Abstract
Introduction
Alzheimer’s disease (AD) is a progressive, neurodegenerative disease and is the most common cause of dementia. Lecanemab is a humanized monoclonal antibody targeting amyloid protofibrils for the treatment of early AD. In the phase II BAN2401-G000-201 trial (NCT01767311), lecanemab reduced amyloid accumulated in the brain and slowed progression on key global and cognitive scales evaluating efficacy after 18 months of treatment.
Methods
A disease simulation model was used to predict the long-term clinical outcomes of lecanemab for patients with early AD [i.e., mild cognitive impairment (MCI) due to AD and mild AD dementia] on the basis of BAN2401-G000-201 trial data and published literature. The model captures the pathophysiology and management of AD, with a focus on simulating the effects of disease modification and early intervention on disease progression. The model compares lecanemab in addition to standard of care (SoC) versus SoC alone.
Results
Lecanemab treatment was estimated to slow the rate of disease progression, resulting in an extended duration of MCI due to AD and mild AD dementia and shortened duration in moderate and severe AD dementia. The mean time to mild, moderate, and severe AD dementia was longer for patients in the lecanemab + SoC group than for patients in the SoC group by 2.51, 3.13, and 2.34 years, respectively. On base-case analysis, lecanemab was associated with 0.73 incremental life years (LY) and 0.75 incremental quality-adjusted LYs (QALY), and the caregiver QALYs lost was reduced by 0.03 years. The model also predicted a lower lifetime probability of admission to institutional care in lecanemab + SoC versus SoC group (25% versus 31%).
Conclusion
The model results demonstrate the potential clinical value of lecanemab for patients with early AD and how it can slow the rate of disease progression and reduce the lifetime probability for institutionalized care.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Alzheimer’s disease (AD) is the most common cause of dementia and the leading cause of death among older populations in the USA, which exerts a significant burden on patients and their caregivers. |
The objective of this analysis was to assess the lifetime health outcomes of lecanemab in the treatment of early AD using a patient-level disease simulation model. |
Lecanemab treatment was estimated to slow the rate of disease progression, increase time with mild cognitive impairment and mild AD, and reduce time with moderate and severe AD; in addition, it was estimated to reduce the lifetime probability of being institutionalized for patients treated with lecanemab in addition to standard of care (SoC) versus SoC only. |
Predicted health outcomes provide a foundation for healthcare decision-makers to understand the potential clinical and socioeconomic value of lecanemab. |
Introduction
Alzheimer’s disease (AD) is a progressive, neurodegenerative disease that affects memory, thinking, and behavior. It is the most common cause of dementia and the leading cause of death among older populations. AD accounts for 60–80% of all dementia cases, affecting an estimated 1 in 14 people over the age of 65 years and 1 in 6 people older than 80 years [1]. AD is the fifth-leading cause of death among Americans age 65 and older, accounting for 120,000 deaths in 2019 [2]. It is projected that the population age 65 and older will reach 74 million by 2030, with 8.5 million living with AD [1].
AD may initially present as mild cognitive impairment (MCI) and advance through mild, moderate, and severe stages of dementia. MCI due to AD is the symptomatic pre-dementia phase of AD; these individuals have subtle cognitive changes and more memory problems than are normal for their age, but their symptoms do not interfere with everyday activities. People with severe AD rely completely on others for their care, which is a significant burden on caregivers. AD is diagnosed by demonstrating the presence of the characteristic neuropathological hallmarks, beta-amyloid (Aβ) plaques and neurofibrillary tangles. Positron emission tomography (PET) imaging of the brain and lumbar puncture are often used to determine the level of Aβ, total tau (T-tau), and phosphorylated tau (P-tau) in cerebrospinal fluid (CSF) during the diagnosis of dementia due to AD [1]. Pharmacologic therapies are the standard of care (SoC) for AD dementia management, and include memantine and cholinesterase inhibitors, such as donepezil, rivastigmine, and galantamine [3, 4]. New studies are focusing on disease-modifying therapies (DMT) to produce an enduring change in the clinical progression of AD by interfering with the underlying pathophysiological mechanisms of the disease process, often through a variety of primary or intermediate mechanisms such as effects on amyloid or tau [5]. DMTs are the most common type among the 120+ agents currently being studied in more than 150 clinical trials of treatments for AD [6]. Aducanumab, a monoclonal antibody directed at Aβ plaques, was approved by the US Food and Drug Administration under the accelerated approval pathway for AD [7]. A recent study assessed the long-term clinical outcomes of aducanumab for patients with early AD; the treatment was predicted to provide a lower lifetime probability of patients transitioning to AD dementia and institutionalized care as well as delays in the median time to transition to AD dementia and institutionalization [8].
Lecanemab, also called BAN2401, is a humanized monoclonal antibody targeting amyloid protofibrils—a soluble aggregated form of Aβ—for the treatment of AD, which was explored in the BAN2401-G000-201 trial (Study 201; NCT01767311) [9]. Two ongoing phase III clinical trials are exploring the safety and efficacy of lecanemab in individuals with preclinical AD (AHEAD 3–45; NCT04468659 [10]) and individuals with early AD (CLARITY AD; NCT03887455) [11]. Study 201 was a randomized, double-blinded, phase II study using a Bayesian adaptive design with response-adaptive randomization to assess the efficacy and safety of lecanemab versus placebo in 856 patients with MCI due to AD and mild AD dementia. All eligible subjects were confirmed amyloid positive via amyloid PET or CSF Aβ1–42. The trial did not meet the 12-month primary endpoint of super-superiority to placebo on the AD Composite Score (ADCOMS), which indicates a > 80% probability of > 25% slowing; however, statistical significance was achieved on key endpoints evaluating efficacy after 18 months of treatment in patients receiving the highest treatment dose (10 mg/kg biweekly) compared with placebo. This included reduction of amyloid by PET standard uptake value ratio (SUVr) accumulated in the brain (−0.30 adjusted mean change from baseline) and slowing cognitive decline measured with ADCOMS by 30%, Clinical Dementia Rating Scale-Sum of Boxes (CDR-SB) by 26%, and AD assessment scale-cognitive subscale 14 by 47% [12]. Dose-dependent changes from baseline were observed across the PET results and the clinical endpoints.
The objective of this analysis was to assess the long-term health outcomes of lecanemab in the treatment of early AD. An evidence-based disease simulation model was developed using data from Study 201 and published literature to compare lecanemab + SoC versus SoC alone in a cohort of patients with MCI due to AD and mild AD dementia. The modeled base case explored the population with early AD who had MCI due to AD and mild AD dementia and confirmed Aβ pathology. Assessments were also conducted in key patient subsets: MCI due to AD; mild AD dementia; and early AD with a mean age of 65 years.
Methods
Model Structure
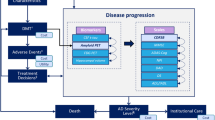
The AD Archimedes condition-event (ACE) model is a patient-level simulator that captures the pathophysiology and management of AD, focusing on the effects of disease modification and early intervention on disease progression. Disease progression is simulated on the basis of changes in the underlying biomarkers (e.g., measures of Aβ and tau levels), and their connections to clinical presentation of AD, which are measured by various patient-level scales of cognition, behavior, function, and dependence (Fig. 1). The relationships among changes in these measures over time are quantified using predictive mixed linear equations derived from long-term observational data from the AD Neuroimaging Initiative (ADNI) [13]. ADNI offers valuable insight into the long-term natural history of AD from patients with subtle cognitive dysfunction to patients with early AD. A total of 1735 patient profiles from ADNI are included in AD ACE with normal cognition through mild AD dementia. Stored baseline characteristics for these patients can be used to select a specific patient subset with certain characteristics (e.g., age, disease severity). The patients in a defined subset are randomly sampled and simulated in the AD ACE model to study disease progression and treatment effects under different scenarios. Full details on the AD ACE model structure and equations have been previously published [14, 15].
High-level model diagram outlining the key relationships in AD ACE simulator. AD Alzheimer’s disease, DMT disease-modifying therapy, ADAS-Cog Alzheimer’s Disease Assessment Scale-Cognitive, ADL activities of daily living, ApoE4 apolipoprotein E4, ARIA-E amyloid-related imaging abnormalities-edema/effusion, CDRSB Clinical Dementia Rating Sum of Boxes, CSF t-tau cerebrospinal fluid total tau, DAD Disability Assessment Scale for Dementia, DS dependence scale, FDG-PET fluorodeoxyglucose–positron emission tomography, IADL instrumental activities of daily living, MMSE Mini-Mental State Examination, NPI Neuropsychiatric Inventory Questionnaire, PET positron emission tomography, SUVr standard uptake value ratio. *Key baseline patient characteristics: age, sex, race, education, ApoE4 status, baseline biomarkers, baseline scales. †DMT effect is directly applied on amyloid PET SUVr level. ‡Includes ARIA-E. §DMT is initiated in patients with confirmed amyloid positive MCI due to AD or mild AD and discontinues once patients progress to moderate AD. ¶Defined by CDRSB thresholds: MCI due to AD < 4.5, mild AD ≥ 4.5 to < 9.5, mild AD ≥ 9.5 to < 16, severe AD ≥ 16
Model Parameters
The BAN2401-G000-201 (Study 201) trial was conducted in accordance with the Declaration of Helsinki and the International Council for Harmonization and Good Clinical Practice guidelines and was approved by the institutional review board or independent ethics committee at each site. All patients provided written informed consent. An independent interim monitoring committee was responsible for oversight and conduct of the interim analyses and response-adaptive randomization design to evaluate the safety routinely and review the futility analysis results.
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. Model parameters were primarily informed by published literature or the Study 201 outcomes.
Base-Case Population and Patient Subsets Explored in Scenario Analyses
Both Study 201 and the base-case population in this modeling work were focused on patients with MCI due to AD and mild AD dementia and confirmed Aβ pathology. Hence, a population of patients with early AD with characteristics similar to the trial population was selected for the base-case analysis. A subset of 429 individual patient profiles were selected from the existing 1735 ADNI patients in the AD ACE simulator that were matched on the key trial inclusion criteria: age range 50–90 years, Mini-Mental State Examination ≥ 22, Clinical Dementia Rating (CDR) 0.5, and amyloid PET SUVr level ≥ 1.1 [9]. The mean baseline characteristics of the selected profiles were closely aligned with the placebo and 10 mg/kg biweekly lecanemab groups of the trial, as shown in Table 1.
Alternative populations defined by age and baseline disease severity were considered in the scenario analyses. Alzheimer’s disease progresses along a continuum of severity and with varying disease trajectories [16]. The extent to which dementia progresses is also highly variable, which affects clinical outcomes. Hence, an average disease trajectory may not truly represent the reality in modeling clinical outcomes. In addition, Petersen et al. [17] reported an estimated 15–20% of adults over 65 years of age met the criteria for MCI, where the prevalence of MCI increased with age from 6.7% for ages 60–64 years to more than 25% for ages 80–84 years. Treatment may then be initiated at a younger age than in clinical trial settings [16, 17].
The impact of early or late treatment on delaying the onset of AD dementia or progression to more severe AD was explored through patient subsets defined on the selected base-case ADNI profiles. Online Supplementary Material Table S1 provides details on each subset. Scenarios included various subsets of patient profiles to explore the impact of earlier treatment initiation. These included: (1) early AD with a median baseline age of 65 years versus 71.5 years in the base case; (2) MCI due to AD; and (3) MCI due to AD with a mean baseline age of 65 years. The impact of later treatment was explored in a subset with mild AD dementia. Additional patient profiles were grouped according to quintiles based on the range of baseline CSF t-tau in the selected base-case ADNI profiles; these subsets explored the impact of early-to-late treatment based on the level of neurodegeneration as measured by CSF t-tau.
Disease Progression
The AD ACE disease equations developed from analyses of longitudinal patient-level data from the ADNI dataset [13] were used to model the natural history progression of AD for patients managed according to SoC. ADNI patients with confirmed Aβ pathology were considered in this modeling simulation to be consistent with the main study inclusion criteria and patient eligibility in the lecanemab trial. Using the AD ACE model, 2000 individual patient profiles were sampled from selected ADNI patients and simulated separately on the lecanemab + SoC and SoC alone arms to capture AD natural history, disease trajectory, and treatment effect.
The model used CDR-SB thresholds to define AD severity levels for patients, as their predicated cognition level deteriorate over time (i.e., MCI due to AD < 4.5, mild AD ≥ 4.5 to < 9.5, mild AD ≥ 9.5 to < 16, severe AD ≥ 16). Patient utility, caregiver disutility, and hazard of mortality changed as patients progressed to more advanced stages of AD.
Mortality
Mortality was modeled in AD ACE by applying hazard ratios (HR) to age-specific US general population survival [18] to naturally increase the probability of death across all health states as patients age. HR estimates from Andersen et al. [19] were used for the base-case settings (Table 1); this randomly drawn population-based cohort study in which participants were followed for 14 years demonstrated that the presence and severity of AD dementia were associated with reduced survival. In base-case analysis, it was assumed that the presence of MCI due to AD did not impact the risk of mortality. Wilson et al. [18] reported an increased risk of mortality for patients with MCI in a US population that was considered in the scenario analysis to explore the potential impact of excess mortality in patients with MCI due to AD. Impact of alternative HRs from Wimo et al. [20] were also explored in the scenario analysis.
Institutionalization
The model used estimates derived from the Consortium to Establish a Registry for Alzheimer’s Disease [21] data to inform the proportion of patients at each AD severity level who were institutionalized (Table 1). Patients were subject to an annual risk of institutionalization that varied according to AD disease severity. No risk of institutionalization was assumed for patients with MCI due to AD due to limited evidence in the literature and potential minimal impact on the outcomes.
Treatment Effect and Dosing
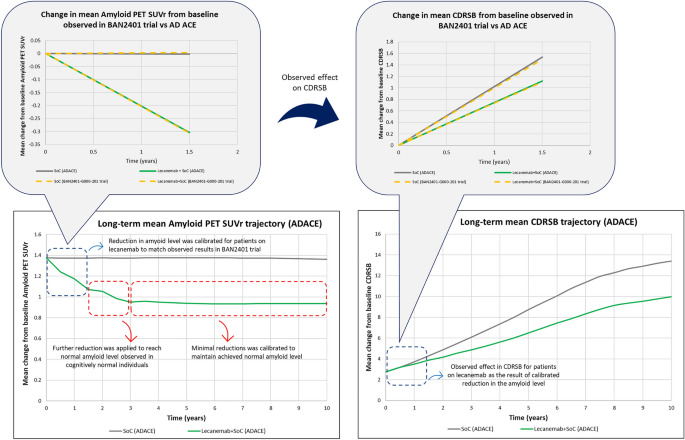
No changes were made to the published AD ACE disease equations to model the treatment effects of Study 201; the model used matching equations to predict progression in the lecanemab + SoC and SoC alone arms over time. A set of calibrated reductions were initially applied to the predicted amyloid PET SUVr values in the model’s SoC arm to match the observed treatment effects in two key trial endpoints over trial duration (i.e., −0.30 mean reduction in amyloid PET SUVr and 26% decline in the rate of change in CDR-SB over 18 months).
AD ACE disease equations predict the change in a given measure over time on the basis of selected baseline characteristics and prior values and rate of change of selected biomarkers and cognitive/behavioral measures. Amyloid PET is a predictor in all AD ACE disease equations; hence any calibrated reductions in amyloid PET SUVr at a given timepoint would impact the prediction of amyloid PET SUVr and other modeled biomarkers and scales at later timepoints.
The calibrated reduction in amyloid PET SUVr resulted in different trajectories of amyloid and CDR-SB for patients in the lecanemab + SoC and SoC arms (Fig. 2). These mean trajectories were generated on the basis of a simulation of 2000 sampled ADNI patients in AD ACE and indicated that the predicted AD natural history in the SoC arm and the calibrated treatment effect applied in the lecanemab + SoC arm of the model were both closely aligned with the trial outcomes. The change in mean CDR-SB of simulated patients in the placebo arm of the model was 1.58 over the 18-month time horizon, similar to the 1.50 CDR-SB change reported by the trial. The reductions achieved in amyloid PET SUVr (−0.30) and CDR-SB (26%) also perfectly matched what was observed in the trial.
Beyond the trial duration, treatment effect was further calibrated on the amyloid PET SUVr level to match its mean value across all simulated profiles to approximately 0.9, i.e., the mean amyloid level observed in cognitively normal individuals in the ADNI dataset. The amyloid level was further maintained at that level until treatment discontinuation, after which amyloid progression followed the natural history. This assumption was reviewed and approved by clinical experts. In the base-case scenario, patients on treatment were assumed to receive a 10 mg/kg intravenous biweekly dose of lecanemab and remain on treatment until progression to moderate AD or a worse health state defined as CDR-SB ≥ 9.5. Treatment stopping rules based on a fixed treatment durations of 1.5, 3, and 5 years were explored in scenario analyses. Recent results from the EMBARK trial for aducanumab indicated that reduction of amyloid plaque levels was maintained during a 1.6-year treatment gap from feeder studies to the EMBARK baseline [22]. Hence, a scenario was included to explore a residual effect for lecanemab even after treatment stoppage (i.e., 1.5 years treatment duration + maintain reduced amyloid level).
Treatment Discontinuation
In the base-case analysis, no treatment discontinuation was assumed to help assess the true efficacy of treatment with lecanemab and its impact on clinical outcomes in study participants who strictly adhered to the protocol and planned treatment. In Study 201, the discontinuation risk was 35.6% (217 subjects) in the lecanemab treatment groups compared with 23.3% (57) in the placebo group over the 18-month trial period [12]. In particular, the risk was higher in the lecanemab 10 mg/kg monthly [36.4% (92)] and biweekly groups [44.1% (71)] for reasons considered unrelated to treatment (i.e., subject choice, treatment-emergent adverse events, or others). In the latter group, many subjects who were apolipoprotein E4 (ApoE4) carriers and were on treatment for less than 6 months were discontinued in accordance with request of one regulatory authority that was related to a safety observation for ApoE4 carriers receiving the highest dose of lecanemab and the risk of developing amyloid-related imaging abnormalities-edema/effusion (ARIA-E) events. Only 24.8% of participants in the 10 mg/kg biweekly group discontinued treatment for reasons considered related to treatment [i.e., adverse events (AEs), subject choice, or inadequate therapeutic effect]. This translated into a 17.3% annual risk of discontinuation, which was explored in the scenario analyses to help assess the predicted outcomes in those not fully adhering to treatment regimen. The protocol for Study 201 mandated treatment discontinuation due to ARIA-E, whereas in the two ongoing phase III studies, treatment can be continued despite asymptomatic mild ARIA-E ; for other ARIA-E events, dosing can be paused until resolved. Annual discontinuation risk levels of 10% and 20% were explored as alternatives in the scenario analysis to account for the potential impact of this adjustment.
AEs
In Study 201, the incidence rates of serious AEs and treatment-emergent AEs other than ARIA-E and infusion reactions aligned with those expected for a population with early AD and were similar across the placebo and lecanemab treatment groups. Infusion reactions observed in the trial were mostly mild to moderate (grade 1 to 2) and typically responded to prophylactic treatment. Sixteen cases of ARIA-E were observed in 161 subjects in the 10 mg/kg biweekly group, three of which were symptomatic cases, consisting of headache, visual disturbances, or confusion [12]. Hence, the model only accounted for the impact of ARIA-E with a 9.9% occurrence rate in the first year, and only 18.8% of these cases were symptomatic. Discontinuation due to ARIA-E was not accounted for in the model, as they resulted in only short treatment interruptions.
Utilities
Utility measures of health-related quality of life are preference values that patients assign to their overall health status that summarize both positive and negative effects of quality of life into one score between 0 (equal to death) and 1 (equal to perfect health). There are different approaches to measure utility based on patient responses to questions about their functional status, or assignment of a single rating to their overall health by means of rating scale, time tradeoff, or willingness to pay. Patient utilities were informed by a fixed-effect meta-analysis conducted using studies identified in a systematic literature review by Landeiro et al. [23], which included utilities for individuals at each AD severity level derived from European Quality of Life-5 Dimensions (EQ-5D) instrument. The same utility estimated on the basis of disease severity was used in the community and institutionalized settings (Table 1). Alternative values from Neumann et al. [24] were considered in the scenario analysis. In addition to the health state utilities, a disutility of −0.14 estimated for headache [25] was applied to patients experiencing ARIA-E for 12 weeks [12]. Caregiver disutilities were informed on the basis of Mesterton et al. [26]; patients were assumed to have one caregiver during their lifetime.
Model Outcomes
Clinical findings have suggested that treatment with lecanemab impacts disease pathology and slows clinical decline in early AD when compared with placebo. The current study aimed to measure the lifetime health outcomes of lecanemab in delaying the onset of AD dementia or clinical worsening in patients with early AD. The mean and median time of delay in each of the current health states and the transition time to the immediate next state were estimated.
The main model outcomes included the mean and incremental health outcomes of lecanemab + SoC versus SoC, and the lifetime impact of lecanemab on delaying the mean/median time to key events such as mild/moderate/severe AD dementia and institutional care. Outcomes related to the impact of treatment on time spent in community/institutional care and proportion of patients progressing to more severe AD, being institutionalized, or discontinuing treatment were also reported by the model. Survival analyses were applied to individual patient results to estimate the mean (area under the curve) and median time to key events. The model also reported on the first time at which 50% of alive patients were in a certain condition, such as progression to mild AD dementia or worse (i.e., median time to event [of those alive]).
The model captured the mean and incremental patient life years (LY) by AD severity level in both community and institutional care settings. An annual discount rate of 3% was applied to overall discounted LYs and quality-adjusted LYs (QALY). The overall QALYs were further broken down to patient QALYs, caregiver QALYs lost, and QALYs lost to ARIA-E AEs. Equal value of LYs (EvLYs) gained was also calculated by applying age-specific general population utility values to periods corresponding to survival gains for lecanemab [27].
Results
Base-Case Analysis
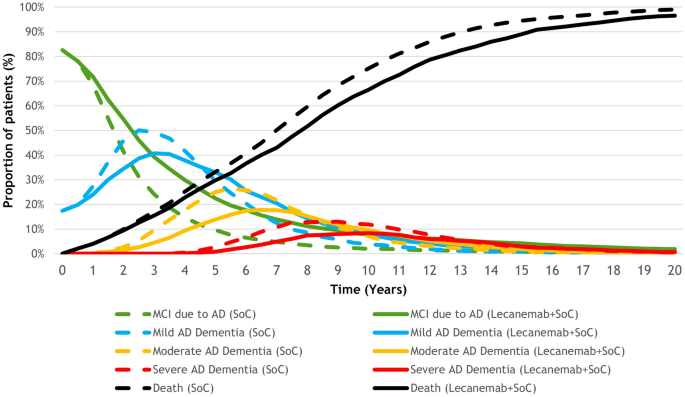
The base-case analysis explored the potential long-term health outcomes of lecanemab for patients with early AD over a lifetime horizon. All base-case results presented in Table 2 were generated on the basis of a lifetime simulation of 2000 patients sampled from the ADNI patient profiles. As illustrated in Fig. 3, the rate of disease progression decreased for patients treated with lecanemab + SoC compared with those treated with SoC alone, and treated patients spent more time in the earlier stages of disease. Compared with SoC alone, the proportions of patients on lecanemab + SoC who progressed to mild AD, moderate AD, and severe AD were reduced by 7%, 13%, and 10%, respectively. The model also predicted a 25% lifetime probability of needing institutional care for patients treated with lecanemab + SoC versus 31% for those who received SoC alone. The estimated mean time on treatment was 6.63 years, and 48% of patients discontinued treatment because of progression to moderate AD.
The mean/median time-to-event outcomes all increased for patients in the lecanemab + SoC group versus SoC alone by increments of 2.51/0.99 years for mild AD; 3.13/1.95 for moderate AD; 2.34/2.01 for severe AD; and 0.74/0.69 for institutional care. The incremental median times to mild and moderate AD (of those alive) were 0.87 and 3.44, respectively, although the median time was not reached for severe AD and institutional care.
In the base-case analysis, patients treated with lecanemab + SoC were estimated to spend 11.6 more years in community care and 0.13 fewer years in institutional care compared with the SoC group, which translated into overall incremental survival of 1.03 years (8.40 years for lecanemab + SoC versus 7.37 years for SoC). In both care settings, lecanemab treatment was estimated to result in longer time with mild AD and shorter time with moderate and severe AD. When accounting for health discount, the model predicted incremental total LYs and QALYs of 0.73 (7.11 versus 6.38) and 0.75 (4.97 versus 4.22), respectively, for lecanemab + SoC versus SoC only. Incremental QALYs were estimated at 0.72 for patients (5.15 versus 4.43), whereas incremental caregiver QALYs lost were −0.03 (0.18 versus 0.21) for patients treated with lecanemab + SoC versus SoC alone. Minimal incremental QALYs lost due to ARIA AEs was estimated from the model.
Scenario Analyses
The impacts of alternative population subsets, model settings, input sources, and assumptions were assessed in scenario analyses. Outcomes, including QALYs, mean time to mild AD dementia, and mean time to moderate AD dementia are presented in Table 3; other detailed outputs from the model are provided in Online Supplementary Material.
When including the subset of patients with MCI due to AD, the model estimated similar incremental mean time to AD dementia (2.53 versus 2.51 years), longer incremental mean time to moderate AD dementia (3.34 versus 3.13 years), and 0.07 additional QALYs over a lifetime compared with the base case. In the mild AD dementia subset, the overall gain in QALYs decreased by 0.39 and the patient progressed to moderate AD dementia faster compared with the base-case scenario where most patients did not have AD dementia at baseline. Initiating lecanemab in patients with early AD with a median baseline age of 65 years resulted in an additional QALY gain of 0.23 over a lifetime and a slight delay in progression to moderate AD dementia. Additional QALY gains of 0.37 over a lifetime and an additional 0.78 years incremental mean time to AD dementia were observed to be higher for patients initiating lecanemab compared with the base case in the subset with MCI due to AD with a mean baseline age of 65 and who were earlier in their disease trajectory. The results for patients were further divided into quintiles based on baseline CSF t-tau level and indicated that lecanemab treatment was associated with better clinical outcomes and QALYs in the lower quintiles that included patients earlier in their tau tangle pathology.
The shorter time horizon of 5 and 10 years resulted in smaller predicated incremental QALYs (0.11 and 0.43 years, respectively) and lower delay in time to mild and moderate AD dementia compared with the lifetime base case. Adjusting the discount rate to 0.0% and 5% resulted in an incremental QALY gain of 1.02 and 0.61, respectively.
Scenario analyses were also conducted using alternative sources to inform the parameters of inputs for mortality and patient utility. Using HRs from Wimo et al. [20] did not significantly impact the incremental QALYs but resulted in a decrease in incremental mean time to moderate AD dementia compared with the base case (2.16 versus 3.13 years). The incremental QALYs declined by 0.08 compared with the base case when Neumann et al. [24] was used to inform patient utilities in different AD severity levels.
The effect of treatment discontinuation and duration was further explored in different scenarios. Patients followed the natural history of disease once they discontinued/stopped treatment in all scenarios except one tested scenario in which the achieved amyloid reduction over treatment duration of 1.5 years was maintained over a lifetime after discontinuation. Longer treatment duration resulted in higher predicted gain in QALYs and a longer delay in time to mild and moderate AD. An annual discontinuation risk of 17.3% was applied to align with the risk observed in the trial; this resulted in 4.86 QALYs for patients treated with lecanemab + SoC versus 4.97 for the base case—a −0.11 change—as well as shorter times to mild and moderate AD (−0.64 and −0.63 years, respectively).
Discussion
This modeling analysis used the AD ACE disease simulator to evaluate the long-term health benefits of treatment with lecanemab. The flexible modeling framework of AD ACE allowed assessment of the impact of different patient subsets and major sources of uncertainty in this study. The base-case results indicated that, for amyloid-positive patients diagnosed with MCI due to AD or mild AD dementia, lecanemab + SoC compared with SoC alone provided a 0.75 additional gain in QALYs, 2.51 years of delay in mean time to progression to AD dementia, a 0.13 decrease in years in institutional care, and 1.16 more years in community care. These results depended on the timeframe of the analysis, as the predicted benefits of lecanemab + SoC accumulated steadily over time, with better health outcomes as the time horizon was increased.
The estimated mean time to mild AD was relatively higher than the estimated median time to mild AD in both studied arms because the distribution of time to mild AD was right-skewed (Fig. 3). The shape of the distribution for time to moderate and severe AD became more symmetrical as patients experienced disease progression, which resulted in closer estimates for the mean and median outcomes.
Lecanemab treatment was estimated to slow the rate of disease progression, resulting in an extended duration in MCI due to AD and mild AD dementia and shortened duration in moderate and severe AD dementia. The mean time in advanced stages of AD was reduced in the lecanemab + SoC arm compared with SoC alone as, with lecanemab treatment, fewer patients progressed to later stages of AD during their lifetime and those who did progress were at an increased risk of background mortality due to their older age at progression.
Scenario analyses indicated that the impact of lecanemab on health outcomes and QALYs was more significant if treatment was initiated at earlier ages in patients with MCI due to AD and in patients earlier in their tau pathology (first, second, and third quintiles). In these scenarios, the estimated gain in QALYs ranged from 0.72 to 0.92, whereas in the mild AD dementia subset, the QALY gain was only 0.36. These outcomes were consistent with the findings in recent studies and the increased recognition that DMTs may be more effective if they are initiated at presymptomatic stages of dementia [28].
Scenario analyses on treatment stopping or discontinuation indicated that longer time on treatment or maintenance of a residual effect after discontinuation resulted in higher long-term health outcomes and QALYs gained. For instance, the maximum 5-year treatment stopping rule scenario resulted in only a 0.06 decrease in QALYs versus the lifetime base-case scenario, although the mean incremental time to AD decreased by 0.54.
Selecting an alternative source to inform the hazard of survival increased QALYs by 0.64 in both arms but did not impact the incremental gain in QALYs. This was mainly derived by lower HRs reported in Wimo et al. [20] versus the base-case source [19]. Considering excessive mortality risk for patients with MCI due to AD resulted in slightly lower gain in QALYs but increased delay in mean time to mild AD dementia. The approach to modeling survival could be a potential source of bias for estimating the benefit of a DMT in the long term. Some published studies modeled survival on the basis of baseline patient characteristics and did not account for the change in hazard of death by disease severity level, which can potentially result in lower survival gains [29, 30].
A key strength of this modeling analysis compared with previous cohort studies was that actual individual patient characteristics were used, rather than mean cohort characteristics, to better capture patient heterogeneity. Disease progression in AD ACE was modeled using a set of disease equations that explicitly tracked the change in amyloid level and CDR-SB, the secondary endpoints in Study 201. This was different from Markov models [8, 31] in which the treatment effect is typically expressed as a reduction in transition probabilities between discrete health states and is assumed to last as long as patients remain on treatment. The treatment effect was also modeled by calibrating the reduction in amyloid level to achieve the effects observed during the trial (Fig. 2).
The model has some limitations that should be considered. First, the efficacy data came from the 18-month Study 201 and expert opinion was used to model the long-term effect of lecanemab; however, ongoing phase III studies will soon be able to inform the model inputs and refine the findings. The AHEAD 3–45 study provides additional evidence on preclinical AD and disease progression at earlier stages, which may further refine the results. Second, there is uncertainly around sources to inform the risk of mortality and patient utility in the literature. This model used the best-available published studies to inform these parameters. Third, the model only explored the indirect effect of treatment on cognition through CDR-SB and did not account for other severity domains (e.g., behavior, function, or dependence) or composite measures such as ADCOMS, which was the primary endpoint of Study 201. Fourth, the model created the simulated population by sampling actual patient profiles from ADNI with matching mean characteristics, which may not closely represent the profile of the Study 201 population. Last, the model projected severity using a single measure of disease severity (CDR-SB) and modeled risk of institutional care and patient utility through simple estimates based on disease severity level.
Longer-term data are needed through clinical trial follow-up and longitudinal real-world data to better validate the key assumptions in this model. It is instrumental to conduct value assessment studies to demonstrate the economic impact of lecanemab and evaluate how the delay in disease progression can result in substantial savings for the patient and caregivers.
Conclusions
This modeling study and the estimated health outcomes provided a foundation for healthcare decision-makers to understand the potential clinical, economic, and societal value of lecanemab for the treatment of patients with early AD. The analysis indicated that treatment with lecanemab would translate to a lower lifetime probability of transitioning to AD dementia or institutional care, and increased mean survival in the community compared with SoC.
References
Alzheimer’s Association. 2021 Alzheimer’s disease facts and figures. Alzheimers Dement. 2021;17:327–406.
K. Kochanek, J. Xu, E. Arias. Mortality in the United States, 2019. NCHS Data Brief, No. 395. Hyattsville, MD: National Center for Health Statistics;2020.
Arvanitakis Z, Shah RC, Bennett DA. Diagnosis and management of dementia: review. JAMA. 2019;322:1589–99.
Cummings JL, Tong G, Ballard C. Treatment combinations for Alzheimer’s disease: current and future pharmacotherapy options. J Alzheimers Dis. 2019;67:779–94.
Cummings J, Fox N. Defining disease modifying therapy for Alzheimer’s disease. J Prev Alzheimers Dis. 2017;4:109–15.
Cummings J, Lee G, Zhong K, Fonseca J, Taghva K. Alzheimer’s disease drug development pipeline: 2021. Alzheimers Dement (N Y). 2021;7:e12179.
Food and Drug Administration, FDA grants accelerated approval for Alzheimer's drug. https://www.fda.gov/news-events/press-announcements/fda-grants-accelerated-approval-alzheimers-drug. Published 2021. Accessed 16 Feb 2022.
Herring WL, Gould IG, Fillit H, et al. Predicted lifetime health outcomes for aducanumab in patients with early Alzheimer’s disease. Neurol Ther. 2021;10:919–40.
Eisai Inc., A study to evaluate safety, tolerability, and efficacy of lecanemab in subjects with early Alzheimer's disease. Clinicaltrials.gov. Published 2013. Updated 2021. Accessed 11 Feb 2022.
Zhou J, Irizarry MC, Kramer LD, et al. AHEAD 3–45 study: Preliminary screening and baseline characteristics from a placebo-controlled, double-blind study evaluating lecanemab in participants with preclinical Alzheimer’s disease and elevated (A45 trial) and intermediate (A3 trial) amyloid. Alzheimers Dement. 2021;17:e053143.
Lynch SY, Irizarry MC, Dhadda S, et al. Baseline characteristics for CLARITY AD: a phase 3 placebo-controlled, double-blind, parallel-group, 18-month study evaluating lecanemab (ban2401) in early Alzheimer’s disease. Alzheimers Dement. 2021;17:e054331.
Swanson CJ, Zhang Y, Dhadda S, et al. A randomized, double-blind, phase 2b proof-of-concept clinical trial in early Alzheimer’s disease with lecanemab, an anti-Aβ protofibril antibody. Alzheimers Res Ther. 2021;13:80.
Alzheimer's Disease Neuroimaging Initiative, Alzheimer's Disease Neuroimaging Initiative [homepage]. http://adni.loni.usc.edu/. Published 2017. Accessed 2 June 2021.
Kansal AR, Tafazzoli A, Ishak KJ, Krotneva S. Alzheimer’s disease Archimedes condition-event simulator: development and validation. Alzheimers Dement (N Y). 2018;4:76–88.
A. Tafazzoli, J. Weng, K. Sutton, et al. Validating simulated cognition trajectories based on ADNI against trajectories from the National Alzheimer's Coordinating Center (NACC) dataset. In: 11th edition of Clinical Trials on Alzheimer's Disease (CTAD); 2018; Barcelona, Spain.
Jack CR Jr, Bennett DA, Blennow K, et al. NIA-AA research framework: toward a biological definition of Alzheimer’s disease. Alzheimers Dement. 2018;14:535–62.
Petersen RC, Lopez O, Armstrong MJ, et al. Practice guideline update summary: mild cognitive impairment: report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology. 2018;90:126–35.
Arias E. United States Life Tables, 2017. Natl Vital Stat Rep. 2019;68:1–66.
Andersen K, Lolk A, Martinussen T, Kragh-Sorensen P. Very mild to severe dementia and mortality: a 14-year follow-up—the Odense study. Dement Geriatr Cogn Disord. 2010;29:61–7.
Wimo A, Handels R, Winblad B, et al. Quantifying and describing the natural history and costs of Alzheimer’s disease and effects of hypothetical interventions. J Alzheimers Dis. 2020;75:891–902.
Neumann PJ, Hermann RC, Kuntz KM, et al. Cost-effectiveness of donepezil in the treatment of mild or moderate Alzheimer’s disease. Neurology. 1999;52:1138–45.
S. Cohen, C. Van Dyck, C. Mummery, et al. Baseline Embark data from Emerge, Enfate, and prime participants in the Embark re-dosing study. Clinical Trials on Alzheimer's Disease (CTAD) 2021; 2021; Boston, MA.
Landeiro F, Mughal S, Walsh K, et al. Health-related quality of life in people with predementia Alzheimer’s disease, mild cognitive impairment or dementia measured with preference-based instruments: a systematic literature review. Alzheimers Res Ther. 2020;12:154.
Neumann PJ, Kuntz KM, Leon J, et al. Health utilities in Alzheimer’s disease: a cross-sectional study of patients and caregivers. Med Care. 1999;37:27–32.
Xu R, Insinga RP, Golden W, Hu XH. EuroQol (EQ-5D) health utility scores for patients with migraine. Qual Life Res. 2011;20:601–8.
Mesterton J, Wimo A, By A, Langworth S, Winblad B, Jonsson L. Cross sectional observational study on the societal costs of Alzheimer’s disease. Curr Alzheimer Res. 2010;7:358–67.
Institute for Clinical and Economic Review (ICER), A guide to ICER's methods for health technology assessment. https://icer.org/wp-content/uploads/2021/01/ICER_HTA_Guide_102720.pdf. Published 2020. Accessed
M. Di Carlo, D. Giacomazza, P.L. San Biagio, Alzheimer's disease: biological aspects, therapeutic perspectives and diagnostic tools. J Phys Condens Matter 2012;24:244102.
Guo S, Getsios D, Revankar N, et al. Evaluating disease-modifying agents: a simulation framework for Alzheimer’s disease. Pharmacoeconomics. 2014;32:1129–39.
Ito K, Chapman R, Pearson SD, Tafazzoli A, Yaffe K, Gurwitz JH. Evaluation of the cost-effectiveness of drug treatment for Alzheimer disease in a simulation model that includes caregiver and societal factors. JAMA Netw Open. 2021;4:e2129392.
Synnott PG, Whittington MD, Lin GA, Rind DM, Pearson SD. The effectiveness and value of aducanumab for Alzheimer’s disease. J Manag Care Spec Pharm. 2021;27:1613–7.
Acknowledgements
Funding
This study and the journal’s Rapid Service Fee was funded by Eisai Inc.
Medical Writing, Editorial, and Other Assistance
The authors would like to acknowledge Evidera's Editor, Geoffrey Heintzelman, for proofreading and editorial services. Geoffrey received funding from Eisai Inc. for his proofreading and editorial services.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Amir Abbas Tahami Monfared and Ali Tafazzoli conceptualized the study and were in charge of overall direction and planning with inputs from Quanwu Zhang. Ali Tafazzoli, Weicheng Ye, and Ameya Chavan developed the model and performed the analyses.
Disclosures
Ali Tafazzoli, Weicheng Ye, and Ameya Chavan are current employees of Evidera, a healthcare research firm that provides consulting and other research services to pharmaceutical, device, government, and non-government organizations. Evidera received funding from Eisai Inc. to conduct the study and develop this manuscript. Amir Abbas Tahami Monfared and Quanwu Zhang are employees of Eisai Inc.
Compliance with Ethics Guidelines
The BAN2401-G000-201 (Study 201) trial was conducted in accordance with Declaration of Helsinki and the International Council for Harmonization and Good Clinical Practice guidelines and was approved by the institutional review board or independent ethics committee at each center. All patients provided written informed consent. An independent interim monitoring committee was responsible for oversight and conduct of the interim analyses and response adaptive randomization design to evaluate the safety routinely and reviewing futility analysis results. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
All data used for this study are provided in the manuscript. Additional details are available from the corresponding author on request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Tahami Monfared, A.A., Tafazzoli, A., Ye, W. et al. Long-Term Health Outcomes of Lecanemab in Patients with Early Alzheimer’s Disease Using Simulation Modeling. Neurol Ther 11, 863–880 (2022). https://doi.org/10.1007/s40120-022-00350-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40120-022-00350-y