Abstract
The present investigation deals with the development of a novel polymer nanocomposite (PNCs) electrodes for simple, selective and sensitive detection of chlorpyrifos (CHL). PNCs were developed using surfactant facilitated polymerization of indole using different concentrations (wt%) of WC ranging 5–30. Formation of PNCs was ascertained through diversified analytical methods. Electrodes were derived from PNCs over stainless steel substrate for electrochemical quantification of CHL. With concentration of WC, the DC conductivity (10−2 × S/cm) of electrodes was increased ranging 3.54–0.75 at 313 K. Electrochemical impedance spectroscopy reveals well stability of electrodes in phosphate buffer (PBS, 0.1 M) at pH 7.4. The performance of electrodes towards detection and quantification of CHL was investigated through square wave voltammetry. Study reveals that detection and quantification of CHL were dependent on concentration of WC in nanocomposites. Square wave voltammetry reveals that the electrode derived from PNCs with 5 wt% of WC has rendered highest limits of detection and quantification of CHL (10−8 mol/L) up to 5.94 and 18. This work describes a viable method of preparation of synergistic blend of WC in PIN matrix having high electrical conductivity, rapid electron shift, huge surface area and enhanced stability for fast and précised electrochemical detection of CHL.
Graphic abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Organophosphates (OPs) are the esters, amide and thiol derivatives of phosphorous-based acid [1,2,3]. Low water solubility and high absorption coefficient impart long residual effect of OPs in soil. This allows the entry of OPs into food chain causing neurotoxicity to animals due to irreversible inhibition of acetyl cholinesterase (AChE) [4,5,6]. For such reasons, development of novel methods of detection of OPs has been the subject of investigation over decades. In this context, various chromatographic, mass spectrometric, electrophoretic, [7,8,9,10] and electrochemical [11,12,13,14] methods were developed for detection of OPs.
Chlorpyrifos (CHL, o,o-diethyl-o-(3,5,6-trichloro-2-pyridinyl)phosphorothioate) belongs to the family of OPs, used for crop protection as insecticide and nematicide. Realizing health hazards imposed over ecosystem, there has been growing concern on advancement of selective, precise, rapid and reliable approach for CHL detection. In this context, the quantification of CHL through chromatographic [15, 16] and electrochemical [17, 18] methods has been well established. Among such methods, based on chromatography involves expensive instrumentation, multiple steps of preparation, immobilization and regeneration of samples, large consumption of chemicals, time along with low sensitivity of detection [19, 20]. However, electrochemical methods, specially based on square wave voltammetry (SWV), have received immense acceptance because of reasonable simplicity, high sensitivity and potential towards selective quantification of detection of CHL down to ng level [21,22,23]. SWV has also been used for detection of a wide range of OPs over nanocomposite-modified GCE. Nanocomposites employed for electrochemical detection of OPs were parathion [24,25,26,27,28], malathion [29, 30] and paraoxon [31, 32]. The nanocomposites employed for modification of GCE for OPs detection were zeolite [24], ZrO2 [25], Au/Graphene [26, 27], ZrO2 [28], SWNT/GO [29], MWCNT [30], Au/Pt [31] and ZrO2/MWCNT [32]. The nanomaterials used for detection of CHL were graphene [17], MWCNT [33,34,35], with nano-TiO2 [36], carbon nitride [37], ferrocene [38], CuO [39]. Conducting polymers have been used as such or in the form of their nanocomposites for detection of CHL through SWV [24, 30]. Literature revealed that a wide range of nano-composites has been employed for the detection of various OPs [24,25,26,27,28,29,30,31,32], including CHL [33,34,35,36,37,38,39]. To the best of our knowledge, no records are available on electrochemical detection of CHL over PNCs derived through immobilization of WC into PIN matrix.
Recently, conducting polymer-based nanostructures containing carbonaceous and inorganic nanomaterials have been used in electrochemical studies for various applications [40,41,42,43,44]. Amongst the family of conducting polymers, polyindole (PIN) has an edge over other, due to its sustained polymerization, high electrical conductivity, low manufacturing cost and less toxicity [45,46,47,48,49]. Sensing behavior of PIN is modified through doping transition metal-based dopants that chanalize the electron transfer mechanism through polaron formation [50]. In this context, common dopants employed for PIN are gold [51], oxides of zinc and nickel [52], tin [53], iron [54], vanadium [55] and copper [56]. Nanocomposite of titanium carbide and poly(3,4-ethylenedioxythiophene) were prepared as an alternative electrocatalyst for dye-sensitized solar cell applications [57,58,59,60]. The present work proposed a novel and economical sensor for the trace level electrochemical detection of CHL, by utilizing WC-doped PIN nanocomposite.
Materials and methods
Materials
CHL with purity (≥ 99.9%), chlorosulfonic acid (> 99%), indole (> 99%) and cetyltrimethylammonium bromide (CTAB) (> 99%) were purchased from Sigma-Aldrich. Rest of the chemicals and solvents (purity > 98%) were indigenously procured and used without further purification. Phosphate buffer (pH 7.4, 0.1 M) and stock solution of CHL (1.0 × 10−4 M) were prepared through traditional methods. In the present results, bare electrode, 5 wt% and 30 wt% PNCs-coated electrode are represented by [I], [II] and [III], respectively.
Preparation of nanocomposites
PIN along with various wt% fractions of WC ranging 5–30 [I–III] was synthesized by cationic surfactant method placed in two-necked glass reaction vessel implemented with mechanical stirrer and dropping funnel. To this, CTAB (2.7 × 10−3 mol) was added and the content was stirred @500 rpm over 6 h. To initiate the polymerization process, a solution of freshly prepared FeCl3 (3.5 M) was added to content @1 mL/min keeping stirring to be continued over 24 h. A dark brown precipitate was produced, that was subsequently filtered and successively washed with deionized water till the filtrate became free from chloride ions. Isolated PNCs was left for 6 h at room temperature and then dried at 40 °C at 400 mmHg for additional 8 h. PIN was also prepared under similar reaction conditions in the absence of WC [61, 62].
Preparation of electrodes
Working electrodes (WE) were prepared through depositing PNCs over stainless steel plates of 1 cm2 area. Prior to deposition of PNCs, the plates were thoroughly finished with emery paper followed by ultrasonicated cleaning with acetone. A suspension of PNCs was prepared through ultrasonicating the composition of SPS (7.5 × 10−3 g) with graphite (5.0 × 10−3 g) and ammonium per sulfate (80 μM) in the presence of PNCs in NMP (1.5 mL) over 2 h. The suspension (125 μL) was applied over electrode and left at room temperature for 8 h, thereafter dried at 40 °C/400 mm Hg for additional 48 h (Scheme 1). The electrodes with mass thickness ranging 1.80–46.15 (× 10−3 g) were obtained and employed for characterization [63].
Instrumentations
SEM images were recorded on JEOL, JSM 6610 LV at 0.2 KX (7 µm) and 15 kV. For this purpose, the electrodes were prepared via mentioned procedure. The SEM images were scanned under identical conditions for comparable results. The conductivity data were recorded over Keithley four-point probe conductivity nanovoltmeter with current (6221 A) and voltage source (2182 V) in the range of 313–373 K.
The activation energy (Ea) was deduced through Arrhenius equation:
where σ is electrical electrodes conductivity, σo is pre-exponential factor k is Boltzmann constant and T is absolute temperature [64].
FT-IR spectra were recorded on Thermo Nicolet ranging 4000–500 cm−1 in KBr. XRD spectra were recorded over Rigaku-Geigerflex, X-Ray diffractometer using Cu-Kα radiation (λ = 0.154 nm) with 2θ ranging 10°–90° at 30 kV and 15 mA. Simultaneous TG–DTA–DTG was conducted over EXSTAR TG/DTA 6300 at sample weight (mg) ranging 10.50–10.55 at 10 °C/min in air.
The electrochemical studies of prepared electrodes were performed in PBS (0.1 M) over IVIUM Potentiostat–Galvanostat using a triple-electrode cell assembly. WE were fabricated through depositing PNCs over SS plates. Pt foil (1 cm2) and Ag/AgCl were used as auxiliary and reference electrodes. All peak currents (µA) and peak potentials (V) were expressed in due units. Calibration curves were obtained from SWV by plotting maximum peak current and respective increasing concentration of CHL.
Limit of detection (LOD) and limit of quantification (LOQ) were calculated from calibration curve using formula: LOQ = 10 s/m and LOD = 3.3 s/m, where s is intercept and m is slope of the calibration curve. Stability of electrodes was investigated through electrochemical impedance spectra (EIS) and circuit simulation was conducted using Randles diagrams.
Results and discussion
Spectral analysis
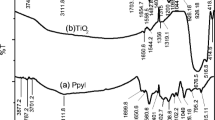
PIN shows characteristic wave numbers (cm−1) corresponding to vN–H (3138.00), vC–H (2925.39), ν δO–H (1617.78) vC–C (1454.64) and δAr–H (745.48). The wave numbers at 2925.39 and 2852.80 attribute to interaction of CTAB with PIN. The wave number at 1110.96, 1334.07 and 1568.39 cm−1 correspond to vC–N, vC=N and δ N–H deformation [65].
WC has shown by the low-frequency fingerprint region at 665.82 cm−1 [66].
[III] shows all the characteristic absorption bands of PIN. EM shows absorptions corresponding to vN–H (3137.12), vC–H (3017.8) and symmetric stretches of WC near the low-frequency fingerprint region (655.84) and out-of-plane deformation for C–H (746.58) (Fig. 1). Interaction of WC has shifted the wave number of PIN to lower values.
XRD
The XRD spectra of the PIN, WC and [III] are shown in Fig. 2. Broad peak at 20.35° attributes to amorphous nature of PIN. Debye–Scherrer calculations provided the corresponding crystallite size of nanoparticles ranging 10–22 nm [67]. Diffraction peaks at 31.68° (2.82), 35.71° (2.51), 48.38° (1.88), 64.05° (1.45), 65.76° (1.42), 73.37° (1.29), 75.63° (1.25), 77.26° (1.23), 84.26° (1.15) correspond to [001], [100], [101], [110], [002], [111], [200], [102], [201] planes of WC. The planes of hexagonal were well matched with JCPDS 73-0471 [68]. Diffraction peaks at 31.03° (2.95), 35.31° (2.54), 47.88° (1.89), 63.65° (1.46), 72.96° (1.29), 75.15° (1.26), 76.87° (1.23), 83.81° (1.15) corresponding to [001], [100], [101], [110], [111], [200], [102], [201] planes reveal the presence of WC in PIN matrix.
Surface morphology of electrodes
Morphology of fabricated electrodes along with quantitative elemental composition was analyzed through SEM–EDX study. To compare the morphology, all electrodes were imaged under identical conditions at 1 KX, 10 μM and camera width of 10 ± 1 nm. EDX spectrum shows the presence of W and high C content in the EM as well as quantity of other elements. EDX represents qualitative detection of elements present in EM. To simulate the emission of X-rays, high-energy beam of charged particles was focused on to the EM. The difference between energies of higher and lower energy shell is released in the form of X-rays which is measured by spectrometer.
SEM and EDX of WC, PIN and PNCs are sequentially shown in Fig. 3a1–a3 and b1–b3, respectively. The dark and bright phases in SEM images represent the morphology of PIN and WC, respectively. Electrodes derived from PIN show a co-continuous morphology without any distinct phase separation. SEM of PIN-coated electrode showed granular structure with rough morphology, which concludes that the nature of particles formed was irregular. Blending of PIN with WC, has afforded with rough morphology. It is observed from SEM images that intercalation of WC into PIN matrix also increases, which leads to roughness in electrode surface. The synergy between the constituents may be weak or strong, which is regulated by the technique used for synthesis of the composite [69]. The reason of surface roughness may be due to the non-homogenized distribution of WC into polymer phase during the procedure of electrode preparation. The morphological structure of PNCs is found to be different from PIN and reveals homogeneous dispersion of WC nanoparticles into PIN matrix.
Thermal stability
Information regarding thermal stability of PIN as well as [III] is provided by thermal studies (Fig. 4). The weight loss caused by evaporation of physically adsorbed moisture and residual solvents is observed in the curves at a temperature lower than 200 °C. The weight residue (Wr) corresponding to TG is expressed as %w/w. DTA signals and rate of degradation of samples in DTG are expressed as mV and mg/Cel, respectively. The heat of fusion data revealed through DTA has been expressed in mJ/mg. TG onset shows thermal stability of WC up to 384 °C. WC shows weight gain of 104.90% due to oxidation at 528 °C [22]. Decomposition of WC was progressed at the rate of − 21.97 × 10−3 mg/°C at 552 °C with DTA signal (0.22 mV) at 548 °C Decomposition of WC was concluded at 597 °C leaving char residue of 114.70 wt% (Fig. 4a).
PIN was decomposed with TG onset at 337 °C leaving 79.4% Wr. Decomposition of PIN was progressed at the rate of 197.6 × 10−3 mg/°C at 559 °C with DTA signal (0.65 mV) at 543 °C. Decomposition of PIN was concluded at 573 °C leaving char residue of 1.90 wt%. DTA reveals fusion of PIN with ∆Hf of − 10.6 × 103 mJ/mg (Fig. 4b). [III] shows single-step decomposition with TG onset at 328 °C leaving 85% Wr. The collective weight loss of 15.0% wt indicates that due to filling of WC, the thermal stability of PIN was compromised. Decomposition of [III] was progressed at the rate of 0.858 mg/°C at 519 °C with DTA signal (0.61 mV) at 520 °C. TG endset of [III] was appeared at 538 °C leaving 36.2% char residue. DTA reveals fusion of [III] with ∆Hf of − 7.44 × 103 mJ/mg (Fig. 4c). Thermal data reveal that due to addition of WC, the thermal stability of [III] was compromised.
Electrical conductivity
The linear variation in I–V (Fig. 5a) at room temperature implies the ohmic conduction behaviour of WC in PIN [70]. The increased conductivity attributes to high conductivity of WC as well as interaction between PIN and WC which favored charge transfer process [71]. Effect of WC on σDC of electrodes was studied under various voltages ranging 1–100 V at room temperature (Fig. 5b). It was found that the conductivity of the [III] increased with concentration of WC and maximum for 100 V which is 3.54 × 10−2 Scm−1. The σDC measurements were also performed at temperature range 313–373 K (Fig. 5c). It was observed that increasing value of σDC with temperature defines the semiconducting behaviour of PNCs and maximum σDC was observed for [III]. Increase in σDC with temperature attributes to enhanced charge mobility across PNCs. Using theoretical Arrhenius model, the calculated activation energies for PIN, WC and [III] were found as 0.035, 0.028 and 0.019 eV (Fig. 5d).
EC behavior of modified electrode
Cyclic voltammetry
CV is the most popular continuous wave technique employed to investigate the redox behavior and electron transfer kinetics of molecules over electrochemically active surface. CV presents the set of anodic (Epa) and cathodic peak (Epc) potentials along with respective anodic (ipa) and cathodic (ipc) peak currents. Potential applied across WE moves back and forth past the formal potential. This leads to current flow across the electrode that renders the redox behavior of analyte [72, 73].
Redox behavior was investigated through scanning the CV of CHL in PBS (0.1 M, pH 7.4) @ 40 mV s−1 over WE derived from PNCs [I] and [II] (Fig. 6). Voltammograms were recorded till fivefold degeneracy in potential window of − 0.11 to + 0.55. [I] has rendered a well-defined redox behavior with peaks peak potentials (− V) ranging 0.089–0.418 V. Under identical conditions, [II] has shown the peak potentials (− V) of 0.093 V and 0.43. Increase in peak current attributes to the formation of electron-conducting tunnel during redox reaction of CHL over [II].The electron transfer mechanism during redox behavior f CHL over [II] has been presented in Fig. 7. Based on peak potential deuced from CV of [II], the redox behavior of CHL over electrodes was found to lead according to one electron transfer mechanism between electrode surface and CHL generating synergy on EC properties [74].
Effect of scan rate on CV
Frequent elevation in scan rate has raised the Ipa (µA) of [I] from 0.084 to 0.279 (Fig. 8a). The coinciding anodic peak potential was observed ranging from − 0.089 to − 0.013 V. At the same time, there was reduction in Ipc (− µA) ranging 0.079–0.235 with corresponding Epc (− V) ranging 0.418–0.454. For [II] with increase in scan rate (Vs−1) from 0.04 to 0.2, there was subsequent increase in Ipa, Epa, Ipc and Epc ranging 0.111–0.329, − 0.093 to − 0.065 V, − 0.099 to − 0.325 and − 0.43 to − 0.52, respectively (Fig. 8b). However, no redox behavior was shown by [III] irrespective of scan rate (Fig. 8c). Figure 9 shows linear regression curve for [II] between Ipa and square root of scan rate. A comparative account reveals higher electrochemical behavior of [II] over [I].
Square wave voltammetry
The SWV experiments have been performed in the potential window (V) from − 2.0 to 2.0 at pulse amplitude 2 mV, scan increment 20 mV and frequency 25 Hz. A comparative SWV voltammograms of electrodes in 0.1 M PBS (pH 7.4) at varying concentrations of CHL are sketched in Fig. 10a–c. The presence of WC in the PIN matrix has raised the peak current that attributes to increase in the sensitivity towards PNCs. With concentration of CHL, the peak was increased over [I] from 6.204 to 7.808 (Fig. 10a). Under identical conditions, the peak currents of [II] and [II] were raised in the range of 7.699–8.99 and 9.892–11.90 (Fig. 10b, c). Linear range of proposed CHL sensor was found to be 25 × 10−7 to 225 × 10−7 mol L−1 and calibration curves obtained between maximum peak current and concentration of CHL added in SWV were used to calculate LOD and limit of quantification LOQ.
The proposed sensor demonstrates acceptable quantification of CHL with significantly low LOD values. The calibration plots for quantification of CHL at [I], [II] and [III] was obtained by studying the effect of increase in CHL concentration on peak current. The LOD obtained for [I], [II] and [III] are 4.8 × 10−8 mol L−1, 5.94 × 10−8 mol L−1 and 4.49 × 10−8 mol L−1. LOQ for [I], [II] and [III] are 14.5 × 10−8 mol L−1, 18 × 10−8 mol L−1 and 13.6 × 10−8 mol L−1 , respectively. The correlations between the concentration and peak current were in linear relation with correlation coefficient (r) for [I], [II] and [III] was 0.979, 0.982, 0.971, respectively, indicating good correlation.
Electrochemical impedance spectroscopy
The impedance data have been expressed in Hz and spectrum was examined by equivalent electrical circuit model (Fig. 11). The elements of the circuit are general electrical components, which include polarization resistance or charge transfer, solution resistance and a double-layer capacitor. EIS measurements were managed by FRA. Nyquist plots at higher and lower frequency range appeared as semicircle attributing to electron transfer resistance (Ret). High impedance in the form of semicircular loop was observed at the surface of electrodes covered with coating of PNCs; whereas, loops were opened within 24 h representing the destruction of coating.
For I, Rp was 8.506 × 102 and |Z| appeared at 1.200 × 103. For [II], Rp was 4.973 × 102 and |Z| appeared at 6.396 × 102. [III] has shown Rp of 2.669 × 102 and |Z| appeared at 2.998 × 102. With the fabrication of electrode with PNCs, the diameter of semicircle decreased, which shows that PNCs possess high electrical conductivity with low charge transfer resistance. It is observed from Fig. 11 that the diameter of the semicircles was successively decreased in the order from [I] to [III], that indicates successive increase in electron transfer at the surface of PNCs-coated electrode. Best semicircular loop in [III] represents highest charge storage and capacitive behavior of [III]. In case of EIS studies [II], opening of semicircular loop indicates the diffusion of electrolyte and that is also responsible for cracking of electrode surface as shown in SEM images (Fig. 12a, b).
Conclusion
A series of electrochemical polymer nanocomposite (PNC)-based electrode was developed by an easy and effective chemical oxidative polymerization method using various concentrations of WC ranging 0–30 wt%. Electrodes from PNCs were developed over SS plates and used for electrochemical quantification of chlorpyrifos (CHL) in phosphate buffer (PBS, 0.1 M) at 7.4. Electroanalytical methods based on square wave voltammetry (SWV) and electrochemical impedance spectroscopy (EIS) reveal immense feasibility of electrodes towards quantification of CHL with stability in PBS. The electrical and electrochemical behavior of electrodes was found synergistic with concentration of WC in PIN. DC conductivity of electrodes was found in increasing order with concentration of WC. Electrodes with 5 wt% of WC have shown enhanced redox behavior of CHL with limits of detection and quantification (10−8 mol L−1) up to 5.94 and 18. Study reveals that incorporation of WC in PIN provides a novel nanohybrid electrode coating material for efficient detection of CHL.
References
Kaur, N., Prabhakar, N.: Current scenario in organophosphates detection using electrochemical biosensors. Trends Anal. Chem. 92, 62–85 (2017)
Ensafi, A.A., Rezaloo, F., Rezaei, B.: Electrochemical determination of fenitrothion organophosphorus pesticide using polyzincon modified-glassy carbon electrode. Electroanal. 29(12), 2839–2846 (2017)
Mehta, J., Vinayak, P., Tuteja, S.K., Chhabra, V.A., Bhardwaj, N., Paul, A.K., Deep, A.: Graphene modified screen printed immunosensor for highly sensitive detection of parathion. Biosens. Bioelectron. 83, 339–346 (2016)
Singh, M., Kashyap, H., Singh, P.K., Mahata, S., Rai, V.K., Rai, A.: AuNPs/Neutral red-biofunctionalized graphene nanocomposite for nonenzymatic electrochemical detection of organophosphate via NO2 reduction. Sensor Actuat. B. 290, 195–202 (2019)
Pearson, J.N., Patel, M.: The role of oxidative stress in organophosphate and nerve agent toxicity. Ann. N. Y. Acad. Sci. 1378(1), 17 (2016)
Berijani, S., Assadi, Y., Anbia, M., Hosseini, M. R. M., Aghaee, E.: Dispersive liquid–liquid microextraction combined with gas chromatography-flame photometric detection: very simple, rapid and sensitive method for the determination of organophosphorus pesticides in water. J. Chrom. A. 1123(1), 1–9 (2006)
Saunders, M., Magnanti, B.L., Carreira, S.C., Yang, A., Alamo-Hernández, U., Riojas-Rodriguez, H., Bartonova, A.: Chlorpyrifos and neurodevelopmental effects: a literature review and expert elicitation on research and policy. Environ. Health. 11(1), S5 (2012)
Guardino, X., Obiols, J., Rosell, M.G., Farran, A., Serra, C.: Determination of chlorpyrifos in air, leaves and soil from a greenhouse by gas-chromatography with nitrogen–phosphorus detection, high-performance liquid chromatography and capillary electrophoresis. J. Chrom. A 823(1–2), 91–96 (1998)
Duffy, G.F., Moore, E.J.: Electrochemical immunosensors for food analysis: a review of recent developments. Anal. Lett. 50(1), 1–32 (2017)
Yang, G., Chen, X., Pan, Q., Liu, W., Zhao, F.: A novel photoelectrochemical sensor for thiamphenicol based on porous three-dimensional imprinted film. Int. J. Electrochem. Sci. 12(8), 7272–7286 (2017)
Zamfir, L.G., Rotariu, L., Bala, C.: Acetylcholinesterase biosensor for drugs based on tetrathiafulvalene–tetracyanoquinodimethane/ionic liquid conductive gels. Biosens. Bioelectron. 46, 61–67 (2013)
Chauhan, N., Narang, J., Pundir, C.S.: Immobilization of rat brain acetylcholinesterase on porous gold-nanoparticle–CaCO3 hybrid material modified Au electrode for detection of organophosphorous insecticides. Int. J. Biol. Macromol. 49(5), 923–929 (2011)
Rackus, D.G., Shamsi, M.H., Wheeler, A.R.: Electrochemistry, biosensors and microfluidics: a convergence of fields. Chem. Soc. Rev. 44(15), 5320–5340 (2015)
Yola, M.L., Atar, N.: A highly efficient nanomaterial with molecular imprinting polymer: carbon nitride nanotubes decorated with graphene quantum dots for sensitive electrochemical determination of chlorpyrifos. J. Electrochem. Soc. 164(6), B223–B229 (2017)
Ramasubramanian, T., Paramasivam, M.: Development and validation of a multiresidue method for the simultaneous determination of organophosphorus insecticides and their toxic metabolites in sugarcane juice and refined sugar by gas chromatography with flame photometric detection. J. Sep. Sci. 39(11), 2164–2171 (2016)
Shamili, A., Dadfarnia, S., Shabani, A.M.H., Saeidi, M., Moghadam, M.R.: High performance liquid chromatographic determination of diazinon after its magnetic dispersive solid-phase microextraction using magnetic molecularly imprinted polymer. Food Anal. Method. 9, 2621–2630 (2016)
Sreedhar, N.Y., Kumar, M.S., Krishnaveni, K.: Sensitive determination of chlorpyrifos using Ag/Cu alloy nanoparticles and graphene composite paste electrode. Sens. Act. B: Chem. 210, 475–482 (2015)
Vera, R., Insa, S., Fontas, C., Antico, E.: A new extraction phase based on a polymer inclusion membrane for the detection of chlorpyrifos, diazinon and cyprodinil in natural water samples. Talanta 185, 291–298 (2018)
Pedrosa, V.D.A., Codognoto, L., Avaca, L.A.: Electroanalytical determination of 4-nitrophenol by square wave voltammetry on diamond electrodes. J. Braz. Chem. Soc. 14(4), 530–535 (2003)
Bebeselea, A., Manea, F., Burtica, G., Nagy, L., Nagy, G.: Electrochemical degradation and determination of 4-nitrophenol using multiple pulsed amperometry at graphite based electrodes. Chem. Bull. Politech. Univ. Timisoara. 53(67), 1–2 (2008)
Viswanathan, S., Radecka, H., Radecki, J.: Electrochemical biosensor for pesticides based on acetylcholinesterase immobilized on polyaniline deposited on vertically assembled carbon nanotubes wrapped with ssDNA. Biosens. Bioelectron. 24(9), 2772–2777 (2009)
Liu, K., Dong, H., Deng, Y.: Recent advances on rapid detection of pesticides based on enzyme biosensor of nanomaterials. J. Nanosci. Nanotech. 16(7), 6648–6656 (2016)
Manisankar, P., Viswanathan, S., Pusphalatha, A.M., Rani, C.: Electrochemical studies and square wave stripping voltammetry of five common pesticides on poly 3, 4-ethylenedioxythiophene modified wall-jet electrode. Anal. Chim. Acta 528(2), 157–163 (2005)
Kaur, B., Srivastava, R.A.: Polyaniline–zeolite nanocomposite material based acetylcholinesterase biosensor for the sensitive detection of acetylcholine and organophosphates. New J. Chem. 39(9), 6899–6906 (2015)
Du, D., Liu, J., Zhang, X., Cui, X., Lin, Y.: One-step electrochemical deposition of a graphene-ZrO2 nanocomposite: preparation, characterization and application for detection of organophosphorus agents. J. Mat. Chem. 21(22), 8032–8037 (2011)
Gong, J., Miao, X., Zhou, T., Zhang, L.: An enzymeless organophosphate pesticide sensor using Au nanoparticle-decorated graphene hybrid nanosheet as solid-phase extraction. Talanta 85(3), 1344–1349 (2011)
Zhu, W., Liu, W., Li, T., Yue, X., Liu, T., Zhang, W., Wang, J.: Facile green synthesis of graphene-Au nanorod nanoassembly for on-line extraction and sensitive stripping analysis of methyl parathion. Electrochim. Acta 146, 419–428 (2014)
Ebrahim, S., El-Raey, R., Hefnawy, A., Ibrahim, H., Soliman, M., Abdel-Fattah, T.M.: Electrochemical sensor based on polyaniline nanofibers/single wall carbon nanotubes composite for detection of malathion. Synth. Met. 190, 13–19 (2014)
Prabhakar, N., Thakur, H., Bharti, A., Kaur, N.: Chitosan-iron oxide nanocomposite based electrochemical aptasensor for determination of malathion. Anal. Chim. Acta 939, 108–116 (2016)
Upadhyay, S., Rao, G.R., Sharma, M.K., Bhattacharya, B.K., Rao, V.K., Vijayaraghavan, R.: Immobilization of acetylcholineesterase–choline oxidase on a gold–platinum bimetallic nanoparticles modified glassy carbon electrode for the sensitive detection of organophosphate pesticides, carbamates and nerve agents. Biosens. Bioelectron. 25(4), 832–838 (2009)
Beleno Cabarcas, M.T., Stoytcheva, M., Zlatev, R., Montero, G., Velkova, Z., Gochev, V.: Chitosan nanocomposite modified OPH-based amperometric sensor for organophosphorus pesticides determination. Curr. Anal. Chem. 14(1), 75–82 (2018)
Xia, N., Gao, Y.: Carbon nanostructures for development of acetylcholinesterase electrochemical biosensors for determination of pesticides. Int. J. Electrochem. Sci. 10, 713–724 (2015)
Chen, D., Liu, Z., Fu, J., Guo, Y., Sun, X., Yang, Q., Wang, X.: Electrochemical acetylcholinesterase biosensor based on multi-walled carbon nanotubes/dicyclohexyl phthalate modified screen-printed electrode for detection of chlorpyrifos. J. Electroanal. Chem. 801, 185–191 (2017)
Zamfir, L.G., Rotariu, L., Bala, C.: A novel, sensitive, reusable and low potential acetylcholinesterase biosensor for chlorpyrifos based on 1-butyl-3-methylimidazolium tetrafluoroborate/multiwalled carbon nanotubes gel. Biosens. Bioelectron. 26(8), 3692–3695 (2011)
Chen, D., Jiao, Y., Jia, H., Guo, Y., Sun, X., Wang, X., Xu, J.: Acetylcholinesterase biosensor for chlorpyrifos detection based on multi-walled carbon nanotubes–SnO2–chitosan nanocomposite modified screen-printed electrode. Int. J. Electrochem. Sci. 10, 10491–10501 (2015)
Kumaravel, A., Chandrasekaran, M.: Electrochemical determination of chlorpyrifos on a nano-TiO2/cellulose acetate composite modified glassy carbon electrode. J. Agri. Food. Chem. 63(27), 6150–6156 (2015)
Yola, M.L., Atar, N.: A highly efficient nanomaterial with molecular imprinting polymer: carbon nitride nanotubes decorated with graphene quantum dots for sensitive electrochemical determination of chlorpyrifos. J. Electrochem. Soc. 164(6), 223–229 (2017)
Jiao, Y., Jia, H., Guo, Y., Zhang, H., Wang, Z., Sun, X., Zhao, J.: An ultrasensitive aptasensor for chlorpyrifos based on ordered mesoporous carbon/ferrocene hybrid multiwalled carbon nanotubes. RSC Adv. 6(63), 58541–58548 (2016)
Xu, G., Huo, D., Hou, C., Zhao, Y., Bao, J., Yang, M., Fa, H.: A regenerative and selective electrochemical aptasensor based on copper oxide nanoflowers-single walled carbon nanotubes nanocomposite for chlorpyrifos detection. Talanta 178, 1046–1052 (2018)
Pal, K., Maiti, U.N., Majumder, T.P., Debnath, S.C., Ghosh, S., Roy, S.K., Otón, J.M.: Switching of ferroelectric liquid crystal doped with cetyltrimethyl ammonium bromide assisted CdS nanostructures. Nanotechnology. 24(12), 125702 (2013)
Elkodous, M.A., El-Sayyad, G.S., Mohamed, A.E., Pal, K., Asthana, N., de Souza Junior, F.G., El-Batal, A.I: Layer-by-layer preparation and characterization of recyclable nanocomposite (CoxNi1–xFe2O4, X = 0.9/SiO2/TiO2). J. Mater. Sci-Mater El. 30(9), 8312–8328 (2019)
Pal, K., Sajjadifar, S., Elkodous, M.A., Alli, Y.A., Gomes, F., Jeevanandam, J., Sigov, A.: Soft, self-assembly liquid crystalline nanocomposite for superior switching. Electron. Mater. Lett. 15(1), 84–101 (2019)
Pal, K., Elkodous, M.A., Mohan, M.M.: CdS nanowires encapsulated liquid crystal in-plane switching of LCD device. J. Mater. Sci-Mater. El. 29(12), 10301–10310 (2018)
Pal, K.: Hybrid Nanocomposites: Fundamentals, Synthesis, and Applications”, CRC Press, 2019
Cai, Z., Yang, G.: Synthesis of polyindole and its evaluation for Li-ion battery applications. Synth. Met. 160, 1902–1905 (2010)
Döşlü, S.T., Mert, B.D., Yazıcı, B.: Polyindole top coat on TiO2 sol–gel films for corrosion protection of steel. Corros. Sci. 66, 51–58 (2013)
Tebyetekerwa, M., Yang, S., Peng, S., Xu, Z., Shao, W., Pan, D., Zhu, M.: Unveiling polyindole: freestanding As-electrospun polyindole nanofibers and polyindole/carbon nanotubes composites as enhanced electrodes for flexible all-solid-state supercapacitors. Electrochim. Acta 247, 400–409 (2017)
Zhou, W., Xu, J.: Progress in conjugated polyindoles: synthesis, polymerization mechanisms, properties, and applications. Polym. Rev. 57(2), 248–275 (2017)
Rita, Mehtab, S., Zaidi, M.G.H., Singhal, K., Arya, B., Siddiqui, T.I.: Polyindole Based Nanocomposites and their Applications: A Review. Mat. Sci. Res. Ind. 16(2), 97–102 (2019)
Taylan, N.B., Sari, B., Unal, H.I.: Preparation of conducting poly(vinyl chloride)/polyindole composites and freestanding films via chemical polymerization. J. Polym. Sci. Pol. Phys. 48(12), 1290–1298 (2010)
Joshi, L., Prakash, R.: One-pot synthesis of Polyindole–Au nanocomposite and its nanoscale electrical properties. Mat. Lett. 65(19), 3016–3019 (2011)
Rajasudha, G., Nancy, A.P., Paramasivam, T., Boukos, N., Narayanan, V., Stephen, A.: Synthesis and characterization of polyindole–NiO-based composite polymer electrolyte with LiClO4. Int. J. Polym. Mater. 60(11), 877–892 (2011)
Kumar, A., Pandey, A.C., Prakash, R.: Electro-oxidation of formic acid using polyindole-SnO2 nanocomposite. Catal. Sci. Tech. 2(12), 2533–2538 (2012)
Ganesan, R., Dhinasekaran, D., Paramasivam, T., Boukos, N., Vengidusamy, N., Arumainathan, S.: Preparation and characterization of polyindole-iron oxide composite polymer electrolyte containing LiClO4. Polym. Plast. Technol. 51(3), 225–235 (2012)
Zhou, X., Chen, Q., Wang, A., Xu, J., Wu, S., Shen, J.: Bamboo-like composites of V2O5/polyindole and activated carbon cloth as electrodes for all-solid-state flexible asymmetric supercapacitors. ACS Appl. Mater. Interfaces. 8(6), 3776–3783 (2016)
Trung, V.Q., Huyen, D.N.: Synthesis, properties and application of polyindole/TiO2 nanocomposites. J. Phy. Conf. Ser. 187(1), 012058 (2009)
He, J., Duffy, N.W., Pringle, J.M., Cheng, Y.B.: Conducting polymer and titanium carbide-based nanocomposites as efficient counter electrodes for dye-sensitized solar cells. Electrochim. Acta 105, 275–281 (2013)
Thirugnanasambandan, T., Pal, K., Sidhu, A., Elkodous, M.A., Prasath, H., Kulasekarapandian, K., Jeevanandam, J.: Aggrandize efficiency of ultra-thin silicon solar cell via topical clustering of silver nanoparticles. Nano-Struct. Nano-Objects 16, 224–233 (2018)
El-Nasser, S.A., Kim, S., Yoon, H., Toth, R., Pal, K., Bechelany, M.: Sodium-assisted TiO2 nanotube arrays of novel electrodes for photochemical sensing platform. Org. Electron. 76, 105443–105450 (2020)
Govindasamy, G., Pal, K., Elkodous, M. A., El-Sayyad, G.S., Gautam, K., Murugasan, P.: Growth dynamics of CBD-assisted CuS nanostructured thin-film: optical, dielectric and novel switchable device applications. J. Mater. Sci-Mater. El. 30(17), 16463–16477 (2019)
Nihmath, A.M., Ramesan, T.: Effect of hydroxylapatite nanoparticles on structural and electrical properties of ethylene propylene diene monomer rubber. J. Chem. Pharm. Sci. 38–44 (2016)
Mudila, H., Rana, S., Zaidi, M.G.H., Alam, S.: Polyindole/graphene oxide nanocomposites: the novel material for electrochemical energy storage. Fuller. Nanotub. Car. N. 23(1), 20–26 (2015)
Mudila, H., Rana, S., Zaidi, M.G.: Supercritical CO2 aided polyindole-graphene nanocomposites for high power density electrode. Adv. Mater. Let. 8(3), 269–275 (2017)
Aziz, S.B., Kadir, M.F.Z., Abidin, Z.H.Z.: Structural, morphological and electrochemical impedance study of CS: LiTf based solid polymer electrolyte: reformulated arrhenius equation for ion transport study. Int. J. Electrochem. Sci. 11, 9228–9244 (2016)
Hoffmann, P., Galindo, H., Zambrano, G., Rincón, C., Prieto, P.: FTIR studies of tungsten carbide in bulk material and thin film samples. Mater. Char. 50(4–5), 255–259 (2003)
Wang, R.X., Fan, Y.J., Wang, L., Wu, L.N., Sun, S.N., Sun, S.G.: Pt nanocatalysts on a polyindole-functionalized carbon nanotube composite with high performance for methanol electrooxidation. J. Power Sources 287, 341–348 (2015)
Lee, S.J., Jeong, J.R., Shin, S.C., Kim, J.C., Chang, Y.H., Lee, K.H., Kim, J.D.: Magnetic enhancement of iron oxide nanoparticles encapsulated with poly (d, l-latide-co-glycolide). Colloid. Surf. A. Physiochem. Eng. Asp. 255(1–3), 19–25 (2005)
Radja, I., Djelad, H., Morallon, E., Benyoucef, A.: Characterization and electrochemical properties of conducting nanocomposites synthesized from p-anisidine and aniline with titanium carbide by chemical oxidative method. Synth. Met. 202, 25–32 (2015)
Doşlu, S.T., Mert, B.D., Yazıcı, B.: The electrochemical synthesis and corrosion behaviour of TiO2/poly (indole-co-aniline) multilayer coating: experimental and theoretical approach. Arab. J. Chem. 11(1), 1–13 (2018)
Ramesan, M.T.: Fabrication and characterization of conducting nanomaterials composed of copper sulfide and polyindole.: Polym. Compos. 33(12), 2169–2176 (2012)
Cai, Z.J., Zhang, Q., Song, X.Y.: Improved electrochemical performance of polyindole/carbon nanotubes composite as electrode material for supercapacitors. Electron. Mat. Lett. 12(6), 830–840 (2016)
Elgrishi, N., Rountree, K.J., McCarthy, B.D., Rountree, E.S., Eisenhart, T.T., Dempsey, J.L.: A practical beginner’s guide to cyclic voltammetry. J. Chem. Educ. 95(2), 197–206 (2017)
Ji, D., Liu, L., Li, S., Chen, C., Lu, Y., Wu, J., Liu, Q.: Smartphone-based cyclic voltammetry system with graphene modified screen printed electrodes for glucose detection. Biosens. Bioelectron. 98, 449–456 (2017)
Cannon, R.D.: Electron transfer reactions. Butterworth-Heinemann ISBN 0-408-10646-8 (2016)
Acknowledgements
Financial support by the Ministry of Defence No. ERIP/ER/0703649/M/01 is hereby acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the author.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Joshi, P., Mehtab, S., Zaidi, M.G.H. et al. Development of polyindole/tungsten carbide nanocomposite-modified electrodes for electrochemical quantification of chlorpyrifos. J Nanostruct Chem 10, 33–45 (2020). https://doi.org/10.1007/s40097-019-00326-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40097-019-00326-9