Abstract
We report a facile synthesis of Titanium dioxide (TiO2) nanoparticles by sol gel technique assisted by biogenic route using the rind of Aloevera and demonstrate the antibacterial assessment against human pathogens causing urinary tract infection. The synthesized nanoparticles were characterized by powder XRD analysis, FT-IR analysis and scanning electron microscopic analysis. The XRD spectrum confirmed that the synthesized TiO2 nanoparticles exhibit anatase phase. Average grain size was calculated using Debye–Scherrer formula and it was found to decrease from 13 to 8 nm with increasing template concentration. FTIR spectrum showed characteristic bands at 1626, 1056 and 1074 cm−1 revealing C–N stretching of amino groups present in the protein cages of Aloevera which assist in the formation of TiO2 nanoparticles. Morphological characterization analyzed by SEM showed nanocoral network and all the networks displayed excellent invitro bioactivity against Staphylococcus aureus, Escherichia coli, Klebsiella pneumoniae, Salmonella typhi and Proteus mirabilis. Drug delivery was assessed by the release of anti-inflammatory streptomycin which is evidenced by the release profile suggesting that it has the potential to provide better deliveries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nanostructured materials have captured the attention of researchers across the world because of the transitional physical and chemical attributes [1]. Metal oxide nanoparticles play a critical and dominant part in diverse areas that are capable of forming diversified oxide compounds adopting many geometrical structures with unique properties due to size and edge corner sites [2]. Titanium dioxide (TiO2) is one of the fascinating biologically compact material, which can live in three nanostructured forms, anatase (tetragonal), brookite (rhombohedral) and rutile (tetragonal) with six coordinate titanium ions possessing high refractive index, low absorption emission in visible and near infrared spectral region [3]. At the bulk state, rutile is thermodynamically stable, whereas anatase and brookite are capable of transforming to rutile by thermal treatment. All the crystalline modifications have different adsorption properties and responses due to different band structures. In nanocrystalline regime, the anatase TiO2 structure is dominant and industrially relevant in many applications such as photo catalysis, photovoltaic cells, sensors and biomedical applications [4–8].
Moreover, TiO2 at anatase phase with its increased thermal stability up to the range of sintering temperature shows enhanced photo catalytic and antimicrobial performances [7]. As the demand for drugs is increasing exponentially worldwide, various mechanisms were adopted to synthesize anatase phase TiO2 nanoparticles for antibiotic resistance against human and animal pathogens. Various synthesis routes such as sol gel, hydrothermal and solvothermal have been adopted to prepare anatase phase TiO2 nanoparticles [9]. Texturing nanocoral morphology seems to be challenging because of the synthesize protocol but it could be achieved by template technique [10]. Instead of employing various biotemplating materials like gelatin, starch, gum, rice straw, egg shell membrane, bamboo membrane, east pollen, etc., It is seen that lignocellulosic material play a vital role as templating agent and it is proved that soft biotemplates enhances the formation of TiO2 nanoparticles successfully [11–13].
Aloe barbadensis is a medicinal plant and a lignocellulosic material whose pulp contains 0.5% solid materials, which is composed of water-soluble compounds, fat-soluble vitamins, minerals, enzymes, polysacrides, organic acids and 99.5% of water [14–16]. In the extracellular synthesis of nanoparticles using plants, the aggregation of nanoparticles is done by stabilization due to the protein-nanoparticles interaction, and therefore, morphology can be influenced by the carbonyl compounds present in Aloevera. Very recently, enhanced textural properties and coral morphology of TiO2 nanoparticles have been discussed and reported while using water storage cells of Aloevera as template medium [17]. However, analysing the related literature reveals that the bactericidal activity of TiO2 against human pathogens has not yet been studied. In this work, Aloevera templated bioactive anatase phase TiO2 nanocorals were studied to evaluate the level of adsorption and release of streptomycin nanocorals, to counter the human pathogens causing urinary tract infection (UTI). Subsequently, we prove that the percentage of templating agent and the calcination temperature enhance the effectiveness of structural and morphological attributes. Interpretation from the antimicrobial activity of pathogens loaded with streptomycin drug suggest that TiO2 assisted by Aloevera could be a better antibacterial agent for biomedical and pharmaceutical applications.
Materials and methods
All chemicals used in this work were purchased from Merck and were used without any further purification. Titanium (IV) isopropoxide and glacial acetic acid were used as main precursor and chelating agent, respectively, and deionized water was used throughout the experiment. Aloevera leaves used in the experiment were washed and the rind parts were finely chopped, shadow dried, ground into fine powder using agate mortar.
Synthesis of TiO2 nanoparticles
TiO2 solution was prepared by adding 2 mL of titanium (IV) isopropoxide in 200 mL of deionized water under continuous magnetic stirring at room temperature. During the preparation of precursor solution, 20 mL of glacial acetic acid acting as the chelating agent was added in drops, which prevent titanium isopropoxide from the nucleophilic attacks by water. The resulting solution was stirred for few hours and then Aloevera rind powder with 0.4, 0.8, 1.2 and 1.6 g concentrations were introduced into the solution (the ratios of titanium (IV) isopropoxide to Aloevera rind powder were chosen at 1 : 0, 1:0.4, 1:0.8, 1:1.2 and 1:1.6 w/w%). The solution was heated at 80 °C, until the xerogel was completely dried and then cooled naturally to room temperature. The dried gel was crushed into fine powder and finally, the powdered samples were calcinated at 500 °C for 5 h in a muffle furnace. Before calcination, FTIR spectra were obtained to study the functional group vibrations.
Result and discussion
Powder XRD analysis
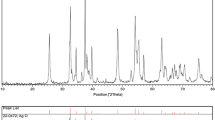
The structure and phase of the synthesized TiO2 nanoparticles were determined using Bruker D8 advance Powder XRD diffractometer and the recorded XRD pattern is shown in the Fig. 1. The predominant peaks observed for various template concentrations of Aloevera in the following ratios of 1:0, 1:0.4, 1:0.8, 1:1.2 and 1:1.6 g calcinated at 500 °C were investigated.
All the diffraction peaks agreed very well with the tetragonal structure, with lattice parameters a = b = 3.784 Å and c = 9.514 Å (JCPDS File No. 78-2486) and the appearance of diffraction planes of (101), (200), (004), (211) and (204) confirms that the exhibited phase was anatase. As the template concentration exceeds the precursor concentration at the ratios from 1:1.2 to 1:1.6 g, a slight transformation starts, which was confirmed by the appearance of additional diffraction peaks at 31.6º and 45.4º corresponding to (111) and (113) diffraction planes indicating that the percentage of Aloevera composition hinders the grain growth of the nanoparticles. Analysis of prominent diffraction peaks using Debye–Scherer’s equation showed that the average grain size decreased from 13 to 8 nm with increasing template concentration with cell parameters a = b = 3.7856 Å and c = 9.514 Å, which agrees very well with the anatase phase of the TiO2 nanoparticles. The XRD pattern shows that the main phase is still anatase, however, when the template concentration is increased, minor peaks of the rutile phase appear and the crystallite size decreases to 8 nm.
FTIR spectroscopy analysis
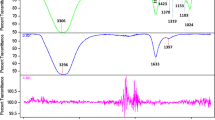
FT-IR spectra of the prepared TiO2 nanoparticles calcinated at 500 °C have been recorded using BRUKER RFS 27 spectrophotometer ranging from 400 to 4000 cm−1 at room temperature (Fig. 2). The broad band appeared at 3406 cm−1 which was assigned to hydrogen bonded OH stretching vibration. The absorption bands in the range of 3000–3500 and 1600 cm−1 in all the spectra indicate the presence of hydroxyl stretching vibration groups. The peaks observed at 1023, 1056 and 1074 cm−1 correspond to Ti–O–C bond which may be due to the increased intensity of template concentration indicating the interaction between organic and inorganic components present in the precursor and the template during condensation and chelation process. The bands that appeared at 1630 and 1626 cm−1 are attributed to amide groups present in Aloevera. A slight shift in the spectrum occurring at 1528 and 1626 cm−1 indicates binding of proteins with the surface of TiO2 nanoparticles at high Aloevera concentrations [15]. Similarly the bands appeared at 1427 and 1074 cm−1 are associated with carboxylic acid and C–N stretching vibration of amine group, respectively. The bands at 649, 652 and 621 cm−1 are attributed to the stretching vibration of TiO2. The broad peak at 564 cm−1 indicates Ti–O–Ti stretching vibration.
SEM analysis
Scanning electron microscopy (SEM) analysis was performed using FEI quanta 200 electron microscope to study the morphology of the synthesized TiO2 nano powder at different template concentration and the images are shown in Fig. 3a, e. It clearly indicates the step wise transition of TiO2 nanoparticles while increasing the template concentration from relatively undefined agglomeration. SEM image of TiO2 nanoparticles without template (Fig. 3a) depicts the constitution of some coral shaped TiO2 nanoparticles associated with agglomeration and scraggy surface, which might be due to the process of simultaneous hydrolysis and condensation. At this initial stage, it is interesting to note that the nanoparticles appear to be agglomerated completely with no sign of well developed faces despite the appearance of anatase phase. It is evident from Fig. 3b, d that the average size of the TiO2 nanoparticles synthesized with varying template concentration varies from 30 to 15 nm, which can be associated with the shrinkage of mesostructure upon protein cages present in the Aloevera. On the contrary, both SEM analysis and XRD analysis suggest that the size of the nanoparticles varies significantly upon the increasing template concentration. However, the increased concentration of Aloevera seems to have decreased agglomeration which might be due to the presence of organic ligands at the surface of the nanoparticles implying that the organic complexes present in Alovera effectively controls the size and morphology of nanoparticles. It is apparent from the Fig. 3b–e that the impact of template is clearly observed on the control of agglomeration and reduction of particle size. However, from Fig. 3d, e it is observed that there is no significant change in morphology which could be attributed to the decreased difference in concentration percentages. Uniformity of particles point out the association of bioconstituents with TiO2 nanoparticles during the synthesis process. The elemental composition of the synthesized TiO2 nanoparticles was confirmed by the energy dispersive X-Ray analysis under SEM (Fig. 3f). It reveals the presence of Ti and O signals with an atomic ratio 2:1. The less significant percentage of oxygen composition is attributed due to the adsorption of oxygen from the Aloevera constituents during the formation of nanoparticles.
The fabrication procedure of TiO2 nanostructures is schematically illustrated in Fig. 4. The results indicate the presence of nucleation-seed-growth step in the formation of nanoparticles. Addition of Aloevera rind powder tends to change the nature of precursor to slurry. This observation is supported by Fig. 3b. Furthermore, the solution was suspended into the template medium and kept static for 48 h. Ti ions are absorbed on the surface of the template, especially on the pores of protein constituents that are present in the phosynthetic cells of the rind part of Aloevera. The protein constituent in the Aloevera functions both as the capping agent and structure directing agent such that the chemisorbed Ti hydroxides are dehydrated to form TiO2 nanocomposites during calcinations. At the calcinations temperature, removal of bioconstituents, as well as spontaneous aggregation takes place that results in the formation of nanocoral morphology.
Antibacterial activity
Various bacterial strains (Staphylococcus aureus, Escherichia coli, Klebsiella pneumoniae, Salmonella typhi and Proteus mirabilis) that cause UTI were maintained on Nutrient agar slants at 4 °C. Antibacterial activity of synthesized TiO2 nanoparticles by Agar well diffusion technique. The pathogens to be tested were spread on the Mueller–Hinton Agar plates with sterile cotton swap. Subsequently, wells were punctured into the agar plates of 7 mm diameter each and filled with 100 mL of synthesized TiO2 nanoparticles loaded with and without streptomycin drug dissolved in 10% DMSO and allowed to diffuse at room temperature for 2 h. Then, the plates were incubated at 37 °C for 48 h. The standard antibiotic disc of streptomycin was used as positive control and the experiments were performed in triplicate each week during a 2 months period. The results were expressed in mean standard deviation. After incubation, the growth incubation zone diameters were measured in mm. Inhibitory concentration of synthesized nanoparticles were maintained constant (10 μL) for all test pathogens and bactericidal activity of synthesized TiO2 nanoparticles loaded streptomycin at 6:4% concentration is shown in (Table 1).
The synthesized TiO2 nanoparticles exhibit high antibacterial activity compared to standard drugs and demonstrates its capability of deactivating cellular enzymes. It is evidenced from Table 1 that synthesized nanoparticles exhibit zone of inhibition for all the test pathogens. However, P. mirabilis does not show antibacterial activity and E. Coli shows no activity for nanoparticles templated at 1.2 and 1.6% probably because the negative carrier of P. mirabilis and E.coli at specific concentration show less affinity towards titanium metal ions. In addition, inhibition of streptomycin loaded nanoparticles at all template concentration exhibit relatively larger zone in comparison with the test control. The statistical interpretation from Table 1 shows that P. mirabilis and E. coli show a remarkable antibacterial profile when, tested with TiO2 nanoparticles loaded with streptomycin. Surprisingly zone width for control is less compared to zone width observed for TiO2 loaded streptomycin and hence the affinity of titanium metal ions in association with the drug against P. mirabilis and E. coli implicates the significant efficacy of drug release.
From Fig. 5f, the dissolution profiles of TiO2 loaded with streptomycin nanoparticles showed a superior increase in the release/dissolution rate that reflected inhibition zone width larger in correlation with synthesized TiO2 nanoparticles and the positive control of streptomycin against human pathogens that are responsible for causing UTI. Diffusion controlled release of TiO2 nanoparticles loaded with streptomycin were studied by dialysis method wherein 10 mg of streptomycin dissolved in 2 µL PBS solution at pH 7.4 (release media) was poured into the inner tube of the dialyzer. The dialyzer tube was suspended into a 200 mL glass beaker containing release media, which was magnetically stirred continually at 500 rpm at room temperature. The percentage of drug released from TiO2 nanocorals at different time intervals is shown in Fig. 5f. It is also evident that increase in the pore size of the TiO2 microspheres on the surface might be due to the release of entrapped protein and deformation in structure, leading to the sustained release of drug. The initial release of streptomycin happens after 60 min. After the first stage, the protonation takes place on the surface of TiO2 nanoparticles, which promotes the release of chemisorbed drug in the medium, so that the release percentage increases to 45 and then to 65% [18].
Conclusion
The study concludes that Aloevera is an effective template for the synthesis of anatase phase TiO2 nanoparticles with an average grain size ranging from 8 to 13 nm. Powder XRD analysis was employed to elucidate the structure and phase of TiO2 nanoparticles. The presence of Ti–O and the biological molecules responsible for the formation of TiO2 nanoparticles were analyzed by FTIR spectroscopic technique. SEM results rationalized the dependence of template concentration on particle size and nanoscale morphology of TiO2 nanoparticles. Interpretation from the antimicrobial activity of pathogens causing UTI loaded with streptomycin drug shows that TiO2 assisted by Aloevera could be a potential antibacterial agent for practical applications. Future perspective of the work could focus on the construction of polymeric nanoarchitectures to improve loading capacity of drugs. Although invitro experiment disclose that TiO2 networks exhibit good response towards improving the efficacy of the drug delivery system, further investigations have to be systematically carried out to expedite the process and broaden the possibility of practical applications in drug delivery and transport mechanism.
References
Kaviyarasu, K., Premanand, D.: Synthesis of Mg doped TiO2 nanocrystals prepared by wet-chemical method: optical and microscopic studies. Int. J. Nanosci. 12(5), 13500331–13500336 (2013)
Gratzel, M.: Photochemical cells. Nature 414, 338–344 (2001)
Gopal, M., Moberly Chan, W.J., De Jonghe, L.C.: Room temperature synthesis of crystalline metal oxides. J. Mater. Sci. 32, 6001–6008 (1997)
Fujishima, A., Honda, K.: Electrochemical photolysis of water at a semiconductor electrode. Nature 238, 37–38 (1972)
Yang, S., Gao, L.: Preparation of titanium dioxide nanocrystallite with high photocatalytic activities. J. Am. Ceram. Soc. 88, 968–970 (2005)
Addamo, M., Augugliaro, V., Di Paola, A., Garcı´a-Lo´pez, E., Loddo, V., Marcı`, G., Molinari, R., Palmisano, L., Schiavello, M.: Preparation, characterization, and photoactivity of polycrystalline nanostructured TiO2 catalysts. J. Phys. Chem. 108, 3303–3310 (2004)
Chun, H., Lan, Y., Jiuhui, Q., Xuexiang, H., Wang, A.: Ag/AgBr/TiO2 visible light photocatalyst for destruction of azodyes and bacteria. J. Phys. Chem. B 110, 4066–4072 (2006)
Sakatani, Y., Grosso, D., Nicole, L., Boissie`re, C., de AA Soler-Illiab, G.J., Sanchez, C.: Optimised photocatalytic activity of grid-like mesoporous TiO2 films: effect of crystallinity, pore size distribution and pore accessibility. J. Mater. Chem. 16, 77–82 (2006)
Rajesh Kumar, S., Pillai, S.C., Hareesh, U.S., Mukundan, P., Warrier, K.G.K.: Synthesis of thermally stable high surface area anatase-alumin a mixed oxides. Mater. Lett. 43, 286–290 (2000)
Malia, S.S., Betty, C.A., Bhosale, P.N., Patila, P.S.: Sensitized solar cells (DSSCs) based on novel nanocoral TiO2: a comparative study. Electrochim. Acta. 59, 113–120 (2012)
Eshun, K., He, Q.: Aloe vera: a valuable ingredient for the food, pharmaceutical and cosmetic industries—a review. Crit. Rev. Food Sci. Nutr. 44, 91–96 (2004)
Boudreau, M.D., Beland, F.A.: An evaluation of the biological and toxicological properties of Aloe barbadensis (miller). Aloe vera J. Environ. Sci. Health. 24, 103–154 (2006)
Kumar, S., Upadhyaya, J.S., Negi, Y.S.: Preparation of nanoparticles from corn cobs by chemical treatment methods. Bio. Res. 5, 1292–1300 (2010)
Hamman, J.H.: Composition and applications of Aloe vera leaf gel. Molecules 13, 1599–1616 (2008)
Venkatesh, K.S., Krishnamoorthi, S.R., Palani, N.S., Thirumal, V., Jose, S.P., Wang, F.M., Ilangovan, R.: Facile one step synthesis of novel TiO2 nanocoral by sol–gel method using Aloe vera plant extract. Indian J. Phys. 89, 445 (2014)
Chekin, F., Bagheri, S., Abd Hamid, S.B.: Synthesis of Pt doped TiO2 nanoparticles: characterization and application for electrocatalytic oxidation of l-methionine. Sen. Actuators B Chem. 177, 898–903 (2013)
Shankar, S.S., Ahmad, A., Sastry, M.: Geranium leaf assisted biosynthesis of silver nanoparticles. Biotechnology 19, 1627–1631 (2003)
Niwa, T., Takeuchi, H., Hino, T., Kunou, N., Kawashima, Y.: In vitro drug release behavior of d, l-lactide/glycolide copolymer (PLGA) nanospheres with nafarelin acetate prepared by a novel spontaneous emulsification solvent diffusion method. J. Pharm. Sci. 83(5), 727–732 (1994)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kalaiarasi, S., Jose, M. Streptomycin loaded TiO2 nanoparticles: preparation, characterization and antibacterial applications. J Nanostruct Chem 7, 47–53 (2017). https://doi.org/10.1007/s40097-016-0213-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40097-016-0213-2