Abstract
Purpose
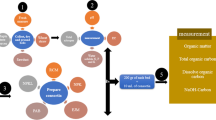
A study was conducted to characterize the ‘Madhyam culture’ (Excel Crop Care Limited.), an aerobic-composting microbial consortium culture, and understand composting dynamics, product quality and use in crop production vis-à-vis vermi-composting (using earthworms).
Methods
16S rDNA analysis was used to characterize aerobic-composting culture. Aerobic-composting and vermi-composting technologies were evaluated to decompose sorghum straw and dung biomass (80:20 ratio; primed with 0.5% urea and 4% rock phosphate) to study days to maturity and composting dynamics in terms of changes in temperature and microbial population. Compost quality was tested for macro-, micro-nutrients and C:N ratio, and evaluated for food production in on-farm trials.
Results
16S rDNA analysis screened sixteen bacterial isolates—eight related to genus Bacillus, three to each Halobacillus and Staphylococcus, one to each Microbacterium and Streptomyces. The population of bacteria was 4.5 cfu ml−1 at 10−7 dilution. Aerobic- and vermi-composts matured in around 50 and 60 days, respectively. Aerobic-composting throughout recorded relatively higher bacterial population, and higher temperatures during the initial phase. Aerobic-compost tested for high nutrient (1.55% N, 0.93% P, 1.00% K) content and stable C:N ratio (10.3) compared to vermi-compost (1.11% N, 0.43% P, 0.96% K and C:N ratio of 11.7). Field evaluation of both composts showed yield benefit and saving of chemical fertilizers up to 25%.
Conclusions
Aerobic-composting (using microbial consortium culture), like vermi-composting, proved to be an effective technology with advantage of no requirement to maintain ambient living conditions in lean periods as is required for earthworms in vermi-composting, but needs more energy/labor for biomass turnings.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
Low soil organic carbon (C) levels and macro-, micro-nutrient deficiencies due to soil mismanagement and misuse are the major challenges for improving productivity in the semi-arid tropics (Chander et al. 2015). Soil organic C is one of the most important parameters governing soil health through influencing soil chemical, physical and biological properties, and thereby influencing crop yields as well (Wani et al. 2003). Moreover, soils are largest terrestrial reservoirs of C (~ 2500 Pg C up to 2 m depth, compared to 600 Pg C in biota and 750 Pg C in atmosphere) (Batjes 1999; Lal and Kimble 1997) and potential C-sink to regulate climate change processes (Lal et al. 2007; Lal 2011). So, practices that add organic matter into soil need to be promoted. In India, ~ 350 million t organic wastes are generated annually from agricultural sources (Pappu et al. 2007), and so recycling C and nutrients contained in such wastes is potential opportunity for soil C-building and cutting costs of chemical fertilizers (Chander et al. 2013; Nagavallemma et al. 2006).
Composting is the technology for conversion of bulky organic wastes into low volume nutrient-enriched and stable product (Yousefi et al. 2013; Kharrazi et al. 2014). Traditional composting (farmers’ practices of heaping straw and dung) is very time consuming and relatively less effective. In such a case, using half decomposed compost/manure/plant residue creates many plant nutrient and pest-related problems, rather than benefits. Vermi-composting is one of the tested technologies to effectively recycle on-farm wastes to produce quality compost for use in crop production (Chander et al. 2013; Wani et al. 2014). Vermi-composting hastens the decomposition process through physical breakdown of the raw biomass coupled with mixing of vast spectrum of microbes with the biomass while passing through earthworm gut. The microbes of earth worm gut are highly potential in digesting the organic materials as well as polysaccharides (Aira et al. 2007; Zhang et al. 2000). However, continuously maintaining ambient living conditions for earthworms is a challenge in vermi-composting and a major cause for many on-farm failures. So, the other option could be to accelerate need-based decomposition of biodegradable materials through the external addition of decomposing microbial consortium culture (aerobic-composting). This may add the convenience of undertaking composting as and when needed without worrying throughout the year about maintaining moisture and feeding material as is needed in vermi-composting. However, relative effectiveness of aerobic-composting via external microbial culture needs to be evaluated vis-à-vis vermi-composting technology, as studies indicate that vermi-composting also renders the advantage of enhancing the diversities of bacterial and fungal communities (Huang et al. 2014). Well-tested scalable composting options are need of the day not only for agricultural wastes, but also for effective disposal of municipal and household wastes (generated to the tune of 125,000 tons per day in a country like India as per the estimates of Central Pollution Control Board (2017) to reduce pollution arising from landfills and open dumps.
Apparently high microbial activity under composting have an indirect role in improving compost nutrient quality by nitrogen fixers, nitrifiers, sulfur oxidizers (Richardson and Simpson 2011) and may also synthesize chemicals which act as plant growth hormones (Pizzeghello et al. 2001; Ghosh et al. 2003; Tomati et al. 1988). Lack of information on standardized practices, processes and products with regard to organic composts is one major drawback in precise use of organics. Therefore, a study was planned to characterize microbial consortia culture (for aerobic-composting) and evaluate its efficacy vis-à-vis vermi-composting in recycling on-farm wastes while understanding microbial dynamics of decomposition process and compost quality and evaluation in field application.
Methodology
Characterization of microbial consortium (Madhyam) culture
‘Madhyam’ is a culture of micro-organisms developed for accelerated aerobic-composting of organic waste by Excel Crop Care Limited.
Enumeration of microorganisms in microbial consortium (Madhyam) culture
Ten grams of ‘Madhyam’ sample was homogenized with 90 ml of sterile 0.85% NaCl solution in water and incubated for 1 h at 120 rpm. Then, the samples were serially diluted (up to 10−8) and plated on several medium such as nutrient agar for bacteria (10−4 to 10−7 dilutions), Bennett’s agar for actinobacteria (10−3 to 10−6 dilutions) and potato dextrose agar for fungi (10−2 to 10−5 dilutions). The plates were incubated at 28 °C for 2–7 days. The colonies that appeared at the end of incubation were enumerated as colony forming units (CFU) per gram of sample on dry weight basis. Characteristics of the colony were noted and representative single colonies were further cultured and purified using respective medium. The single colonies were stored at 4 °C for subsequent analysis.
Cellulose degradation capability of microbial consortium (Madhyam) culture
Cellulase degrades cellulose in the plant cell wall and cellulase production by bacteria was determined by cellulose congo red agar medium. The test microbes were inoculated on above medium and incubated for 4 days at 28 ± 2 °C. Halo zone around the microbial colonies indicates cellulase production and the observations were documented on a 0–4 rating scale as, 0 = no change; 1 = positive; 2 = halo zone of 1–3 mm; 3 = halo zone of 4–6 mm and 4 = halo zone of 7 mm and above (Hendricks et al. 1995; Gopalakrishnan et al. 2015).
Molecular identification of microbial consortium (Madhyam) culture
The potential cellulose-degrading bacteria identified previously were grown until log phase is reached. The genomic DNA was isolated as per Bazzicalupo and Fani (1995). The 16S rDNA gene was amplified through universal bacterial primer 907R (5′-CCGTCAATTCMTTTRAGTTT-3′) and 785F (5′-GGATTAGATACCCTGGTA-3′) according to the conditions of Pandey et al. (2005). Resulting PCR products were sequenced at Macrogen Inc. (Seoul, Korea). The sequences obtained were compared with those from the GenBank using BLAST program (Altschul et al. 1990) and aligned through Clustal W software (Thompson et al. 1997). The phylogenetic trees were constructed using neighbor-joining method according to Saitou and Nei (1987).
Evaluation of composting methods
Vermi- and aerobic-composting methods were evaluated for their efficacy in decomposing on-farm wastes in open field conditions at ICRISAT during 2014. The raw biomass was decomposed under the shade by spreading on cement floor base and maintaining moisture through watering the alternate days. Thus, there were 2 treatments: (1) vermi-composting, and (2) aerobic-composting. Under each of the composting method, the raw biomass used was sorghum straw and cow dung (in the ratio 80–20). Sorghum straw was chopped with the help of tractor-run chopper. Urea (0.5%) priming of sorghum biomass was done before 1 week to lower C:N (Carbon:Nitrogen) ratio. In addition, rock phosphate @ 4 kg per 100 kg biomass was also added at start of the experiment. In case of vermi-composting, Eudrilus eugeniei and Eisenia fetida species of earth worms were used for decomposition of biomass, while in case of aerobic-composting; microbial culture (Madhyam, Excel Crop Care Ltd) @ 1 kg per ton was used.
Raw biomass (~ 1 ton) was filled in layers of sorghum (0.30 m) and cowdung slurry. Vermi-composting was started on a harvested bed of ~ 0.15 m having earthworm culture. In aerobic-composting, microbial culture was spread between the layers and a heap of ~ 0.75 m was prepared on day-1 itself. However, in case of vermi-composting, heap was filled up to 0.75 m height in stages at weekly intervals at days 1, 7 and 14. Intense microbial activity in aerobic-composting leading to high temperatures is required to be regulated through enhanced aeration by regular biomass turnings; so, turnings were done at 10 days interval up to 50 days to maintain the aerobic conditions.
Both composting methods were evaluated for earliness to maturity as judged from color, texture and stabilization of temperature. Other parameters evaluated were microbial dynamics, changes in temperature and chemical analysis of composts (see sections ahead). Bio-washes from vermi- and aerobic-compost were also collected during 30–45 days in plastic containers through outlets for cement floor base at lower end of slope and stored at 4 °C. Bio-washes were also analyzed for nutrient contents and plant growth hormones as described in subsequent sections.
Microbial dynamics during composting
To study microbial dynamics during composting, the samples of vermi- and aerobic-compost were collected at 1, 20 and 55 days of the study in polythene bags and stored at 4 °C. During sampling a treatment at any time, three composite samples of about 100 g each were collected at 0.15–0.20 m depth along length of the heap and thoroughly mixed together for microbial analysis.
For microbial isolation, nutrient agar was used for bacteria, potato dextrose agar for fungi and actinomycetes isolation agar for actinomycetes. All the media were prepared as per standard procedure and sterilized in an autoclave at 121 °C and 15 lb pressure for 20 min. After sterilization, media was dispersed into Petri plates under a sterile atmosphere in laminar air flow and after solidification, these plates were used for the isolation of microorganisms. For enumerating microbial population, dilution plate technique was followed (Ariffin et al. 2008). One gram of compost sample was added to 9 ml of sterile distilled water in a test tube to get 10−1 dilution which was serially diluted. Spread plate technique was followed for isolation of microorganisms through spreading 0.1 ml of the desired dilution to the surface of solid agar medium. To check dehydration of medium, the agar plates were wrapped with parafilm, and kept in incubator at 28 + 2 °C. The period of incubation varied for different microorganisms: 24–48 h for bacteria, 72 h for fungi and 72–96 h in case of actinomycetes. The colonies grown in each plate were enumerated with the help of a colony counter as CFU. The total CFU were worked out by taking into account the dilution factor.
Changes in temperature during composting
Daily mean temperature was recorded in both composting types at two depths (0.15 and 0.30 m) using automatic sensors.
Chemical analysis of compost
For estimation of nitrogen (N), phosphorus (P), potassium (K), calcium (Ca) and magnesium (Mg) in compost, 0.5 g of samples was digested using 3.5 ml sulfuric acid–selenium mixture (Sahrawat et al. 2002a). N and P in the digests were estimated with the help of auto-analyzer, and K, Ca and Mg with help of atomic absorption spectrophotometer (AAS). For micronutrients, zinc (Zn), copper (Cu), iron (Fe) and manganese (Mn), 0.2 g compost samples was digested with 2 ml tri-acid and contents in the digests were estimated using AAS (Sahrawat et al. 2002b). S and B in samples were estimated through digesting 0.2 g samples with 2 ml nitric acid plus 0.5 ml hydrogen peroxide and determining the contents in the digest through Inductive Coupled Plasma Atomic Emission Spectrophotometer (ICP-AES) (Mills and Jones Jr 1996). The pH was measured in 1:2 water suspension (20 g sample and 40 ml water) using glass electrode and total C by Skalar TN/TC analyzer through using 1 g sample (PrimacsSNC100 series).
Similar process was followed for macro, micro nutrient chemical analysis in bio-wash samples.
Plant growth hormones analysis in compost bio-washes
Plant growth hormones in compost bio-washes were extracted using standard method by Miezah et al. (2008). A 100 ml of compost washing was taken in 500 ml extraction bottle and then added 200 ml of methanol. This mixture was kept in the shaker for 24 h. The supernatant was then centrifuged for 10 min at 5000 rpm and organic part was stored in freezer. Again, 200 ml methanol was added in aqueous phase and same process followed 4 times to get 800 ml of methanol extract. Then with help of by rotary vacuum evaporator, the methanol extract was concentrated to 2 ml. It was then passed through anhydrous sodium sulfate to make it moisture free and kept in freezer before analysis.
Thin layer chromatography (TLC) technique was used for hormone identification viz. auxins, gibberellins and cytokinins. Each bio-wash extract along with its standards were spotted onto chromatographic plates of 20 cm × 20 cm which were coated with silica gel (60F254) of thickness 0.25 mm. After 4 h of each run, the TLC plates were air dried and then sprayed with coloring reagent, i.e., H2SO4. Once the plates were oven dried, Rf value (distance traveled by compound/distance traveled by solvent) of each spot was calculated.
The plant growth hormone standards were used for validating method for auxins (1 ppm), gibberellins (1 ppm) and cytokinins (2 ppm). For isolation of auxins and gibberellins under TLC, iso-propyl alcohol: ammonium hydroxide: water (90:10:10) was used on volume basis, whereas, in case of cytokinins, butyl alcohol: acetic acid: water (80:10:10) was used (Miezah et al. 2008).
On-farm evaluation of composts
The composts prepared following this methodology were evaluated in on-farm studies in India—vermi-compost in Sagar district during 2011–2012 post-rainy season (6 trials with chickpea, 9 trials with wheat) and aerobic-compost in Kadapa district during 2016 rainy season (40 trials with paddy crop). The treatments evaluated were, (1) Balanced nutrition (BN) comprising of soil test-based addition of N, P and K along with deficient micro- and secondary nutrients (i.e. S, Zn and B), and (2) Integrated nutrient management (INM) i.e. 75% of BN inputs + compost. The vermi- or aerobic-compost was added on the basis to replace 25% of N requirement in case of non-legumes and 25% of P requirement in legumes. The on-farm treatment size was 2000 m2 side by side. All other crop management practices were similar in both the treatments. Composts and all required nutrients except N in non-legumes were added as basal application. In case of N in non-legumes, 50% was added as basal and 25% after 1 month and rest 25% after 2 months. Crop yields were estimated from 3 m × 3 m = 9 m2 plots, and calculated into kg ha−1.
Results and discussion
Enumeration of microorganisms in microbial consortium (Madhyam) culture
A total of 21 bacterial isolates including one actinomycete were isolated from nutrient agar plates and Bennett’s agar plates based on the colony morphology, color and shape. Fungal colonies were not found on the PDA plates while single actinomycete was found on the Bennett’s agar. The bacterial population was found to be 4.5 cfu ml−1 at 10−7 dilution.
Cellulose degradation capability of bacteria isolated from microbial consortium (Madhyam) culture
Of the 21 isolates studied for cellulose degradation, a total of 16 isolates produced the cellulase enzyme. A clear halo zone was found around the colonies in the congo red agar plates. Of the 21 bacteria studied, two (MP-2 and MP-20) were found to produce maximum cellulase (Table 1).
Molecular identification of microbial consortium (Madhyam) culture
The 16 cellulase-producing bacteria were identified through 16S rDNA sequencing. A neighbor-joining dendrogram was generated with the partial sequences of 16 cellulase producing bacteria (1400 bp) and representative sequences from the databases. Phylogenetic analysis of the 16 isolates matched with Streptomyces (1), Microbacterium (1), Halobacillus (3), Staphylococcus (3), and Bacillus (8 isolates) (Fig. 1; Table 2). Bacillus and Halobacillus are reported to have various traits including N-fixation, P-solubilization, plant growth promotion, biological control and biofortification (Singh et al. 2008; Richardson and Simpson 2011; Sharma et al. 2013; Gopalakrishnan et al. 2015, 2016). Streptomyces sp. are efficient in degrading cellulose and promote crop growth (El-Tarabily 2008; Gopalakrishnan et al. 2011, 2014, 2015, 2016). All the 16 isolates were able to grow at neutral pH (6.8–7.5) and mesophilic (28–35 °C) temperature (data not shown).
Evaluation of composting methods
Results revealed that aerobic-composting, like vermi-composting, is an effective technology to recycle on-farm wastes into valuable composts in a reasonably short time. Aerobic-compost was ready by around 50th day and vermi-compost by around 60th day. Breakdown of biomass while passing through earthworm gut and mixing of several microbes in coelomic fluid apparently accounted for fast decomposition in vermi-composting. While, high microbial population through added culture and regular turnings at 10-day interval apparently led to fast decomposition in aerobic-composting.
Microbial dynamics during composting
Results revealed that bacteria were the predominant microorganisms found throughout the study followed by actinomycetes and the fungi the least. In general, bacteria occupied 68–92% of the microbial population followed by actinomycetes by 8–32% and fungi the negligible counts (Table 3).
As regards bacterial population, throughout the study, a higher population (21.1–65.3 × 1010 CFU/g) was observed under aerobic-composting compared with vermi-composting (19.7–61.6 × 1010 CFU/g). The highest population in aerobic-composting was apparently due to external addition of microbial consortium culture dominated by bacterial isolates (see Table 1). In vermi-composting also, there is evidence that different microbes in coelomic fluid get mixed with the biomass while passing through their body and apparently accounts for reasonably higher bacterial number. With passage of time, the bacterial count declined which may be expected due to decomposition of preferable food components of bacteria and also pasteurization as result of high temperatures (Vivas et al. 2009). Bacillus spp. was predominant in both the compost types.
Fungi comprised very little component of microbial population (5–9 × 103 CFU/g on day 1, 5–10 × 103 CFU/g on day 20 and 4–4 × 103 CFU/g on day 55) (Table 3). Similar to bacterial count, the fungi population also decreased over the period of time. However, contrary to bacterial population trends, relatively less number of fungal colonies was observed under aerobic-composting apparently due to competition with corresponding higher bacterial population.
Similar to fungi, relatively higher population (71–237 × 109 CFU/g) of actinomycete was observed under vermi-composting compared with aerobic-composting (56–75 × 109 CFU/g) (Table 3). Mixing of earthworm coelomic fluid with the biomass could be the reason for consistently higher actinomycete population observed during vermi-composting. Further, lower temperature during the vermi-composting also supports the growth of actinomycetes.
Changes in temperature during composting
At 0.15 m depth, up to 30 days, the relatively higher daily mean temperature was recorded under aerobic-composting (38–67 °C) as compared with the vermi-compost (29–48 °C). Under aerobic-composting, it was relatively quite high during the initial 15 days apparently because of high microbial activity, thus indicating rapid decomposition. The temperature reached up to 67 °C by the 9th day (Fig. 2); the turnings at 10th day, however, regulated it to escalate beyond limit which could otherwise negatively affect microbial population and composting process as such and decreased it to 52 °C. By contrast to aerobic-composting, the temperature in vermi-composting remained below 40 °C during 1st week, which may be due to the fact that vermi-composting heap was filled in layers at 0, 7, 14 days. After 1 week, temperature increased and maintained between 40 and 48 °C almost up to 1 month. Between 30 and 60 days (till maturity), the temperatures were almost similar under both the composting processes and ranged between 31 and 39 °C.
At 0.30 m depth, the trends observed as such were similar to that at 0.15 m depth; however, slightly higher temperature was recorded at 0.30 m depth (Fig. 3). Up to 1 month, a higher temperature was observed under aerobic-composting which varied between 44 and 67 °C compared to vermi-composting between 32 and 51 °C. Similar to depth at 0.15 m, the corresponding temperature at 0.30 m depth beyond 30 days was almost at par in both the composting processes.
Chemical analysis of compost
Results showed aerobic-compost in general having relatively higher nutrient contents w.r.t N, P, K, Ca, S and Zn (Table 4). High nutrient contents in aerobic-compost may be due to better decomposition as a result of higher microbial activity observed in this study. However, vermi-compost was found to contain higher contents of Mg, Cu, Fe, Mn and B. Total C was found to be 160 mg g−1 in aerobic-compost and 130 mg g−1 in vermi-compost with C:N ratio of 10.3 and 11.7, respectively. The pH was almost same at 6.75 in aerobic-compost and 6.73 in vermi-compost. Reasonably higher nutrient contents in the compost types were probably due to better decomposition (Shah et al. 2015), and enrichment with rock phosphate and urea.
The bio-washes of vermi-compost and aerobic-compost were almost at par in nutritional quality (Table 5). Relatively higher N content in biowash collected from aerobic-compost is apparently due to better decomposition due to higher microbial activity through added culture. Thus, these washings may serve as good nutrient source for foliar application to manage nutrient deficiencies in any standing crop.
In standardizing hormones identification method using 25% sulphuric acid spray, a light pink color spot (0.75 Rf value) was observed for auxins, greenish yellow for gibberellins (Rf = 0.59) and light brown for cytokinins (Rf = 0.82). However, as regards the plant growth hormones in bio-washes, only auxin was detected in vermi-wash, while none of other hormones were detected in any of the bio-washes.
On-farm evaluation of composts
The on-farm evaluation of vermi-compost in wheat and chickpea crops, and aerobic-compost in paddy crop showed that with use of composts, the crop yields are not only maintained or increased over the balanced nutrient management solely through chemical fertilizers (Fig. 4), but also result in savings of chemical fertilizers up to 25%. The integrated use of vermi-compost along with chemical fertilizers recorded higher yield by 5% in wheat (4450 vs 4250 kg ha−1) and 9% in chickpea (2270 vs 2090 kg ha−1). Similarly, the use of aerobic-compost along with chemical fertilizers recorded higher paddy yield by about 7% (3570 vs 3330 kg ha−1) compared to sole use of balanced chemical fertilizers. Other studies have shown similar benefits with INM along with improvement in food nutritional quality through producing nutrient dense food and resilience-building of production systems (Chander et al. 2013; Mengistu et al. 2017).
Thus, aerobic-composting is a sound scalable technology as is vermi-composting for recycling on-farm wastes and suitability of any one is more dependent on farm situations. While continuously feeding the biomass and maintaining moisture is required in vermi-composting to maintain earthworms, which is not the case in aerobic-composting, but may involve recurring cost on the microbial culture and labor for turning the biomass to maintain aerobic conditions.
Conclusions
Aerobic- and vermi-composting proved effective technologies to recycle on-farm wastes into valuable manure. Aerobic-compost matured in around 50 days and vermi-compost in around 60 days. Thus, aerobic-composting may be a good option to handle large quantities of on-farm wastes as and when required without having to constantly maintain ambient living conditions as required for earthworms in vermi-composting. But, it needs more labor or energy for minimum four turnings required during the composting process. Both aerobic- and vermi-composting supported a diverse microbial population of bacteria, fungi and actinomycetes. Growth hormone auxin was detected in vermi-compost bio-wash. Also, both the composts produced had a good nutrient value and high stability. Thus, as per farmers’ requirements, both aerobic- and vermi-composting methods have a potential to be used as effective technologies to recycle the plant nutrients in on-farm wastes and help reduce the load of inorganic fertilizers. Its scaling-up will boost the economy and help in maintaining the sustainability of ecosystem.
References
Aira M, Monroy F, Dominguez J (2007) Earthworms strongly modify microbial biomass and activity triggering enzymatic activities during vermicomposting independently of the application rates of pig slurry. Sci Total Environ 385:252–261. https://doi.org/10.1016/j.scitotenv.2007.06.031
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Ariffin H, Abdullah N, Umikalsom MS, Shirai Y, Hassan MA (2008) Production of bacterial endoglucanase from pretreated oil palm empty fruit bunch by Bacillus pumilus EB3. J Biosci Bioeng 106:231–236. https://doi.org/10.1263/jbb.106.231
Batjes NH (1999) Management options for reducing CO2 concentrations in the atmosphere by increasing carbon sequestration in the soil. ISRIC, Wageningen, The Netherlands. https://www.researchgate.net/profile/Niels_Batjes/publication/228068233_Management_Options_for_Reducing_CO2_Concentrations_in_the_Atmosphere_by_Increasing_Carbon_Sequestration_in_the_Soil/links/0912f50c0a50b2b348000000/Management-Options-for-Reducing-CO2-Concentrations-in-the-Atmosphere-by-Increasing-Carbon-Sequestration-in-the-Soil.pdf
Bazzicalupo M, Fani R (1995) The use of RAPD for generating specific DNA probes for microorganisms. In: Clapp JP (ed) Methods in molecular biology. Humana, Totowa, pp 155–175. https://doi.org/10.1385/0-89603-323-6:155
Central Pollution Control Board, Delhi (2017) Status report on municipal solid waste management. http://www.cpcb.nic.in/divisionsofheadoffice/pcp/MSW_Report.pdf. Accessed Nov 2017
Chander G, Wani SP, Sahrawat KL, Kamdi PJ, Pal CK, Pal DK, Mathur TP (2013) Balanced and integrated nutrient management for enhanced and economic food production: case study from rainfed semi-arid tropics in India. Arch Agron Soil Sci 59(12):1643–1658. https://doi.org/10.1080/03650340.2012.761336
Chander G, Wani SP, Sahrawat KL, Pardhasaradhi G (2015) Soil test based nutrient management for sustainable intensification and food security: case from Indian semi-arid tropics. Commun Soil Sci Pl Anal 46(S1):20–33. https://doi.org/10.1080/00103624.2014.988087
El-Tarabily KA (2008) Promotion of tomato (Lycopersicon esculentum Mill.) plant growth by rhizosphere competent 1-aminocyclopropane-1-carboxylic acid deaminase–producing Streptomycete actinomycetes. Plant Soil 308:161–174. https://doi.org/10.1007/s11104-008-9616-2
Ghosh S, Penterman JN, Little RD, Chavez R, Glick BR (2003) Three newly isolated plant growth promoting bacilli facilitate the seedling growth of canola, Brassica campestris. Plant Physiol Biochem 41:277–281. https://doi.org/10.1016/S0981-9428(03)00019-6
Gopalakrishnan S, Pande S, Sharma M, Humayun P, Kiran BK, Sandeep D, Vidya MS, Deepthi K, Rupela O (2011) Evaluation of actinomycete isolates obtained from herbal vermicompost for biological control of Fusarium wilt of chickpea. Crop Prot 30:1070–1078. https://doi.org/10.1016/j.cropro.2011.03.006
Gopalakrishnan S, Vadlamudi S, Bandikinda P, Sathya A, Vijayabharathi R, Rupela O, Kudapa H, Katta K, Varshney RK (2014) Evaluation of Streptomyces strains isolated from herbal vermicompost for their plant growth-promotion traits in rice. Microbiol Res 169:40–48. https://doi.org/10.1016/j.micres.2013.09.008
Gopalakrishnan S, Srinivas V, Alekhya G, Prakash B, Kudapa H, Rathore A, Varshney RK (2015) The extent of grain yield and plant growth enhancement by plant growth-promoting broad-spectrum Streptomyces sp. in chickpea. SpringerPlus 4:31. https://doi.org/10.1186/s40064-015-0811-3
Gopalakrishnan S, Srinivas V, Srinivasan S, Sameer Kumar CV (2016) Plant growth-promotion and biofortification of chickpea and pigeonpea through inoculation of biocontrol potential bacteria, isolated from organic soils. SpringerPlus 5:1882. https://doi.org/10.1186/s40064-016-3590-6
Hendricks CW, Doyle JD, Hugley B (1995) A new solid medium for enumerating cellulose-utilizing bacteria in soil. Appl Environ Microbiol 61:2016–2019. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1388450/
Huang K, Li F, Wei Y, Fu X, Chen X (2014) Effects of earthworms on physicochemical properties and microbial profiles during vermicomposting of fresh fruit and vegetable wastes. Bioresour Technol 170:45–52. https://doi.org/10.1016/j.biortech.2014.07.058
Kharrazi SM, Younesi H, Abedini-Torghabeh J (2014) Microbial biodegradation of waste materials for nutrients enrichment and heavy metals removal: an integrated composting-vermicomposting process. Int Biodeterior Biodegrad 92:41–48. https://doi.org/10.1016/j.ibiod.2014.04.011
Lal R (2011) Sequestring carbon in soils of agro-ecosystems. Food Policy 36:S33–S39. https://doi.org/10.1016/j.foodpol.2010.12.001
Lal R, Kimble JM (1997) Conservation tillage for carbon sequestration. Nutr Cycl Agroecosyst 49:243–253. https://doi.org/10.1023/A:1009794514742
Lal R, Follett RF, Stewart BA, Kimble JM (2007) Soil carbon sequestration to mitigate climate change and advance food security. Soil Sci 172(12):943–956. https://doi.org/10.1097/ss.0b013e31815cc498
Mengistu T, Gebrekidan H, Kibret K, Woldetsadik K, Shimelis B, Yadav H (2017) The integrated use of excreta-based vermicompost and inorganic NP fertilizer on tomato (Solanum lycopersicum L.) fruit yield, quality and soil fertility. Int J Recycl Org Waste Agric 6(1):63–77. https://doi.org/10.1007/s40093-017-0153-y
Miezah K, Ofosu-Anim J, Budu GKO, Enu-Kwesi L, Cofie O (2008) Isolation and identification of some plant growth promoting substances in compost and co-compost. Int J Virol 4(2):30–40. https://doi.org/10.3923/ijv.2008.30.40
Mills HA, Jones JB Jr (1996) Plant analysis handbook II: a practical sampling, preparation, analysis and interpretation guide. Micro-Macro Publishing, Athens, GA. https://books.google.co.in/books/about/Plant_Analysis_Handbook_II.html?id=AzorngEACAAJ&redir_esc=y
Nagavallemma KP, Wani SP, Stephane Lacroix, Padmaja VV, Vineela C, Babu Rao M, Sahrawat KL (2006) Vermicomposting: recycling wastes into valuable organic fertiliser. SAT eJ 2(1):1–16. http://ejournal.icrisat.org/agroecosystem/v2i1/v2i1vermi.pdf
Pandey P, Kang SC, Maheshwari DK (2005). Isolation of endophytic plant growth promoting Burkholdeina sp. MSSP from root nodules of Mimosa pudica. Curr Sci 89:177–180. http://www.iisc.ernet.in/currsci/jul102005/177.pdf
Pappu A, Saxena M, Asolekar SR (2007) Solid wastes generation in India and their recycling potential in building materials. Build Environ 42(6):2311–2320. https://doi.org/10.1016/j.buildenv.2006.04.015
Pizzeghello D, Nicolini G, Nardi S (2001) Hormones-like activity of humin substances in Fagus sylvaticae forest. New Phytol 151:647–657. https://doi.org/10.1046/j.0028-646x.2001.00223.x
Richardson AE, Simpson RJ (2011) Soil microorganisms mediating phosphorus availability. Plant Physiol 156:989–996. https://doi.org/10.1104/pp.111.175448
Sahrawat KL, Kumar Ravi G, Rao JK (2002a) Evaluation of triacid and dry ashing procedures for determining potassium, calcium, magnesium, iron, zinc, manganese, and copper in plant materials. Commun Soil Sci Plant Anal 33:95–102. https://doi.org/10.1081/CSS-120002380
Sahrawat KL, Kumar Ravi G, Murthy KVS (2002b) Sulfuric acid-selenium digestion for multi-element analysis in a single digest. Commun Soil Sci Plant Anal 33:3757–3765. https://doi.org/10.1081/CSS-120015920
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Shah RU, Abid M, Qayyum MF, Ullah R (2015) Dynamics of chemical changes through production of various composts/vermicompost such as farm manure and sugar industry wastes. Int J Recycl Org Waste Agric 4(1):39–51. https://doi.org/10.1007/s40093-015-0083-5
Sharma A, Shankhdhar D, Shankhdhar SC (2013) Enhancing grain iron content of rice by the application of plant growth-promoting rhizobacteria. Plant Soil Environ 59:89–94. http://www.agriculturejournals.cz/publicFiles/82693.pdf
Singh N, Pandey P, Dubey RC, Maheshwari DK (2008) Biological control of root rot fungus Macrophomina phaseolina and growth enhancement of Pinus roxburghii by rhizosphere competent Bacillus subtilis BN1. World J Microbiol Biotechnol 24:1669–1679. https://doi.org/10.1007/s11274-008-9680-z
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882. https://doi.org/10.1093/nar/25.24.4876
Tomati U, Grappelli A, Galli E (1988) The hormone-like effect of earthworm casts on plant growth. Biol Fertil Soils 5(4):288–294. https://doi.org/10.1007/BF00262133
Vivas A, Moreno B, Garcia-Rodriguez S, Benitez E (2009) Assessing the impact of composting and vermicomposting on bacterial community size and structure, and microbial functional diversity of an olive-mill waste. Bioresour Technol 100(3):1319–1326. https://doi.org/10.1016/j.biortech.2008.08.014
Wani SP, Pathak P, Jangawad LS, Eswaran H, Singh P (2003) Improved management of Vertisols in the semi-arid tropics for increased productivity and soil carbon sequestration. Soil Use Manag 19:217–222. https://doi.org/10.1111/j.1475-2743.2003.tb00307.x
Wani SP, Chander G, Vineela C (2014) Vermicomposting: recycling wastes into valuable manure for sustained crop intensification in the semi-arid tropics. In: Ramesh, Chandra, Raverkar KP (ed) Bioresources for sustainable plant nutrient management. Satish Serial Publishing House, Delhi, India, pp 123–151. https://www.researchgate.net/publication/270528480_Vermicomposting_Recycling_Wastes_into_Valuable_Manure_for_Sustained_Crop_Intensification_in_the_Semi-Arid_Tropics. http://hdl.handle.net/10568/75837
Yousefi J, Younesi H, Ghasempoury SM (2013) Co-composting of municipal solid waste with sawdust: improving compost quality. Clean Soil Air Water 41(2):185–194. https://doi.org/10.1002/clen.201100315
Zhang BG, Li GT, Shen TS, Wang JK, Sun Z (2000) Changes in microbial biomass C, N, and P and enzyme activities in soil incubated with the earthworms Metaphire guillelmi or Eisenia foetida. Soil Biol Biochem 32:2055–2062. https://doi.org/10.1016/S0038-0717(00)00111-5
Acknowledgements
Funding support in on-farm scaling-out/evaluation from Government of Andhra Pradesh and Sir Dorabji Tata Trust is gratefully acknowledged. Authors acknowledge help from Mr K Srinivas in assisting to automatically record composting biomass temperatures; and Mr G Pardhasaradhi in chemical analysis of samples. Help from Mr LS Jangawad and Mr Krishna Reddy in facilitating to conduct this study and Dr A Rathore in statistically analyzing yield data is duly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to urisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Chander, G., Wani, S.P., Gopalakrishnan, S. et al. Microbial consortium culture and vermi-composting technologies for recycling on-farm wastes and food production. Int J Recycl Org Waste Agricult 7, 99–108 (2018). https://doi.org/10.1007/s40093-018-0195-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40093-018-0195-9