reported a higher RSVH rate in infants aged <12 months [29, 30, 36, 38]. In a retrospective study by Boyce et al. [29], the estimated number of hospitalizations per 1000 children with CHD aged 6 to <12 months and 12 to <24 months were 63.5 and 18.2, respectively. In another retrospective study performed in Switzerland [30], RSVH rates (per 100 child–years) in CHD patients aged <6, <12, 12–24, and <24 months of age were 2.5 (95% CI: 0.8–5.6), 2.0 (0.8–3.8), 0.5 (0.1–1.8), and 1.3 (0.6–2.3), respectively. The relative risk in comparison with non-CHD patients was 1.4 (0.6–3.1), 1.6 (0.8–3.2), 2.7 (0.7–9.7), and 1.8 (1.0-3.3), respectively [30]. A significant decrease in frequency of RSV LRTI and RSVH was also observed with increasing age in the Canadian PICNIC study [36]. The RSVH rates in children with CHD aged 0–3, 3–6, 6–12 and 12–24 months were 360, 60, 0 and 10 per 1000, respectively [36]. In contrast, Altman et al. [10] found that RSV disease necessitating hospitalization occurred in children with CHD well into the second year of life. The average age at admission for RSV infection in children with CHD was 16 ± 12 months, with children >12 months of age accounting for 61% (34/56) of the cohort [10]. The aforementioned studies included data from 1989–2003 [10, 29, 30, 36, 38]. It might be expected that, with corrective surgery now mostly occurring in the first few months of life, rates of RSVH in the second year of life would be lower than what has been reported here. However, we were unable to identify any data to support this assertion. There is recently published evidence from the US and Canada which indicates that infants with complex cardiac conditions remain at substantial risk of RSVH in the second year of life [48, 49].
In multivariate analyses, CHD has been found to be an independent risk factor for RSVH [27, 34, 38, 50]. Data on a population of Danish children with heart disease revealed that cardiomyopathy [odds ratio (OR) 5.84, 95% CI 1.26–27.16] and HS-CHD (OR 1.53, 95% CI 1.04–2.26) were both significant predictors of RSVH [38]. In a retrospective study by Boyce et al. [29], CHD was found to be an independent risk factor for RSVH in the first year of life with an incidence rate ratio of 2.8 (95% CI 2.3–3.3) versus children born at term with no underlying medical condition (low-risk group). While HS-CHD has been shown to have a significantly higher RSVH rate compared to other CHDs (92 vs. 33 per 1000; P = 0.01) [8], recent data from the PONI study [50] suggest that children diagnosed with CHD that is not hemodynamically significant (non-hsCHD) suffer a substantial burden of RSV disease that seems to be underestimated and underreported in the literature. During the 2013–2014 RSV season, 2390 preterm infants (33 weeks + 0 days to 35 weeks + 6 days) aged ≤6 months were prospectively followed across 23 countries in Western Europe. RSVH rates (per 1000 infant–years) for the study cohort were 41 and 61 during the study period and RSV season, respectively. Non-hsCHD diagnosis in this premature population was associated with an increased risk of RSV-related LRTI hospitalization in multivariable analyses (P = 0.0077) [50]. Verification of non-hsCHD as an independently significant risk factor for RSVH in non-premature populations is required.
Morbidity and Healthcare Resource Utilization
The disease burden associated with RSVH in infants and young children (<6 years) with CHD is considerable (Table 2). Length of stay in hospital and ICU admissions vary among studies, but, on average, children with CHD and severe RSV infection spent an average of 4.4–14 days in hospital [5, 6, 8, 10, 27, 30, 33, 38,39,40, 42, 45, 47, 48, 52]. Up to 53% were admitted to ICU with a median stay of 9.5–11 days [5, 6, 10, 27, 30, 44]. A retrospective, single-center study of 30 children with CHD and severe RSV infection reported that more than half (53.3%) were admitted to PICU for treatment [6]. The majority (87.5%) of PICU admissions were in infants ≤2 years of age and the median number of days spent in PICU was 11 days (range 1–43 days). The majority (87.7%) of these children had not received RSV immunoprophylaxis. During hospitalization, 15 children (50%) required respiratory support: 9 required mechanical ventilation and 6 required continuous positive airway pressure (CPAP) or non-invasive positive pressure ventilation. In addition, a third (33.3%) of the children required supplemental oxygen. Of the 24 infants in the study aged ≤2 years, 14 had non-hsCHD. The overall hospital LOS for all patients was 10 days (range: 1–65 days). Hospitalized children were susceptible to major complications following RSV infection: 20% were found to have concurrent bacterial sepsis, 16.7% electrolyte abnormalities, and 13.3% worsening of pulmonary hypertension [6].
Duppenthaler et al. [30] observed that complications leading to ICU admission, supplemental oxygen and ventilation appear to be more common in infants aged <2 years with CHD than in those without CHD. In addition, infants with CHD were significantly more often admitted to the ICU than infants with BPD or prematurity ≤35 weeks gestational age (50% vs. 6.7% and 20%, respectively), but not in comparison with otherwise healthy infants <1 month of age (32%) (Table 3) [30]. Baysal et al. [42] also reported that PICU admission and mechanical ventilation rates were significantly higher for infants with CHD aged <2 years as compared to infants without CHD infected with RSV (P = 0.01). In a further study by Kristensen et al. [38], cardiac decompensation (including the need for anticongestive therapy) was identified as a predictor for respiratory support (supplemental oxygen, nasal CPAP or mechanical ventilation) during RSVH [relative risk (RR): 1.81, 95% CI 1.02–3.23].
Recently published data from the US specifically examined the risk of RSVH in the second year of life in infants with CHD [48]. In total, 4468 RSVHs among infants 12–23 months of age with CHD were identified over a 16-year period (1997–2012). The mean LOS for RSVH was 4.4 days, with 11.4% requiring mechanical ventilation. For those without CHD, the comparative rates were 2.3 days and 2.3%, respectively. Several specific CHD diagnoses were associated with a longer LOS and higher rates of mechanical ventilation, with congestive heart failure having the worst overall morbidity (LOS: 8.2 days; mechanical ventilation: 31%) [48].
Nosocomial outbreaks of RSV infection in ICUs also represent an important cause of morbidity in this specific, high-risk population [53, 54]. Children on long-term mechanical ventilation may acquire RSV infection by transmission through droplets or caregivers and face an increased risk of a severe course of RSV infection [53]. A German study prospectively documented 1568 RSV infections in 1541 pediatric patients of whom 20 (1.3%) had acquired the RSV infection while being treated by mechanical intervention for other reasons. Thirty-five percent of the children (median age 4.2 months, range 0.5–97 months) who acquired the RSV infection whilst mechanically ventilated had CHD [53]. In a UK study reporting on a RSV outbreak in a PICU, 27.8% (15/54) of the children acquired the RSV infection whilst in the PICU [54]. In this study, PICU-acquired RSV infection was defined as having occurred when a child admitted to the PICU was RSV-negative or from whom no samples were taken because they did not exhibit signs of bronchiolitis, and who then was found to be RSV-positive ≥5 days after the admission [54]. Nosocomially-acquired RSV infection has also been documented during hospitalization in infants aged <2 years in an Italian study [45]. These data confirm the high risk of infants and children developing a severe RSV infection during hospitalization and the importance of adhering to strict infection control measures to prevent further spread of RSV in clinical settings.
Further data from a retrospective study in Canada demonstrated that children with CHD hospitalized for LRTI (0.6% RSV) in infancy had an almost two-fold increase in risk of childhood chronic respiratory morbidity (asthma, chronic bronchitis or chronic lung disease) by age 10 compared to CHD children not hospitalized for LRTI [58.5% (244/417) vs. 31.5% (884/2805), respectively] [55]. Among CHD children, LRTI hospitalization was associated with a 3-fold increase in the risk of childhood chronic respiratory morbidity [adjusted OR 3.0 (2.3–3.9)] and a 6-fold increased risk of hospitalization for chronic respiratory morbidity [adjusted OR 5.7 (4.0–8.1)] [55]. The nature, incidence and impact of long-term respiratory morbidity associated with RSVH in infancy in Western countries will be covered in more detail in a subsequent publication in the REGAL series.
Impact of RSV Infection on Cardiac Surgery for CHD
Surgical outcomes in children with CHD have improved over the past two decades. However, a significant number of children are exposed to RSV, which can result in substantial morbidity and mortality [10, 56,57,58]. In a post hoc analysis of a multicenter, randomized trial [59], Tulloh et al. [57] included all children who underwent cardiac surgery comparing outcomes for those who acquired RSV infection with those who did not (controls), matched for demographics (age and weight at operation) and physiology of cardiac morphology. It was found that RSV infection more than 6 weeks before cardiopulmonary bypass caused significant morbidity, but there was no indirect evidence of pulmonary hypertension after RSVH. This analysis also found that the duration of heart failure medication tended to be longer (by 6 months in >50% of children) if the children were hospitalized for RSV than if not [57]. Khongphatthanayothin et al. [56] reported on 25 children with CHD who had cardiac surgery within 6 months after RSV infection. Surgery for CHD performed during the symptomatic period of RSV infection was associated with a higher risk of postoperative complications (particularly pulmonary hypertension) than if surgery was undertaken electively after being discharged following RSV infection [56]. Altman et al. [10] reported that post-operative RSV infection in children with CHD can cause significant morbidity, resulting in prolonged hospital stays (2.1 times longer vs. historical, age-matched controls with comparable cardiac lesions) and time spent in ICU. RSV infection also resulted in delayed cardiac surgery in 35% (12/34) of patients in need of surgery during the RSV season [10]. Any delays in corrective surgery caused by RSV infection may increase cardiac-associated morbidity in children with CHD, though no evidence is available to adequately quantify this impact.
Case Fatality Rates
Few studies have specifically investigated mortality due to RSV in young children with CHD. Available data from the published literature suggest that the case fatality rate for RSV is relatively low among infants and children with CHD in Western countries, ranging from 0 to 3.3% [5, 6, 8, 10, 30, 38,39,40, 42, 44, 47, 48, 52]. In a retrospective, single-center study of 30 children with CHD by Butt et al. [6], conducted over a period of 7 years, only one death (3.3%) was attributed to RSV. Meberg et al. [8] reported one death related to RSV infection among 500 RSVH children with CHD; a 4-month-old premature, male infant with Down syndrome, suffering from CLD, atrioventricular septal defect and heart failure. In a US study of 4486 RSVHs among infants 12–23 months of age with various CHD diagnoses, the overall case fatality rate was 1.6%; however, certain diagnoses were associated with substantially higher rates (transposition of great vessels: 10.6%; congestive heart failure: 9.6%; cardiomyopathy: 9.5%; Ebstein’s anomaly: 8.8%) [48]. A study published in 2009, undertaken to determine the mortality rate and risk factors for death in children with severe RSV infection, found that pre-existing disease/comorbidity, in particular multiple pre-existing diseases and cardiac anomaly, was associated with a significantly higher risk of death from severe RSV infection [9]. All the RSV deaths had pre-existing medical conditions/comorbidity (27% cardiac lesions) [9]. Similar data come from a recently published US study which reported that the majority (76–79%) of RSV-associated deaths occurred in infants with complex chronic conditions [60]. Cardiovascular conditions were the most frequent single chronic condition identified, being associated with 37–45% of all RSV-related deaths [60].
Limitations
It should be recognised that the evolving definitions of CHD over time may have affected comparisons between studies and interpretation of results. Additionally, it is difficult to measure the impact of improved surgical practice in this population on the subsequent outcome of RSV infection. There were also few studies identified specifically addressing children with CHD, with the majority of studies including mixed populations of children. Other factors, such as improvements over time in both medical and surgical practice and RSV surveillance, will also have influenced interpretation of the results. Future studies should use the current, accepted definition of CHD, as described in “Methods”. Research areas of particular interest include studies investigating how delays in surgery caused by RSV impact CHD-related morbidity and studies on the epidemiology and associated morbidity of severe RSV LRTI in infants with CHD in the second year of life.
Summary Box
Key statements/findings | Level of evidencea |
|---|---|
CHD, in particular HS-CHD, is a significant risk factor for severe RSV infection with RSVH rates ranging from 14–357 per 1000 | Level 1 (Level 1 studies: n = 9; Risk of biasb: very low) |
Children with CHD spend an average of 4.4–14 days in hospital for RSV infection, with up to 53% requiring admission to the ICU | Level 1 (Level 1 studies: n = 8; Risk of biasb: very low) |
Children with CHD have a more severe disease course (increased ICU admission and ventilation) than children without CHD | Level 1 (Level 1 studies: n = 2; Risk of biasb: very low) |
RSV infection can delay and impact surgery for CHD, increasing post-operative complications, such that the timing of surgery is an important consideration | Level 2 (Level 1 studies: n = 1; Level 2 studies: n = 1; Level 3 studies: n = 2 Risk of biasb: very low) |
Case fatality rates associated with RSVH in children with CHD are reported to range from 0–3.3% | Level 1 (Level 1 studies: n = 6; Risk of biasb: very low) |
Key areas for research | |
Further research and specific studies are needed to determine the longer-term effects of severe RSV infection in infants and young children with HS-CHD as well as those with CHD that is not hemodynamically significant. Additional data are also required to assess outcomes of HS-CHD and non-hsCHD in children, independent of chromosomal/non-chromosomal anomalies and other serious pre-existing medical disorders | |
Conclusions
Infants and children with CHD are at high risk for severe RSV infection, particularly in the first year of life. Available data from the published literature suggest that, while the case fatality rate for RSV in this vulnerable population is relatively low, the burden of RSV in terms of hospitalization and the need for ICU admission is high. Nosocomially acquired RSV-infection in CHD children results in substantial morbidity. In addition, cardiac surgery performed during the symptomatic period of RSV infection has been associated with a high risk of postoperative complications, particularly postoperative pulmonary hypertension. Data suggest that early surgery significantly reduces the risk of RSVH during the first RSV season. Conversely, RSV infection may delay corrective cardiac surgery.
Most studies have focussed on HS-CHD, but the definition across the reviewed studies is not standardized. Recent data suggest that infants with CHD that is not hemodynamically significant are also at increased risk of RSVH and suffer a substantial burden of RSV disease. Moreover, infants with CHD seem to remain at risk for RSVH during their second year of life, particularly those with complex cardiac conditions, although this risk may be diminishing with earlier surgical intervention and improving outcomes. Further research and specific studies are needed to determine the longer-term effects of severe RSV infection in infants and young children with HS-CHD, as well as those with CHD that is not hemodynamically significant, in order to reduce the burden and improve outcomes in this patient population.
References
Fahed AC, Gelb BD, Seidman JG, Seidman CE. Genetics of congenital heart disease: the glass half empty. Circ Res. 2013;112:707–20.
van der Linde D, Konings EE, Slager MA, Witsenburg M, Helbing WA, Takkenberg JJ, et al. Birth prevalence of congenital heart disease worldwide: a systematic review and meta-analysis. J Am Coll Cardiol. 2011;58:2241–7.
Geskey KM, Cyran SE. Managing the morbidity associated with respiratory viral infections in children with congenital heart disease. Int J Pediatr. 2012;2012:646780.
Nair H, Nokes DJ, Gessner BD, Dherani M, Madhi SA, Singleton RJ, et al. Global burden of acute lower respiratory infections due to respiratory syncytial virus in young children: a systematic review and meta-analysis. Lancet. 2010;375:1545–55.
Medrano López C, García-Guereta L, CIVIC Study Group. Community-acquired respiratory infections in young children with congenital heart disease in the palivizumab era: the Spanish 4-season civic epidemiologic study. Pediatr Infect Dis J. 2010;29:1077–82.
Butt M, Symington A, Janes M, Steele S, Elliott L, Chant-Gambacort C, et al. Respiratory syncytial virus prophylaxis in children with cardiac disease: a retrospective single-centre study. Cardiol Young. 2014;24:337–43.
Zachariah P, Simões EAF. Respiratory syncytial virus and congenital heart disease. South Afr J Epidemiol Infect. 2008;23:17–9.
Meberg A, Bruu AL. Respiratory syncytial virus infections in congenital heart defects-hospitalizations and costs. Acta Paediatr. 2006;95:404–6.
Thorburn K. Pre-existing disease is associated with a significantly higher risk of death in severe respiratory syncytial virus infection. Arch Dis Child. 2009;94:99–103.
Altman CA, Englund JA, Demmler G, Drescher KL, Alexander MA, Watrin C, et al. Respiratory syncytial virus in patients with congenital heart disease: a contemporary look at epidemiology and success of preoperative screening. Pediatr Cardiol. 2000;21:433–8.
Bollani L, Baraldi E, Chirico G, Dotta A, Lanari M, Del Vecchio A, Italian Society of Neonatology, et al. Revised recommendations concerning palivizumab prophylaxis for respiratory syncytial virus (RSV). Ital J Pediatr. 2015;41:97.
American Academy of Pediatrics Committee on Infectious Diseases; American Academy of Pediatrics Bronchiolitis Guidelines Committee. Updated guidance for palivizumab prophylaxis among infants and young children at increased risk of hospitalization for respiratory syncytial virus infection. Pediatrics. 2014;134:415–20.
Robinson JL, Le Saux N, Canadian Paediatric Society, Infectious Diseases and Immunization Committee. Preventing hospitalizations for respiratory syncytial virus infection. Paediatr Child Health. 2015;20:321–33.
Figueras Aloy J, Carbonell Estrany X, Comité de Estándares de la SENeo. Update of recommendations on the use of palivizumab as prophylaxis in RSV infections. An Pediatr (Barc). 2015;82:199e1–2.
Nakazawa M, Saji T, Ichida F, Oyama K, Harad K, Kusuda S. Guidelines for the use of palivizumab in infants and young children with congenital heart disease. Pediatr Int. 2006;48:190–3.
Resch B, Michel-Behnke I. Respiratory syncytial virus infections in infants and children with congenital heart disease: update on evidence of prevention with palivizumab. Curr Opin Cardiol. 2013;28:85–91.
Pinter M, Geiger R. Empfehlungen zur RSV-prophylaxe bei kindern mit angeborenem herzfehler. Konsensuspapier der Arbeitsgruppe für Kinderkardiologie der österreichischen Gesellschaft für Kinder- und Jugendheilkunde, 2004. Monatsschr Kinderheilkd. 2005;153:878–80.
Bont L, Checchia P, Fauroux B, Figueras-Aloy J, Manzoni P, Paes B, et al. Defining the epidemiology and burden of severe respiratory syncytial virus infection among infants and children in Western countries. REGAL: RSV Evidence—a Global Archive of the Literature. Infect Dis Ther. 2016;5:271–98.
Figueras-Aloy J, Manzoni P, Paes B, Simões EA, Bont L, Checchia PA, et al. Defining the risk and associated morbidity and mortality of severe respiratory syncytial virus infection among preterm infants without chronic lung disease or congenital heart disease. Infect Dis Ther. 2016;5:417–52.
Paes B, Fauroux B, Figueras-Aloy J, Bont L, Checchia PA, Simões EA, et al. Defining the risk and associated morbidity and mortality of severe respiratory syncytial virus infection among infants with chronic lung disease. Infect Dis Ther. 2016;5:453–71.
Feltes TF, Sondheimer HM, Tulloh RM, Harris BS, Jensen KM, Losonsky GA, et al. A randomized controlled trial of motavizumab versus palivizumab for the prophylaxis of serious respiratory syncytial virus disease in children with hemodynamically significant congenital heart disease. Pediatr Res. 2011;70:186–91.
Abman SH, Hansmann G, Archer SL, Ivy DD, Adatia I, Chung WK, et al. Pediatric pulmonary hypertension: guidelines from the American Heart Association and American Thoracic Society. Circulation. 2015;132:2037–99.
Douwes JM, Humpl T, Bonnet D, Beghetti M, Ivy DD, Berger RM, et al. Acute vasodilator response in pediatric pulmonary arterial hypertension: current clinical practice from the TOPP registry. J Am Coll Cardiol. 2016;67:1312–23.
OCEBM Levels of Evidence Working Group. The Oxford 2011 levels of evidence. Oxford Centre for Evidence–Based Medicine. http://www.cebm.net/index.aspx?o=5653. Accessed March 2016.
OCEBM Levels of Evidence Working Group. The Oxford 2009 levels of evidence. Oxford Centre for Evidence-Based Medicine http://www.cebm.net/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/. Accessed March 2016.
Viswanathan M, Berkman ND, Dryden DM, L Hartling. Assessing risk of bias and confounding in observational studies of interventions or exposures: further development of the RTI item bank. Methods Research Report. AHRQ Publication No. 13-EHC106-EF. Rockville, MD: Agency for Healthcare Research and Quality; August 2013. www.effectivehealthcare.ahrq.gov/reports/final.cfm. Accessed March 2016.
Resch B, Kurath-Koller S, Hahn J, Raith W, Köstenberger M, Gamillscheg A. Respiratory syncytial virus-associated hospitalizations over three consecutive seasons in children with congenital heart disease. Eur J Clin Microbiol Infect Dis. 2016;35:1165–9.
Resch B, Eibisberger M, Morris N, Müller W. Respiratory syncytial virus- and influenza virus-associated hospitalizations in infants less than 12 months of age. Pediatr Infect Dis J. 2011;30:797–9.
Boyce TG, Mellen BG, Mitchel EF Jr, Wright PF, Griffin MR. Rates of hospitalization for respiratory syncytial virus infection among children in Medicaid. J Pediatr. 2000;137:865–70.
Duppenthaler A, Ammann RA, Gorgievski-Hrisoho M, Pfammatter JP, Aebi C. Low incidence of respiratory syncytial virus hospitalisations in haemodynamically significant congenital heart disease. Arch Dis Child. 2004;89:961–5.
Grimaldi M, Cornet B, Milour C, Gouyon JB. Prospective regional study of an epidemic of respiratory syncytial virus (RSV) bronchiolitis. Arch Pediatr. 2002;9:572–80.
Granbom E, Femlund E, Sunnegårdh J, Lundell B, Naumburg E. Evaluating national guidelines for the prophylactic treatment of respiratory syncytial virus in children with congenital heart disease. Acta Paediatr. 2014;103:840–5.
Willson DF, Landrigan CP, Horn SD, Smout RJ. Complications in infants hospitalised for bronchiolitis or respiratory syncytial virus pneumonia. J Pediatr. 2003;143:S142–9.
Cilla G, Sarasua A, Montes M, Arostegui N, Vicente D, Pérez-Yarza E, et al. Risk factors for hospitalization due to respiratory syncytial virus infection among infants in the Basque Country, Spain. Epidemiol Infect. 2006;134:506–13.
Bonillo Perales A, DíezDelgado Rubio J, Ortega Montes A, Infante Márquez P, Jiménez Liria M, Batlles Garrido J, et al. Perinatal history and hospitalization for bronchiolitis. A comparison with the impact-RSV Study Group. An Esp Pediatr. 2000;53:527–32.
Wang EE, Law BJ, Robinson JL, Dobson S, al Jumaah S, Stephens D, et al. PICNIC (Pediatric Investigators Collaborative Network on Infections in Canada) study of the role of age and respiratory syncytial virus neutralizing antibody on respiratory syncytial virus illness in patients with underlying heart or lung disease. Pediatrics. 1997;99:E9.
Kristensen K, Hjuler T, Ravn H, Simões EAF, Stensballe LG. Chronic diseases, chromosomal abnormalities and congenital abnormalities as risk factors for respiratory syncytial virus hospitalization: a population-based cohort study. Clin Infect Dis. 2012;54:810–7.
Kristensen K, Stensballe LG, Bjerre J, Roth D, Fisker N, Kongstad T, et al. Risk factors for respiratory syncytial virus hospitalisation in children with heart disease. Arch Dis Child. 2009;94:785–9.
Medrano C, Garcia-Guereta L, Grueso J, Insa B, Ballesteros F, Casaldaliga J, CIVIC Study Group from the Spanish Society of Pediatric Cardiology and Congenital Heart Disease, et al. Respiratory infection in congenital cardiac disease. Hospitalizations in young children in Spain during 2004 and 2005: the CIVIC Epidemiologic Study. Cardiol Young. 2007;17:360–71.
Eriksson M, Bennet R, Rotzén-Ostlund M, von Sydow M, Wirgart BZ. Population-based rates of severe respiratory syncytial virus infection in children with and without risk factors, and outcome in a tertiary care setting. Acta Paediatr. 2002;91:593–8.
Chang RK, Chen AY. Impact of palivizumab on RSV hospitalizations for children with haemodynamically significant congenital heart disease. Pediatr Cardiol. 2010;31:90–5.
Baysal K, Kilinc A, Aygun C, Sungur M. The frequency of respiratory syncytial virus on congenital heart disease patients, its impacts, and efficacy of monoclonal antibody prophylaxis in reducing respiratuar sinsitial virus infection. Eur Heart J. 2013;34:691–2.
Pezzotti P, Mantovani J, Benincori N, Mucchino E, Di Lallo D. Incidence and risk factors of hospitalization for bronchiolitis in preterm children: a retrospective longitudinal study in Italy. BMC Pediatr. 2009;10:56.
Simoes EA, Sondheimer HM, Top FH Jr, Meissner HC, Welliver RC, Kramer AA, The Cardiac Study Group, et al. Respiratory syncytial virus immune globulin for prophylaxis against respiratory syncytial virus disease in infants and children with congenital heart disease. J Pediatr. 1998;133:492–9.
Lanari M, Rossi GA, Merolla R, di Luzio Paparatti U. High risk of nosocomial-acquired RSV infection in children with congenital heart disease. J Pediatr. 2004;145:140.
Tatochenko V, Uchaikin V, Gorelov A, et al. Epidemiology of respiratory syncytial virus in children ≤2 years of age hospitalized with lower respiratory tract infections in the Russian Federation: a prospective, multicenter study. Clin Epidemiol. 2010;2:221–7.
Hervás D, Reina J, Yañez A, et al. Epidemiology of hospitalization for acute bronchiolitis in children: differences between RSV and non-RSV bronchiolitis. Eur J Clin Microbiol Infect Dis. 2012;31:1975–81.
Friedman D, Fryzek J, Jiang X, Bloomfield A, Ambrose CS, Wong P. Respiratory syncytial virus hospitalization risk in the second year of life by specific congenital heart disease diagnoses. Presented at the Pediatric Academic Societies Annual Meeting; April 30–May 3, 2016; Baltimore, MD, USA.
Li A, Wang DY, Lanctôt KL, Mitchell I, Paes BA, CARESS Investigators. Comparing first- and second-year palivizumab prophylaxis in patients with hemodynamically significant congenital heart disease in the CARESS database (2005–2015). Pediatr Infect Dis J. 2016. doi:10.1097/INF.0000000000001357
Straňák Z, Saliba E, Kosma P, Posfay-Barbe K, Yunis K, Farstad T, et al. Predictors of RSV LRTI hospitalization in infants born at 33 to 35 weeks gestational age: a large multinational study (PONI). PLoS ONE. 2016;11:e0157446.
Meberg A, Otterstad JE, Frøland G, Lindberg H, Sørland SJ. Outcome of congenital heart defects—a population based study. Acta Paediatr. 2000;89:1344–51.
Fjaerli HO, Farstad T, Bratlid D. Hospitalisations for respiratory syncytial virus bronchiolitis in Akershus, Norway, 1993–2000: a population-based retrospective study. BMC Pediatr. 2004;4:25.
von Renesse A, Schildgen O, Klinkenberg D, Müller A, von Moers A, Simon A, DSM RSV Study Group. Respiratory syncytial virus infection in children admitted to hospital but ventilated mechanically for other reasons. J Med Virol. 2009;81:160–6.
Thorburn K, Kerr S, Taylor N, van Saene HK. RSV outbreak in a paediatric intensive care unit. J Hosp Infect. 2004;57:194–201.
Vo P, Szabo SM, Gooch KG, Korol EE, Bradt P, Mitchell I, et al. Hospitalization for lower respiratory tract infection increases the risk of childhood respiratory morbidity among children with congenital heart disease. Cardiol Young. 2012;22:S40.
Khongphatthanayothin A, Wong PC, Samara Y, Newth CJ, Wells WJ, Starnes VA, et al. Impact of respiratory syncytial virus infection on surgery for congenital heart disease: postoperative course and outcome. Crit Care Med. 1999;27:1974–81.
Tulloh R, Flanders L, Henderson J, Thompson R, Feltes T. Does RSV infection cause pulmonary hypertension in children undergoing cardiac surgery. Arch Dis Child. 2011;96(Suppl 1):A1–100.
Spaeder MC, Carson KA, Vricella LA, et al. Impact of the viral respiratory season on postoperative outcomes in children undergoing cardiac surgery. Pediatr Cardiol. 2011;32:801–6.
Feltes TF, Cabalka AK, Meissner HC, Piazza FM, Carlin DA, Top FH Jr, et al. Palivizumab prophylaxis reduces hospitalization due to respiratory syncytial virus in young children with hemodynamically significant congenital heart disease. J Pediatr. 2003;143:532–40.
Byington CL, Wilkes J, Korgenski K, Sheng X. Respiratory syncytial virus-associated mortality in hospitalized infants and young children. Pediatrics. 2015;135:e24–31.
Acknowledgements
Sponsorship and article processing charges for this study were funded by AbbVie. Dr Joanne Smith, Julie Blake (Reviewers 1 and 2) and Dr Barry Rodgers-Gray (Reviewer 3), from Strategen Limited, undertook the systematic review following the protocol approved by the authors. AbbVie provided funding to Strategen to undertake the systematic review. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval to the version to be published. Editorial assistance in the preparation of this manuscript was provided by Julie Blake and Dr Barry Rodgers-Gray. Julie Blake and Barry Rodgers-Gray developed a first draft of the manuscript, based on the results of the systematic review and input/approval from all authors, which was initially edited by Xavier Carbonell-Estrany and Paul Checchia and then circulated among the other authors for input, further edits and subsequent approval. Support for this assistance was funded by AbbVie. AbbVie had the opportunity to review and comment on the completed manuscript but final editorial control rested fully with the authors.
Disclosures
The institute of Louis Bont received money for investigator-initiated studies by MeMed, AstraZeneca, AbbVie, and Janssen. The institute of Louis Bont received money for consultancy by Astra Zeneca, AbbVie, MedImmune, Janssen, Gilead and Novavax. Paul Checchia has acted as an expert advisor and speaker for AbbVie and has received honoraria in this regard. He has also received research grant funding from AstraZeneca. Brigitte Fauroux has received compensation as a neonatology board member from AbbVie. Josep Figueras-Aloy has acted as an expert advisor and speaker for AbbVie and has received honoraria in this regard. Paolo Manzoni has acted as a speaker for AbbVie, and as an expert advisor for AbbVie, TEVA, MedImmune, AstraZeneca, Janssen, and has received honoraria in this regard. Bosco Paes has received research funding from AbbVie Corporation and compensation as an advisor or lecturer from AbbVie and MedImmune. Eric Simões has received grant funding to his institution from MedImmune Inc, Glaxo Smith Kline Inc, and received consultancy fees to the institution, from AbbVie. Xavier Carbonell-Estrany has acted as an expert advisor and speaker for AbbVie and has received honoraria in this regard.
Compliance with Ethics Guidelines
The analysis in this review article is based on previously published studies and does not involve any new studies of human subjects performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article go to http://www.medengine.com/Redeem/F6E4F06044E2A44B.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Checchia, P.A., Paes, B., Bont, L. et al. Defining the Risk and Associated Morbidity and Mortality of Severe Respiratory Syncytial Virus Infection Among Infants with Congenital Heart Disease. Infect Dis Ther 6, 37–56 (2017). https://doi.org/10.1007/s40121-016-0142-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40121-016-0142-x