Abstract
Purpose
The aim of this study was to prove that the Fe-containing by-products (compost, sewage and lime sludge) have potential influence on the correction of Fe deficiency in sunflower and maize.
Methods
The physiological parameters used were the dry matter (DM), the relative chlorophyll content, and the content of photosynthesis pigments (chlorophyll a, chlorophyll b and carotenoids).
Results
The results showed that sewage sludge was effective even when applied at low concentration (2 g L−1) for both sunflower and maize plants. In maize, however, chlorophyll a was the only pigment induced after sewage sludge treatments in contrast to sunflower, which could be the reason for lower DM in maize. Compost could only be effective when applied at higher concentrations (4 g L−1) for sunflower because of its low-Fe content, further showing that maize plants require more Fe. Despite its high-Fe content, lime sludge could not be effective in alleviating Fe deficiency for both sunflower and maize plants because of its high pH, which makes Fe unavailable for uptake by the plants.
Conclusions
Although the investigated by-products can be hazardous depending on their origin, their use as micro-element fertilizer can be a very economical way of utilization in the agricultural fields, which may limit the use of high quantities of expensive Fe-containing fertilizers. Among the three waste products, sewage sludge application proved to have a great potential of correcting Fe deficiency by inducing accumulation of the photosynthesis pigments, leading to high photosynthesis and eventually high DM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Iron (Fe) plays a crucial role in various physiological and biochemical processes such as plant metabolism (acts as an electron carrier), synthesis of chlorophyll and maintaining chloroplast structure as well as function, which makes it one of the essential trace elements in plants. An imbalance between its solubility in the soil and the demand by the plant leads to Fe deficiency (Guerinot and Yi 1994; Rout and Sahoo 2015). This is a widespread phenomenon in plants because Fe availability is limited by its chemical properties in aerated environments, forming Fe3+, which is a constituent of oxyhydroxide polymers with extremely low solubility (López-Millán et al. 2013). Disorders associated with Fe deficiency in crops include decreases in vegetative growth, yield and quality. Chlorosis, which usually appears in the youngest leaves and shoots, is another symptom associated with Fe deficiency in calcareous soils (Von Wiren et al. 1994; Abadía et al. 2011). Iron deficiency may also cause economics problems due to reduction in plant growth and yield (Santos et al. 2015). On the other hand, Fe deficiency symptoms can appear in people if they do not consume appropriate quality agricultural products (Prentice et al. 2017).
Nitrification/denitrification phases of wastewater treatment process result in high nitrogen (N) and phosphorus (P) in sewage sludge, suggesting the potential use of sewage sludge in the agricultural sector as a fertilizer, although it may also contain other elements and heavy metals (Tchobanoglous and Burton 1991), which can inhibit photosynthesis and Fe uptake (Fodor 2002). An accelerated effort to combat this problem has been adopted by the developed countries. This involves requirements for more stringent industrial pre-treatment of wastewater and removal of contaminants such as heavy metals before disposal, which leads to improved quality of sewage sludge, making it a viable option for recycling carbon and nutrients back into the agricultural production chain (Jamali et al. 2008; Yoshida et al. 2015).
Considering the above, this study was conducted to investigate the effect of the three pre-treated industrial waste products (compost, sewage and lime sludge) on the correction of Fe deficiency in sunflower (Helianthus annuus L. cv Arena, a C3 plant) and maize (Zea mays L. cv Norma SC, a C4 plant). The plants were selected for this research because their Fe requirements would differ. According to Sharma and Sanwal (1992), C4 plants have higher Fe supply requirements than C3 plants. The results will, therefore, give an insight on the response of these plants when exposed to the same quantity of Fe-containing material. Since Fe deficiency has higher impact on photosynthesis, the physiological parameters used for this investigation were dry matter (DM), relative chlorophyll (RC) content and the content of individual photosynthesis pigments. Although some studies have indicated that some industrial by-products can be recycled back into the industrial production chain, they did not focus on Fe deficiency alleviation. This report will also shed light on the suitable application dose of the used materials for the different plant types.
Materials and methods
Plant growth
Sunflower (Helianthus annuus L. cv Arena) and maize (Zea mays L. cv Norma SC) seeds were surface sterilized with 18% (v/v) hydrogen peroxide (H2O2), washed in distilled water and germinated on moistened filter paper at 25 °C for 5 days. For maize, the seeds were first placed in 10 mM CuSO4 4 h prior to germination. Seedlings were then transferred to aerated modified Hoagland nutrient solution (1 L) containing 2.0 mM Ca(NO3)2, 0.7 mM K2SO4, 0.5 mM MgSO4, 0.1 mM KH2PO4, 0.1 mM KCl, 1 µM H3BO3, 1 µM MnSO4, 0.25 µM CuSO4 and 0.01 µM (NH4)6Mo7O24 (Cakmak and Marschner 1990). The negative control seedlings were grown in aerated nutrient solution without Fe(III)-ethylenediaminetetraacetic acid (EDTA) for the duration of the experiment. For the positive controls, nutrient solution was supplemented with 100 µM of Fe(III)-EDTA on the 6th (for maize) or the 12th (for sunflower) day of growth. To investigate the effect of the three industrial by-products (compost, lime and sewage sludge), each by-product was added separately to the nutrient medium in two doses (2 and 4 g L−1) on the 6th or the 12th days. The seedlings were further grown for 6 days post-treatment under controlled environmental conditions (light/dark regime 10/14 h at 24/20 °C, relative humidity of 65–70% and a photon flux density of 300 μmol m−2 s−1) and used for the different physiological measurements. Each pot consisted of four plants. The data were collected from three independent replications with four samplings per pot.
Sewage sludge and compost originated from Alkaloida Chemicals Co. Ltd. Compost was made from this sewage sludge and waste wood of various sizes. Lime sludge was provided by Ore, Mineral and Waste Recycling Works of Borsod Private Company Limited. The analytical grade chemicals used were supplied by Merck and BDH chemical companies.
Iron (Fe) content of the investigated materials
To measure the Fe content of the examined by-products, 15 mL of 65% (v/v) HNO3 was added to 1 g of each sample and incubated overnight at room temperature. Afterwards, the samples were pre-digested for 30 min at 60 °C. To this solution, 5 mL of 30% (v/v) H2O2 was added and further incubated for 270 min at 120 °C. The solutions were made up to 50 mL with double-distilled water, homogenized and filtered through Filtrak 388 filter paper. The iron content of filtrate samples was measured at 238 nm with an OPTIMA 3300DV inductively coupled plasma-optical emission spectrometry (ICP-OES) (Kovács et al. 1996).
Dry matter accumulation
The measurement of shoots and roots DM in either sunflower or maize was achieved through the use of thermal gravimetric analysis, after drying at 80 °C for 48 h (Brower and Zar 1984).
The relative chlorophyll content (Soil–Plant Analyses Development, SPAD, units)
A non-destructive handheld chlorophyll meter, SPAD-502 (Minolta, Japan), was used for the measurement of the relative chlorophyll content of the second sunflower and maize leaves. Five measurements were done per plant.
Chlorophyll a, chlorophyll b, and carotenoid content
Fresh weight (50 mg) of the second sunflower and maize leaves was homogenized in N,N-dimethyl formamide (5 mL) and incubated at 4 °C for 72 h. Quantification of chlorophyll a, chlorophyll b and carotenoids was performed at 664 nm, 647 nm and 480 nm, respectively, using a Meterek SP 80 Spectrometer. The data were analyzed mathematically according to Moran and Porath (1980) as well as Wellburn (1994).
Statistical analysis
For the statistical analysis of the results, a student t test was performed with Sigma Plot 12.0 program to determine the significance of treatments against the Fe-deficient controls. Three independent replications were used. Treatments were considered to be significant at p < 0.01 and highly significant at p < 0.001. Mann–Whitney Rank Sun test was used to evaluate the SPAD units and photosynthetic pigment content.
Results and discussion
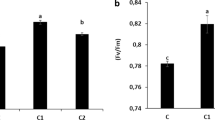
High-iron (Fe) content in the waste materials (compost, sewage and lime sludge; Table 1) indicates their potential use in the correction of Fe deficiency for plants because Fe is usually not readily available for various physiological and biochemical processes especially in aerated environments (López-Millán et al. 2013). Sobik-Szoltysek et al. (2017) reported that Fe bioavailability increased when sewage and coal sludge was applied as fertilizer. Sewage and lime sludge had higher Fe concentration than compost (2.1-fold and 12-fold high, respectively). In addition, the used waste materials had very low heavy metal content (unpublished results). This may lead to less Fe uptake inhibition and eventually high photosynthesis (Fodor 2002), since Fe also plays a very important role in the electron transport chain. The Fe content in these materials were in agreement with a study by Anderson (1982), which showed that raw or composted animal, human and industrial wastes generally contain large amounts of Fe. Furthermore, Fodor et al. (2017) reported that Fe content of the shoot of energy grass increased substantially when dried municipal sewage sludge was added to the quarter strength Hoagland nutrient solution.
Dry matter (DM) content for sunflower and maize shoots/roots were measured (Table 2). Significantly high DM levels were recorded for sunflower and maize when treated with the Fe-containing industrial by-products. For both plants, shoots generally had higher DM content than the roots. Dry matter content of the positive controls was substantially higher (p < 0.01) than that of the negative controls for both sunflower and maize shoots. Compost treatment was highly significant for the induction of DM accumulation for both shoots and roots of sunflower only when the dosage was increased (4 g L−1). In contrast, higher compost dose (4 g L−1) did not yield significant DM induction for maize seedlings. Sewage sludge application at 2 g L−1 induced significant increases in DM accumulation for sunflower shoots (43%) and roots (49%). Similarly, maize had significantly high increases in the DM of shoots (62%) and roots (45%) after sewage sludge treatment (2 g L−1). Although significantly high compared to the Fe-deficient controls, no tangible increases in the shoot and root DM were observed when sewage sludge concentration was increased in sunflower and maize contrary to compost. Treatment of sunflower and maize seedlings with lime sludge had no substantial effect on the DM accumulation despite increasing its concentration. Induction of DM accumulation after treatment of sunflower and maize seedlings with the Fe-containing materials strongly indicated their potential in the correction of Fe deficiency. This argument was substantiated by high DM in the positive controls for both plants. Higher DM accumulation in the shoots than roots of both plants could be the result of high photosynthesis in the leaves. Since compost had the least Fe content compared to the other waste materials used, it only induced significant increases in DM accumulation for sunflower when the concentration was increased to 4 g L−1. Contrary, the high dose was not sufficient to significantly induce DM in maize. This could probably be due to the fact that the C4 plants generally have higher demands for Fe than C3 plants (Sharma and Sanwal 1992), indicating that an umbrella approach cannot be used when it comes to application of these waste materials for alleviation of Fe deficiency. Sunflower and maize seedlings responded positively to sewage sludge application by inducing highly significant DM accumulation, showing the potential role of this treatment in the correction of Fe deficiency. Although there was still a significant induction in DM compared to the Fe-deficient controls when higher concentration was used, there was no big difference between the two concentrations for both plants. The reason for the non-substantial increase in DM at higher dosage could be linked to Fe toxicity because Fe can act catalytically via the Fenton reaction to generate hydroxyl radicals, which can damage lipids, proteins and DNA (Connolly and Guerinot 2002). Lime sludge had the highest Fe content (Table 1); however, treatment of sunflower and maize seedlings with different concentrations of this material had no substantial effect on the DM accumulation (Table 2). This could be as a result of the high pH of this material (10.77), which was approximately twofold higher than that of the sewage sludge and compost (results not shown). Such conditions may trigger an imbalance between uptake and usage (Rout and Sahoo 2015). Iron deficiency is highly likely in calcareous soils (Von Wiren et al. 1994; Abadía et al. 2011) and plays an important role in the biosynthesis pathway (leading to more DM accumulation) because most of the leaf Fe is localized mainly in complexes involved in the photosynthetic electron transport chain (Terry and Abadía 1986).
Relative chlorophyll (RC) content was significantly high (p < 0.001) for the positive than negative sunflower controls (Table 3). Compost induced the RC content substantially compared to the negative control, with a noted increase (30%) observed when 4 g L−1 was used. Sewage sludge application at both concentrations induced highly significant (p < 0.001) increases in the RC content. The highest values were obtained in this treatment compared to the compost and lime sludge. Increasing the sewage sludge concentration increased the RC content of sunflower seedlings only slightly (4% increase). Although lime sludge induced some significant increases in the RC content of sunflower, it was the least and decreased with an increase in concentration. The increased RC content of the positive maize control was highly significant. Contrary to sunflower, RC content did not increase with increasing compost concentrations in maize. Sewage sludge treatment on the other hand induced highly significant increases in the RC content irrespective of the concentration. In agreement with sunflower, lime sludge application led to no significant increases in the RC content for maize seedlings.
In sunflower, positive controls had highly substantial increases in chlorophyll a, chlorophyll b and carotenoid content. In the case of compost treatments, the concentrations of all three chloroplast pigments increased with increasing application doses, except for chlorophyll b, which was not significantly induced at 2 g L−1. Sewage sludge (at a lower dosage) induced highly significant increase in the chlorophyll content but not of carotenoids. Although all three pigments were substantially high at increased sewage sludge concentrations compared to the Fe-deficient controls, differences in the chloroplast pigments for the two concentrations were not significant. Lime sludge, on the other hand induced non-significant increases in all measured parameters in sunflower (Table 4).
Positive maize controls had significantly high increases in chlorophyll a content. Other pigments were not substantially induced by Fe-EDTA in contrast to sunflower. Compost treatment did not induce substantial accumulation of any of the individual chloroplast pigments despite increasing the concentration. Maize seedlings treated with lower concentration (2 g L−1) significantly induced chlorophyll a content by 43%. Similarly, chlorophyll a content of sunflower increased with an increase in sewage sludge concentration, though the differences between the concentrations were not substantial. Carotenoid content was only substantially induced when the sewage sludge concentration increased, in agreement with the sunflower treatments. Chlorophyll b was not significantly induced by all treatments in maize. Lime sludge did not have any substantial effect on all tested chloroplast pigments (Table 5).
Chloroplast pigments trap light energy and store it as chemical energy, which is achieved by catalyzing oxidation–reduction process where hydrogens are boosted from water to organic matter (Rabinowitch and Govindjee 1965). Highly significant increases in the RC content of the positive sunflower (3.9-fold) and maize (1.4-fold) controls (Table 3) clearly indicated the importance of Fe in chlorophyll synthesis and photosynthesis. In agreement with the DM results for sunflower, RC content increased with an increase in compost concentrations. Among the three used waste products, sewage sludge induced the most significant increases (closer to the positive controls) in the RC content for both sunflower and maize seedlings, further supporting the use of this material (especially at 2 g L−1) in alleviation of Fe deficiency. Re-greening of the Fe-deficient plants was more evident in the sewage sludge (2 g L−1) treated sunflower and maize (results not shown), further suggesting the potential use of this treatment for the correction of chlorosis associated with Fe deficiency. In disagreement, Singh and Agrawal (2007) reported that sewage sludge-amended soils led to decreased photosynthesis rates and chlorophyll content in beetroot. However, addition of Fe to deficient cucumber plants led to recovery of the first leaf from chlorosis, albeit not completely. The newly expanded second leaf of Fe-supplied plants had chlorophyll concentrations typical of Fe-sufficient plants on the fifth day after Fe addition (Johnson and Barton 2007). For both plant types, lime sludge results corroborated with the DM results where induction of the RC content was not significant.
Compost treatments induced significant increases in all the measured photosynthesis pigments, especially when a high dose (4 g L−1) was used in sunflower (Table 4). At this concentration, the levels of individual pigments were close to those of the sewage sludge treatments, which were found to be the best treatment for Fe deficiency alleviation according to the DM and RC content results. This further supports the above suggestion that for sunflower, compost can only be effective when applied at high concentrations. Contrary to sunflower, compost was not effective in inducing accumulation of these pigments in maize, further agreeing that this can be attributed to the low-Fe content in compost since C4 plants have high-Fe requirements. Lime sludge was not effective in correcting Fe deficiency for both sunflower and maize, further showing that the high pH of this sludge may affect Fe uptake, thereby influencing the synthesis of photosynthesis pigments (Rout and Sahoo 2015).
Conclusions
Among the three waste products, sewage sludge application was found to have a great potential of correcting Fe deficiency by inducing accumulation of the photosynthesis pigments, leading to high photosynthesis and eventually high DM accumulation. The results explicitly showed that for this material to work, it does not have to be applied at high concentrations to alleviate chlorosis, especially in sunflower. For maize, chlorophyll a was the only pigment induced after sewage sludge treatments. Compost could only be effective when applied at higher concentrations (4 g L−1), and which according to the results, is more applicable for sunflower. Even though lime sludge had the highest Fe content, it only induced the least DM and photosynthesis pigment contents for both plants because of its high pH. Since all of the by-products used in this study are usually generated in high volumes, they can be suitable for solving iron-deficiency problems in the agricultural fields and their use in agriculture offer an alternative to the expensive chemical fertilizers, also contributing to the green economy.
References
Abadía J, Vázquiz S, Rellán-Álvarez R, El-Jendoubi H, Abadía A, Álvaerz-Fernández A, López-Millán AF (2011) Towards knowledge-based correction of iron chlorosis. Plant Physiol Biochem 49:471–482. https://doi.org/10.1016/j.plaphy.2011.01.026
Anderson WB (1982) Diagnosis and correction of iron deficiency in field crops: an overview. J Plant Nutr 5:785–795. https://doi.org/10.1080/01904168209363008
Brower JE, Zar JH (1984) Field and laboratory methods for general ecology. Dubuque, Iowa
Cakmak I, Marschner H (1990) Decrease in nitrate uptake and increase in proton release in zinc deficient cotton, sunflower and buckwheat plants. Plant Soil 129:261–268. https://doi.org/10.1007/BF00032421
Connolly EL, Guerinot ML (2002) Iron stress in plants. Genome Biol 3:reviews1024.1–1024.4. https://doi.org/10.1186/gb-2002-3-8-reviews1024
Fodor F (2002) Physiological responses of vascular plants to heavy metals. In: Prasad MNV, Strzalka K (eds) Physiology and biochemistry of metal toxicity and tolerance in plants. Kluwer Academic Publishers, Dordrecht, pp 149–177
Fodor F, Tóth B, Solti Á, Gy S, Fodor F (2017) Responses of Szarvasi-1 energy grass to sewage sludge treatments in hydroponics. Plant Physiol Biochem 118:627–633. https://doi.org/10.1016/j.plaphy.2017.07.027
Guerinot ML, Yi Y (1994) Iron: nutritious, noxious, and not readily available. Plant Physiol 104:815–820. https://doi.org/10.1104/pp.104.3.815
Jamali MK, Kazi TG, Arain MB, Afridi HI, Memon AR, Jalbani N, Shah A (2008) Use of sewage sludge after liming as fertilizer for maize growth. Pedosphere 18:203–213. https://doi.org/10.1016/S1002-0160(08)60008-9
Johnson GV, Barton LL (2007) Inhibition of iron deficiency stress response in cucumber by rare earth elements. Plant Physiol Biochem 45:302–308. https://doi.org/10.1016/j.plaphy.2007.03.012
Kovács B, Győri Z, Prokisch J, Loch J, Dániel P (1996) A study of plant sample preparation and inductively coupled plasma emission spectrometry parameters. Commun Soil Sci Plant Anal 27:1177–1198. https://doi.org/10.1080/00103629609369625
López-Millán AF, Grusak MA, Abadía A, Abadía J (2013) Iron deficiency in plants: an insight from proteomic approaches. Front Plant Sci 4:1–7. https://doi.org/10.3389/fpls.2013.00254
Moran R, Porath D (1980) Chlorophyll determination in intact tissues using N,N-dimethyl-formamide. Plant Physiol 65:478–479. https://doi.org/10.1104/pp.65.3.478
Prentice AM, Mendoza YA, Pereira D, Cerami C, Wegmuller R, Constable A, Spieldenner J (2017) Dietary strategies for improving iron status: balancing safety and efficiency. Nutr Rev 75(1):49–60. https://doi.org/10.1093/nutrit/nuw055
Rabinowitch EI, Govindjee (1965) The role of chlorophyll in photosynthesis. Sci Am 213:74–83
Rout GR, Sahoo S (2015) Role of iron in plant growth and metabolism. Rev Agric Sci 3:1–24. https://doi.org/10.7831/ras.3.1
Santos CS, Roriz M, Carvalho SMP, Vasoncelos MW (2015) Iron partitioning at an early growth stage impacts iron deficiency responses in soybean plants (Gylcine max L.). Front Plant Sci 6:325. https://doi.org/10.3389/fpls.2015.00325
Sharma S, Sanwal GG (1992) Effects of Fe deficiency on the photosynthetic system of maize. J Plant Physiol 140:527–530. https://doi.org/10.1016/S0176-1617(11)80782-0
Singh RP, Agrawal M (2007) Effects of sewage sludge amendment on heavy metal accumulation and consequent responses of Beat vulgaris plants. Chemosphere 67(1):2229–2240. https://doi.org/10.1016/j.chemosphere.2006.12.019
Sobik-Szoltysek J, Wystalska K, Grobelak A (2017) Effect of addition of sewage sludge and coal sludge on bioavailability of selected metals in the waste from the zinc and lead industry. Environ Res 156:588–596. https://doi.org/10.1016/j.envres.2017.04.011
Tchobanoglous G, Burton FL (1991) Wastewater engineering: treatment, disposal, and reuse. McGraw-Hill, New York
Terry N, Abadía J (1986) Function of iron in chloroplasts. J Plant Nutr 9:609–646. https://doi.org/10.1080/01904168609363470
Von Wiren N, Mori S, Marschner H, Römheld V (1994) Iron inefficiency in the maize mutant ysl (Zea mays L. cv Yellow-stripe) is caused by defect in uptake of iron phytosiderophores. Plant Physiol 106:71–77. https://doi.org/10.1104/pp.106.1.71
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144(3):307–313. https://doi.org/10.1016/S0176-1617(11)81192-2
Yoshida H, Nielsen MP, Scheutz C, Jensen LS, Christensen TH, Nielsen S, Bruun S (2015) Effects of sewage sludge stabilization on fertilizer value and greenhouse gas emissions after soil application. Acta Agric Scand B Soil Plant Sci 65:506–516. https://doi.org/10.1080/09064710.2015.1027730
Acknowledgements
We would like to thank the following companies for supplying the material used for this research (compost, lime and sewage sludge): Alkaloida Chemicals Co. Ltd. and Ore, Mineral and Waste Recycling Works of Borsod Private Co. Ltd. We would also like to acknowledge Dr. Béla Kovács for helping with the ICP analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Tóth, B., Moloi, M.J. The use of industrial waste materials for alleviation of iron deficiency in sunflower and maize. Int J Recycl Org Waste Agricult 8 (Suppl 1), 145–151 (2019). https://doi.org/10.1007/s40093-019-0284-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40093-019-0284-4