Abstract
Purpose
Membrane filtration is recently applied to recover nutrients and dischargeable water from anaerobic sludge. The purpose of this study is to quantify nutrient separation, membrane performance, and process stability and to increase the economical applicability of the process by energetic optimisation.
Methods
At the site of a 2.5 MWe agricultural biogas plant, a membrane pilot plant was operated over a period of 7 months. It consisted of a screw press separator, a decanter centrifuge, an ultrafiltration unit, and a three-stage reverse osmosis unit. Mass and nutrient balances were generated by sampling and analysing every process stream. Process performance was analysed by monitoring separation efficiencies, membrane flux, cleaning intervals, and energy demand.
Results
Solid/liquid separation resulted in separation efficiencies of 70% for total solids and 80% for phosphorus. The solid fraction contained high concentrations of organics and particle-ligated nutrients (20% TS, 8 kg t−1 Ntotal, 5.5 kg t−1 P2O5). The retentate of the reverse osmosis had high concentrations of dissolved ammonia and potassium (4 kg t−1 NH4–N and 10 kg t−1 K2O). 38% of the sludge volume was recovered as clean water.
Conclusion
The membrane pilot plant successfully produced a solid N/P-fertiliser, a liquid N/K-fertiliser and clean water. The results contribute to a sound understanding and growing database for future adaption of the process chain. Hydrodynamic optimisation within the pilot plant reduced the energy demand of the ultrafiltration step by 50%, which considerably contributes to the economy of the process.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
As a result of the German Renewable Energies Act, enacted in 2000, the number and size of biogas plants in Germany has constantly increased. In 2017, more than 9300 biogas plants were operating in Germany with an electrical capacity of P ≈ 4500 MWe (German Biogas Association 2017). Consequently, a considerable amount of anaerobic sludge (digestate) is produced. In 2011, the output of digestate amounted to 65.5 million m3 containing approximately 390,000 t of nitrogen, 74,000 t of phosphorus, and 330,000 t of potassium (Möller and Müller 2012). In terms of concentration, raw digestate contains about 1.2–9.1 kgN t−1 of total nitrogen with about 44–81% ammonia, 0.4–2.6 kgP t−1 phosphorus and 1.2–11.5 kgK t−1 potassium (Möller and Müller 2012).
Due to its high nitrogen and phosphorus content, anaerobic digestion products are mostly used for local field applications. Anaerobic sludge is characterised by high biological stability and appropriate fertilising quality. Soil and air characteristics are not negatively affected using anaerobic sludge products when applying correct manure procedures with subsurface injection to limit ammonia emissions (Riva et al. 2016). Furthermore, fertilisers based on anaerobic digestion products achieve comparable or even higher biomass production rates compared to synthetic mineral fertiliser for, e.g., the production of wheat (Abubaker et al. 2012) or silage corn (Riva et al. 2016). Local over-fertilisation results in an excess of nutrients in the soil and nutrient leaching into the groundwater (Lee and Oa 2013; Schoumans and Chardon 2014). To guarantee a good fertilising procedure without local over-fertilisation and maintain high groundwater qualities, recent revisions of the fertiliser ordinance in Germany limit field applications of organic residues to 170 kgN ha−1 a−1 (DüV 2017). Regarding phosphorus, no surplus application on fields with high or very high phosphate content will be allowed (saldo = 0 kgP ha−1 a−1). Good fertilising practice thus often requires transportation of nutrients between different areas. Given that anaerobic sludge contains more than 90% water, transportation is neither economically nor ecologically sensible. Therefore, a number of different digestate treatment processes emerge which often originate from manure or wastewater treatment (e.g. Fuchs and Drosg 2013; Drosg et al. 2015; Vaneeckhaute et al. 2017; Bolzonella et al. 2017). The combination of mechanical solid/liquid separation and membrane treatment offers the advantage of producing dischargeable or reusable process water on the one hand and concentrated, transport-worthy nutrient products on the other hand.
Solid/liquid separation of organic slurries is achieved by screw press separators and/or decanter centrifuges. Decanter centrifuges achieve high efficiencies of solid and phosphorus separation of about 60–90% for slurries and biogas digestate (Balsari et al. 2006; Lukehurst et al. 2010). According to Lukehurst et al. (2010), separation efficiency for both total solids and nutrients can be improved with the use of stable polymeric flocculants during the centrifugal process. Further treatment of the liquid phase after solid/liquid separation is mostly carried out as a process combination of a micro- or ultrafiltration step (MF, UF) followed by a reverse osmosis step (RO). In Germany, required water specifications for water discharge from biogas plants depend on the quality of receiving water bodies and are defined by the appropriate environmental agency. The requirements for effluents from wastewater treatment plants (size 5) are limited to, i.a., chemical oxygen demand < 75 mg L−1, total nitrogen concentration < 13 mg L−1, and phosphorus concentration < 1 mg L−1 (AbwV 2005).
In lab scale experiments, different authors investigated the ultrafiltration performance for anaerobic digester effluents with varying digester feedstock (Waeger et al. 2010; Choo et al. 1992; Zacharof and Lovitt 2014; Meixner et al. 2015; Bhattacharya et al. 2013). The ultrafiltration step has the highest operational energy demand and consequently the highest operational costs (Drosg et al. 2015, Gienau et al. 2018). Membrane material, cross-flow velocity and transmembrane pressure were detected to have the strongest influence on the flux performance of the membrane process, while the flux rates were generally low and the filtration performance limited by organic fouling. This is in accordance with experience from membrane filtration of activated sludges, where organic macromolecules such as proteins and polysaccharides were identified as major membrane foulants (e.g., Rosenberger et al. 2006; Jiang et al. 2008). Waeger et al. (2010) recommended ceramic ultrafiltration membranes with pore sizes of 20–50 nm for biogas digestate filtration applications. Due to pronounced organic fouling, repeated permeate backflushing operations and intensive chemical cleaning strategies were required to ensure stable, high membrane performances.
Only few authors published results of pilot or full scale trials (Chiumenti et al. 2013, Ledda et al. 2013). While Chiumenti et al. (2013) report nutrient balances of a digestate treatment chain at the site of an agricultural biogas plant fed with swine and poultry manure and corn silage, Ledda et al. (2013) report results with digested cattle manure and with digested swine manure. Apart from different feedstock characteristics, the experimental set-ups consisted of different membranes used for the ultrafiltration step. Chiumenti et al. (2013) used ceramic modules with 0.1 µm pore size and Ledda et al. (2013) used 40 kDa grafted polyacrylonitrile membranes. Although the set-up was quite similar, the results of the mass and nutrient balances varied considerably. Ledda et al. (2013) separated the digestates into 38–51% water, 12–32% solid fertiliser, and 30–37% liquid concentrate, while Chiumenti et al. (2013) reported 48% water, 35% solid fertiliser, and 17% liquid concentrate. In the study of Ledda et al. (2013), the different mass distributions occur due to different biogas plant feedstock. Screenings on different biogas digestates reveal the large spectrum of digestate properties (Gienau et al. 2018; Vaneeckhaute et al. 2017). Similar problems have also been reported by Masse et al. (2007) in a review on membrane-based nutrient recovery from manure. The authors conclude that the technology is technically feasible, but that the experimental database is not big enough for a sound technology analysis. Technology reviews on the membrane-based digestate treatment also rely on small available databases. (Drosg et al. 2015, Camilleri-Rumbau et al. 2013; Vaneeckhaute et al. 2017; Bolzonella et al. 2017). When comparing different nutrient recovery technologies, the authors not only emphasize the high technical potential of the membrane treatment chain but also state the relatively high overall costs compared to other technologies such as stripping and drying. Published specific treatment costs vary between 16 and 25 kWh m3 (Fuchs and Drosg 2013; Chiumenti et al. 2013).
The objective of this paper is to contribute to a sound database on the membrane-based conditioning process for different biogas effluents and to define potential for future reduction of operational costs. Over a period of 7 months, about 1500 m3 of raw digestate were separated by a screw press separator, a decanter centrifuge, an ultrafiltration unit and a reverse osmosis unit. Besides the characterisation of all process streams, mass and nutrient balances were determined and efficiencies of all process units were calculated. Further focus was on process performance and optimisation of the most energy-intensive process step of the membrane chain, which is the ultrafiltration step.
Materials and methods
Biogas plant
Pilot trials were carried out at the site of an agricultural biogas plant in Bad Bentheim, Lower Saxony (Germany). The biogas plant has a total installed electric power of Ptotal = 2.5 MWe. Four combined heat and power units (CHP) are installed on site (P = 1.01 MWe) and five further satellite CHP units are located close to local heat-demanding areas in the city (P = 1.49 MWe). The biogas plant is fed with about 50 t day−1 of cattle manure and dung, with 50 t day−1 of corn silage and with 38 t day−1 of sugar beet and seasonal crops. The biogas plant is equipped with four fermenters (2 × 1500 m3 and 2 × 1900 m3), a post fermenter (2200 m3), and three storage tanks (2700 m3, 3,400 m3 and 9700 m3). The average overall retention time in the fermenters and post fermenter is 60–65 days; the fermenters operate at mesophilic conditions between 40 and 42 °C. The total digestate output of the plant varies from 35,000 to 55,000 t a−1.
Pilot plant for nutrient recovery
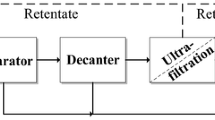
The pilot plant for nutrient recovery consists of a two-stage solid/liquid separation unit followed by ultrafiltration (UF) and reverse osmosis (RO) units (Fig. 1).
Process scheme of the total conditioning process with ultrafiltration membrane and reverse osmosis; DIG digestate, SPL liquid screw press separator, SPS solid screw press separator, DECL liquid decanter, DECS solid decanter, UFP permeate ultrafiltration, UFR retentate ultrafiltration, ROR retentate reverse osmosis, ROP permeate reverse osmosis
The digestate (DIG) from the biogas plant is separated with a screw press separator (agriKomp, Quetschprofi, 3 kW) with a mesh size of 500 µm to detach organic fibres and larger particles (Fig. 2, left). A decanter centrifuge dewaters the remaining liquid of the screw press (SPL) (Fig. 2, right, GEA, Westphalia Separator AD 0509, relative centrifugal force: 3,400 g). The liquid/solid separation is improved by cationic polymeric flocculants with a molecular weight of 3 × 106 to 4 × 106 g mol−1. The applied concentration of polymers was 4–6 gPolymer kg −1TR . These polymers are certified by the manufacturer for 20% biodegradability in 2 years. The solid fractions of the screw press and the decanter centrifuge (SPS and DECS) of the digestate were used as solid organic fertiliser. The supernatant from the centrifuge (DECL) was fed to the ultrafiltration unit (Fig. 3, left). The ultrafiltration module (αAl2O3/TiO2, pore size = 50 nm, A = 7.3 m2, Atech, Germany) was operated at transmembrane pressure differences of 4.5–5 bars, cross-flow velocities of 4–5 m s−1, a permeate yield of 60–70%, and temperatures between 40 and 70 °C. The UF permeate (UFP) was further treated by a three-stage reverse osmosis unit (Fig. 3, right, DOW, PA, Atotal = 137.2 m2) at a transmembrane pressure difference of 12–60 bars, a permeate yield of 70–80% and temperatures between 30 and 35 °C. The retention of ammonia was improved by setting the pH value of the UF permeate to 6.6–6.8 based on the ammonia equilibrium.
The retentate of the RO process (ROR) represents a liquid fertiliser. Clean process water (ROP) is collected and reused for the polymer solution and other applications on site. The screw press separator and the decanter have a capacity of 5–6 m3 h−1, which represents full-scale concerning a digestate output of 35,000–50,000 t a−1. The membrane process has a capacity of 0.3–0.5 m3 h−1 (scale down factor approx. 1:10), which is depending on temperature and digestate composition. The excess of about 4–5 m3 h−1 from the decanter centrifuge is discharged to the digestate storage tank and/or used as liquid fertiliser.
Analysis and instrumentation
Total solids (TS in weight percent—wt%) and volatile solids (VS in % of TS) were analysed according to the European standards EN 12880 and EN 12879, respectively. Densities were quantified with a pycnometer (25 cm3, BRAND) or a volumetric flask (500 cm3, BRAND), depending on the roughness of the material. Measurements were carried out as double determination and have a relative error of < 1% for both TS and density.
Volume flow was recorded with magnetic inductive flow meters for almost all liquid streams. Based on the literature reports on organic fouling on membranes (e.g., Rosenberger et al. 2006, Jiang et al. 2008), polysaccharide and protein concentrations were analysed in the feed and the retentate of the ultrafiltration membrane according to the methods of Dubois (Dubois et al. 1956) and Bradford (Bradford 1976), respectively. All measurements were carried out as double determination and have a relative error of ≤ 5%.
The nutrient and organic carbon concentrations of all liquid streams were measured with standard Hach vial tests LCK 302 (ammonia), LCK 338 (total nitrogen), LCK 228 (potassium), LCK 350 (phosphorus), and LCK 914 (COD). Nutrient concentrations of solid components were analysed by Agrolab institute using standard procedures (Kjeldahl-N: DIN 19684-4, Dumas-N: DIN EN 16168, NH4–N: DIN 38406-5-2 [E 5-2], P2O5: DIN EN ISO 11885 [E 22], K2O: DIN EN ISO 11885 [E 22]). The relative errors of measurement named by Agrolab institute for nitrogen, ammonia, phosphorus and potassium are < 16.2%, < 15.6%, < 17.2% and < 8.4%, respectively.
Determination of mass balances and separation efficiency
Mass balances
Based on the analysed concentrations of TS, VS and nutrient components, mass balances were set up for both the entire process and each separation unit. As nearly all process streams and intermediates were analysed and each measurement comprises the method’s standard deviation, mass balance equations were over-determined, leading to positive or negative balance gaps. Unknown mass flows (e.g., solid fertilisers) were calculated according to TS-concentration or density balances. The mass balances were fitted to minimise balance gaps: mean concentrations were iteratively adapted to fulfil mass balance equations for each single unit operation as well as for the entire process. As boundary condition, each concentration had to remain within the standard deviation of the mean value. The method allowed to almost close the balance gaps (100% of total solids and on average 95.2% of all nutrients).
Separation efficiency
The average values and standard deviations for the separation efficiencies \(\eta_{i}\) were calculated according to (Eq. 1) to benchmark the efficiency of each unit operation. The efficiency compares the concentration of the component \(i\) for each unit operation \((c_{{i, {\text{unit}}}} )\) with the initial concentration of the digestate \((c_{{i, {\text{DIG}}}} )\).
Results and discussion
Chemical and physical parameters
Visual inspections revealed significant differences between the process fractions. Solid fractions were of dark brown colour, paste-like, and fibrous. Liquid fractions from screw press separator (SPL), decanter (DECL), and retentate of the ultrafiltration step (UFR) were characterised by pronounced turbidity and brown colour (see Fig. 4). The UF permeate (UFP) is optically free of organic particles and of lighter colour. Retentate of the reverse osmosis (ROR) is intensified in colour. The permeate of the last reverse osmosis step (ROP) optically appears like clean water without organic particles.
Rheological measurements showed that the original digestate is of high viscosity with pseudoplastic properties. The liquid fraction of the decanter is of lower viscosity, but still well above water viscosity, and also shows pseudoplastic rheological behaviour (Gienau et al. 2018). Table 1 shows the chemical and physical parameters of all process streams. The density of all process streams varied between 1000 and 1029 kg m−3. The pH values were in the range of 5.6–8.3. Concentrations of total solids, volatile solids, nutrients and other organic components constantly decreased at each separation unit. TS of the raw digestate (DIG) decreased from 5.8 wt% to no detectable solids in the reverse osmosis permeate (ROP). Most solids were detached by the screw press separator and the decanter, with TS concentrations of 19–21 wt%. Solids from the screw press separator (SPS) had a higher volatile solid content than the decanter solid fraction (DECS). The permeate was particle free after passing the ultrafiltration membrane with a pore size of 50 nm. The remaining COD and TS in the UF permeate can be attributed to salts and low molecular weight organic substances such as saccharides, proteins, and humic substances (Gienau et al. 2018).
The nutrient concentrations varied for all process streams. Measured concentrations of input material (i.e., 4.0 kg t−1 Ntotal, 1.6 kg t−1 P2O5, and 4.6 kg t−1 K2O) are well in accordance with published values (Möller and Müller 2012). Highest concentrations of total nitrogen and phosphorus were found in the solid streams from the screw press separator (SPs) and the decanter centrifuge (DECs) with 6.0 kg t−1 and 9.7 kg t−1, respectively. The retentate of the reverse osmosis (ROR) showed the highest values of ammonia, nitrogen and potassium (4.0 kg t−1 NH4–N and 10.1 kg t−1 K2O, respectively). The reverse osmosis membranes retained almost all organic, inorganic and nutrient substances. Chemical oxygen demand of the permeate was reduced from approx. 63 g L−1 to < 15 mg L−1 in the last reverse osmosis step. The quality of the permeate (ROP) was characterised by COD < 15 mg L−1, Ntotal = 14 mg L−1, NH4–N = 10 mg L−1 and P2O5 < 0.1 mg L−1. The RO permeate thus is of high quality. All concentrations apart from total nitrogen even fulfil WWTP size 5 effluent requirements in Germany (legal limit WWTP size 5: Ntotal = 13 mg L−1). In Germany, there is no legal limit for the discharge of treated digestate effluent. Depending on the quality of the local receiving water body, the local authorities decide on the necessary effluent quality. In a full-scale treatment plant, the RO step can be individually designed to meet the necessary requirements. The obtained effluent concentrations are lower than the values reported in the literature: Ledda et al. (2013) and Chiumenti et al. (2013) report mean effluent concentrations of 20 mg L−1 Ntotal and 85 mg L−1, respectively, which could be critical regarding water discharge.
Mass balance
Based on measured mass flow as well as physical and chemical parameters, Fig. 5 shows a Sankey diagram of the complete process, extrapolated to a daily mass flow of 150 t day−1 digestate. The input of 150 t day−1 digestate contained 94% water (141.3 t day−1). The screw press separator delivered a solid output of 16 t day−1. Before the decanter, an aqueous solution of polymers (7.0 t day−1) was mixed into the liquid stream of the screw press separator to improve separation efficiency of the particularly fine grain fraction. Thus, the decanter separated another 16 t day−1 of solids. With significant amounts of total solids, total nitrogen, and phosphorus, both solid fractions represented 32 t day−1 of a high-content organic N/P fertiliser. The supernatant of the decanter centrifuge (125 t day−1) was fed to the ultrafiltration unit. The retentate of the ultrafiltration amounted to about 47 t day−1, mainly consisting of water (45.4 t day−1). The UF retentate was reduced in the particulate nutrients phosphorus and organic nitrogen. Ammonia and potassium concentrations are similar to the original digestate. The permeate of the ultrafiltration was nearly free of solids and contained dissolved nutrients/salts and low molecular weight organic components such as proteins and saccharides (compare Table 1). All dissolved components were further transferred to the retentate of the reverse osmosis (22 t day−1). The dischargeable permeate of the reverse osmosis amounted to 56.0 t day−1.
Regarding the complete nutrient separation process, 20% of the input mass flow volume (i.e., from screw press separator and decanter centrifuge) were transferred into a solid N/P-fertiliser (Fig. 6). About 30% of the digestate can be found in the retentate of the ultrafiltration. The retentate of the reverse osmosis amounted to about 14%. The biggest process stream was the treated water after reverse osmosis (35.8%). The UF retentate can be used as liquid N/K-fertiliser. As an alternative, it is often recirculated to the decanter or the digester. Including an internal recirculation loop to the decanter, an RO permeate share of 53.9% of the input mass flow was calculated (Fig. 6). The pilot scale results thus support the literature reports [i.e., 46% (Bolzonella et al. 2017); 48% (Chiumenti et al. 2013) and 50% (Drosg et al. 2015)].
Separation efficiencies
The separation efficiency of each process step can be benchmarked by comparing the concentration of each component with the initial concentration of the digestate. Figure 7 shows the cumulated separation efficiency of the organic fractions. The combined liquid/solid separation (SPS and DECS) detached about 67% of TS and nearly 74% of organic matter (VS). For the screw press separator, high standard deviations for the measured parameters occurred. Obviously, these variations were based on fluctuating input quality, composition and TS/VS values of the digestate. The combined liquid/solid separation with supporting flocculants represented an appropriate separation technique for significant TS and VS reduction to enable ultrafiltration processes. The separation of TS and VS after ultrafiltration amounted to 83% and 92%, respectively. After the three-stage reverse osmosis process, no TS and VS were detectable; the separation efficiency was 100%. Total COD reduction was 92.5% after UF and > 99.9% after RO (see Table 1). In comparison, Waeger et al. (2010) report COD removal rates of 80–85% after MF or UF for digestate from a biogas plant fed with organic waste. Chiumenti et al. (2013) report 91.7% COD removal after UF and > 99.9% after RO for a similar biogas plant feedstock to this study.
The cumulated separation efficiency of the nutrients total nitrogen (TN), ammonia (NH4–N), potassium (K2O) and phosphorus (P2O5) constantly increased at each separation step (Fig. 8). For total nitrogen, a linear separation trend is visible starting with 8% after the screw press separator over 55% after UF to > 98% after reverse osmosis. Ammonia was first separated after the decanter centrifuge (13%) and not significantly affected by the ultrafiltration membrane. As the ammonia equilibrium is not strictly located on the side of ammonia, the centrifugal field can detach gaseous, dissolved components. By shifting the ammonia equilibrium towards ammonia with pH values < 7, the ammonia separation efficiency reached 99.4% after the reverse osmosis step. Waeger et al. (2010) report total nitrogen removal of 5–18%, Ledda et al. (2013) of 60–80%, and Chiumenti et al. (2013) of 61%. The differences can be explained by the percentage of dissolved ammonia nitrogen on total nitrogen due to different feedstock and process conditions. In all studies, removal efficiencies after RO were > 98%.
For potassium, a quite similar separation behaviour can be observed. Potassium concentration in the liquid phase was nearly constant through solid/liquid separation and UF, while separation efficiency reached nearly 100% by reverse osmosis. Phosphorus was constantly separated to 27, 79, 98 and 100% for screw press separator, decanter, ultrafiltration, and reverse osmosis, respectively. Large standard deviations were detected for the screw press separator. As phosphorus is essentially particulate and thus connected to the solid fraction (Lukehurst et al. 2010; Gienau et al. 2018), separation efficiency is linked to the detachment of total solids (compare Fig. 7).
Membrane performance and stability
The ultrafiltration step needs high cross-flow velocities to minimise organic fouling on the membrane surface. UF amounts to more than 50% of total operation costs of the treatment chain (Fuchs and Drosg 2013). Therefore, UF performance was constantly monitored and optimised. During standard operation procedure, UF feed temperature was 42 °C (supported by mesophilic fermenter conditions) and the cross-flow velocity was set to 4–5 m s−1 at a transmembrane pressure difference (TMP) of 4 bars (see Test 1 in Table 2). With a permeate yield of 70%, a mean permeate flux of 20.5 L m−2 h−1 could be achieved. In comparison, Waeger et al. (2010) report membrane fluxes between 20 and 50 L m−2 h−1 for digestate from an organic waste biogas plant at TMPs of 1 bar with total permeate recirculation (yield = 0). The differences in flux rates can be explained by the different permeate yield and different feedstock to the biogas plant. Generally, organic waste biogas plants tend to show higher flux rates (Gienau et al. 2018). Besides cross-flow velocity and TMP, the ultrafiltration performance depends on total solid concentration, concentration of biopolymers, and apparent viscosity of the separated anaerobic sludge (Rosenberger et al. 2006; Jiang et al. 2008; Zacharof and Lovitt 2014). For efficient back transport of particles from the membrane surface, the cross-flow velocity has to be sufficiently high to achieve Reynolds numbers (Re) above the laminar flow regime.
The standard operation mode (reference case–100% relative energy demand) was first improved by increasing the feed temperature to 65 °C (Test 2), which reduces fluid viscosity and increases Re. Savings were 28% of process energy due to a significantly improved membrane flux of 29.4 L m−2 h−1. In a third test, high temperature was combined with a lower cross-flow velocity of 3 m s−1. As a result of the high temperature, viscosity of the sludge changed to lower values and enabled lower cross-flow velocities for sufficiently turbulent flow regimes. The measured flux of Test 3 was 23.3 L m−2 h−1, which is + 14% compared to the reference case (Test 1). By combination of flux improvement and reduction of volume flow rate through the tubular membrane channels, the relative energy demand of UF could be reduced to 48%. Test 3 was found to be the most economic and efficient optimisation case. Long-term studies with technical equipment are planned for the future.
Above results were obtained with frequent chemical membrane cleaning. Cleaning intervals were 1–2 weeks. Chemical cleaning was carried out fully automated with acids and bases for about 2–3 h. The chemicals have no negative impact on the material of the ultrafiltration plant, i.e., stainless steel pipes and ceramic ultrafiltration membranes. Initial membrane flux was achieved after each cleaning step. Availability of the plant was > 90% including maintenance. Life time of the ceramic ultrafiltration membrane and the polymeric reverse osmosis membrane are expected to be 7–9 years and 1–2 years, respectively.
Conclusion
The performed experimental tests demonstrate the successful separation of anaerobic sludge from an agricultural biogas plant over a 7-months operation period. Main products of the process are solid and liquid fertilisers, and dischargeable water, representing about 20, 44 and 36% of total mass flow, respectively. It was shown that an internal recirculation of UF retentate increases dischargeable water yield from 36 to 54%. Solid/liquid separation by screw press separator and decanter centrifuge in combination with polymeric flocculants provided a reliable pre-treatment for the following ultrafiltration unit. The solids from the combined solid/liquid separation were characterised by high total solid concentrations (19–21%), but also by concentrations of 4.7–6.1 kg t−1 phosphorus and 6.0–9.7 kg t−1 total nitrogen. Thus, the solids represent a high value organic N/P-fertiliser for local-field applications.
The membrane treatment was successfully realised by ceramic ultrafiltration units and a three-stage reverse osmosis unit. For the ultrafiltration membrane, a flux performance of 20–30 L m−2 h−1, depending on cross-flow velocity and process temperature, was measured. The retentate of the reverse osmosis was rich in dissolved nutrient salts such as K+ and NH4+ and represented a directly available liquid N/K-fertiliser. The RO permeate in this study almost achieved the German effluent limits for wastewater treatment plants size 5. In Germany, the required effluent quality for discharge of treated digestate is defined for each individual case and depends on the quality of the receiving water body. It is possible to adjust the clear water quality by design of the RO train and pH adaptation. A comparison of obtained nutrient and mass balances with the literature revealed a spread of results, probably depending on feedstock to the biogas plant and on process configuration. For a sound understanding and increasing applicability of the process train, a broader database based on onsite trials is needed. The literature on membrane performance is very limited and mainly focusses on lab-scale trials.
Besides the quality of the nutrient products, the economy of the process depends on its energy consumption. First optimisation schemes resulted in energy reductions of 50% for the ultrafiltration unit. This can be realised by adjusting process temperature and flow conditions in the tubular membrane modules. Future investigations should focus on the long-time behaviour of the ultrafiltration performance and the energy consumption in full scale.
References
Abubaker J, Risberg K, Pell M (2012) Biogas residues as fertilisers—effects on wheat growth and soil microbial activities. Appl Energy 99:126–134. https://doi.org/10.1016/j.apenergy.2012.04.050
AbwV (2005) Ordinance on requirements for the discharge of waste water into waters (Waste Water Ordinance—AbwV). 1–121
Balsari P, Santoro E, Dinuccio E, Gioelli F (2006) Assessment of the performances of different mechanical solid-liquid separators for pig and cattle slurries. Proc 12th Ramiran Int Conf Technol Recycl Manure Org Residues Whole-Farm Perspect 2:157–159
Bhattacharya P, Ghosh S, Mukhopadhyay A (2013) Combination technology of ceramic microfiltration and biosorbent for treatment and reuse of tannery effluent from different streams: response of defence system in Euphorbia sp. J Recycl Org Waste Agric. https://doi.org/10.1186/2251-7715-2-19
Bolzonella D, Fatone F, Gottardo M, Frison N (2017) Nutrients recovery from anaerobic digestate of agro-waste: techno-economic assessment of full scale applications. J Environ Manage. https://doi.org/10.1016/j.jenvman.2017.08.026
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Camilleri-Rumbau MS, Norddahl B, Kjær Nielsen AK, Christensen KV, Fjerbæk Søtoft L (2013) Comparative techno-economical study between membrane technology systems for obtaining concentrated fertilizers from biogas plant effluents. 15th Ramiran Int Conf 8:7–10
Chiumenti A, Da Borso F, Teri F, Chiumente R, Piaia B (2013) Full-scale membrane filtration system for the treatment of digestate from a co-digestion plant. Appl Eng Agric 29:985–990. https://doi.org/10.13031/aea.29.10117
Choo K, Lee C, Pek U, Koh U, Kim S, Koh J (1992) Characteristics of membrane filtration as a post treatment to anaerobic digestion. J Korean Ind Eng Chem 3(4):730–738
Drosg B, Fuchs W, Al Seadi T, Madsen M, Linke B (2015) Nutrient recovery by biogas digestate processing. In: IEA Bioenergy. http://www.iea-biogas.net/files/daten-redaktion/download/TechnicalBrochures/NUTRIENT_RECOVERY_RZ_web1.pdf. Accessed 8 Feb 2018
Dubois M, Gilles K, Hamilton J, Rebers P, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
DüV (2017) Fertiliser Ordinance: (in German) Verordnung über die Anwendung von Düngemitteln, Bodenhilfsstoffen, Kultursubstraten und Pflanzenhilfsmitteln nach den Grundsätzen der guten fachlichen Praxis beim Düngen (Düngeverordnung—DüV)
Fuchs W, Drosg B (2013) Assessment of the state of the art of technologies for the processing of digestate residue from anaerobic digesters. Water Sci Technol. https://doi.org/10.2166/wst.2013.075
German Biogas Association (2017) Biogas sector statistics 2016/2017. https://de.statista.com/statistik/daten/studie/167676/umfrage/branchendaten-fuer-biogas-in-deutschland. Accessed 13 Nov 2017
Gienau T, Brüß U, Kraume M, Rosenberger S (2018) Nutrient recovery from biogas digestate by optimised membrane treatment. Waste Biomass Valori. https://doi.org/10.1007/s12649-018-0231-z
Jiang T, Myngheer S, De Pauw D, Spanjers H, Nopens I, Kennedy MD, Amy G, Vanrolleghem PA (2008) Modelling the production and degradation of soluble microbial products (SMP) in membrane bioreactors (MBR). Water Res 42:4955–4964. https://doi.org/10.1016/j.watres.2008.09.037
Ledda C, Schievano A, Salati S, Adani F (2013) Nitrogen and water recovery from animal slurries by a new integrated ultrafiltration, reverse osmosis and cold stripping process: a case study. Water Res 47:6157–6166. https://doi.org/10.1016/j.watres.2013.07.037
Lee Y, Oa S (2013) Nutrient transport characteristics of livestock manure in a farmland. Int J Recycl Org Waste Agric 2:1–6. https://doi.org/10.1186/2251-7715-2-1
Lukehurst C, Frost P, Al Seadi T (2010) Utilisation of digestate from biogas plants as biofertiliser. In: IEA Bioenergy. http://task37.ieabioenergy.com/files/daten-redaktion/download/publi-task37/Digestate_Brochure_Revised_12-2010.pdf. Accessed 8 Feb 2018
Masse L, Masse DI, Pellerin Y (2007) The use of membranes for the treatment of manure: a critical literature review. Biosyst Eng 98:371–380. https://doi.org/10.1016/j.biosystemseng.2007.09.003
Meixner K, Fuchs W, Valkova T, Svardal K, Lodereer C, Neureiter M, Bochmann G, Drosg B (2015) Effect of precipitating agents on centrifugation and ultrafiltration performance of thin stillage digestate. Sep Purif Technol 145:154–160. https://doi.org/10.1016/j.seppur.2015.03.003
Möller K, Müller T (2012) Effects of anaerobic digestion on digestate nutrient availability and crop growth: a review. Eng Life Sci 12:242–257. https://doi.org/10.1002/elsc.201100085
Riva C, Orzi V, Carozzi M, Acutis M, Boccasile G, Lonati S, Tambone F, D’Imporzano G, Adani F (2016) Short-term experiments in using digestate products as substitutes for mineral (N) fertilizer: agronomic performance, odours, and ammonia emission impacts. Sci Total Environ 547:206–214. https://doi.org/10.1016/j.scitotenv.2015.12.156
Rosenberger S, Laabs C, Lesjean B, Gnirss R, Amy G, Jekel M, Schrotter C (2006) Impact of colloidal and soluble organic material on membrane performance in membrane bioreactors for municipal wastewater treatment. Water Res 40:710–720. https://doi.org/10.1016/j.watres.2005.11.028
Schoumans OF, Chardon WJ (2014) Phosphate saturation degree and accumulation of phosphate in various soil types in The Netherlands. Geoderma 237:325–335. https://doi.org/10.1016/j.geoderma.2014.08.015
Vaneeckhaute C, Lebul V, Michels E, Belia E, Vanrolleghem PA, Tack F, Meers E (2017) Nutrient recovery from digestate: systematic technology review and product classification. Waste Biomass Valori 8:21–40. https://doi.org/10.1007/s12649-016-9642-x
Waeger F, Delhaye T, Fuchs W (2010) The use of ceramic microfiltration and ultrafiltration membranes for particle removal from anaerobic digester effluents. Sep Purif Technol 73:271–278. https://doi.org/10.1016/j.seppur.2010.04.013
Zacharof M, Lovitt RW (2014) The filtration characteristics of anaerobic digester effluents employing cross flow ceramic membrane micro filtration for nutrient recovery. Desalination 341:27–37. https://doi.org/10.1016/j.desal.2014.02.034
Acknowledgements
This research project is supported and financed by the German Federal Environmental Foundation (Deutsche Bundesstiftung Umwelt), Grant no. AZ 31276. The scientific research was organised at Osnabrück, University of Applied Science in cooperation with the company A3 Water Solution GmbH. The pilot plant was provided by A3 Water Solutions GmbH and the local farmer in Bad Bentheim. The authors gratefully acknowledge the entire team of the farm for their practical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Gienau, T., Brüß, U., Kraume, M. et al. Nutrient recovery from anaerobic sludge by membrane filtration: pilot tests at a 2.5 MWe biogas plant. Int J Recycl Org Waste Agricult 7, 325–334 (2018). https://doi.org/10.1007/s40093-018-0218-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40093-018-0218-6