Abstract
Introduction
The aim was to evaluate the clinical characteristics and prognostic significance of subclinical seizures (SCSs) on scalp video-electroencephalogram (VEEG) monitoring with or without intracranial electroencephalogram (IEEG) monitoring in patients who had epilepsy surgery.
Methods
We reviewed 286 epileptic patients who underwent subsequent epilepsy surgery during scalp-VEEG evaluation with or without IEEG monitoring between 2013 and 2020, with a minimum follow-up of 1 year. The prevalence and clinical characteristics of SCSs, as well as their prognostic significance, were analyzed.
Results
A total of 286 patients were enrolled for analysis, and 80 patients had IEEG implanted. SCSs were recorded in 9.79% of the patients based on VEEG and 50% based on IEEG. In the VEEG group (n = 286), younger seizure onset (P = 0.004) was associated with the presence of s-SCSs (SCSs detected on scalp VEEG). In the IEEG group (n = 80), temporal lobe epilepsy (P = 0.015) was associated with the presence of i-SCSs (SCSs detected on IEEG). Of 286 patients, 208 (72.73%) were seizure-free in the VEEG group, and 56 0f 80 patients (70%) were seizure-free in the IEEG group through the last follow-up. In the VEEG group, the presence of s-SCSs did not affect seizure outcome; predictors of seizure recurrence were longer epilepsy duration (P = 0.003, OR 1.003, 95% CI 1.001–1.005), history of focal to bilateral tonic–clonic seizure (P = 0.027, OR 1.665, 95% CI 1.060–2.613), nonspecific pathology (P = 0.018, OR 2.184, 95% CI 1.145–4.163), and incomplete resection (P = 0.004, OR 2.705, 95% CI 1.372–5.332). In the IEEG group, i-SCSs were significantly associated with seizure outcome (P = 0.028, OR 0.371, 95% CI 0.153–0.898).
Conclusion
The rate of SCSs captured on IEEG monitoring was higher than that on VEEG monitoring during presurgical evaluation. SCSs detected on VEEG monitoring were associated with younger seizure onset. SCSs detected on IEEG monitoring were associated with temporal lobe epilepsy and also predicted surgical outcomes in focal epilepsy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Subclinical seizures (SCSs) are not uncommonly detected on both video-electroencephalogram (VEEG) and intracranial electroencephalogram (IEEG) monitoring. Understanding subclinical seizure characteristics is important in our appreciation of epilepsy. |
SCSs detected on VEEG monitoring were associated with younger seizure onset. |
SCSs detected on IEEG monitoring were associated with temporal lobe epilepsy and predicted surgical outcomes in focal epilepsy. |
Introduction
Epilepsy is a disease caused by abnormal firing of neurons in the brain, which affects about 11.1 per 1000 people in lower- and middle-income countries and 7.0 per 1000 in high-income countries [1]. The diagnosis of epilepsy depends mainly on clinical history and findings of electroencephalography (EEG) monitoring. Up until now, the clinical focus has mainly been on clinical seizures (CSs) and interictal epileptic discharges. Subclinical seizures (SCSs) are usually not noticed and are ignored by both patients and doctors. SCSs are defined as electrographic seizures with rhythmic ictal discharges that evolve in frequency and space that lack any objective or subjective alteration in behavior or consciousness [2, 3]. SCSs are not uncommonly detected on both scalp video-electroencephalogram (VEEG) and intracranial electroencephalogram (IEEG) monitoring during epilepsy presurgical evaluation [2, 4,5,6]. Recently, research on SCSs has garnered increasing attention [2, 5, 7,8,9,10]. However, there is still much about SCSs that remains unknown. First, the prevalence of SCSs has previously been unclear. Quite a few studies have been based on children [6, 11, 12], in whom the prevalence of SCSs was found to be higher than in epileptic adults [3, 4, 6]. Our previous studies on SCSs were based on nonoperative patients [4, 13], whose EEG recordings were relatively short, decreasing the SCS detection rate. Second, there have been few studies on SCSs based on IEEG findings [2, 8, 11, 14]. The prevalence of SCSs recorded by IEEG was supposed to be higher than that recorded by scalp EEG, which was closer to the real prevalence of SCSs. In addition, there are currently no studies comparing the SCS detection rates or analysis of the clinical characteristics of SCSs based on both VEEG and IEEG findings in the same population. Third, the significance of SCSs in epileptic patients with surgery remains unclear, with some studies finding that the presence of SCSs had no influence on the seizure outcome in focal epileptic patients with resection [11, 14], while others found that the presence of SCSs was associated with favorable prognosis for patients undergoing temporal lobe resection [8].
To address the aforementioned limitations, we aimed to explore the clinical characteristics of SCSs in patients with focal epilepsy who underwent subsequent surgery during scalp-VEEG evaluation with or without IEEG monitoring and assess the prognostic significance.
Methods
Patient Selection
We retrospectively reviewed the records of consecutive epileptic patients who underwent subsequent epilepsy surgery during scalp-VEEG evaluation with or without IEEG monitoring at the Epilepsy Center, Second Affiliated Hospital of Zhejiang University, from 2013 to 2020. This retrospective study was approved by the institutional review boards of the Second Affiliated Hospital of Zhejiang University (2013-032).
The inclusion criteria were as follows: (1) epileptic patients with VEEG monitoring, (2) undergoing subsequent surgery, (3) with at least 1-year follow-up. The exclusion criteria were as follows: (1) those who had experienced vagus nerve stimulation, (2) those who had secondary surgery (secondary surgery refers to a second operation when the results of the first surgery were deemed inadequate or the patient had intracranial surgery previously), (3) missing at follow-up, (4) incomplete clinical data.
VEEG and IEEG Monitoring
VEEG was performed using digital VEEG systems (Nicolet, VIASYS, USA, and Biologic, NATUS, USA), with scalp electrodes placed according to the international 10-20 system, including anterior temporal electrodes and sphenoidal electrodes if necessary. All the patients were monitored for at least 24 h. The intracranial electrode location was designed based on the multidisciplinary discussions in our epilepsy center. IEEG was performed with a 256-channel NK system or a 128-channel XLTEK system. Antiepileptic drugs (AEDs) were administered at normal or reduced doses during the VEEG or IEEG recordings according to different demands. SCSs were defined as electrographic seizures with rhythmic ictal discharges that evolve in frequency and space and lack any objective or subjective alteration in behavior or consciousness [2, 3]. CSs were defined as clinically evident seizures, possibly captured on VEEG or IEEG monitoring [3]. Each SCS and CS was assessed by two professional electroencephalographers (Wenjie Ming and Zhongjin Wang) who were blind to the outcome. If there was any disagreement, then the EEG epochs were re-reviewed by the epileptologist (Shuang Wang). If SCSs were detected on scalp VEEG, they were defined as s-SCSs, and if SCSs were detected on IEEG, they were defined as i-SCSs. If CSs were detected on scalp VEEG, they were defined as s-CSs, and if CSs were detected on IEEG, they were defined as i-CSs.
Data Collection
Clinical, VEEG, IEEG, postoperative histopathology, and surgical outcome data were all collected. Epilepsy was classified into temporal lobe epilepsy (TLE) and extratemporal lobe epilepsy (ETLE), which included the frontal lobe, parietal lobe, occipital lobe, insular lobe, hypothalamus, and multilobe. The groups that were monitored by VEEG were classified into two subgroups: patients that had SCSs captured by VEEG (s-SCSs group) and patients that did not have SCSs captured by VEEG (non-s-SCSs group). The groups that were monitored by IEEG were classified into two subgroups: patients that had SCSs captured by IEEG (i-SCSs group) and patients that did not have SCSs captured by IEEG (non-i-SCSs group). Localization of SCSs and CSs was based on VEEG and IEEG finalized phase reports, which were done by board-certified adult or pediatric epileptologists and re-reviewed at patient management conferences. Concordance between SCSs and CSs was summarized into five categories [7]: SCS and CS localizations were consistent (SCSs = CSs); the localization of SCSs was contained within that of CSs (SCSs < CSs); the localization of CSs was included within that of SCSs (CSs < SCSs); SCSs and CSs had overlapping localizations (SCSs ∩ CSs); SCSs and CSs had different, nonoverlapping localizations (SCSs ≠ CSs). Patients were recommended for epilepsy surgery based on multidisciplinary discussions, and the surgical strategy was determined collectively by neurosurgeons and epileptologists. As for the definition of “incomplete resection,” there were several scenarios. When the lesion was magnetic resonance imaging (MRI)-positive, “incomplete resection” was defined as “the lesion was not totally removed, and the residual tissue can be identified on postoperative MRI.” When the routine MRI was negative, positron emission tomography (PET)/MRI co-registration and MRI postprocessing (morphometry analysis program) was performed to help localize the epileptogenic zone. When the imaging postprocessing findings were consistent with VEEG findings and semiology, then the “incomplete resection” was also defined as “the residual lesion can be identified on both postoperative MRI and preoperative imaging modalities.” When all presurgical imaging modalities failed to find the potential lesion, IEEG was performed. We determined the resection area based on the seizure onset of IEEG. If the seizure onset brain zone identified by the planted electrodes was removed, it was considered “complete resection”; if not, then “incomplete resection.” The pathological diagnoses were then categorized as follows: hippocampal sclerosis, focal cortical dysplasia, tumor, hemangioma/vascular malformation, arachnoid cyst, dermoid cyst, colloid cyst, and nonspecific. Seizure outcomes were evaluated according to the last visit or telephone interviews as specified in the Engel classification [15], and further classified as being in either the seizure-free group (Engel class Ia) or the non-seizure-free group (Engel class Ib–IV).
Statistical Analysis
The data were presented as median (interquartile range) via SPSS version 23.0. Nonparametric Mann–Whitney U test was used to compare group differences on continuous variables. Categorical variables were analyzed by Pearson’s chi-square test or Fisher’s exact test. Univariate analysis results were then entered into a multivariable binary logistic regression/Cox regression model with statistical significance at P < 0.1. A logistic regression model was applied to determine the unique related factors of several variables on SCSs with statistical significance set at P < 0.05. To evaluate the predictors of seizure outcome, a Cox regression model was applied with statistical significance set at P < 0.05. Odds ratio (OR) and 95% confidence interval (CI) were also calculated for each of these parameters. The Kaplan–Meier survival analysis was used to calculate the seizure-free duration probability after surgery. To identify the threshold of continuous variables that could predict seizure outcomes, continuous variables were stratified, and the cutoff values were determined according to the Youden index in a receiver operating curve (ROC) analysis.
Results
Cohort Characteristics
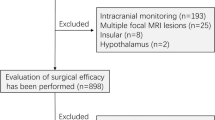
Of the 351 patients who initially met this study’s inclusion criteria, those with vagal nerve stimulation (n = 22), secondary surgery (n = 24), missing follow-up (n = 14), or incomplete clinical data (n = 5) were ultimately excluded. In total, 286 patients were enrolled for analysis (151 male, 135 female). There were 28 (9.79%) patients in the s-SCSs group and 253 (90.21%) in the non-s-SCSs group. The median duration of VEEG monitoring was 2 (interquartile range 1–4) days. The total number of patients with IEEG monitoring was 80, with 40 having had i-SCSs and 40 having had no i-SCSs captured (50%; Fig. 1). IEEG recordings identified 35 cases of SCSs that were not captured by VEEG, 18 of which (51.43%) were TLE patients. The median duration of IEEG monitoring was 8 (interquartile range 6.25–12) days. There were 166 patients (58.04%) who had pharmacoresistant epilepsy. The median age of seizure onset was 12 (interquartile range 6–22) years and the median epilepsy duration was 72 (interquartile range 18–168) months. There were 171 patients with TLE and 115 with ETLE (73 frontal, 16 parietal, 7 occipital, 2 insular, 3 hypothalamus, and 14 multilobe). The pathological types were classified as hippocampal sclerosis (n = 59), focal cortical dysplasia (n = 77), tumor (n = 86), hemangioma/vascular malformation (n = 31), arachnoid cyst (n = 3), dermoid cyst (n = 1), colloid cyst (n = 1), and nonspecific pathology (n = 25) (Table 1).
Study flow chart. VEEG video-electroencephalogram, IEEG intracranial electroencephalogram, s-SCSs subclinical seizures recorded by scalp VEEG, non-s-SCSs no subclinical seizures recorded by scalp VEEG, i-SCSs subclinical seizures recorded by IEEG, non-i-SCSs no subclinical seizures recorded by IEEG, TLE temporal lobe epilepsy, ETLE extratemporal lobe epilepsy
There were 24 patients (85.71%) who had SCSs recorded in the first 2 days among 28 patients with s-SCSs, and 32 (80%) patients who had SCSs recorded in the first 2 days among 40 patients with i-SCSs, proving that SCSs can generally be detected in the first 2 days through VEEG/IEEG recordings. We also compared the success rates in recording SCSs by the two different methods over the same time period (Table 2), and found that the detection rate of s-SCSs was always lower than that of i-SCSs. We further calculated the prevalence of SCSs at different monitoring times in all patients and found that the rate of patients with SCSs did not increase significantly with the extension of VEEG/IEEG recording time during the presurgical evaluation (Supplementary Fig. 1).
Localization of SCSs and CSs
In the 28 patients with s-SCSs, five patients had no s-CSs. Among these five patients, s-SCSs helped to localize the epileptogenic foci to some extent. The localization of s-SCSs in the first two patients was consistent with imaging findings and symptomatology, and they became seizure-free after direct resection up through the last follow-up. The postoperative pathology of these two patients found a dysembryoplastic neuroepithelial tumor in the left parieto-occipital lobe and cavernous hemangioma in the left frontal lobe, respectively. In the third case, the localization of s-SCSs and interictal epileptic discharges suggested that the epileptogenic zone might be in the right frontal lobe, and MRI suggested focal cortical dysplasia in the right frontal lobe. This patient underwent direct resection but had seizure recurrence (Engel class II) in the first year after surgery when reducing the dose of antiepileptic drugs. The MRI imaging of the fourth patient suggested a tumor in the left parietal lobe, but the localization of s-SCSs was in the left temporal lobe. Considering that patient symptoms were consistent with MRI findings, this patient underwent direct resection and only had a few auras through 3-year follow-up. The remaining patient had deep electrodes implanted due to the negative MRI imaging, and the localization of s-SCSs was fully consistent with the resection. The postoperative pathology was focal cortical dysplasia type IIB in the right frontal lobe, and this patient was seizure-free through 2-year follow-up.
The remaining 23 patients had both s-SCSs and s-CSs. S-SCSs in three patients provided a localization value during the diagnosis process, while s-CSs did not. S-CSs in the other three patients showed localization value while s-SCSs did not. Finally, of the last 17 patients with both s-SCSs and s-CSs, 12 patients (70.59%) had identical localization between two ictal events (Table 3).
In the 40 patients with i-SCSs, one patient did not have i-CSs recorded. The seizure onset zone of i-SCSs in this patient suggested the left posterior central gyrus, consistent with imaging findings and patient symptoms. This patient was seizure-free within 2-year follow-up after resection with the pathology of focal cortical dysplasia type IIB. In the remaining 39 patients, the seizure onset zone of i-SCSs was totally consistent with the i-CSs in 21 patients (53.85%) (Table 3). These above cases suggest that the seizure onset zone of SCSs can provide useful localization information, especially when CSs are not captured.
Predictors of SCSs
Univariate analysis in patients who underwent VEEG monitoring found younger seizure onset (P = 0.001), children (P = 0.030), pharmacoresistant epilepsy (P = 0.056) and daily seizure (P = 0.032) were more common in patients with s-SCSs (Table 1). Logistic regression analysis found that seizure onset age (P = 0.004, OR 0.931, 95% CI 0.886–0.978) was significantly associated with the presence of s-SCSs (Table 4).
We further compared the clinical characteristics between the two groups based on IEEG monitoring. Univariate analysis revealed that reducing the dose of AEDs during IEEG recordings (P = 0.091), left lesion (P = 0.072), temporal lobe epilepsy (P = 0.012) and hippocampal sclerosis (P = 0.077) were associated with i-SCSs (Table 5). Logistic regression analysis showed that temporal lobe epilepsy (P = 0.015, OR 3.324, 95% CI 1.264–8.740) was significantly associated with the presence of i-SCSs (Table 6; Fig. 2). And among the 21 patients with temporal epilepsy in the i-SCS group, 17 (80.95%) had mesial temporal lobe epilepsy.
Proportion of epilepsy types among four groups. The asterisk represents a difference between i-SCS and non-i-SCS groups (P = 0.012). s-SCSs subclinical seizures recorded by scalp VEEG, non-s-SCSs no subclinical seizures recorded by scalp VEEG, i-SCSs subclinical seizures recorded by IEEG, non-i-SCSs no subclinical seizures recorded by IEEG, TLE temporal lobe epilepsy, ETLE extratemporal lobe epilepsy
We further analyzed the relationship between lesion locations in MRI and SCSs in both the VEEG group (Supplementary Table 1) and the IEEG group (Supplementary Table 2), and found there was no difference in the VEEG group, while the IEEG group showed an MRI-positive lesion in the temporal lobe was associated with i-SCSs (P = 0.015), which was similar to the results from Table 5. In the VEEG group, there were some patients that had no PET data from their pre-surgical assessment, especially in patients with positive MRI. Therefore, we provided PET (positron emission tomography) data of the IEEG group and further analyzed the correlation between abnormalities from the PET scan and occurrence of SCSs in the IEEG group (Supplementary table 3), but no difference was found.
Predictors of Seizure Outcome
In the VEEG group, 208 of 286 (72.73%) patients remained seizure-free until the last follow-up. The median follow-up was 32 (interquartile range 18–52.25) months. The presence of s-SCSs did not affect seizure outcome. Univariate analysis found that longer duration (P = 0.040), history of focal to bilateral tonic–clonic seizure (P = 0.009), nonspecific pathology (P = 0.049), and incomplete resection (P = 0.012) through the last follow-up were associated with seizure recurrence (Table 7). Cox regression analysis also revealed that longer duration (P = 0.003, OR 1.003, 95% CI 1.001–1.005), history of focal to bilateral tonic–clonic seizure (P = 0.027, OR 1.665, 95% CI 1.060–2.613), nonspecific pathology (P = 0.018, OR 2.184, 95% CI 1.145–4.163), and incomplete resection (P = 0.004, OR 2.705, 95% CI 1.372–5.332) were statistically significant negative predictive factors for surgery outcomes through the last follow-up (Table 8).
In the IEEG group, 56 of 80 (70%) patients remained seizure-free through the last follow-up. The median follow-up was 23 (interquartile range 13–43.5) months. Univariate analysis found patients with i-SCSs (P = 0.015) and female patients (P = 0.062) had better seizure outcome through the last follow-up (Table 9). Cox regression analysis revealed i-SCSs was significantly associated with seizure outcome (P = 0.028, OR 0.371, 95% CI 0.153–0.898; Table 10).
Kaplan–Meier survival curves were used to illustrate postsurgical seizure freedom over time, across the study population (Fig. 3). Although patients in the IEEG group showed a lower rate of seizure freedom than the VEEG group, the difference was not statistically significant (Fig. 3a). By ROC analysis, we found that the epilepsy duration cutoff was 162 months; patients with longer seizure durations (especially > 162 months) were significantly more likely to have seizure recurrence (Fig. 3b). Regarding patients characterized as with/without a history of focal to bilateral tonic–clonic seizure (Fig. 3c), nonspecific pathology (Fig. 3d) and incomplete resection (Fig. 3e), the probability of seizure freedom showed distinct differences between groups. In the IEEG group, the non-i-SCSs group showed a sharp decrease in the probability of seizure freedom compared to the i-SCSs group (Fig. 3f).
Kaplan–Meier curves exhibiting distinct seizure outcomes among different groups. a The seizure outcomes between the whole cohort and IEEG cohort showed no distinct difference. Patients with longer epilepsy duration (b; P = 0.003), history of FBTCS (c; P = 0.027), nonspecific pathology (d; P = 0.018), incomplete resection (e; P = 0.004), had worse seizure outcome than those without through the follow-up. Patients with SCSs recorded by IEEG (f) had better seizure outcomes than those without through the follow-up (P = 0.028). VEEG video-electroencephalogram, IEEG intracranial electroencephalogram, FBTCS focal to bilateral tonic–clonic seizures, i-SCSs subclinical seizures recorded by IEEG, non-i-SCSs had no subclinical seizures recorded by IEEG
Discussion
To our knowledge, this is the first study to systematically analyze SCSs based on both VEEG and IEEG monitoring in patients who had focal epilepsy surgery. The prevalence of s-SCSs is supposed to be much lower than i-SCSs, possibly because the skull and extracranial tissue attenuate EEG signals and the spatial localization of VEEG is inherently limited [16]. In our cohort, VEEG captured SCSs in 9.79% of patients, while IEEG showed a greater sensitivity, capturing SCSs in 50% of patients, consistent with previous studies that showed the prevalence of SCSs was 5–18% in VEEG recording [4, 6, 12, 13] and approximately 60% in IEEG recording [11, 14].
In the VEEG group, logistic regression analysis showed that younger seizure onset (P = 0.004) was significantly associated with s-SCSs. We suspect that immature brain development might play a role in producing SCSs. Thus, the clinical reports of seizure frequency may underestimate the true frequency of seizures in patients with younger seizure onset. Previous studies reported that SCSs were more common in epileptic children [3, 6], patients with pharmacoresistant epilepsy [4, 6], and patients with frequent seizures [6]. However, we did not find this to be true in our study, which might be due to a difference in the study’s patient population. In the IEEG group, we found that temporal lobe epilepsy was significantly associated with i-SCSs (P = 0.015). In addition, IEEG detected 49.30% of SCSs that VEEG failed to find, about half of which originated from the temporal lobes (83.3% were mesial temporal lobes), demonstrating that discharges from mesial temporal lobes do not propagate as easily to symptom-producing areas [3], which might be due to the limited hippocampal connections to the ipsilateral neocortex and the contralateral hemisphere [8, 17]. The hippocampus has been previously shown to have a low seizure threshold, making it easier for SCSs to occur [18]. However, one previous study also reported that patients with extratemporal lobe epilepsy had twice the number of SCSs as patients with temporal lobe epilepsy, and the authors attributed this to an increased number of electrodes implanted in the extratemporal areas in their cohort [2]. In general, the dominant view is that the temporal lobe is the most common location that produces SCSs [5, 6, 8, 11, 14].
The presence of s-SCSs did not significantly affect seizure outcome, while i-SCSs were a positive predictor of surgery outcome in focal epilepsy. VEEG recordings might underestimate the prevalence of SCSs, making them unpredictable in seizure outcome. SCSs recorded by IEEG might indicate that the electrodes were placed near the epileptogenic zone, which is then well-defined for resective surgery [8], or the presence of SCSs implied the preservation of the brain’s ability to restrict seizure development and suppress spread [8]. However, a recent study reported that the presence of SCSs recorded by IEEG did not influence surgical outcome [11], and the difference from our results might be explained by the population they chose for their study, which was pediatric patients with refractory focal epilepsy, who are known to have a higher prevalence of SCSs than adults, making SCSs not specific in predicting seizure outcome. More studies with large sample size are needed for further verification.
The localization of SCSs can provide useful localization information, especially when CSs are not captured or cannot help localize the epileptogenic zone due to movement artifacts. In our study, the consistent rate of s-SCSs and s-CSs was 70.59%, which was much higher than the consistent rate of i-SCSs and i-CSs (53.85%), possibly because we utilized a “simplistic” lobar classification scheme for VEEG localization that produces less precise results compared to IEEG [7]. Previous studies reported that colocalization of SCSs and CSs was associated with improved surgical outcome in both VEEG and IEEG recordings [3, 7, 14]. In our cohort, the number of patients with colocalization values of SCSs and CSs was not large in either the VEEG or IEEG subgroup, so we did not compare such a relationship. It is widely accepted that inconsistent epileptogenic origin represents a more complicated epileptogenic network, worsening surgical outcome [7]. In our study, we did find two of four patients with i-SCSs ≠ i-CSs in IEEG recordings who had seizure recurrences after resective surgery.
It was observed that SCSs rarely propagated beyond the site of origin [14]. However, the generation mechanism that causes SCSs to differ from CSs is unclear. One previous study reported that the mean duration of SCSs was shorter than complex partial seizures and focal to bilateral tonic–clonic seizures but similar to simple partial seizures [14]. It has been reported that only 7% of neurons showed increased firing rates during SCSs, whereas 14% showed increased firing rates during auras and 36% had increased firing rates during seizures with loss of consciousness, movements, and postictal confusion [19], indicating that the neuron firing rate correlates with clinical symptoms. Further studies on IEEG signal analysis may help to explain how SCSs generate.
Although SCSs have no apparent clinical manifestation, they may result in irreversible damage to the brain’s function [20,21,22]. One case report indicated that the rapidity of recall of a well-learned word list was impaired when SCSs occurred in the left hippocampus [21]. In addition, SCSs can even be detected in patients with Alzheimer’s disease, leading to accelerated cognitive decline [22]. One previous study on functional MRI reported that the hemodynamic response function did not show a return to baseline of the BOLD signal until 30 s after SCSs end [23]. Severe hypoxemia can occur in association with SCSs involving the temporal lobe and after scalp EEG seizure activity had stopped [10]. Timely intervention of SCSs may yield considerable benefits [5], but the therapeutic management of SCSs still remains unknown. Further efforts in this aspect are needed.
SCSs can also be used to evaluate the severity of certain diseases. It was reported that SCSs were a sign of recurrence or progression in gliomas in patients who underwent tumor resection surgery [24]. SCSs showed a relatively high incident rate (16.1%) in children with traumatic brain injury through continuous video-EEG monitoring, the presence of which was associated with lower discharge King’s Outcome Scale for Childhood Head Injury score [25]. SCSs captured in neonates after cardiac surgery with cardiopulmonary bypass were a marker of greater illness severity and increased mortality [26]. The clinical value of SCSs still needs to be explored in various aspects.
Important limitations cannot be disregarded in this study. First, the limited EEG recording time cannot reflect the true prevalence of SCSs, and long-term follow-up EEG monitoring is needed in future studies. Second, our sample was not large enough for us to analyze the relationship between surgical prognosis and the concordance between SCSs and CSs localization. Third, we could not directly compare the detection rate of SCSs between the VEEG group and the IEEG group because they were not recorded at the same time, and not all of the included patients had IEEG recordings. Currently, our epilepsy center is conducting research in this area and will further explore the differences in seizure onset patterns between VEEG and IEEG.
Conclusion
The rate of SCSs captured on IEEG monitoring was higher than that on VEEG monitoring during presurgical evaluation. SCSs detected on VEEG monitoring were associated with younger seizure onset. SCSs detected on IEEG monitoring were associated with temporal lobe epilepsy and could also predict surgery outcome in focal epilepsy.
References
Thurman DJ, Begley CE, Carpio A, et al. The primary prevention of epilepsy: a report of the Prevention Task Force of the International League Against Epilepsy. Epilepsia. 2018;59:905–14.
Farooque P, Duckrow R. Subclinical seizures during intracranial EEG recording: are they clinically significant? Epilepsy Res. 2014;108:1790–6.
De Groen AC, Bolton J, Bergin AM, Sahin M, Peters JM. The evolution of subclinical seizures in children with tuberous sclerosis complex. J Child Neurol. 2019;34:770–7.
Jin B, Wang S, Yang L, et al. Prevalence and predictors of subclinical seizures during scalp video-EEG monitoring in patients with epilepsy. Int J Neurosci. 2017;127:651–8.
Feldwisch-Drentrup H, Ihle M, Quyen Mle V, Schulze-Bonhage A, Schelter B, et al. Anticipating the unobserved: prediction of subclinical seizures. Epilepsy Behav. 2011;22(Suppl 1):S119–26.
Akman CI, Montenegro MA, Jacob S, et al. Subclinical seizures in children diagnosed with localization-related epilepsy: clinical and EEG characteristics. Epilepsy Behav. 2009;16:86–98.
Wang S, Wang ZI, Tang Y, et al. Localization value of subclinical seizures on scalp video-EEG in epilepsy presurgical evaluation. Epilepsia. 2019;60:2477–85.
Sperling MR, O’Connor MJ. Auras and subclinical seizures: characteristics and prognostic significance. Ann Neurol. 1990;28:320–8.
Cuaycong M, Engel M, Weinstein SL, et al. A novel approach to the study of hypoxia-ischemia-induced clinical and subclinical seizures in the neonatal rat. Dev Neurosci. 2011;33:241–50.
Maglajlija V, Walker MC, Kovac S. Severe ictal hypoxemia following focal, subclinical temporal electrographic scalp seizure activity. Epilepsy Behav. 2012;24:143–5.
Tsuboyama M, Harini C, Liu S, Zhang B, Bolton J. Subclinical seizures detected on intracranial EEG: Patient characteristics and impact on surgical outcome in a single pediatric epilepsy surgery center. Epilepsy Behav. 2021;121:108040.
Velkey A, Siegler Z, Janszky J, Duray B, Fogarasi A. Clinical value of subclinical seizures in children with focal epilepsy. Epilepsy Res. 2011;95:82–5.
Wang S, Jin B, Yang L, et al. Clinical value and predictors of subclinical seizures in patients with temporal lobe epilepsy undergoing scalp video-EEG monitoring. J Clin Neurosci. 2017;44:214–7.
Zangaladze A, Nei M, Liporace JD, Sperling MR. Characteristics and clinical significance of subclinical seizures. Epilepsia. 2008;49:2016–21.
Engel JJ, Van Ness P, Rasmussen T. Outcome with respect to epileptic seizure. In: Engel J, editor. Surgical treatment of the epilepsies. New York: Raven Press; 1993. p. 609–21.
Smith SJ. EEG in the diagnosis, classification, and management of patients with epilepsy. J Neurol Neurosurg Psychiatry. 2005;76(Suppl 2):ii2-7.
Amaral DG, Insausti R, Cowan WM. The commissural connections of the monkey hippocampal formation. J Comp Neurol. 1984;224:307–36.
Myers RR, Shapiro HM. Local cerebral metabolism during enflurane anesthesia: identification of epileptogenic foci. Electroencephalogr Clin Neurophysiol. 1979;47:153–62.
Babb TL, Wilson CL, Isokawa-Akesson M. Firing patterns of human limbic neurons during stereoencephalography (SEEG) and clinical temporal lobe seizures. Electroencephalogr Clin Neurophysiol. 1987;66:467–82.
Kanazawa K, Matsumoto R, Shimotake A, et al. Persistent frequent subclinical seizures and memory impairment after clinical remission in smoldering limbic encephalitis. Epileptic Disord. 2014;16:312–7.
Bridgman PA, Malamut BL, Sperling MR, Saykin AJ, O’Connor MJ. Memory during subclinical hippocampal seizures. Neurology. 1989;39:853–6.
Vossel KA, Tartaglia MC, Nygaard HB, Zeman AZ, Miller BL. Epileptic activity in Alzheimer’s disease: causes and clinical relevance. Lancet Neurol. 2017;16:311–22.
Kobayashi E, Hawco CS, Grova C, Dubeau F, Gotman J. Widespread and intense BOLD changes during brief focal electrographic seizures. Neurology. 2006;66:1049–55.
Fernandez-Torre JL, Hernandez-Hernandez M, Martino J, Hinojo C. Subclinical focal seizures as a sign of progression in gliomas. Epileptic Disord. 2014;16:546–53.
Arndt DH, Lerner JT, Matsumoto JH, et al. Subclinical early posttraumatic seizures detected by continuous EEG monitoring in a consecutive pediatric cohort. Epilepsia. 2013;54:1780–8.
Naim MY, Gaynor JW, Chen J, et al. Subclinical seizures identified by postoperative electroencephalographic monitoring are common after neonatal cardiac surgery. J Thorac Cardiovasc Surg. 2015;150:169–80.
Acknowledgements
Funding
This work was supported by the National Natural Science Foundation of China [grant numbers: 82001365, 82071443]. The Rapid Service Fee was funded by the corresponding author, Shan Wang.
Author Contributions
Chenmin He, Cong Chen, Shuang Wang and Shan Wang contributed to the conception and design of the study. Chenmin He, Cong Chen, Yuyu Yang, Lingli Hu, Bo Jin, Wenjie Ming, Zhongjin Wang, Yao Ding collected and analyzed the original data. Chenmin He, Cong Chen, Shuang Wang and Shan Wang revised the manuscript. Meiping Ding provides the technique and material support. All authors contributed to drafting, the paper and were involved in the approval of the final version.
Disclosures
Chenmin He, Cong Chen, Yuyu Yang, Lingli Hu, Bo Jin, Wenjie Ming, Zhongjin Wang, Yao Ding, Meiping Ding, Shuang Wang and Shan Wang have nothing to disclose.
Compliance with Ethics Guidelines
This retrospective study involving human participants were reviewed and approved by the Institutional Review Boards of The Second Affiliated Hospital of Zhejiang University (2013-032).
Data Availability
All data generated or analyzed during this study are included in this published article/as supplementary information files.
Author information
Authors and Affiliations
Corresponding authors
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
He, C., Chen, C., Yang, Y. et al. Clinical Characteristics and Prognostic Significance of Subclinical Seizures in Focal Epilepsy: A Retrospective Study. Neurol Ther 11, 763–779 (2022). https://doi.org/10.1007/s40120-022-00342-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40120-022-00342-y