Abstract
Introduction
The preference for using transradial access (TRA) over transfemoral access (TFA) in patients requiring percutaneous coronary intervention (PCI) is based on evidence suggesting that TRA is associated with less bleeding and fewer vascular complications, shorter hospital stays, improved quality of life, and a potential beneficial effect on mortality. We have limited study data comparing the two access routes in a patient population with atrial fibrillation (AF) undergoing PCI, who have a particular increased risk of bleeding, while AF itself is associated with an increased risk of thromboembolism.
Methods
Using data from the RIVA-PCI registry, which includes patients with AF undergoing PCI, we analyzed a high-bleeding-risk (HBR) cohort. These patients were predominantly on oral anticoagulants (OAC) for AF, and the PCI was performed via radial or femoral access. Endpoints examined were in-hospital bleeding (BARC 2–5), cerebral events (TIA, hemorrhagic or ischemic stroke) and coronary events (stent thrombosis and myocardial infarction).
Results
Out of 1636 patients, 854 (52.2%) underwent TFA, while 782 (47.8%) underwent the procedure via TRA, including nine patients with brachial artery puncture. The mean age was 75.5 years. Groups were similar in terms of age, sex distribution, AF type, cardiovascular history, risk factors, and comorbidities, except for a higher incidence of previous bypass surgeries, heart failure, hyperlipidemia, and chronic kidney disease (CKD) with a glomerular filtration rate (GFR) < 60 ml/min in the TFA group. No clinically relevant differences in antithrombotic therapy and combinations were present at the time of PCI. However, upon discharge, transradial PCI patients had a higher rate of triple therapy, while dual therapy was preferred after transfemoral procedures. Radial access was more frequently chosen for non-ST-segment elevation myocardial infarction (NSTEMI) and unstable angina pectoris (UAP) cases (NSTEMI 26.6% vs. 17.0%, p < 0.0001; UAP 21.5% vs. 14.5%, p < 0.001), while femoral access was more common for elective PCI (60.3% vs. 44.1%, p < 0.0001). No differences were observed for ST-segment elevation myocardial infarction (STEMI). Both groups had similar rates of cerebral events (TFA 0.2% vs. TRA 0.3%, p = 0.93), but the TFA group had a higher incidence of bleeding (BARC 2–5) (4.2% vs. 1.5%, p < 0.01), mainly driven by BARC 3 bleeding (1.5% vs. 0.4%, p < 0.05). No significant differences were found for stent thrombosis and myocardial infarction (TFA 0.2% vs. TRA 0.3%, p = 0.93; TFA 0.4% vs. TRA 0.1%, p = 0.36).
Conclusions
In HBR patients with AF undergoing PCI for acute or chronic coronary syndrome, the use of TRA might be associated with a decrease in in-hospital bleeding, while not increasing the risk of embolic or ischemic events compared to femoral access. Further studies are required to confirm these preliminary findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Limited studies exist comparing transradial (TRA) and transfemoral access (TFA) in patients with atrial fibrillation undergoing percutaneous coronary intervention (PCI), who face elevated risk of bleeding and thromboembolism. |
This analysis is based on data from the RIVA-PCI registry, involving patients who were predominantly on oral anticoagulation for atrial fibrillation. The patients were subjected to PCI either via TRA or TFA for acute or chronic coronary syndrome. |
The study aims to explore the influence of the access site on in-hospital bleeding and cerebral and coronary events. |
Out of 1636 patients studied, 854 (52.2%) underwent TFA, and 782 (47.8%) received TRA. The study found no significant differences in cerebral (transient ischemic attack [TIA], stroke) or coronary events (myocardial infarction, stent thrombosis) between the two groups. However, the TFA group exhibited a higher incidence of in-hospital bleeding (BARC 2-5) (4.2% vs. 1.5%, p < 0.01). |
The preliminary findings suggest that in high-bleeding-risk (HBR) patients with atrial fibrillation undergoing PCI, the use of TRA might be associated with a decrease in in-hospital bleeding without increasing the risk of embolic or ischemic events compared to femoral access. Further studies are needed to confirm these initial results. |
Introduction
Coronary artery disease (CAD) is a major contributor to illness and death worldwide. Patients with CAD and non-valvular atrial fibrillation (AF) who are treated with percutaneous coronary intervention (PCI) require oral anticoagulant (OAC) and additional antiplatelet therapy to prevent thromboembolic events and are therefore exposed to a bleeding risk [1], while AF itself is associated with an increased thromboembolic risk.
According to current guidelines, treatment with novel oral anticoagulants is recommended for these patients, with the optimal additional antithrombotic therapy being the subject of current clinical research. In addition to periprocedural anticoagulation and antiplatelet therapy, periprocedural complications can be related to multiple factors. One of the recently studied predictors, especially for in-hospital bleeding events, is the access site utilized for PCI. The preference of TRA over TFA for coronary catheterization has been endorsed by European and American guidelines [2,3,4]. This is based on evidence suggesting that TRA is linked to a decreased incidence of major bleeding and vascular complications at the access site, shorter hospital stays, and better quality of life [5]. While the majority of the data supporting these findings derive from patients undergoing coronary intervention in the setting of acute coronary syndrome (ACS)—including both non-ST-segment elevation myocardial infarction (NSTEMI) and ST-segment elevation myocardial infarction (STEMI), data regarding patients with AF undergoing coronary intervention in the setting of anticoagulation therapy is scarce [6]. Patients with AF undergoing PCI are more likely to experience adverse outcomes, including in-hospital stroke, compared to those without AF [7]. Moreover, in the recently published SAFARI-STEMI study, there was a trend towards an increased stroke rate in the TRA group compared to the TFA group, with an RR of 2.24. However, the difference was not significant (p-value of 0.12) in this trial [8].
The objective of this analysis was to examine the rates of in-hospital bleeding (BARC 2–5), cerebral events (transient ischemic attack [TIA], hemorrhagic or ischemic stroke) and coronary events (stent thrombosis and myocardial infarction) with respect to the selected access route for coronary intervention in a large, real-world, prospective cohort of patients with AF undergoing PCI (RIVA-PCI registry).
Methods
The “Rivaroxaban in Patients with Atrial Fibrillation Undergoing PCI” (RIVA-PCI) was a prospective, non-interventional, multicenter observational study of consecutive patients with non-valvular AF undergoing PCI that examined the current antithrombotic treatment regimen in Germany after PCI in a 14-month follow-up of a real-world population registry. A total of 1636 patients (elective in 52.6%, non-ST-segment elevation acute coronary syndrome [NSTE-ACS] in 39.3%, ST-elevation myocardial infarction in 8.2% of cases) from 51 German hospitals were enrolled in the study. This cohort was not limited to taking specific OACs or platelet aggregation inhibitors. Patients were enrolled between 2018 and 2020, and data have recently been published [9]. The inclusion criteria were as follows: signed written informed consent before or after PCI; age ≥ 18 years; known or newly diagnosed non-valvular AF; PCI with stent implantation during index hospital stay. The exclusion criteria were as follows: participation in any randomized trial influencing the antithrombotic therapy, and patients who were compulsorily detained for treatment of either a psychiatric or physical illness. Informed consent could be signed either before or after PCI. This study demonstrated that the preferred treatment after PCI in patients with AF involves dual antithrombotic therapy (DAT) with a non-vitamin K antagonist oral anticoagulant (NOAC) and clopidogrel. Ischemic and bleeding events during the hospital stay were infrequent, and the recommended durations for combination therapy vary considerably.
In this sub-analysis, patients from the aforementioned study were compared with respect to the site utilized for vascular access during the index PCI.
Endpoints examined were in-hospital bleeding (BARC 2–5), cerebral events (TIA, hemorrhagic or ischemic stroke) and coronary events (stent thrombosis and myocardial infarction).
Additionally, we examined the all-cause mortality, duration of hospital stay, and antithrombotic regimen to gain a more comprehensive understanding of these patient populations.
Stroke was characterized as sudden onset of neurological dysfunction, either focal or global, due to vascular damage of blood vessels in the brain, spinal cord, or retina caused by either hemorrhage or infarction. Ischemic stroke was defined as sudden onset of focal dysfunction of the central nervous system tissue, be it cerebral, spinal, or retinal, caused by infarction. Hemorrhagic stroke refers to a sudden onset of cerebral or spinal dysfunction, either focal or global, caused by intraparenchymal, intraventricular, or subarachnoid hemorrhage. In cases where there was insufficient information to categorize a stroke as either ischemic or hemorrhagic, it was defined as an undetermined stroke. Bleeding episodes were classified according to the Bleeding Academic Research Consortium (BARC) bleeding definitions in this analysis [9].
This study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the ethics committee of Landesärztekammer Rheinland-Pfalz, Germany (no. 837.448.17).
Statistical Analysis
The data for this study were collected and analyzed from the RIVA-PCI study database. We used the Shapiro–Wilk test for normality checks on continuous variables and employed the Mann–Whitney (or Wilcoxon) test for non-normally distributed continuous variables like age and hospitalization days. The chi-squared test was used for categorical variables. A p-value < 0.05 was considered statistically significant. Binary, categorical, and ordinal parameters are described using absolute numbers and percentages, while continuous/numerical variables are described by means of standard statistics. The sample size for this sub-analysis was determined based on the overall sample size of the RIVA-PCI study, which included a total of 1636 patients. Statistical calculations were performed using IBM SPSS Statistics and GraphPad Prism software.
Results
Baseline Characteristics
In the RIVA-PCI study, a total of 1636 patients with AF undergoing PCI were enrolled, of which 854 (52.2%) underwent surgery via TFA, while 782 (47.8%) underwent the procedure via TRA, including nine patients with a brachial artery puncture. These 782 patients were categorized as the radial access group. The mean age of patients in the femoral group was 75.1 years, while the mean age in the radial access group was 75.9 years.
Our analysis revealed no significant differences regarding baseline characteristics between groups, except for a higher prevalence of heart failure (48.1% vs. 38.7%, p < 0.001), a history of previous bypass surgery (15.6% vs. 7.7%, p < 0.0001), hyperlipidemia (64.5% vs. 58.2%, p < 0.01), glomerular filtration rate (GFR) < 60 ml/min (49.4% vs. 42.1%, p < 0.01), and patients with dual therapy at baseline (16.3% vs. 12.3%, p < 0.05) in the femoral access group.
When considering the antithrombotic therapy of these patients with AF undergoing PCI, there was no difference between the TRA and TFA groups regarding those who did not receive any antithrombotic therapy prior to treatment or who received single antiplatelet agent, novel oral anticoagulants (NOAC) only, or vitamin K antagonist (VKA) only. However, a significant number of patients in the TFA group had a dual therapy with acetylsalicylic acid (ASA)/P2Y12-inhibitor + VKA/NOAC (16.3% vs. 12.3%, p < 0.05) (Table 1). An overview of periprocedural anticoagulation and data on coronary intervention is provided in the supplementary material (Tables S1 and S2).
In cases of non-ST-segment elevation myocardial infarction (NSTEMI) and unstable angina pectoris (UAP), the radial approach was more frequently chosen (NSTEMI 26.6% vs. 17.0%, p < 0.0001; UAP 21.5% vs. 14.5%, p < 0.001), while the femoral approach was more commonly used for elective PCI (60.3% vs. 44.1%, p < 0.0001). There was no difference between the two groups in cases of ST-segment elevation myocardial infarction (STEMI) (Table 1). A vascular closure device was used more frequently after femoral puncture (73.8% vs. 17.4%, p < 0.0001).
In both groups, the median CHA2DS2-VASc score was 5, while the median HAS-BLED score was 3 in the femoral access group and 2 in the radial access group. Figure 1 provides a detailed overview of the distributions of both scores.
Examined Endpoints
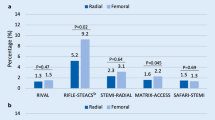
Our results show that there was no significant difference in the incidence of in-hospital stroke or TIA between the two groups (two ischemic strokes in the TFA group and one ischemic stroke and one stroke of unknown type in the TRA group). There was a significant difference in the incidence of bleeding events, with significantly more bleeding that required transfusion or surgical therapy (BARC 3) in the TFA group than in the TRA group (1.5% vs. 0.4%; p < 0.05). No fatal bleeding events occurred (Table 2). There were no differences observed between the femoral access and radial access groups in terms of coronary events. The incidence of myocardial infarction was 0.4% in the femoral access group and 0.1% in the radial access group (p = 0.36), and the occurrence of stent thrombosis was 0.2% in the femoral access group and 0.3% in the radial access group (p = 0.93).
Additionally, the rate of all-cause mortality was similar between the two groups (0.9% [TFA] vs. 0.5% [TRA], p = 0.31). The average length of the hospital stay until discharge for patients undergoing PCI was 4 days in both the femoral access and radial access groups. Upon discharge, transradial PCI patients had a higher rate of triple therapy (33.2% vs. 19.3%, p < 0.0001), while dual therapy was preferred after transfemoral procedures (72.7% vs 59.2%, p < 0.0001).
Discussion
This sub-analysis of the RIVA-PCI trial [9] aimed to investigate in-hospital bleeding and cerebral and coronary event rates depending on femoral or radial access for coronary intervention in patients with AF. Meta-analyses showed that in patients undergoing coronary angiography and PCI, radial access (TRA) was associated with a significant reduction in the risk of bleeding, vascular complications, and mortality compared to femoral access (TFA). The risk of stroke or MI was comparable in patients with radial or femoral access [6, 10]. A considerable number of these studies investigated coronary interventions performed in the setting of acute coronary syndrome (ACS). Moreover, it is still controversial whether the benefits of radial versus femoral access for coronary angiography and PCI are based on the choice of access site itself, the experience of the operator, or other mechanisms.
The data underlying the present investigation derive from the RIVA-PCI registry and therefore address a cohort of patients with AF who underwent PCI in the setting of both acute and chronic coronary syndrome. The study was conducted at multiple centers, allowing any significant differences in the selected parameters of bleeding and cerebral event rate to be attributed with high confidence to the selected access route. The study focused on a homogeneous patient population with AF, which adds to the growing evidence comparing these two access sites in patients undergoing PCI.
In this study, the use of femoral access for PCI was associated with a higher bleeding rate (BARC 2–5) compared to the radial access group (4.2% vs. 1.5%, p < 0.01). This is caused mainly by significantly more bleeding that requires transfusion or surgical therapy (BARC 3). These differences were observed peri-procedurally. This is particularly remarkable considering that in the TRA group, significantly more patients were prescribed triple therapy at the time of discharge relative to the TFA group, which predominantly received dual therapy. Our results are consistent with the data from a recently published meta-analysis of 31 randomized controlled trials (RCTs) comparing radial versus femoral access sites for coronary angiography and PCI. In that meta-analysis, radial access was associated with a significant reduction in major bleeding compared to femoral access (odds ratio [OR] 0.53, 95% CI 0.42–0.66). These findings were consistent regardless of clinical characteristics or whether coronary angiography was performed with or without PCI [6]. A similar OR of 0.55 was calculated in the meta-analysis by Gargiolu et al., which was published in 2022 [5]. The OR for the defined endpoint of BARC 2–5 bleeding in our analysis is 0.36 [95% CI 0.18–0.68]. These findings should be taken into consideration when selecting the optimal access site for PCI, particularly in patients who may be at higher risk for bleeding complications. In our analysis, the number needed to treat (NNT) was 37 to avoid relevant bleeding that needed to be treated (BARC 2–5). Regardless of the puncture technique used and any additional measures taken to limit vascular complications, there is mounting evidence that the superior safety profile of TRA over TFA persists. While advancements in ultrasound guidance and micropuncture needles may reduce the incidence of vascular complications after TFA, TRA still appears to yield better outcomes in terms of safety [11, 12]. Regarding the use of ultrasound guidance for the TFA procedure, we did not record data on its utilization in the registry. Therefore, we do not have specific information on whether ultrasound guidance was employed or its potential impact on bleeding events.
The incidence of TIA and stroke, however, was similar between the groups. Although the TRA is associated with an increased rate of subclinical cerebral embolism, no differences in the rates of clinically relevant strokes have been demonstrated in large randomized studies [8, 13, 14]. This may be due to the low incidence of periprocedural strokes. With radial access, there could be an increased risk of stroke due to catheter manipulation in the extracranial cervical vessels, but on the other hand, this risk also exists with femoral access and catheter manipulation in the aortic arch, especially in the presence of plaques in the thoracic aorta. In our study, two strokes occurred in each group, two ischemic strokes in the femoral access group (0.2%) and one ischemic stroke and one stroke of unknown type in the patient cohort with radial access (0.3%). As no systematic neurological examination was performed post-intervention, as in most of the other studies, the true event rate may be higher. The following consideration should be mentioned at this point: even to investigate a 50% increase in stroke rate with a stroke incidence of 0.2% and a statistical power of 80%, nearly 80,000 patients would be required.
The findings of this study also suggest that the selection of access site, either TFA or TRA, did not significantly impact the risk of stent thrombosis or myocardial infarction. The occurrence of stent thrombosis was observed in 0.2% of patients in the TFA group and 0.3% of patients in the TRA group, with no statistically significant difference between the two groups (p = 0.93). Similarly, myocardial infarction was seen in three cases in the TFA group and one case in the TRA group, with no significant difference between them (p = 0.36). This is consistent with previous research indicating comparable outcomes between the femoral and radial approaches in terms of coronary events [5]. The studied data additionally revealed that there was no difference in all-cause mortality between groups. During the time until discharge, 12 out of 1636 patients died—eight in the TFA group (six due to cardiac death, two unknown) and four in the TRA group (three due to cardiac death, one unknown). There was no statistically significant difference between groups (0.9% vs. 0.5%, p = 0.31). The mean length of stay after PCI until discharge was 4 days, and there was no significant difference between groups. A meta-analysis published in 2022 showed that all-cause mortality was lower with TRA than with TFA at 30 days (1.6% versus 2.1%; HR, 0.77 [95% CI 0.63–0.95]; p = 0.012), with a number needed to treat to benefit (NNTB) of 214. Landmark analyses demonstrated that the benefit in favor of TRA was mainly observed in the first few days after the index intervention (0.5% vs. 0.8%; HR 0.64 [95% CI 0.46–0.90]; 0.010) [5]. Our data roughly correspond to these results, although our sample size was significantly smaller, thereby increasing the susceptibility to type II error. Major bleeding can be considered as a mediator of all-cause mortality (indirect effect), especially in patients with moderate or severe baseline anemia [15].
The access route was chosen by the interventionalist, and therefore it is possible that sicker, possibly more complex patients were more likely to receive their PCI through the femoral access. As shown in Fig. 1, patients with high CHA2DS2VASc-score (5–9) were more likely to receive a femoral approach. Patients treated via the TFA also had a higher prevalence of heart failure and previous CABG surgeries. Interventionalists may have opted for the femoral access route more frequently for sicker patients. In a noteworthy study, a model was developed and internally validated to predict the risk of vascular access site complications during percutaneous coronary intervention (PCI) via a transfemoral approach. The findings of this study illustrated that patients with the lowest risk for access site complications predominantly received transradial arterial access, whereas those with the highest risk were more likely to undergo transfemoral arterial access. This underscores a potential discrepancy in the allocation of arterial access strategies, highlighting that the rational employment of transradial arterial access as a means to mitigate vascular access site complications is suboptimal and warrants considerable enhancement in its application. This insight is crucial as it sheds light on the need for refined strategies to optimize the use of transradial arterial access, particularly for patients at higher risk, to prevent complications and improve overall patient outcomes [16].
Analysis of in-hospital bleeding, cerebral and coronary events in the RIVA-PCI trial population, based on the access route (femoral/radial) for coronary intervention. In addition to the absolute numbers of affected patients, event rates are given as percentages. The case number of patients with femoral access (TFA) was 854, that with radial access (TRA) 782
Overall, this study suggests that using femoral access during PCI in patients with AF may be associated with a higher in-hospital bleeding rate compared to radial access, but there is no difference in cerebral or coronary event rate. These data is in line with previously published results of comparisons between both access routes, which, however, predominantly originated from patients who received PCI for acute coronary syndrome and were not all affected by AF. The data here provided constitute an examination of “real world” practices in an environment with similar rates of radial and femoral access and inclusion of a full spectrum of CAD presentations. Therefore, further research is needed to confirm these results and to determine which patients are most likely to benefit from each access site.
Study Limitations
The RIVA-PCI study was a registry-based observational study. Registry studies have some limitations that should be considered to interpret the results appropriately. These limitations include selection bias, where certain groups of patients may be less likely to be included in the registry or may be less willing to participate. Additionally, information bias is possible, although this registry had high accuracy and completeness of data. Like all registry studies, it is not easy to account for the influence of other factors on the outcomes being studied (confounding). The calculations performed in this study were retrospective, provide valuable information, and generate hypotheses, but randomization into TRA and TFA groups as in an RCT was not performed. Furthermore, the group sizes were not a priori powered to detect a small difference in mortality, coronary or cerebral events. A more detailed analysis of the cause-and-effect relationship appears unfeasible to us due to the exploratory nature of this registry-based observational study.
Conclusions
In conclusion, this sub-analysis of the RIVA-PCI trial demonstrates that using radial access for coronary intervention in patients with AF might be associated with a decrease in in-hospital bleeding (BARC 2–5) compared to femoral access, while not increasing the risk of embolic or ischemic events. Notably, this study included patients with both acute and chronic coronary syndromes undergoing PCI. These findings support the use of transradial access in patients with AF undergoing PCI, particularly in those at higher risk for bleeding complications.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Lopes RD, et al. Antithrombotic therapy after acute coronary syndrome or PCI in atrial fibrillation. N Engl J Med. 2019;380(16):1509–24.
Ibanez B, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: the task force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39(2):119–77.
Collet JP, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42(14):1289–367.
Lawton JS, et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation. 2022;145(3):e18–114.
Gargiulo G, et al. Effects on mortality and major bleeding of radial versus femoral artery access for coronary angiography or percutaneous coronary intervention: meta-analysis of individual patient data from 7 multicenter randomized clinical trials. Circulation. 2022;146(18):1329–43.
Chiarito M, et al. Radial versus femoral access for coronary interventions: an updated systematic review and meta-analysis of randomized trials. Catheter Cardiovasc Interv. 2021;97(7):1387–96.
Patil S, et al. Prevalence and determinants of atrial fibrillation-associated in-hospital ischemic stroke in patients with acute myocardial infarction undergoing percutaneous coronary intervention. Am J Cardiol. 2021;144:1–7.
Le May M, et al. Safety and efficacy of femoral access vs radial access in ST-segment elevation myocardial infarction: the SAFARI-STEMI randomized clinical trial. JAMA Cardiol. 2020;5(2):126–34.
Zeymer U, et al. Current status of antithrombotic therapy and in-hospital outcomes in patients with atrial fibrillation undergoing percutaneous coronary intervention in Germany. Herz. 2023;48(2):134–40.
Ferrante G, et al. Radial versus femoral access for coronary interventions across the entire spectrum of patients with coronary artery disease: a meta-analysis of randomized trials. JACC Cardiovasc Interv. 2016;9(14):1419–34.
Nguyen P, et al. Standard versus ultrasound-guided radial and femoral access in coronary angiography and intervention (SURF): a randomised controlled trial. EuroIntervention. 2019;15(6):e522–30.
Sandoval Y, et al. Contemporary arterial access in the cardiac catheterization laboratory. JACC Cardiovasc Interv. 2017;10(22):2233–41.
Valgimigli M, et al. Radial versus femoral access and bivalirudin versus unfractionated heparin in invasively managed patients with acute coronary syndrome (MATRIX): final 1-year results of a multicentre, randomised controlled trial. Lancet. 2018;392(10150):835–48.
Jolly SS, et al. Radial versus femoral access for coronary angiography and intervention in patients with acute coronary syndromes (RIVAL): a randomised, parallel group, multicentre trial. Lancet. 2011;377(9775):1409–20.
Leonardi S, et al. Prognostic implications of declining hemoglobin content in patients hospitalized with acute coronary syndromes. J Am Coll Cardiol. 2021;77(4):375–88.
Wimmer NJ, et al. Risk-treatment paradox in the selection of transradial access for percutaneous coronary intervention. J Am Heart Assoc. 2013;2(3): e000174.
Acknowledgements
We thank the participants of the study.
Funding
The RIVA-PCI registry has been supported by an unrestricted grant of the Bayer-Vital GmbH, Germany. The authors funded the journal's rapid service fee.
Author information
Authors and Affiliations
Contributions
MB and RT conceived and authored this work, including the development of its concept and the writing of the manuscript with contributions and final reviews by all authors (MB, UZ, HW, H-PH, AC, RB, OR, BH, KH, MH, WS, PL, HM, MS, P-LS, CL, HT, CC, TR, RZ, LI, GR, RT). The statistical analysis was largely carried out by TR and individual analyses were performed by MB.
Corresponding author
Ethics declarations
Conflict of Interest
The article is original, with no portion under simultaneous consideration for publication elsewhere or previously published. All authors have read and approved the submission. Martin Borlich, Uwe Zeymer, Harm Wienbergen, Hans-Peter Hobbach, Alessandro Cuneo, Raffi Bekeredjian, Oliver Ritter, Birgit Hailer, Klaus Hertting, Marcus Hennersdorf, Werner Scholtz, Peter Lanzer, Harald Mudra, Markus Schwefer, Peter-Lothar Schwimmbeck, Christoph Liebetrau, Holger Thiele, Christoph Claas, Thomas Riemer, Ralf Zahn, Leon Iden, Gert Richardt and Ralph Toelg declare no conflicts of interest.
Ethical Approval
This study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the ethics committee of Landesärztekammer Rheinland-Pfalz, Germany (no. 837.448.17). Patient consent to participate in the RIVA-PCI study has been obtained.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Borlich, M., Zeymer, U., Wienbergen, H. et al. Impact of Access Site on Periprocedural Bleeding and Cerebral and Coronary Events in High-Bleeding-Risk Percutaneous Coronary Intervention: Findings from the RIVA-PCI Trial. Cardiol Ther 13, 89–101 (2024). https://doi.org/10.1007/s40119-023-00343-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40119-023-00343-4